FDA Approves PolyPid's Trial Design for D-PLEX
2023年5月22日 - 10:54PM
Dow Jones News
By Dean Seal
The U.S. Food and Drug Administration has agreed to PolyPid's
Phase 3 trial design examining D-PLEX for the prevention of
abdominal colorectal surgical site infections.
The late-stage biopharmaceutical company said Monday that the
revised trial will recruit patients undergoing colorectal resection
surgery with large incisions that are greater than 20
centimeters.
"We are pleased with FDA feedback to our revised protocol and
are excited to resume SHIELD II trial recruitment," Chief Executive
Dikla Czaczkes Akselbrad said.
Recruitment for the trial will resume imminently, PolyPid said.
An estimated 550 additional patients will join the 40 who have
already been recruited.
Shares jumped 18.3% to 45 cents in premarket trading.
Write to Dean Seal at dean.seal@wsj.com
(END) Dow Jones Newswires
May 22, 2023 09:39 ET (13:39 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
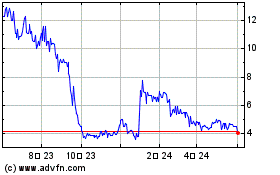
PolyPid (NASDAQ:PYPD)
過去 株価チャート
から 4 2024 まで 5 2024
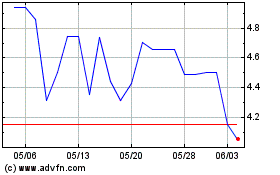
PolyPid (NASDAQ:PYPD)
過去 株価チャート
から 5 2023 まで 5 2024