false 0001817713 0001817713 2023-07-17 2023-07-17
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): July 17, 2023
Janux Therapeutics, Inc.
(Exact name of Registrant as Specified in Its Charter)
|
|
|
|
|
| Delaware |
|
001-40475 |
|
82-2289112 |
| (State or Other Jurisdiction of Incorporation) |
|
(Commission File Number) |
|
(IRS Employer
Identification No.) |
|
|
|
| 10955 Vista Sorrento Parkway, Suite 200 |
|
|
| San Diego, California |
|
92130 |
| (Address of Principal Executive Offices) |
|
(Zip Code) |
Registrant’s Telephone Number, Including Area Code: (858) 751-4493
N/A
(Former Name or Former Address, if Changed Since Last Report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| ☐ |
Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| ☐ |
Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| ☐ |
Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| ☐ |
Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
|
|
|
|
|
| Title of each class |
|
Trading
Symbol(s) |
|
Name of each exchange on which registered |
| Common Stock, $0.001 par value per share |
|
JANX |
|
Nasdaq Global Market |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ☒
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☒
| Item 2.02 |
Results of Operations and Financial Condition. |
As of the date hereof, based on currently available information, Janux Therapeutics, Inc (“we,” “us,” “our,” and the “Company”) expects its cash, cash equivalents, restricted cash and short-term investments as of June 30, 2023 to be approximately $304.1 million. Inclusive in this amount is $0.8 million of restricted cash that is not available for current use.
The preliminary results set forth above are based on management’s initial review of our operations for the quarter ended June 30, 2023 and are subject to completion of financial closing procedures. Actual results may differ materially from these preliminary results as a result of the completion of financial closing procedures, final adjustments, and other developments arising between now and the time that our financial results are finalized. In addition, these preliminary results are not a comprehensive statement of our financial results for the quarter ended June 30, 2023, should not be viewed as a substitute for full financial statements prepared in accordance with generally accepted accounting principles, and are not necessarily indicative of our results for any future period. The preliminary financial results included in this Item 2.02 have been prepared by, and are the responsibility of, our management. Ernst & Young LLP has not audited, reviewed, compiled, or applied agreed-upon procedures with respect to the preliminary financial results. Accordingly, Ernst & Young LLP does not express an opinion or any other form of assurance with respect thereto.
| Item 7.01 |
Regulation FD Disclosure. |
On July 17, 2023, the Company issued a press release announcing interim clinical data. A copy of the press release is attached as Exhibit 99.1 to this report.
The information in this Item 7.01 of this report (including Exhibit 99.1) is furnished and shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended, or subject to the liabilities of that section or Sections 11 and 12(a)(2) of the Securities Act of 1933, as amended. The information shall not be deemed incorporated by reference into any other filing with the Securities and Exchange Commission (the “SEC”) made by the Company, whether made before or after today’s date, regardless of any general incorporation language in such filing, except as shall be expressly set forth by specific references in such filing.
The Company is making available a corporate presentation reporting interim clinical data. A copy of the corporate presentation is attached as Exhibit 99.2 to this report, and is incorporated herein by reference. The corporate presentation will also be available under the “Investors” section of the Company’s website.
The corporate presentation reports the Company’s interim clinical data as of June 28, 2023 for PSMA-TRACTr (JANX007) in adult subjects with metastatic castration-resistant prostate cancer. The data from eight patients from the first three cohorts of the dose escalation portion of the Phase 1a clinical trial, as of June 28, 2023 show that JANX007 has been generally well tolerated, with no dose-limiting toxicities. JANX007 has been dosed at 300µg flat dose, which is above the projected maximum tolerable dose of the parental T cell engagers. JANX007 showed clinical activity at both 100ug and 300ug flat doses and yielded best overall PSA reductions between 31% and 67% in four of the five patients who received a flat dose. Grade 1 or 2 cytokine release syndrome (“CRS”) was observed only in patients who demonstrated PSA reductions, suggesting cytokine release resulting from anti-tumor activity was associated with CRS. No Grade 3 CRS has been observed. The most common, non-CRS related adverse event observations have been generally consistent with tumor-specific activity and reduced PSMA(+) healthy tissue activity. No transaminitis was observed. JANX007 clinical development has moved into step-dosing and dose optimization with the goal to enhance efficacy while maintaining suitable safety results.
The information in Item 2.02 above is incorporated by reference into this Item 8.01.
| Item 9.01 |
Financial Statements and Exhibits. |
(d) Exhibits
Forward-Looking Statements
This report contains certain forward-looking statements that involve risks and uncertainties that could cause actual results to be materially different from historical results or from any future results expressed or implied by such forward-looking statements. Such forward-looking statements include statements regarding, among other things, our ability to bring new treatments to cancer patients in need, expectations regarding the timing, scope and results of our development activities, including our ongoing and planned clinical trials and that the interim data results supporting continued clinical development of JANX007, and the potential benefits of our product candidates and platform technologies. Factors that may cause actual results to differ materially include the risk that interim results of a clinical trial are not necessarily indicative of final results and one or more of the clinical outcomes may materially change as patient enrollment continues, following more comprehensive reviews of the data and as more patient data becomes available, the risk that compounds that appear promising in early research do not demonstrate safety and/or efficacy in later preclinical studies or clinical trials, the risk that we may not obtain approval to market our product candidates, uncertainties associated with performing clinical trials, regulatory filings and applications, risks associated with reliance on third parties to successfully conduct clinical trials, the risks associated with reliance on outside financing to meet capital requirements, and other risks associated with the process of discovering, developing and commercializing drugs that are safe and effective for use as human therapeutics, and in the endeavor of building a business around such drugs. You are urged to consider statements that include the words “may,” “will,” “would,” “could,” “should,” “believes,” “estimates,” “projects,” “promise,” “potential,” “expects,” “plans,” “anticipates,” “intends,” “continues,” “designed,” “goal,” or the negative of those words or other comparable words to be uncertain and forward-looking. For a further list and description of the risks and uncertainties we face, please refer to our periodic and other filings with the SEC, which are available at www.sec.gov. Such forward-looking statements are current only as of the date they are made, and we assume no obligation to update any forward-looking statements, whether as a result of new information, future events or otherwise.
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
|
|
|
|
|
|
|
|
|
|
|
JANUX THERAPEUTICS, INC. |
|
|
|
|
|
|
|
|
By: |
|
/s/ David Campbell, Ph.D. |
|
|
|
|
|
|
David Campbell, Ph.D. |
|
|
|
|
|
|
President and Chief Executive Officer |
Date: July 17, 2023
Exhibit 99.1

Janux Therapeutics Announces Positive Interim Clinical Data from Phase 1a Dose Escalation for PSMA-TRACTr JANX007 and an
Update on Pipeline Programs
| |
• |
|
Multiple patients treated with PSMA-TRACTr JANX007 have achieved meaningful PSA drops coupled with manageable
safety and CRS |
| |
• |
|
PSMA-TRACTr JANX007 and EGFR-TRACTr JANX008 have been generally well tolerated at levels above the projected
maximum tolerable dose of the parental T cell engagers |
| |
• |
|
JANX007 preliminary data showed encouraging safety results consistent with
on-tumor activity with no dose-limiting toxicities |
| |
• |
|
PK exposure demonstrated TRACTr activation with lack of TCE accumulation |
| |
• |
|
No treatment-emergent ADA titers observed |
| |
• |
|
Janux to host virtual investor event today at 4:00 PM Eastern Time |
SAN DIEGO, July 17, 2023 – Janux Therapeutics, Inc. (Nasdaq: JANX) (Janux), a clinical-stage biopharmaceutical company developing a broad pipeline
of novel immunotherapies by applying its proprietary technology to its Tumor Activated T Cell Engager (TRACTr) and Tumor Activated Immunomodulator (TRACIr) platforms, today announced interim Phase 1 clinical data for PSMA-TRACTr JANX007 in adult
subjects with metastatic castration-resistant prostate cancer (mCRPC) and provided a pipeline update. Janux will host a virtual investor event today at 4:00 PM ET. To register for the event, please click here.
