UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 6-K
REPORT OF FOREIGN ISSUER
PURSUANT TO RULE 13a-16 OR 15d-16
OF THE SECURITIES EXCHANGE ACT OF 1934
Date of Report: November [ ], 2023
(Commission File No. 001-39308)
CALLIDITAS THERAPEUTICS AB
(Translation of registrant’s name into
English)
Kungsbron 1, D5
SE-111 22
Stockholm, Sweden
(Address of registrant’s principal executive office)
Indicate by check mark whether the registrant files or will file annual
reports under cover of Form 20-F or Form 40-F.
Form 20-F
x Form 40-F ¨
Company Announcement and Interim Report
On November 7, 2023, the Company announced its unaudited results
for the nine months ended September 30, 2023, which are further described in the Company’s Interim Report Q3 2023 and press
release, copies of which are attached hereto as Exhibits 99.1 and 99.2, respectively, and are incorporated by reference herein.
The information contained in this Form 6-K, including Exhibits
99.1 and 99.2 is hereby incorporated by reference into the registrant’s Registration Statements on Form F-3 (File No. 333-265881)
and Form S-8 (File Nos. 333-240126 and 333-272594).
EXHIBIT INDEX
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934,
the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| |
CALLIDITAS THERAPEUTICS AB |
| |
|
|
| Date: November 7, 2023 |
By: |
/s/ Fredrik Johansson |
| |
|
Fredrik Johansson
Chief Financial Officer |
Exhibit 99.1

| INTERIM REPORT
JANUARY – SEPTEMBER
2023
Q3 |

| 2 Calliditas Therapeutics | Interim Report Q3: January - September 2023
284 130% 787
Key Takeaways from Q3
JULY – SEPTEMBER 2023
(COMPARED TO JULY – SEPTEMBER 2022)
JANUARY – SEPTEMBER 2023
(COMPARED TO JANUARY – SEPTEMBER 2022)
JUL – SEP 2023
“We are looking forward to a potential
full approval of TARPEYO for the
treatment of IgA Nephropathy on our
PDUFA date of December 20, 2023.”
JUL – SEP 2023 JUL – SEP 2023
MSEK
TARPEYO net sales Renée Aguiar-Lucander / CEO
TARPEYO net sales
growth (vs Q3 2022)
MSEK
Cash position on
30 September 2023
Interim Report
January – September 2023
• Net sales amounted to SEK 294.6 million, of which
TARPEYO® net sales amounted to SEK 283.6 million,
for the three months ended September 30, 2023. For
the three months ended September 30, 2022 net sales
amounted to SEK 260.1 million, of which TARPEYO net
sales amounted to SEK 123.4 million.
• Operating loss amounted to SEK 159.6 million and SEK
36.2 million for the three months ended September 30,
2023 and 2022, respectively.
• Loss per share before and after dilution amounted to SEK
3.14 and SEK 0.17 for the three months ended September
30, 2023 and 2022, respectively.
• Cash amounted to SEK 786.9 million and SEK 736.2
million as of September 30, 2023 and 2022, respectively.
• In August, the FDA granted priority review for
the application for full approval of TARPEYO
(budesonide) delayed release capsules for the
treatment of IgA nephropathy.
• In August, full data from the Phase 3 NefIgArd
study with Nefecon® (TARPEYO®/Kinpeygo®)
was published in The Lancet.
• In September STADA and Calliditas announced
the filing of the application for full marketing
authorisation of Kinpeygo in the EU.
• PDUFA date set as December 20, 2023: potential
full approval of TARPEYO in the complete study
population for IgAN patients.
• Regulatory decision in China regarding market
authorization for Nefecon.
• Regulatory decision in EU regarding full
marketing authorisation for Kinpeygo.
• Full data from Phase 2 proof-of-concept study
of setanixib in head and neck cancer, expected in
H1 2024.
• Net sales amounted to SEK 755.3 million, of which
TARPEYO net sales amounted to SEK 728.5 million, for
the nine months ended September 30, 2023. For the nine
months ended September 30, 2022 net sales amounted to
SEK 373.8 million, of which TARPEYO net sales amounted
to SEK 205.0 million.
• Operating loss amounted to SEK 414.8 million and SEK
454.4 million for the nine months ended September 30,
2023 and 2022, respectively.
• Loss per share before and after dilution amounted to SEK
8.34 and SEK 7.72 for the nine months ended September
30, 2023 and 2022, respectively.
