NEW
YORK, July 31, 2024 /PRNewswire/ -- The
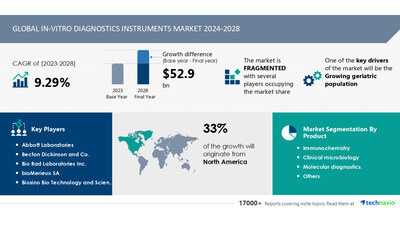
global in-vitro diagnostics instruments market size is
estimated to grow by USD 52.9 billion
from 2024-2028, according to Technavio. The market is estimated to
grow at a CAGR of 9.29% during the forecast period. Growing
geriatric population is driving market growth, with a
trend towards potential opportunities for physician office
laboratories (POL). However, strained laboratory budgets and
lowering reimbursements affecting instrument sales poses a
challenge. Key market players include Abbott Laboratories,
Becton Dickinson and Co., Bio Rad
Laboratories Inc., bioMerieux SA, Biosino Bio Technology and
Science Inc, CPC Diagnostics Pvt Ltd., DAAN Gene Co. Ltd., Danaher
Corp., F. Hoffmann La Roche Ltd., Guangzhou Wondfo Biotech Co.
Ltd., Hologic Inc., Myriad Genetics Inc., QIAGEN NV, QuidelOrtho
Corp., Shanghai Kehua Bio Engineering Co. Ltd, Shenzhen Mindray
BioMedical Electronics Co. Ltd, Siemens AG, Sysmex Corp., and
Thermo Fisher Scientific Inc..
Get a detailed analysis on regions, market
segments, customer landscape, and companies- View the
snapshot of this report
|
In-Vitro Diagnostics
Instruments Market Scope
|
|
Report
Coverage
|
Details
|
|
Base year
|
2023
|
|
Historic
period
|
2017 - 2021
|
|
Forecast
period
|
2024-2028
|
|
Growth momentum &
CAGR
|
Accelerate at a CAGR of
9.29%
|
|
Market growth
2024-2028
|
USD 52.9
billion
|
|
Market
structure
|
Fragmented
|
|
YoY growth 2022-2023
(%)
|
8.25
|
|
Regional
analysis
|
North America, Europe,
Asia, and Rest of World (ROW)
|
|
Performing market
contribution
|
North America at
33%
|
|
Key
countries
|
US, Germany, China,
Canada, and UK
|
|
Key companies
profiled
|
Abbott Laboratories,
Becton Dickinson and Co., Bio Rad Laboratories Inc., bioMerieux SA,
Biosino Bio Technology and Science Inc, CPC Diagnostics Pvt Ltd.,
DAAN Gene Co. Ltd., Danaher Corp., F. Hoffmann La Roche Ltd.,
Guangzhou Wondfo Biotech Co. Ltd., Hologic Inc., Myriad Genetics
Inc., QIAGEN NV, QuidelOrtho Corp., Shanghai Kehua Bio Engineering
Co. Ltd, Shenzhen Mindray BioMedical Electronics Co. Ltd, Siemens
AG, Sysmex Corp., and Thermo Fisher Scientific Inc.
|
Market Driver
In-Vitro Diagnostics (IVD) instruments market growth is driven
by the increasing adoption of Point of Care (POL) testing in
primary and outpatient care. POL testing, particularly in
infectious disease markers, cardiovascular diseases, and diabetes
care, is gaining popularity due to its convenience and quick
results. Portable IVD instruments are advancing technically,
enabling clinicians to perform tests in-house. The CLIA testing for
molecular assays in POL practices is expected to fuel market
growth. However, certain regulatory aspects regarding CLIA-waived
testing may impact market expansion. Despite these challenges, the
global IVD instruments market is projected to experience average
growth due to the increasing trend towards POL testing.
The In Vitro Diagnostics Instruments Market is experiencing
significant growth due to the rising demand for accurate and rapid
medical tests. With a focus on infectious diseases such as Malaria
and Chikungunya, the market is seeing increased adoption of
advanced technologies like Next-Generation Sequencing (NGS) for
pathogen detection. Laboratory technicians play a crucial role in
this sector, handling various samples like blood, serum, plasma,
urine, saliva, and tissues. Blood banks, home care settings,
pharmaceutical companies, biotechnology firms, academic institutes,
and non-communicable disease segments are key end-users. Infectious
diseases and chronic conditions including cardiology,
gastroenterology, and cancer are major application areas. The aging
population and the rise of molecular diagnostics for diseases like
Alzheimer's and various cancers further fuel market expansion. In
vitro diagnostics instruments offer efficiency, accuracy, and
convenience in the laboratory environment, making them an essential
tool for healthcare providers.
Discover 360° analysis of this market. For
complete information, schedule your consultation - Book
Here!