“These data showing tumor-activated T cell engagement in patients with prostate cancer represent the first
in-human data for the TRACTr platform and give us confidence for continuing clinical development of JANX007 and advancing future programs from this platform. We are encouraged by these positive interim
clinical data for JANX007, which displayed PSA reductions coupled with low-grade and transient CRS, which we believe to be consistent with the TRACTr mechanism of action” said David Campbell, Ph.D.
“The concept of tumor-specific activation as a new therapeutic strategy for T cell engagers in solid tumors has taken an important step forward, and the JANX007 program has hit an important milestone in its clinical development.”
Interim Clinical Data for PSMA-TRACTr JANX007 in mCRPC as of June 28, 2023
The data from eight patients from the first three cohorts of the dose escalation portion of the Phase 1a clinical trial, as of June 28, 2023 show that
JANX007 has been generally well tolerated, with no dose-limiting toxicities. JANX007 has been dosed at 300µg flat dose, which is above the projected maximum tolerable dose of the parental T cell engagers. JANX007 showed clinical activity at
both 100µg and 300µg flat doses and yielded best overall PSA reductions between 31% and 67% in four of the five patients who received a flat dose. Grade 1 or 2 cytokine release syndrome (CRS) was observed only in patients who
demonstrated PSA reductions, suggesting cytokine release resulting from anti-tumor activity was associated with CRS. No Grade 3 CRS has been observed. The most common, non-CRS related adverse event
observations have been generally consistent with tumor-specific activity and reduced PSMA(+) healthy tissue activity. No transaminitis was observed. JANX007 clinical development has moved into step-dosing and dose optimization with the goal to
enhance efficacy while maintaining suitable safety results.
The TRACTr technology is designed to create potent T cell engagers (TCEs) by tumor-specific
activation via mask cleavage by tumor proteases. Plasma levels in patients exhibited prolonged TRACTr exposure, clear evidence of activation as measured by a specific cleavage fragment, and lack of accumulation of the active TCE in the blood. The
lack of TCE accumulation shows consistency with TRACTr design principles and suggest that observed PSA reductions have been a result of tumor activation and not systemic TCE exposure.
Conference Call Information
Janux will host a live conference call and webcast today at 4:00 PM ET. A live question and answer session will follow the formal presentation.
Conference call participant joining details:
United States: 1
(888) 412-4131
United States (Local): 1 (646) 960-0134
International: 1 (888) 412-4131
Access code: 7426984
To access the live webcast, please visit
the Investors section of the Company’s website. A replay of the webcast presentation will be available on the Company’s website at https://investors.januxrx.com for approximately 30 days.
Janux’s TRACTr and TRACIr Pipeline
Janux’s
first clinical candidate, JANX007, is a TRACTr that targets PSMA and is being investigated in a Phase 1 clinical trial in adult subjects with metastatic castration-resistant prostate cancer (mCRPC). Janux’s second clinical candidate, JANX008,
is a TRACTr that targets EGFR and is being studied in a Phase 1 clinical trial for the treatment of multiple solid cancers including colorectal cancer, squamous cell carcinoma of the head and neck, non-small
cell lung cancer, and renal cell carcinoma. Janux’s TRACIr drug candidate, JANX009, is designed for targeting both the programmed death-ligand 1 (PD-L1) receptor as well as the costimulatory CD28 receptor
on T cells and is being investigated in preclinical studies for the treatment of solid tumors. Janux is also applying its proprietary technology to develop a TRACTr designed to target TROP2, a clinically validated anti-tumor target that is
overexpressed in various cancer types, such as breast, lung, urothelial, endometrial, ovarian, prostate, pancreatic, gastric, colon, head and neck, and glioma. In addition to named programs, Janux is generating a number of unnamed TRACTr and TRACIr
programs for potential future development.
About Janux Therapeutics
Janux is a clinical-stage biopharmaceutical company developing tumor-activated immunotherapies for cancer. Janux’s proprietary technology enabled the
development of two distinct bispecific platforms: Tumor Activated T Cell Engagers (TRACTr) and Tumor Activated Immunomodulators (TRACIr). The goal of both platforms is to provide cancer patients with safe and effective therapeutics that direct and
guide their immune system to eradicate tumors while minimizing safety concerns. Janux is currently developing a broad pipeline of TRACTr and TRACIr therapeutics directed at several targets to treat solid tumors. Janux has two TRACTr therapeutic
candidates in clinical trials, the first targeting PSMA is in development for prostate cancer, and the second targeting EGFR is being developed for colorectal, lung, head and neck, and renal cancers. For more information, please
visit www.januxrx.com and follow us on LinkedIn.
Forward-Looking Statements
This news release contains certain forward-looking statements that involve risks and uncertainties that could cause actual results to be materially different
from historical results or from any future results expressed or implied by such forward-looking statements. Such forward-looking statements include statements regarding, among other things, Janux’s ability to bring new treatments to cancer
patients in need, expectations regarding the timing, scope and results of Janux’s development activities, including its ongoing and planned clinical trials and that the interim data results supporting continued clinical
development of JANX007, and the potential benefits of Janux’s product candidates and platform technologies. Factors that may cause actual results to differ materially include the risk that
interim results of a clinical trial are not necessarily indicative of final results and one or more of the clinical outcomes may materially change as patient enrollment continues, following more comprehensive reviews of the data and as more patient
data becomes available, the risk that compounds that appear promising in early research do not demonstrate safety and/or efficacy in later preclinical studies or clinical trials, the risk that Janux may not obtain approval to market its product
candidates, uncertainties associated with performing clinical trials, regulatory filings and applications, risks associated with reliance on third parties to successfully conduct clinical trials, the risks associated with reliance on outside
financing to meet capital requirements, and other risks associated with the process of discovering, developing and commercializing drugs that are safe and effective for use as human therapeutics, and in the endeavor of building a business around
such drugs. You are urged to consider statements that include the words “may,” “will,” “would,” “could,” “should,” “believes,” “estimates,” “projects,”
“promise,” “potential,” “expects,” “plans,” “anticipates,” “intends,” “continues,” “designed,” “goal,” or the negative of those words or other comparable words
to be uncertain and forward-looking. For a further list and description of the risks and uncertainties Janux faces, please refer to Janux’s periodic and other filings with the Securities and Exchange Commission, which are available at
www.sec.gov. Such forward-looking statements are current only as of the date they are made, and Janux assumes no obligation to update any forward-looking statements, whether as a result of new information, future events or otherwise.
Contacts
Investors:
Andy Meyer
Janux Therapeutics
ameyer@januxrx.com
(202)
215-2579
Media:
Jessica Yingling, Ph.D.
Little Dog
Communications Inc.
jessica@litldog.com
(858) 344-8091

Program Updates July 2023 Exhibit
99.2

Forward-looking statements and
disclaimers This presentation includes certain forward-looking statements that involve risks and uncertainties that could cause actual results to be materially different from historical results or from any future results expressed or implied by such
forward-looking statements regarding Janux Therapeutics, Inc. (the “Company”). These forward-looking statement include, but are not limited to, those regarding the Company’s ability to bring new treatments to patients in need, the
progress and expected timing of the Company’s drug development programs, clinical development plans and timelines, the timing of and plans for regulatory filings, market size and opportunity, the Company’s strategy and intellectual
property matters, and estimates regarding the Company’s expenses, capital requirements, and needs for additional financing. Because such statements are subject to risks and uncertainties, actual results may differ materially from those
expressed or implied by such forward-looking statements. Factors that may cause actual results to differ materially include the risk that compounds that appeared promising in early research do not demonstrate safety and/or efficacy in later
preclinical studies or clinical trials, the risk that the Company may not obtain approval to market its product candidates, uncertainties associated with performing clinical trials, regulatory filings and applications, risks associated with reliance
on third parties to successfully conduct clinical trials, the risks associated with reliance on outside financing to meet capital requirements, and other risks associated with the process of discovering, developing and commercializing drugs that are
safe and effective for use as human therapeutics, and in the endeavor of building a business around such drugs. Also, interim results of a clinical trial are not necessarily indicative of final results and one or more of the clinical outcomes may
materially change as patient enrollment continues, following more comprehensive reviews of the data and more patient data become available. In light of these risks, uncertainties, contingencies and assumptions, the events or circumstances referred
to in the forward-looking statements may not occur. For a further list and description of the risks and uncertainties that the Company faces, please refer to the Company’s periodic and other filings with the Securities and Exchange Commission,
which are available at www.sec.gov. Such forward-looking statements are current only as of the date they are made, and the Company assumes no obligation to update any forward-looking statements, whether as a result of new information, future events,
or otherwise. The trademarks included herein are the property of the owners thereof and are used for reference purposes only. Such use should not be construed as an endorsement of such products. This presentation concerns therapeutic product
candidates that are in preclinical and clinical development and which have not yet been approved for marketing by the U.S. Food and Drug Administration. They are currently limited by federal law to investigational use, and no representation is made
as to their safety or effectiveness for the purposes for which they are being investigated.