Expected Key Events
upcoming 6 months |

| 3 Calliditas Therapeutics | Interim Report Q3: January - September 2023
Calliditas
– pioneering new
treatments for rare
diseases
Our Values
Calliditas Therapeutics leverages scientific expertise and disease-specific insights to improve the lives of patients. We are a com-mercial-stage biopharma company that researches, develops and
commercializes novel therapies that address significant unmet
needs in rare diseases. We are committed to expanding treatment
options and establishing new standards of care for patients with
rare diseases, reflected by our pipeline of innovative medicines
that target unmet medical needs.
Our lead product provides provides a treatment option that we
believe is disease-modifying for IgA nephropathy (IgAN) – also
known as Berger’s Disease – a progressive autoimmune disease
of the kidney that for many patients leads to end-stage renal
disease (ESRD), requiring dialysis or organ transplantation. This
drug product, developed under the name Nefecon®, was granted
accelerated approval by the FDA in 2021, and is today marketed
in the US under the brand name TARPEYO®. It has also been
granted conditional marketing authorisation by the European
Commission under the brand name Kinpeygo® in the European
Economic Area (EEA) and in the UK. TARPEYO and Kinpeygo are
currently being reviewed by the FDA and EMA for full approval,
with the US PDUFA date set for December 20, 2023.
Nefecon has also been approved in Macau and is being reviewed
by regulators in China, Singapore and South Korea, and Calliditas
has recently entered into a partnership to develop and commer-cialize Nefecon in Japan.
AGILITY
We are flexible and able to rapidly pivot and adapt to
changing situations and requirements.
EXPERTISE
We leverage our strong internal experience and com-petencies while complementing our strengths through
knowledge sharing and external collaborations as
needed.
INTEGRITY
We take responsibility for our actions and hold ourselves
to the highest ethical standards, guided by our moral
principles to make the right decisions.
PIONEER
We explore novel approaches and empower each other
to find new ways of operating in a compliant, innovative
and pragmatic manner.
IgA nephropathy is the largest of the glomerular nephritis
diseases, so the market potential for Nefecon is substantial,
as evidenced by out-licensing deals with potential payments
exceeding USD 300 million, encompassing upfront payments and
predefined milestones, as well as ongoing royalty obligations.
Our late-stage pipeline is based on a first-in-class platform
of NOX inhibitors. Our lead compound, setanaxib, inhibits
enzymes involved in inflammation and fibrosis pathways
and is the first drug of this class to reach the clinical stage.
Setanaxib is currently undergoing clinical trials targeting rare
diseases characterized by inflammation and fibrosis, including
IPF and PBC, and Calliditas is also planning to launch a trial with
setanaxib in Alport syndrome. Additionally, based on promising
preclinical findings, we are conducting a proof-of-concept trial in
head and neck cancer to further support the mode of action of
this drug class.
While our headquarters are in Stockholm, Sweden, we maintain a
significant presence in the United States, with offices in New York
and New Jersey. We also have offices in France and Switzerland,
where our discovery team is based. Calliditas Therapeutics ordi-nary shares were listed on NASDAQ Stockholm in 2018 (CALTX)
and subsequently American Depositary shares representing our
ordinary shares were listed on the NASDAQ Global Select Market
in the United States in 2020 (CALT). |

| 4 Calliditas Therapeutics | Interim Report Q3: January - September 2023
Key Figures
Investment highlights 2023
Jul–Sep Jan–Sep Jan–Dec
(SEK in thousands, except per share amount or as otherwise indicated) 2023 2022 2023 2022 2022
Net sales 294,592 260,056 755,327 373,837 802,879
Of which TARPEYO product sales 283,591 123,400 728,522 204,989 372,247
Operating profit/(loss) (159,573) (36,227) (414,819) (454,438) (421,943)
Profit/(loss) before income tax for the period (164,082) (15,958) (442,760) (419,483) (409,417)
Earnings/(loss) per share before and after dilution (SEK) (3.14) (0.17) (8.34) (7.72) (7.78)
Cash flow used in operating activities (62,529) (124,725) (457,500) (541,383) (311,354)
As of As of
(SEK in thousands, except per share amount or as otherwise indicated) 30.09.2023 30.09.2022 31.12.2022
Total registered shares, including shares held by Calliditas, at the end of the period 59,580,087 59,157,587 59,580,087
Equity attributable to equity holders of the Parent Company at the end of the period 352,435 725,936 766,264
Equity ratio at the end of the period in % 22% 48% 39%
Cash at the end of the period 786,883 736,161 1,249,094
RECENT AND ANTICIPATED VALUE DRIVERS
NEFECON
Setanaxib
Positive readout of
topline NefIgArd data
Readout of biomarker data
from Ph2 POC trial with
setanaxib in head and neck
cancer
Launch of trial in
Alport syndrome