Market Challenges
- In the In-Vitro Diagnostics Instruments Market, reimbursement
policies for diagnostic testing vary across geographies and are
trending downward. This puts pressure on hospitals and diagnostic
laboratories to reduce expenditure on IVD instruments to
maintain revenue. Workforce shortages and lost health insurance
cards lead to fewer patient visits and limited testing volumes.
Lean laboratories are emerging in some countries, prioritizing
essential capital equipment purchases. Reduced reimbursements for
certain instruments, such as SMBG in Europe and IVD instruments in the US, impact
sales growth. IVD companies respond by focusing on emerging markets
and adapting to market conditions. These factors are expected to
restrict the growth of the global IVD Instruments Market during the
forecast period.
- The In-Vitro Diagnostics (IVD) Instruments Market is
experiencing significant growth due to advances in Next Generation
Sequencing, Liquid Biopsy, and Personalized Medicine. Genetic and
molecular diagnostic technologies are driving innovation in
Clinical Profiles, Companion Diagnostics, and various disease areas
such as Hospitals and Healthcare Facilities. IVD equipment in
Immunodiagnostics, Hematology, Tissue Diagnostics, Clinical
Chemistry, and Cardiac Diseases are in high demand. Challenges
include Norovirus, HIV, Lyme disease, and other infectious
diseases. Reagents and kits, Software and Services, and
Manufacturing Technology are essential components of this market.
Diabetes, Oncology, Sexually Transmitted Diseases, and Geriatric
Population are key applications. Distribution channels include
hypermarkets, pharmacies, drug stores, online providers, standalone
laboratories, and Clinical Laboratories. Tropical diseases and
Endocrine Disorders are also targeted markets. High blood
cholesterol, Heart Disease, Stroke, and Chronic Conditions are
significant disease areas.
For more insights on driver and
challenges - Request a sample report!
Segment Overview
This in-vitro diagnostics instruments market report extensively
covers market segmentation by
- Product
- 1.1 Immunochemistry
- 1.2 Clinical microbiology
- 1.3 Molecular diagnostics
- 1.4 Others
- End-user
- 2.1 Hospitals and diagnostic laboratories
- 2.2 Academic and research institutes
- 2.3 Home care
- Geography
- 3.1 North America
- 3.2 Europe
- 3.3 Asia
- 3.4 Rest of World (ROW)
1.1 Immunochemistry- The In-Vitro Diagnostics (IVD)
Instruments Market's highly automated segment of clinical
laboratories holds the largest test volume compared to others.
However, the demand for immunochemistry analyzers is declining in
the US and Europe due to
consolidation in France, late
payments from Italy, Spain, and Portugal, and reductions in expenditure in the
UK, France, and Germany. In contrast, the rise of rural
hospitals in China and
India lacking basic diagnostics
infrastructure presents a growth opportunity. Over 80 immunoassay
analyzer models are available, with manufacturers responding to
competition through frequent new launches or analyzer upgrades.
Reduced European reimbursement is attributed to delayed spending by
some medical laboratories, while the Affordable Care Act (ACA)
provides greater certainty for investments. IVDs, like Ortho
Clinical Diagnostics' VITROS immunodiagnostic products for HIV-1+2
detection, aid in early COVID-19 virus detection. Roche Diagnostics
received FDA approval for the first HIV-1/HIV-2 qualitative test on
cobas 6800/8800 systems. Innovative products and advanced
technologies continue to enter the market, intensifying competition
during the forecast period.
For more information on market segmentation with geographical
analysis including forecast (2024-2028) and historic data
(2017-2021) - Download a Sample Report
Research Analysis
In vitro diagnostics refer to medical tests conducted outside of
a living organism, using samples of bodily fluids, tissues, or
other specimens. These tests play a crucial role in the detection
and diagnosis of various diseases, including infectious diseases
such as Zika, Malaria, and Chikungunya, as well as chronic
conditions like cardiovascular diseases, cancer, and endocrine
disorders. The laboratory environment is essential for the accurate
and reliable performance of these tests. In vitro diagnostics are
used extensively in clinical laboratories to diagnose
non-communicable diseases such as heart disease, stroke, high blood
cholesterol, and Alzheimer's disease. Molecular diagnostics and
pathogen detection are advanced areas of in vitro diagnostics,
offering rapid and accurate results for the diagnosis of various
conditions. Laboratory technicians play a vital role in the
collection, preparation, and analysis of samples for in vitro
diagnostic tests. In vitro diagnostics are used in various medical
specialties, including cardiology, gastroenterology, and neurology,
to aid in the early detection and treatment of diseases.