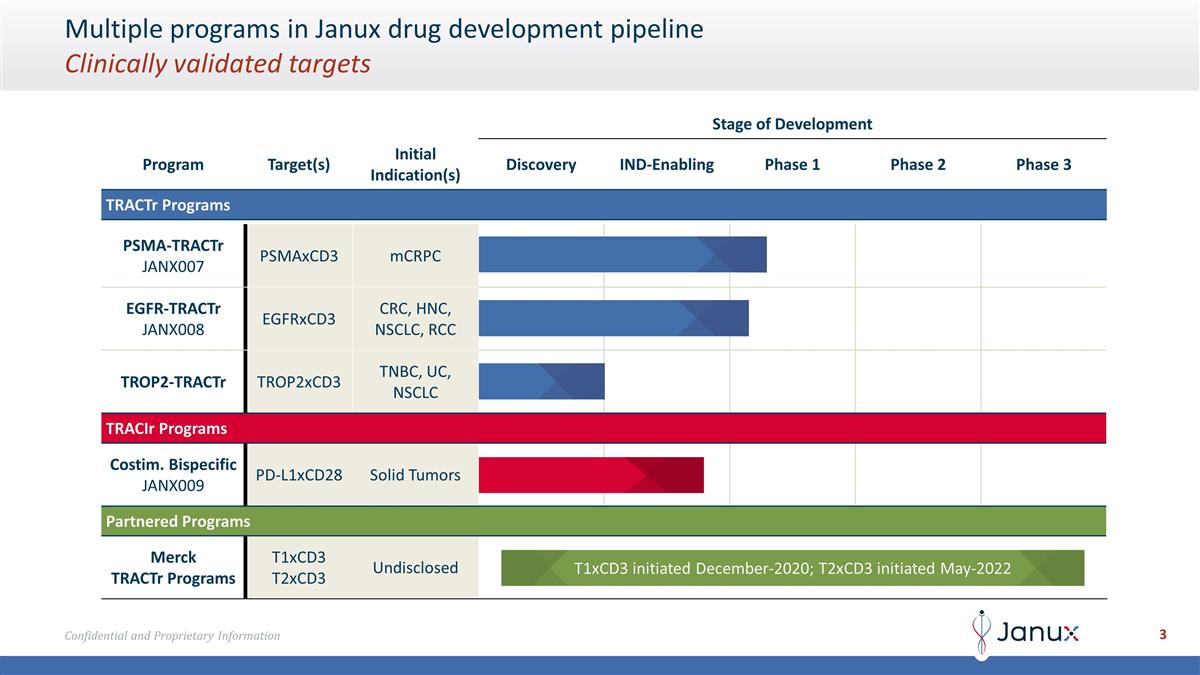
Multiple programs in Janux drug
development pipeline Clinically validated targets Stage of Development Program Target(s) Initial Indication(s) Discovery IND-Enabling Phase 1 Phase 2 Phase 3 TRACTr Programs PSMA-TRACTr JANX007 PSMAxCD3 mCRPC EGFR-TRACTr JANX008 EGFRxCD3 CRC, HNC,
NSCLC, RCC TROP2-TRACTr TROP2xCD3 TNBC, UC, NSCLC TRACIr Programs Costim. Bispecific JANX009 PD-L1xCD28 Solid Tumors Partnered Programs Merck TRACTr Programs T1xCD3 T2xCD3 Undisclosed T1xCD3 initiated December-2020; T2xCD3 initiated May-2022
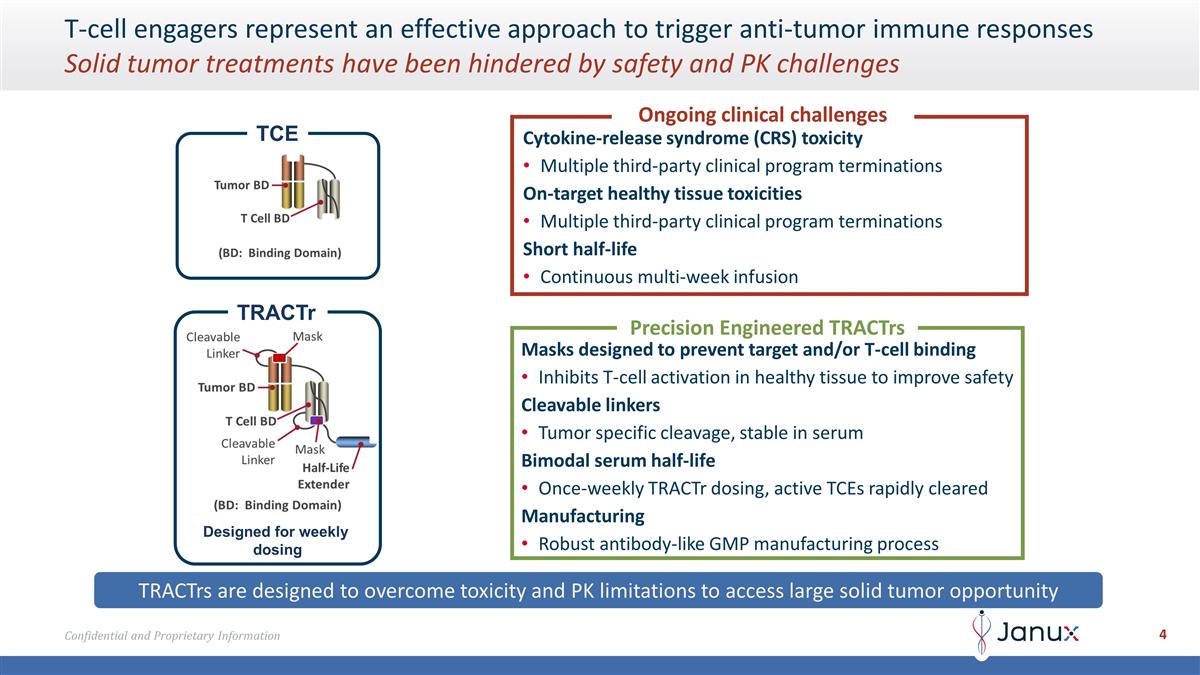
T-cell engagers represent an effective
approach to trigger anti-tumor immune responses Solid tumor treatments have been hindered by safety and PK challenges Ongoing clinical challenges Cytokine-release syndrome (CRS) toxicity Multiple third-party clinical program terminations On-target
healthy tissue toxicities Multiple third-party clinical program terminations Short half-life Continuous multi-week infusion TCE Precision Engineered TRACTrs Masks designed to prevent target and/or T-cell binding Inhibits T-cell activation in healthy
tissue to improve safety Cleavable linkers Tumor specific cleavage, stable in serum Bimodal serum half-life Once-weekly TRACTr dosing, active TCEs rapidly cleared Manufacturing Robust antibody-like GMP manufacturing process Designed for weekly
dosing TRACTr TRACTrs are designed to overcome toxicity and PK limitations to access large solid tumor opportunity

TRACTr platform update overview
Interim clinical data provides first insights into TRACTr performance in humans* PSMA-TRACTr clinical data consistent with TRACTr mechanism-of-action Meaningful PSA drops coupled with manageable safety and CRS profile PK exposure demonstrated TRACTr
activation with lack of TCE accumulation No significant treatment-emergent ADA titers observed Both PSMA-TRACTr and EGFR-TRACTr have been generally well tolerated at dose levels above the projected MTD of the parental TCEs Development programs