Final data readout of
Ph2 trial in head and
neck cancer
Data readout
of Ph2 IPF trial
Data readout
of Ph2b PBC trial
Filing for full approval
in IgAN in the US and
Europe
Potential approval
in IgAN in China
PDUFA Date
20 December:
Potential full
approval in
IgAN in US
Commercial ramp
of TARPEYO in
the US
Commercial ramp of
Nefecon in China by
Everest Medicines
H1 2023 H2 2023 H1 2024 H2 2024
Commercial ramp of
Kinpeygo in the EU
by STADA
Potential full
approval in IgAN
in EU |

| 5 Calliditas Therapeutics | Interim Report Q3: January - September 2023
CEO STATEMENT
In August we were excited to see the full data set from our global,
randomized, placebo controlled, double-blind Phase 3 clinical
trial, NefIgArd, published in The Lancet. As previously reported,
the trial successfully met its primary endpoint, providing evidence
that 9 months of treatment with Nefecon 16 mg/day led to a
significant reduction in the decline of eGFR, a measure of kidney
function, over 2 years in adult patients with primary IgA nephrop-athy (IgAN). In addition, a durable reduction in proteinuria was
observed for the entire period and the positive effect of Nefecon
on eGFR was independent of baseline proteinuria and resulted in
an approximate 50% reduction in the decline of eGFR compared
with placebo over the 2-year period. We believe these results
support the thesis that the drug has the potential to provide a
clinically meaningful delay in the need for dialysis or transplanta-tion. We are looking forward to the potential of a full approval of
TARPEYO on our PDUFA date of December 20, 2023.
In September we had the opportunity to attend the IIgANN bi-annual conference, which this year was held in Tokyo, Japan.
At the summit we presented 5 posters, including confirmatory
biomarker analysis showing significant reduction of Gd-IgA1
and circulating immune complexes (IgA-IC), which supports the
disease modifying mechanism of the drug. We also had 2 oral
presentations, including one looking at racial differences and one
showing that treatment with Nefecon increased the number of
patients without microhematuria from 34% to 60%, versus 32%
to 39% in placebo, which was statistically significant (p=0.0001).
As expected, the third quarter was impacted by summer season-ality, and also saw some turnover in the sales team, resulting in
a slight decrease in new enrolments notwithstanding continued
growth in new prescribers. Based on early Q4 trends however,
we are very encouraged with regards to significant enrolment
growth. Total Q3 revenues were SEK 295 million, out of which
net revenues from TARPEYO amounted to SEK 284m (USD
26.3m). The operating loss for the quarter was SEK 160m
which included SEK 52m of R&D costs of one-time nature
mainly stemming from changes to the TRANSFORM trial and
discontinuation of a research project. Net decrease in cash in
the quarter was SEK 73m and our cash position remains strong
with SEK 787m on the balance sheet, which we believe is suffi-cient to take us to cash flow break even, based on revenue pro-jections for TARPEYO. We have also started to see improved
market access statistics with new patients starts taking on
average 15-20 days to fill, reflecting our efforts to continuously
improve patient access.
Following positive feedback regarding the complete NefIgArd trial
results from multiple US advisory board meetings and dozens of
face-to-face interactions with nephrologists at recent conferences,
Lancet publication of
full Phase 3 data set
we have decided to launch a targeted US focused investment
program in order to appropriately support and strengthen our
category leadership position. This will involve adding resources
across relevant commercial functions in order to fully capitalize
on the potential for a full approval in late 2023. We believe that
this will maximize our ability to drive significant revenue growth
in 2024, with limited cost impact in 2023. Over the next several
months we plan to selectively complement our existing teams with
the goal to continue to enhance our medical education, patient
support and market access activities to facilitate patient access
and continue to build a leading franchise based on the NefIgArd
trial data set. This increase is targeted and incremental and is not
expected to materially increase the commercial cost base in 2024.
I also had the opportunity during the quarter to visit our partner,
Everest Medicines, in China and learn more about their pre
commercial activities and preparations in expectation of a
potential approval later in the year, as well as the highly success-ful Early Access Patient (EAP) program which was launched in
April. Several hundreds of patients signed up for the program,
making it one of the most successful EAP programs launched
in China. We are hopeful for an approval for Nefecon later this
year in China, where the high prevalence of glomerulonephritis
leads to many young people ending up in dialysis and where the
need for an effective treatment for patients is therefore critical.
