FDA Approves Valneva's Chikungunya Virus Vaccine
2023年11月10日 - 8:35AM
Dow Jones News
By Stephen Nakrosis
The Food and Drug Administration said it granted approval of
Ixchiq, the first vaccine for chikungunya virus, to Valneva
Austria.
Valneva Austria's parent company is France's Valneva SE.
The FDA said Thursday chikungunya is a mosquito-borne disease,
which causes symptoms including headache, fever, joint pain and
rash. The virus is an emerging global health threat, with at least
5 million cases of infection reported over the past 15 years,
according to the FDA.
Ixchiq was approved for those 18 years of age and older who are
at increased risk of exposure to chikungunya virus, the FDA
said.
Trade in Valneva's America depositary receipts was halted after
hours Thursday because of pending news.
Write to Stephen Nakrosis at stephen.nakrosis@wsj.com
(END) Dow Jones Newswires
November 09, 2023 18:20 ET (23:20 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
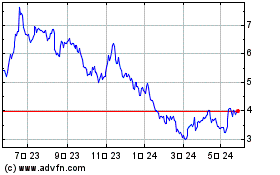
Valneva (EU:VLA)
過去 株価チャート
から 4 2024 まで 5 2024
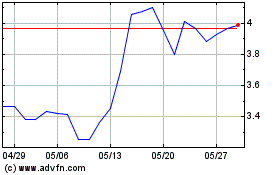
Valneva (EU:VLA)
過去 株価チャート
から 5 2023 まで 5 2024