progressing on-schedule PDL1xCD28-TRACIr IND enabling studies and manufacturing ongoing TROP2-TRACTr development compound identification in-progress Pipeline development Multiple TCE and CD28 bispecific programs progressing *28 June 2023 data
cutoff; MTD maximum tolerated dose; TCE T-cell engager; ADA anti drug antibody

PSMA-TRACTr Program
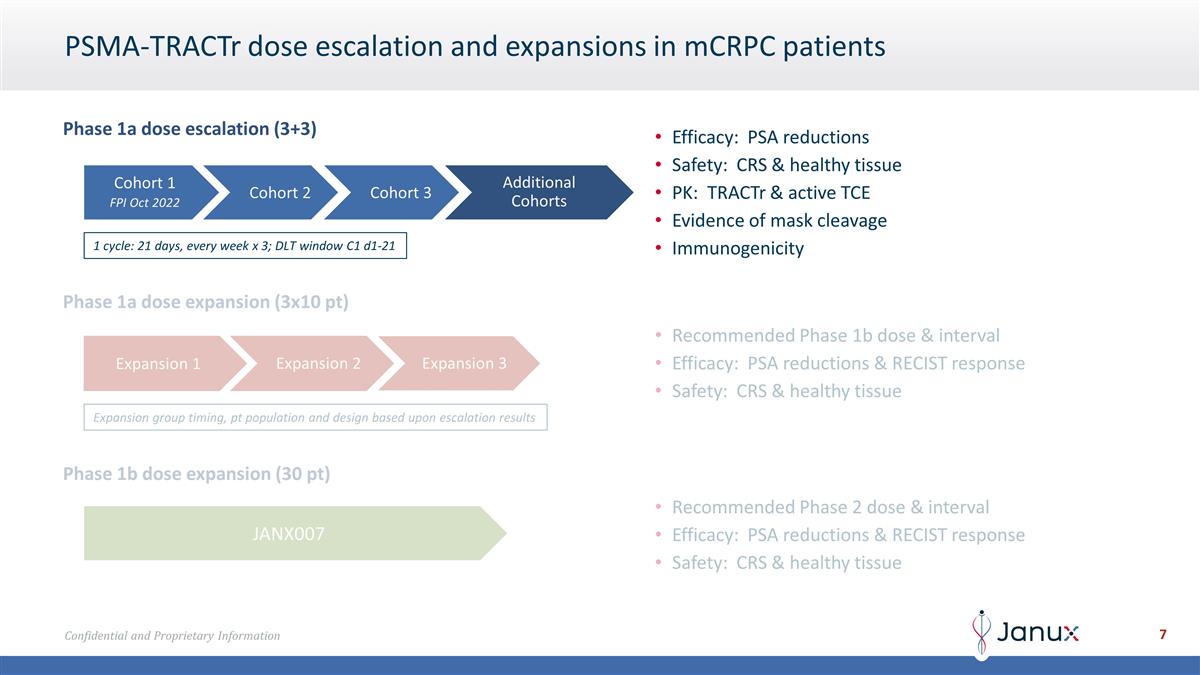
PSMA-TRACTr dose escalation and
expansions in mCRPC patients Phase 1a dose escalation (3+3) Cohort 1 FPI Oct 2022 Cohort 2 Additional Cohorts Cohort 3 Efficacy: PSA reductions Safety: CRS & healthy tissue PK: TRACTr & active TCE Evidence of mask cleavage Immunogenicity 1
cycle: 21 days, every week x 3; DLT window C1 d1-21 Phase 1a dose expansion (3x10 pt) Recommended Phase 1b dose & interval Efficacy: PSA reductions & RECIST response Safety: CRS & healthy tissue Phase 1b dose expansion (30 pt) JANX007
Recommended Phase 2 dose & interval Efficacy: PSA reductions & RECIST response Safety: CRS & healthy tissue Expansion group timing, pt population and design based upon escalation results Expansion 1 Expansion 2 Expansion 3
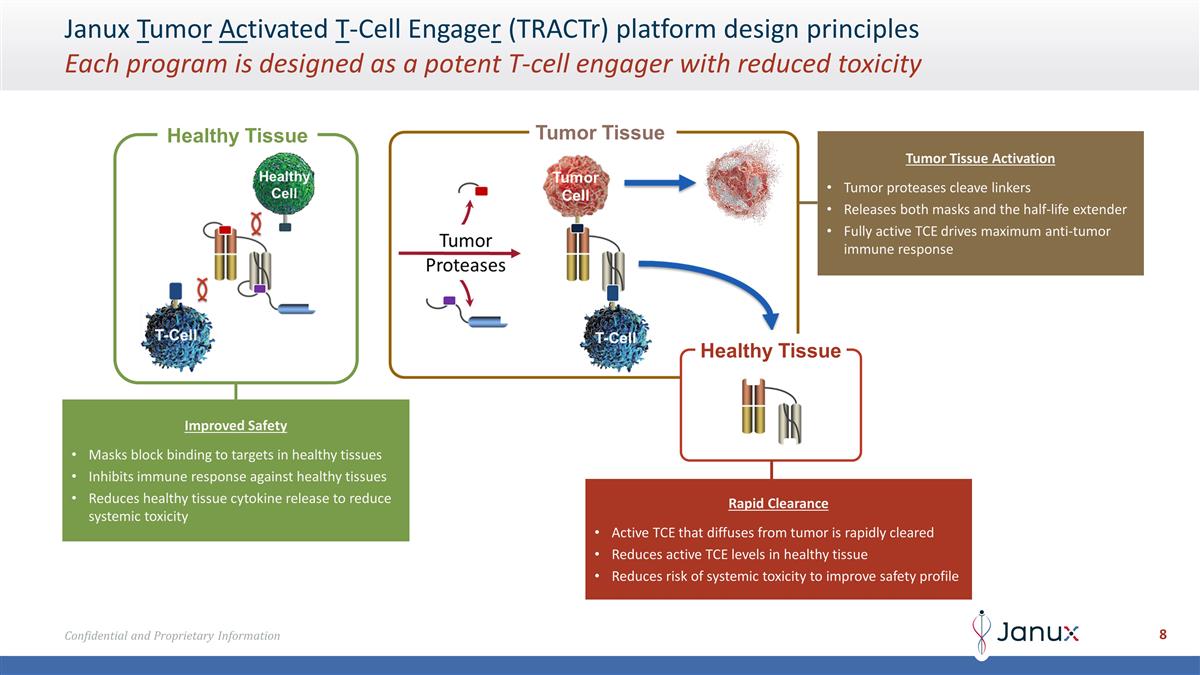
Janux Tumor Activated T-Cell Engager
(TRACTr) platform design principles Each program is designed as a potent T-cell engager with reduced toxicity Healthy Tissue Healthy Tissue Improved Safety Masks block binding to targets in healthy tissues Inhibits immune response against healthy
tissues Reduces healthy tissue cytokine release to reduce systemic toxicity Tumor Tissue Activation Tumor proteases cleave linkers Releases both masks and the half-life extender Fully active TCE drives maximum anti-tumor immune response Rapid
Clearance Active TCE that diffuses from tumor is rapidly cleared Reduces active TCE levels in healthy tissue Reduces risk of systemic toxicity to improve safety profile Tumor Tissue