Post quarter activities included the American Society of Nephrol-ogy Kidney Week meeting in Philadelphia, which provided us with
numerous opportunities to meet and engage with nephrologists
across the country. The overall sentiment as well as the many
encounters with nephrologists were very positive and further
cemented our belief that the strong long-term data from our
Phase 3 trial in combination with published registry data such as
the RaDaR publication have started an important conversation in
the nephrology community concerning the need for earlier inter-vention and treatment of IgAN. More on this in my Q4 report.
We hold our guidance for 2023 and look forward to the potential
of a full approval of TARPEYO in Q4 and an exciting 2024.
*Linear spline mixed-effect model for slope-based endpoints in clinical trials of chronic kidney disease. Renée Aguiar-Lucander, CEO |

| 6 Calliditas Therapeutics | Interim Report Q3: January - September 2023
BUSINESS OVERVIEW
Our Pipeline
Exciting Journey Ahead
Calliditas’ lead product, developed under the name Nefecon, has been approved in the US and
Europe and is awaiting approval in China. Our pipeline consists of development programs based on a
first-in-class NOX inhibitor platform. The lead compound, setanaxib, is the first NOX inhibitor to reach
the clinical stage and is a selective NOX 1 and NOX 4 inhibitor. Calliditas is presently running trials
with setanaxib in Primary Biliary Cholangitis (PBC) and in Squamous Cell Carcinoma of the Head &
Neck (SCCHN). There is also an ongoing investigator-led trial in Idiopathic Pulmonary Fibrosis (IPF)
and we are planning to initiate a Phase 2 trial in Alport syndrome.
December 20 2023 PDUFA
date; FDA decision on full
approval of TAPREYO in
primary IgA nephropathy for
patients at risk of progression
Regulatory decision in China
expected in 2023
Potential full approval of
Kinpeygo in EU in 1H 2024
Q4 23: Targeting initiation of
Alport syndrome clinical trial
1H 24: Full data from phase
2 study in head and neck
cancer
NEFECON* Setanaxib
Preclinical Phase 1 Phase 2 Phase 3 Marketed Rights Commercial region Upcoming milestones
* Approved under accelerated approval in the US under the tradename TARPEYO® to reduce proteinuria in adults with primary IgAN at risk of rapid disease progression, gener-ally a urine protein-to-creatinine ratio (UPCR) ≥ 1.5 g/g, and granted conditional marketing authorization in the EEA and UK under the tradename Kinpeygo® for the treatment
of primary IgAN in adults at risk of rapid disease progression with a urine protein-to-creatinine ratio (UPCR) ≥1.5 g/gram.
Commercial Ongoing Trial Planned Trial Investigator Led Trial
IgAN Japan
IgAN United States
ROW ex partnered
PDUFA Date for potential full
approval: December 20 2023
IgAN
China, Hong Kong, Macau,
Taiwan, Singapore,
South Korea
NDA accepted and FTD
received in China. Decision
expected in Q4 2023
Awaiting EMA review of
application for full marketing
authorisation. IgAN KINPEYGO 4 mg Modified-release hard capsules
budesonide
Europe, UK, Switzerland
Global
SCCHN Global
IPF Global
Alport Global
PBC |

| 7 Calliditas Therapeutics | Interim Report Q3: January - September 2023
BUSINESS OVERVIEW
Our Commercial Product
Calliditas’ lead product, which was
granted accelerated approval by the US
Food and Drug Administration (FDA)
in December 2021 and conditional
marketing authorisation by the European
Commission (EC) in July 2022, was the first
ever approved medication and is the only
treatment specifically designed to target the
origin of the autoimmune kidney disease
IgA nephropathy (IgAN) and to be disease-modifying.
IgAN is a serious progressive disease, in which up to 50% of
patients end up at risk of developing end-stage renal disease
(ESRD) within ten to twenty years. This product, which was
developed under the name Nefecon®, is approved under the
brand name TARPEYO® in the United States and under the
brand name Kinpeygo® in Europe.
Disease Background
Although IgAN manifests in the kidney, the evidence indicates
that it is a disease that starts in the distal part of the intestine,
specifically in the ileum. Peyer’s patches, which are concen-trated within the gut-associated lymphoid tissue in the ileum,
have been identified as a major source of mucosal-type IgA
antibodies. Patients with IgA nephropathy have elevated levels
of mucosal-type IgA, which – in contrast to the majority of the
IgA in the blood - are predominately dimeric or polymeric and
are galactose-deficient. In IgAN patients, a combination of a
genetic predisposition and environmental, bacterial and dietary
factors is presumed to lead to an increased production of these
galactose-deficient IgA antibodies. This increased production,
potentially in conjunction with increased intestinal permeability,
leads to these secretory antibodies appearing in the blood.