Market Research Overview
In Vitro Diagnostics Instruments Market: Overview The In Vitro
Diagnostics Instruments Market refers to the industry that produces
and supplies diagnostic tools and equipment used in medical
laboratories for analyzing samples derived from bodily fluids,
tissues, and other specimens. These diagnostic instruments play a
crucial role in detecting various diseases, including infectious
diseases such as Norovirus, HIV, Lyme disease, and Shigellosis,
chronic conditions like diabetes, cardiovascular diseases, cancer,
and Alzheimer's disease, and genetic disorders. The market caters
to various end-users, including hospitals, healthcare facilities,
pharmacies, drug stores, online providers, standalone laboratories,
and clinical laboratories. The In Vitro Diagnostics Instruments
Market encompasses a wide range of diagnostic technologies,
including Immunodiagnostics, Hematology, Tissue Diagnostics,
Clinical Chemistry, Cardiac Diseases, Immune System Disorders,
Nephrological Diseases, Gastrointestinal Diseases, and others. The
market is segmented into various product categories, including IVD
equipment, Diagnostic Technologies, Reagents and Kits, Software and
Services, and Molecular Diagnostics. Molecular Diagnostics further
includes Next-Generation Sequencing (NGS), Liquid Biopsy, and
Personalized Medicine. The market is also influenced by various
factors such as the aging population, the rise in non-communicable
diseases, and the increasing prevalence of chronic conditions. The
In Vitro Diagnostics Instruments Market is driven by the growing
demand for early and accurate diagnosis, the increasing focus on
personalized medicine, and the increasing adoption of advanced
diagnostic technologies such as NGS, liquid biopsy, and companion
diagnostics. The market is also expected to witness significant
growth due to the increasing prevalence of diseases such as
diabetes, cancer, and cardiovascular diseases, and the growing
awareness and acceptance of genetic testing. The market is highly
competitive, with various players offering a wide range of
diagnostic instruments and solutions. The market is also witnessing
significant innovation, with new technologies and products being
developed to address the evolving needs of healthcare providers and
patients. Some of the key trends in the market include the
increasing use of automation, the integration of artificial
intelligence and machine learning, and the growing focus on
point-of-care testing. The In Vitro Diagnostics Instruments Market
is expected to grow at a significant CAGR during the forecast
period, driven by the increasing demand for accurate and timely
diagnosis, the growing prevalence of chronic diseases, and the
increasing adoption of advanced diagnostic technologies. The market
is also expected to be influenced by various factors such as
regulatory frameworks, reimbursement policies, and technological
advancements. In conclusion, the In Vitro Diagnostics Instruments
Market plays a vital role in the healthcare industry by providing
accurate and timely diagnosis, enabling personalized medicine, and
contributing to better patient outcomes. The market is highly
dynamic and is expected to witness significant growth in the coming
years, driven by various factors such as the increasing prevalence
of chronic diseases, the growing focus on personalized medicine,
and the increasing adoption of advanced diagnostic
technologies.
Table of Contents:
1 Executive Summary
2 Market Landscape
3 Market Sizing
4 Historic Market Size
5 Five Forces Analysis
6 Market Segmentation
- Product
-
- Immunochemistry
- Clinical Microbiology
- Molecular Diagnostics
- Others
- End-user
-
- Hospitals And Diagnostic Laboratories
- Academic And Research Institutes
- Home Care
- Geography
-
- North America
- Europe
- Asia
- Rest Of World (ROW)
7 Customer Landscape
8 Geographic Landscape
9 Drivers, Challenges, and Trends
10 Company Landscape
11 Company Analysis
12 Appendix
About Technavio
Technavio is a leading global technology research and advisory
company. Their research and analysis focuses on emerging market
trends and provides actionable insights to help businesses identify
market opportunities and develop effective strategies to optimize
their market positions.
With over 500 specialized analysts, Technavio's report library
consists of more than 17,000 reports and counting, covering 800
technologies, spanning across 50 countries. Their client base
consists of enterprises of all sizes, including more than 100
Fortune 500 companies. This growing client base relies on
Technavio's comprehensive coverage, extensive research, and
actionable market insights to identify opportunities in existing
and potential markets and assess their competitive positions within
changing market scenarios.
Contacts
Technavio Research
Jesse Maida
Media & Marketing Executive
US: +1 844 364 1100
UK: +44 203 893 3200
Email: media@technavio.com
Website: www.technavio.com/
 View original content to download
multimedia:https://www.prnewswire.com/news-releases/in-vitro-diagnostics-instruments-market-size-is-set-to-grow-by-usd-52-9-billion-from-2024-2028--growing-geriatric-population-boost-the-market-technavio-302210037.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/in-vitro-diagnostics-instruments-market-size-is-set-to-grow-by-usd-52-9-billion-from-2024-2028--growing-geriatric-population-boost-the-market-technavio-302210037.html
SOURCE Technavio