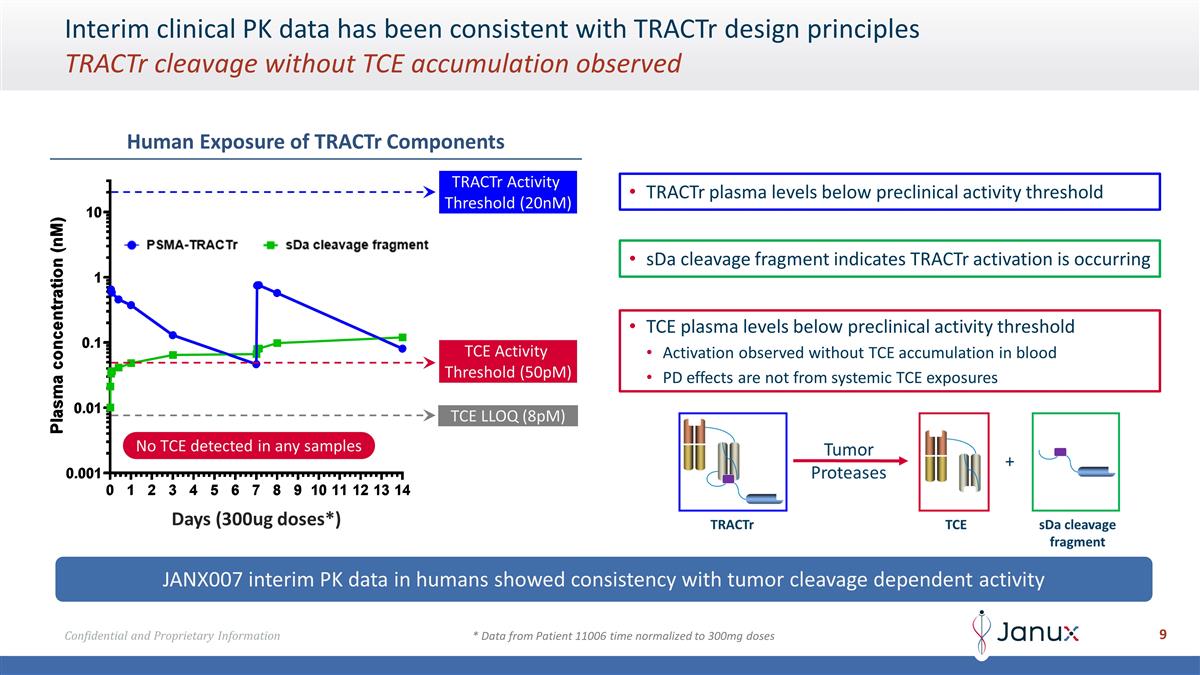
Interim clinical PK data has been
consistent with TRACTr design principles TRACTr cleavage without TCE accumulation observed JANX007 interim PK data in humans showed consistency with tumor cleavage dependent activity Human Exposure of TRACTr Components TRACTr plasma levels below
preclinical activity threshold TRACTr Activity Threshold (20nM) TCE Activity Threshold (50pM) TCE LLOQ (8pM) No TCE detected in any samples TCE plasma levels below preclinical activity threshold Activation observed without TCE accumulation in blood
PD effects are not from systemic TCE exposures sDa cleavage fragment indicates TRACTr activation is occurring Days (300ug doses*) * Data from Patient 11006 time normalized to 300mg doses
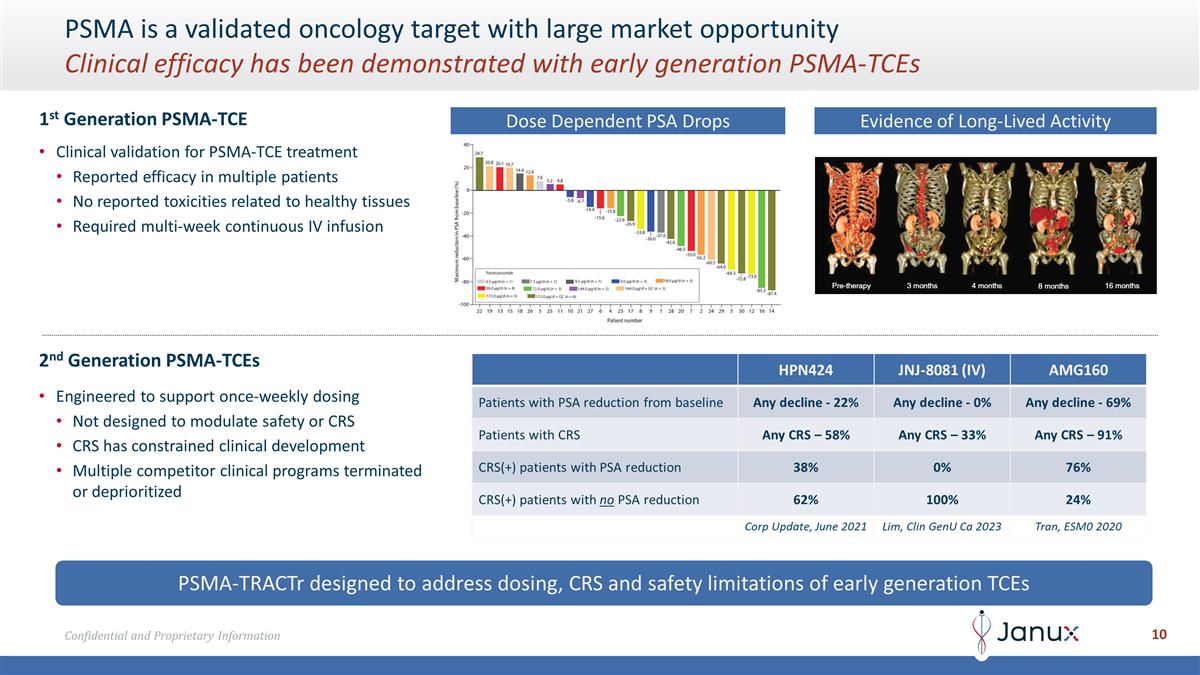
PSMA-TRACTr designed to address
dosing, CRS and safety limitations of early generation TCEs PSMA is a validated oncology target with large market opportunity Clinical efficacy has been demonstrated with early generation PSMA-TCEs Clinical validation for PSMA-TCE treatment Reported
efficacy in multiple patients No reported toxicities related to healthy tissues Required multi-week continuous IV infusion 1st Generation PSMA-TCE Engineered to support once-weekly dosing Not designed to modulate safety or CRS CRS has constrained
clinical development Multiple competitor clinical programs terminated or deprioritized 2nd Generation PSMA-TCEs Dose Dependent PSA Drops Evidence of Long-Lived Activity
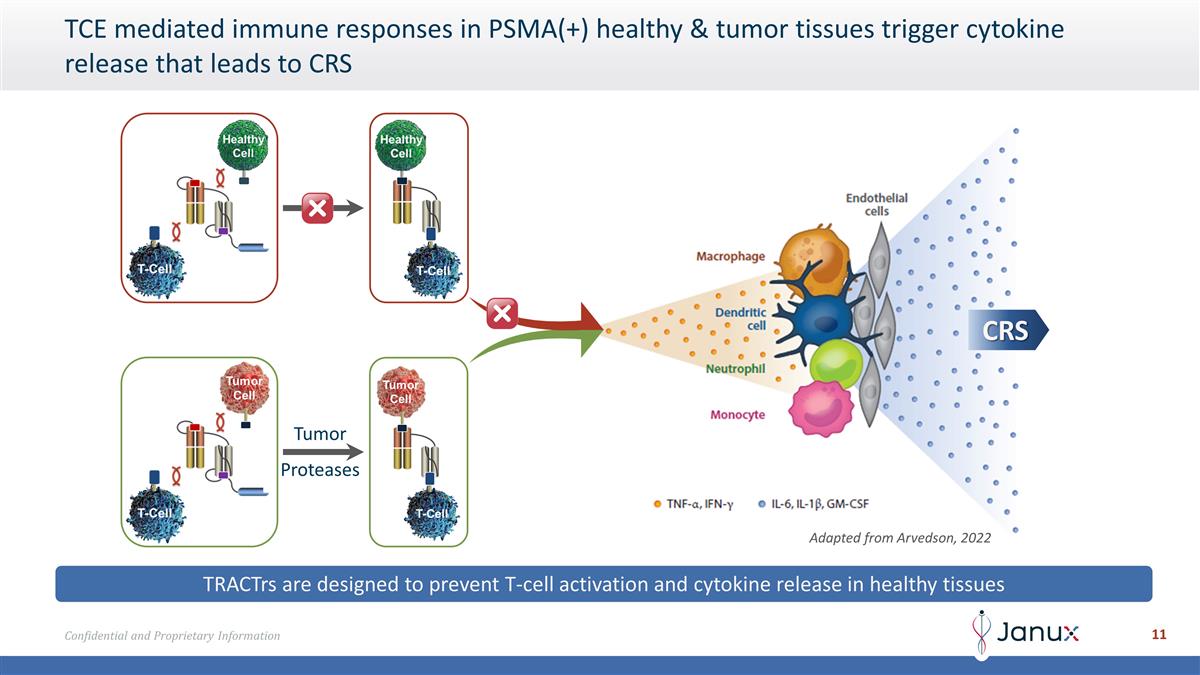
TCE mediated immune responses in
PSMA(+) healthy & tumor tissues trigger cytokine release that leads to CRS TRACTrs are designed to prevent T-cell activation and cytokine release in healthy tissues Adapted from Arvedson, 2022 CRS Tumor Proteases
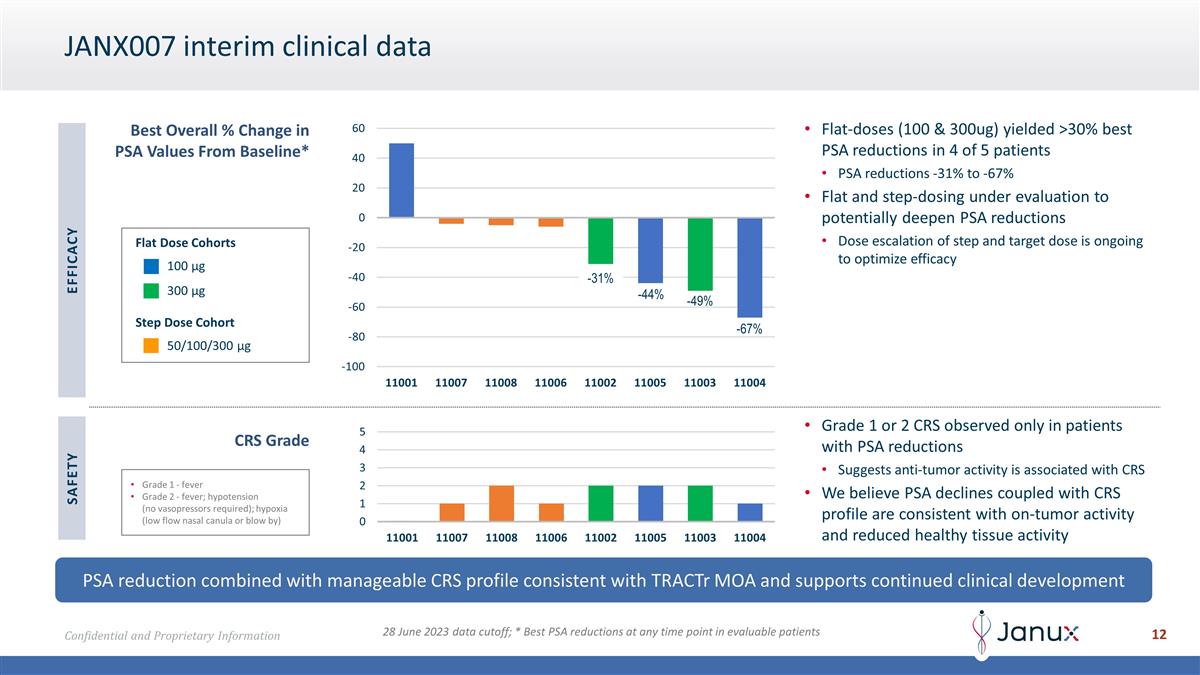
JANX007 interim clinical data Best