Strong data from the full Phase 3 trial readout
NefIgArd is the first Phase 3 trial specifically designed for IgA
nephropathy to show a statistically significant and clinically rele-vant kidney protective effect as measured by eGFR. Calliditas’
supplemental New Drug Application to the FDA was based on
the data from this trial, with Calliditas filing for full approval on
the basis of the strong eGFR data readout.
The full Phase 3 NefIgArd trial consisted of a total of 364
patients, including 200 patients from the interim analysis,
based upon which Calliditas successfully filed for accelerated
and conditional approval with the FDA and EMA, respectively.
The full trial included 9 months of treatment and a 15-month
post-treatment observational period for all study participants
to confirm long-term renal protection. The endpoint of the full
Phase 3 trial assessed the difference in kidney function between
treated and placebo patients, as measured by eGFR, over a
two-year period from the start of dosing of each patient. The
data read-out took place in March 2023, and in August 2023
was published in The Lancet.
The primary endpoint of the Phase 3 trial was a time-weighted
average of eGFR observed at each time point over two years.
The primary endpoint was successfully met with a highly statis-tical p value of <0.0001. Over the two-year period of treatment
and observation, the mean decline in eGFR was 2.47 mL/
min/1.73 m2 for patients who received Nefecon compared with
7.52 mL/min/1.73 m2 for patients who received placebo. On
average, over the two-year period, there was therefore a 5.05
mL/min/1.73 m2 eGFR treatment benefit in favor of Nefecon
compared to placebo (p<0.0001).
There was a cumulative improvement in proteinuria in patients
treated with Nefecon versus placebo during the 9-month
treatment period, which continued to significantly improve at
12 months, declining by over 50%. At month 24, proteinuria
levels in patients who had received Nefecon were still at a
reduced level, similar to that observed at the 9-month time
point, reflecting the durability of the proteinuria reduction of a
9-month course of treatment.
Filing for full approval
On the basis of this positive data, Calliditas submitted a sNDA
to the FDA seeking full approval of TARPEYO for the entire
study population from the Phase 3 NeflgArd study. TARPEYO
is currently approved in the US under accelerated approval to
reduce proteinuria in adults with primary IgAN at risk of rapid
disease progression, generally a urine protein-to-creatinine
ratio (UPCR) ≥1.5g/g. The FDA accepted this filing and granted
priority review, setting a PDUFA date of December 20 2023.
In September 2023, Calliditas’ partner STADA filed with the
EMA for full marketing authorisation of Kinpeygo in the EU,
and in October also filed with the UK MHRA.
1Watson S, Padala SA, Hashmi MF, et al. Alport Syndrome. [Updated 2023 Feb 19]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-Available from: https://www.ncbi.nlm.nih.gov/books/NBK470419/ |

| 8 Calliditas Therapeutics | Interim Report Q3: January - September 2023
IgA nephropathy
- a significant market opportunity
Our commercial partnerships
• IgAN is a rare disease, but it is the most common
primary glomerulonephritis. Prevalence is estimated
to range from 130,000 to 150,000 patients in the US,
to be around 200,000 patients in Europe and up to 5
million patients in China.
• In the United States, we estimate there are around
12,000 nephrologists, of which up to two thirds treat
patients with IgAN. The majority of patients are seen
by approximately 4,000 to 5,000 specialists. About
40% of the patients are treated in academic settings
while the remaining are treated in community settings.1
BUSINESS OVERVIEW
EU
Nefecon® was granted conditional
marketing authorisation (CMA) by the
European Commission in July 2022,
and subsequently by the Medicines and
Healthcare products Regulatory Agency
(MHRA) of the United Kingdom in February
2023, under the brand name Kinpeygo®
for the treatment of IgAN in adults at risk
of rapid disease progression with a urine
protein-to-creatinine ratio (UPCR) ≥1.5 g/
gram, becoming the first and only approved
treatment for IgAN in EU.
Kinpeygo will be marketed in the European
Economic Area (EEA), the UK and
Switzerland, if approved in this jurisdic-tion, exclusively by STADA Arzneimittel
AG, with whom Calliditas entered into a
license agreement in July 2021 to register
and commercialize Kinpeygo in Europe.
STADA launched Kinpeygo in Germany in
September 2022, with additional Euro-pean countries to follow. In Germany it
is estimated that 3.1 people per 100,000
develop IgAN each year.