Overall % Change in PSA Values From Baseline* PSA reduction combined with manageable CRS profile consistent with TRACTr MOA and supports continued clinical development Grade 1 - fever Grade 2 - fever; hypotension (no vasopressors required); hypoxia
(low flow nasal canula or blow by) Flat-doses (100 & 300ug) yielded >30% best PSA reductions in 4 of 5 patients PSA reductions -31% to -67% Flat and step-dosing under evaluation to potentially deepen PSA reductions Dose escalation of step and
target dose is ongoing to optimize efficacy CRS Grade Grade 1 or 2 CRS observed only in patients with PSA reductions Suggests anti-tumor activity is associated with CRS We believe PSA declines coupled with CRS profile are consistent with on-tumor
activity and reduced healthy tissue activity EFFICACY SAFETY 100 µg 300 µg 50/100/300 µg Flat Dose Cohorts Step Dose Cohort 28 June 2023 data cutoff; * Best PSA reductions at any time point in evaluable patients -31% -44% -49%
-67%
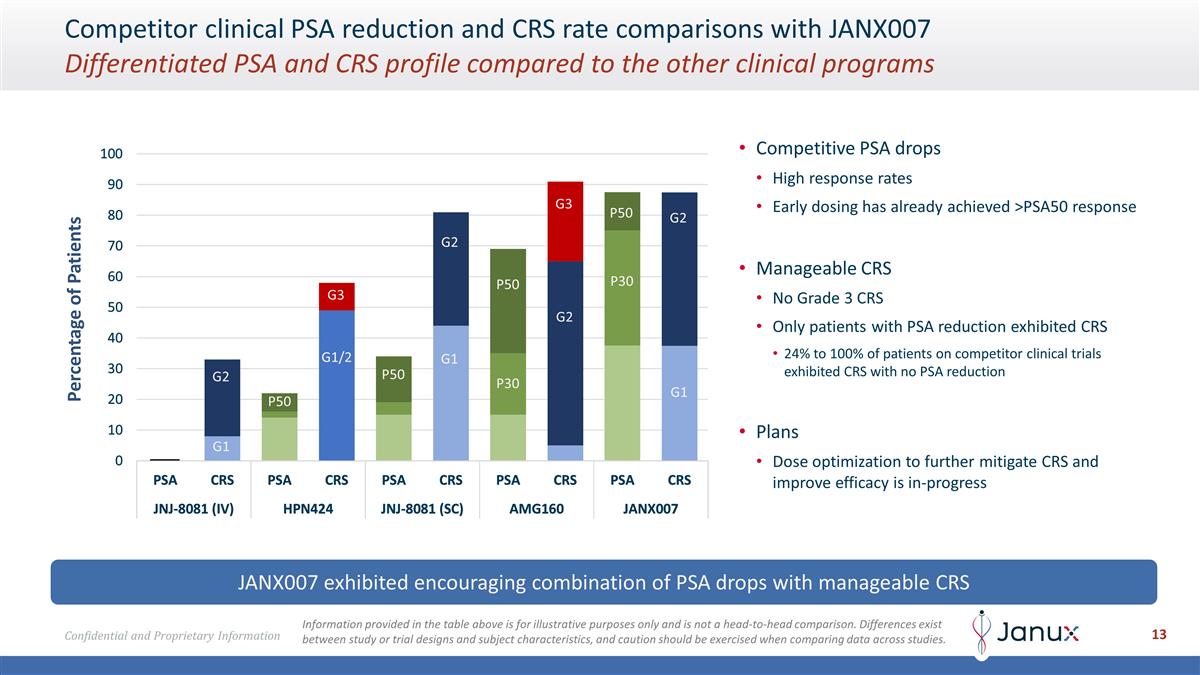
P50 P50 P50 P50 P30 P30 G3 G3 G2 G2
G2 G2 G1 G1 G1 G1/2 Competitor clinical PSA reduction and CRS rate comparisons with JANX007 Differentiated PSA and CRS profile compared to the other clinical programs JANX007 exhibited encouraging combination of PSA drops with manageable CRS
Competitive PSA drops High response rates Early dosing has already achieved >PSA50 response Manageable CRS No Grade 3 CRS Only patients with PSA reduction exhibited CRS 24% to 100% of patients on competitor clinical trials exhibited CRS with no
PSA reduction Plans Dose optimization to further mitigate CRS and improve efficacy is in-progress Percentage of Patients Information provided in the table above is for illustrative purposes only and is not a head-to-head comparison. Differences
exist between study or trial designs and subject characteristics, and caution should be exercised when comparing data across studies.