Following the positive data readout from
the full NefIgArd trial and the submission of
an sNDA to the FDA, Calliditas is collabo-rating with STADA to seek full approval of
Kinpeygo by the European Commission and
the MHRA in the full study population.
Greater China
Calliditas entered into a license agreement
to develop and commercialize Nefecon for
IgAN in Greater China and Singapore with
Everest Medicines (HKEX 1952.HK) in
2019. In March 2022, this agreement was
expanded to include South Korea.
Everest’s New Drug Application (NDA)
for Nefecon was accepted by the Chinese
regulatory authority National Medical Prod-ucts Administration (NMPA) in November
2022, and in December the Center for
Drug Evaluation (CDE) of the NMPA
recommended Priority Review. A regulatory
decision is expected in Q4 2023.
Everest launched Nefecon in China’s
Hainan Boao Pilot Zone as a First-in-Dis-ease therapy for IgA nephropathy in April
2023. This program allows innovative
• The IgAN patient population at risk of disease progression
as defined by KDIGO guidelines is estimated to amount
to between 45,000 and 60,000 patients in the US.2
• Today the majority of these patients are treated princi-pally with supportive care such as generic ACEs and/or
ARBs to control blood pressure, complemented with other
broadly indicated cardio and kidney protective drugs.
• As availability and familiarity of approved drugs
specifically indicated and approved for IgAN increase
and physicians consider more active intervention to
preserve kidney function, we estimate the global IgAN
market to amount to USD 5 – 8 billion.
overseas drugs and medical devices that
have been approved in other territories
to be sold and used in real-world clin-ical settings in Hainan Province before
regulatory approval by the NMPA. Several
hundreds of patients signed up for this
early access program, making it one of the
most successful EAP programs launched in
China.
In October 2023, Everest received approval
for Nefecon in IgAN patients at risk of
disease progression from the Pharmaceu-tical Administration Bureau of the Macau
Special Administrative Region, making it
the first region in Everest territories that
received Nefecon approval.
Japan
At the end of 2022, Calliditas entered into
a partnership to commercialize Nefecon in
Japan with Viatris Pharmaceuticals Japan,
a subsidiary of Viatris Inc. (Nasdaq: VTRS).
Viatris is a global healthcare company which
is headquartered in the United States and
has a presence in over 165 countries and
territories, and also operates approximately
40 manufacturing facilities.
1Veeva OpenData for 2023, including all active HCPs where the primary specialty is Nephrology
2Spherix RealWorld Dynamix |

| 9 Calliditas Therapeutics | Interim Report Q3: January - September 2023
Nefecon Has the Potential to
Establish a New Standard of Care in IgAN
BUSINESS OVERVIEW
Targeted immuno-modulator designed
to locally target origin
of disease
In combination with
optimized RASi ther-apy; option of inter-mittent, rather than
chronic treatment
Durable eGFR benefit
and sustained pro-teinuria disease-mod-ifying effects in IgAN
Well characterized
active ingredient and
safety profile
Mechanism of Action Patient focus Efficacy Safety
Nefecon® was the first-ever medication approved by the FDA and European Commission for
IgAN, and the only treatment specifically designed to target the origin of IgA nephropathy
(IgAN) to be disease-modifying.
• A genetic predisposition is required
but not sufficient; most patients
are diagnosed in their 20s and 30s
• More than 50% are at risk of
developing ESRD within 10-20
years, leading to kidney transplant
• The treatment goal is to preserve
eGFR – kidney function
• Proteinuria levels > 1g/24h
indicate a risk of disease progression
and a worse outlook
IgAN Patients: |

| 10 Calliditas Therapeutics | Interim Report Q3: January - September 2023
Continued TARPEYO Sales Growth
BUSINESS OVERVIEW
During the third quarter of 2023, Calliditas’ commercial team continued to build on the
achievements from the previous quarter, further reinforcing TARPEYO’s position as a
transformative treatment option for IgA nephropathy (IgAN). Notably, this quarter the
FDA granted priority review of our supplemental New Drug Application for full approval of
TARPEYO, with a PDUFA date set for December 20, 2023. The team is preparing for a new
indication launch in anticipation of FDA approval for the use of TARPEYO in the full IgAN patient
population studied in the NefIgArd trial.
Exciting Journey Ahead
7 abstracts accepted for
presentations at ASN.
Opportunity for P2P edu-cation and scientific data
exchange at ASN, November
1-4 2023, in Philadelphia.
December 20 2023
PDUFA Date
Potential for full
approval.