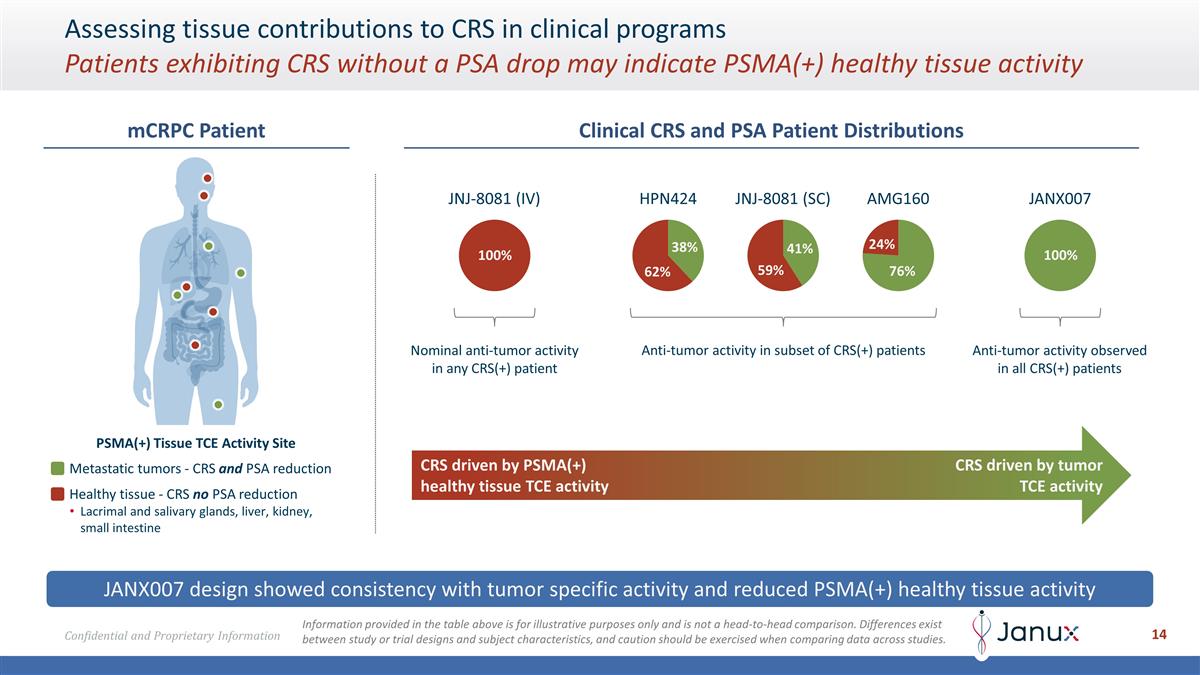
Assessing tissue contributions to
CRS in clinical programs Patients exhibiting CRS without a PSA drop may indicate PSMA(+) healthy tissue activity JANX007 design showed consistency with tumor specific activity and reduced PSMA(+) healthy tissue activity mCRPC Patient PSMA(+) Tissue
TCE Activity Site Metastatic tumors - CRS and PSA reduction Healthy tissue - CRS no PSA reduction Lacrimal and salivary glands, liver, kidney, small intestine Clinical CRS and PSA Patient Distributions CRS driven by PSMA(+) healthy tissue TCE
activity CRS driven by tumor TCE activity JNJ-8081 (IV) 100% Nominal anti-tumor activity in any CRS(+) patient JANX007 100% Anti-tumor activity observed in all CRS(+) patients AMG160 76% 24% HPN424 38% 62% 41% 59% JNJ-8081 (SC) Anti-tumor activity
in subset of CRS(+) patients Information provided in the table above is for illustrative purposes only and is not a head-to-head comparison. Differences exist between study or trial designs and subject characteristics, and caution should be
exercised when comparing data across studies.
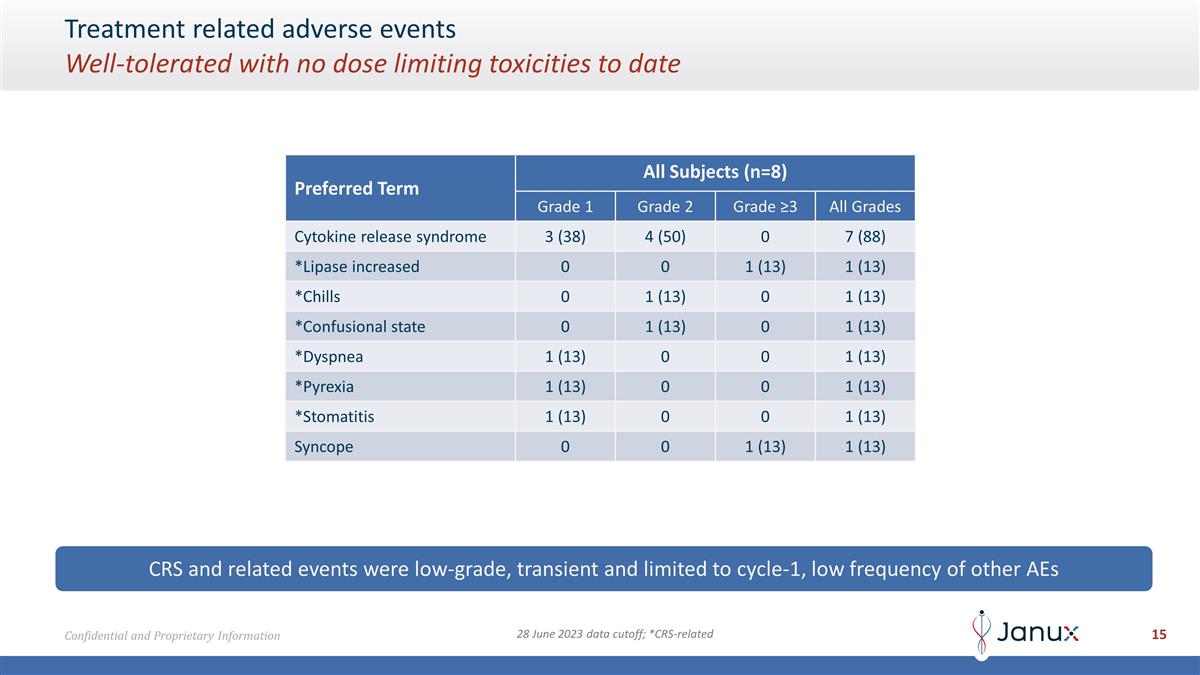
Treatment related adverse events
Well-tolerated with no dose limiting toxicities to date CRS and related events were low-grade, transient and limited to cycle-1, low frequency of other AEs 28 June 2023 data cutoff; *CRS-related Preferred Term All Subjects (n=8) Grade 1 Grade 2
Grade ≥3 All Grades Cytokine release syndrome 3 (38) 4 (50) 0 7 (88) *Lipase increased 0 0 1 (13) 1 (13) *Chills 0 1 (13) 0 1 (13) *Confusional state 0 1 (13) 0 1 (13) *Dyspnea 1 (13) 0 0 1 (13) *Pyrexia 1 (13) 0 0 1 (13) *Stomatitis 1 (13) 0
0 1 (13) Syncope 0 0 1 (13) 1 (13)
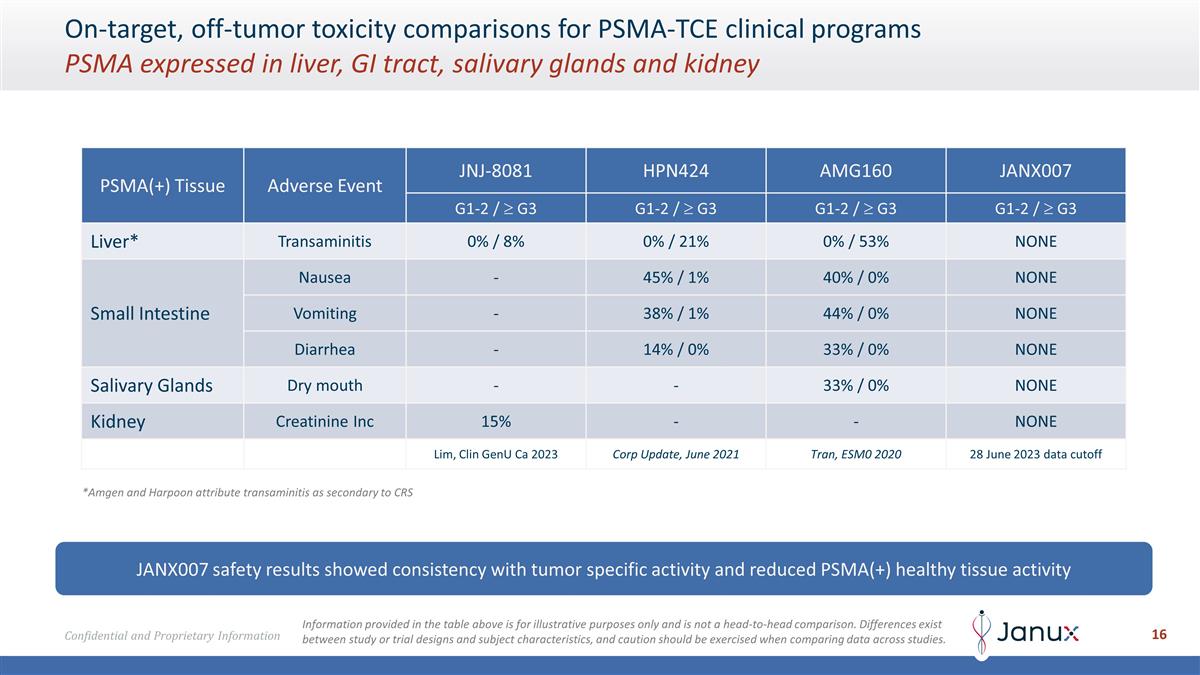
On-target, off-tumor toxicity
comparisons for PSMA-TCE clinical programs PSMA expressed in liver, GI tract, salivary glands and kidney PSMA(+) Tissue Adverse Event JNJ-8081 HPN424 AMG160 JANX007 G1-2 / ³ G3 G1-2 / ³ G3 G1-2 / ³ G3 G1-2 / ³ G3 Liver*
Transaminitis 0% / 8% 0% / 21% 0% / 53% NONE Small Intestine Nausea - 45% / 1% 40% / 0% NONE Vomiting - 38% / 1% 44% / 0% NONE Diarrhea - 14% / 0% 33% / 0% NONE Salivary Glands Dry mouth - - 33% / 0% NONE Kidney Creatinine Inc 15% - - NONE Lim, Clin
GenU Ca 2023 Corp Update, June 2021 Tran, ESM0 2020 28 June 2023 data cutoff JANX007 safety results showed consistency with tumor specific activity and reduced PSMA(+) healthy tissue activity *Amgen and Harpoon attribute transaminitis as secondary
to CRS Information provided in the table above is for illustrative purposes only and is not a head-to-head comparison. Differences exist between study or trial designs and subject characteristics, and caution should be exercised when comparing data