New indication promotional
launch - new label for full
NefIgArd Phase 3 trial pop-ulation, based on evidence
of slowing kidney function
decline*.
New Patients enrolled in Q3
2023
YTD patients enrolled: 1,198
New Prescribers in Q3
2023
LTD Prescribers: 1,338
*Does not include patients that are still waiting final insurance decision
*Subject to review and approval by FDA
Of patients enrolled in
TARPEYO Touchpoints got
TARPEYO* YTD
Net sales of TARPEYO in Q3
FDA grants priority
review for full approval
of TARPEYO in the
NefIgArd Pahse 3 trial
population – PDUFA
December 20, 2023.
Full Results from
the NefIgArd Phase 3
trial published in The
Lancet.
Engaging with the
IgAN community at
the SPARK 2023
symposium hosted by
the IgA Nephropathy
Foundation
>200 patients attended.
7 presentations at the
IIGaNN conference,
reporting results of
NeflgArd Phase 3 trial
and biomarker data.
QUARTERLY HIGHLIGHTS
KEY METRICS Q3
367 197 86% $26.3M |

| 11 Calliditas Therapeutics | Interim Report Q3: January - September 2023
BUSINESS OVERVIEW
Calliditas’ pipeline consists of development
programs based on a first-in-class NOX inhib-itor platform. Calliditas is presently running
clinical trials with lead compound setanaxib
in Squamous Cell Carcinoma of the Head &
Neck (SCCHN), which read out interim data in
July, as well as in Primary Biliary Cholangitis
(PBC). We also plan to launch a clinical trial in
Alport syndrome in Q4 2023.
NOX Enzyme Inhibitors
NOX enzyme inhibitors are a set of promising novel experimental
drugs in a new therapeutic class, recognized by the WHO since
2019 when it approved “naxib” as a new stem. Nicotinamide
adenine dinucleotide phosphate (NADPH) oxidases, otherwise
known as NOX enzymes, are the only known enzymes that are
solely dedicated to producing reactive oxygen species (ROS) as
their primary function. They are transmembrane enzymes that
transfer electrons from NADPH in the cytoplasm across the cell
membrane, which results in the formation of ROS.
At appropriate concentrations, ROS have essential functions
in cellular signaling processes, but disruption of the redox
homeostasis has been implicated in multiple disease pathways.
When a cell is injured, excess NOX activity is triggered and
redox homeostasis becomes unbalanced, leading to activation
of pro-fibrogenic pathways. Cancer-associated fibroblasts in the
tumor microenvironment also express NOX enzymes, which can
result in tumours with low immunological activity and relative
resistance to the effects of immuno-oncologic agents, such as
checkpoint inhibitors.
Setanaxib, which is the first NOX inhibitor to reach the clinical
stage, inhibits NOX1 and NOX4, enzymes that are implicated in
fibrosis and inflammation pathways and that represent a high-potential therapeutic target.
Alport Syndrome
Alport syndrome is a genetic disorder arising from the mutations
in the genes that code for type 4 collagen. .The type 4 collagen
alpha chains are primarily located in the kidneys, eyes, and
cochlea, and thus the condition is characterized by kidney
disease, loss of hearing, and eye abnormalities. Eventually,
patients present with proteinuria, hypertension, progressive loss
of kidney function (gradual decline in GFR), and end-stage renal
disease (ESRD).
Pipeline: NOX Inhibitor Platform
It is estimated that 30,000 to 60,000 people in the United
States have this disorder, and it is a significant cause of chronic
kidney disease (CKD), leading to ESRD in adolescents and young
adults and accounting for 1.5% to 3.0% of children on renal
replacement therapies in EU and the US.1
Based on significant and supportive in vivo pre-clinical work, Callid-itas plans to launch a randomized, placebo-controlled clinical trial
in Alport syndrome involving around 20 patients, evaluating overall
safety as well as impact on proteinuria. We expect the study to be
initiated in Q4 2023 and on the basis of the data readout we will
decide on a full regulatory program in Alport. Calliditas was granted
orphan drug designation by the FDA for the treatment of Alport
syndrome with setanaxib in September 2023, and in October
2023 the EMA Committee for Orphan Medicinal Products (COMP)
issued a positive opinion on the company’s application for orphan
drug designation in the EU for setanaxib in Alport syndrome.
Primary Biliary Cholangitis
PBC is a progressive and chronic autoimmune disease of the liver
that causes a cycle of immune injury to biliary epithelial cells,
resulting in cholestasis and fibrosis. It is an orphan disease and,
based on its known prevalence rates, we estimate that there are
approximately 140,000 patients in the US, where the annual
incidence ranges from 0.3 to 5.8 cases per 100,000. Calliditas
received FDA Fast Track Designation for setanaxib in PBC in
August 2021.