across studies.
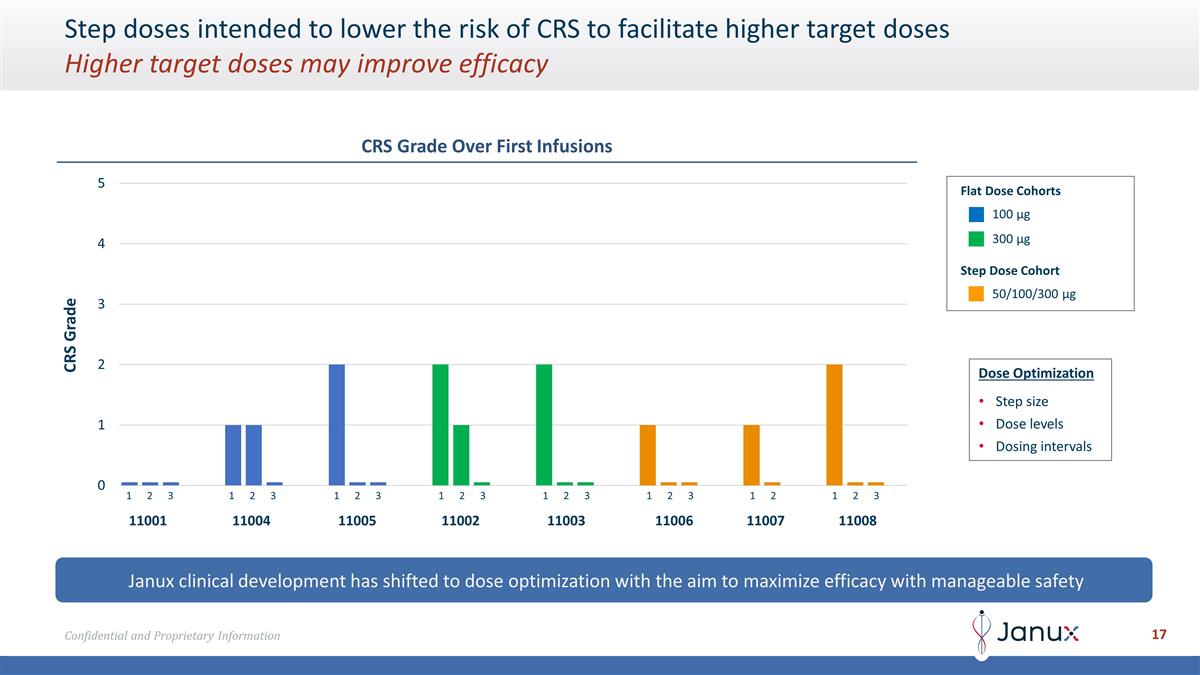
Step doses intended to lower the
risk of CRS to facilitate higher target doses Higher target doses may improve efficacy Janux clinical development has shifted to dose optimization with the aim to maximize efficacy with manageable safety CRS Grade Over First Infusions Dose
Optimization Step size Dose levels Dosing intervals 100 µg 300 µg 50/100/300 µg Flat Dose Cohorts Step Dose Cohort 11001 11004 11002 11003 11005 11007 11006 11008 123 123 123 123 123 123 123 12

Update summary TRACTr platform
clinical proof-of-principle supported by interim data First evidence of tumor-activated T cell engagement for the TRACTr platform in patients with prostate cancer Both PSMA-TRACTr and EGFR-TRACTr have been generally well tolerated at dose levels
above the projected MTD of the parental TCEs PK exposure demonstrated TRACTr activation with lack of TCE accumulation Meaningful PSA drops coupled with manageable safety and CRS profile No significant treatment-emergent ADA titers observed
PSMA-TRACTr clinical program continuing to progress Multiple patients have had substantial declines in serum PSA and activity has been observed in all CRS(+) patients Suggests reduced healthy tissue activity JANX007 preliminary data showed
encouraging initial safety results; no dose-limiting toxicities and cytokine-related adverse events have been transient and manageable Minimal healthy tissue/non-CRS related safety issues as of data cutoff Manageable CRS profile that appears linked
to anti-tumor activity Evidence of TRACTr activation without TCE accumulation 28 June 2023 data cutoff

David Campbell, Ph.D. President and
CEO 10955 Vista Sorrento Parkway San Diego, CA 92130 dcampbell@januxrx.com
v3.23.2
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 2 such as Street or Suite number
| Name: |
dei_EntityAddressAddressLine2 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the state or province.
| Name: |
dei_EntityAddressStateOrProvince |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:stateOrProvinceItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 13e
-Subsection 4c
| Name: |
dei_PreCommencementIssuerTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14d
-Subsection 2b
| Name: |
dei_PreCommencementTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Section 14a
-Number 240
-Subsection 12
| Name: |
dei_SolicitingMaterial |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Securities Act
-Number 230
-Section 425
| Name: |
dei_WrittenCommunications |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
Janux Therapeutics (NASDAQ:JANX)
過去 株価チャート
から 4 2024 まで 5 2024

Janux Therapeutics (NASDAQ:JANX)
過去 株価チャート
から 5 2023 まで 5 2024