Ursodeoxycholic acid, a generic drug also known as ursodiol
or UDCA, and obeticholic acid, known as Ocaliva, are the only
treatments for PBC approved by the FDA and the European
Commission. However, despite these treatment options, there
is still an unmet medical need among PBC patients, in particular
when it comes to important quality of life outcomes.
Phase 2 data from a trial with setanaxib in PBC with 111 patients
demonstrated that setanaxib had a more pronounced effect on
fibrosis and ALP reduction (alkaline phosphatase, a widely estab-lished independent predictor of prognosis in PBC) in patients with
an estimated liver fibrosis stage of F3 or higher. Patients with
elevated liver stiffness are at greater risk of disease progression.
Calliditas is conducting a randomized, placebo-controlled, double-blind Phase 2b trial in PBC patients with elevated liver stiffness.
We have submitted a protocol amendment to the FDA to adapt
the design of this trial, and are expecting to read out data in
mid-2024.
1. Watson S, Padala SA, Hashmi MF, et al. Alport Syndrome. [Updated 2023 Aug 14]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-.
Available from: https://www.ncbi.nlm.nih.gov/books/NBK470419/ |

| 12 Calliditas Therapeutics | Interim Report Q3: January - September 2023
BUSINESS OVERVIEW
Phase 2 Proof-of-Concept Study: Interim Data
Readout
In July 2023, Calliditas read out interim data from the trial, which
reflected encouraging early clinical progression-free survival
(PFS) results and supports the presumed anti-fibrotic mode of
action of setanaxib. The basis for the analysis consisted of a data
set of 20 patients with recurrent or metastatic SCCHN, of which
16 patients had evaluable tumor size and PFS-related results.
Twelve patients had tumor biopsies before and after treatment
that were evaluable for the biomarker analysis, which included
transcriptomic analysis and evaluated pathology markers such
as SMA, Foxp3 regulatory T cells and PDL-1 CPS.
The transcriptomic analysis showed that the two top pathways
impacted by the treatment were fibrosis‑related signalling pathways
(the Idiopathic Pulmonary Fibrosis Signaling Pathway and Hepatic
Fibrosis/Hepatic Stellate Cell Activation Pathway), providing
support for the presumed mode of action on activated cancer
Setanaxib in Squamous Cell Carcinoma of the Head
and Neck
Calliditas is evaluating setanaxib in head and neck cancer, building
on promising in vivo preclinical data that suggests that setanaxib
could significantly enhance the effects of immune-oncology thera-pies. We are conducting a double-blind, randomized, placebo-con-trolled, proof-of-concept Phase 2 study, which is investigating
the effect of setanaxib 800mg twice daily in conjunction with
pembrolizumab 200mg IV, administered every 3 weeks, in at least
50 patients with relapsed or metastatic SCCHN and tumors with
moderate or high levels of cancer-associated fibroblasts.
Pipeline: NOX Inhibitor Platform
28 days
No treatment
Screening Treatment Follow up
~50 Patients
≤ 28 Days
Tumor accessible for
tissue biopsy
Positive CAF level
(defined as CAFs level
in tumors ≥5%)
No treatment
Randomized, Double-Blind
All Enrolled Patients
Tumor Biopsy
9 weeks
(±1 week)
Pembrolizumab 200mg IV, every 3 weeks
Setanaxib 800mg PO, twice daily
Matching placebo
CAF = Cancer-Associated Fibroblast
IV = Intraveneously
PO = Per os (orally)
Randomization
A tumor biopsy will be taken prior to randomization and again
after approximately 9 weeks of treatment. Treatment will continue
until unacceptable toxicity or disease progression, in keeping with
standard practice for oncology trials. Calliditas read out interim data
from the study in July 2023, and expects to read out final trial data
in 1H 2024.
Further details of this study can be found at www.clinicaltrials.gov, with the
reference NCT05323656.
associated fibroblasts in head and neck cancer, as well as a poten-tial anti-fibrotic effect in Calliditas’ other ongoing clinical programs.
Pathology analysis showed preliminary evidence of an increase
in immunological activity within tumors of patients treated with
setanaxib, with favorable changes in Foxp3 and PDL-1 CPS. As
SMA levels at baseline were not balanced between the groups,
and tumor biopsy samples were generally small, it was not possible
to draw any conclusions regarding setanaxib’s impact on SMA
reduction.
In terms of PFS, 7 out of the 16 evaluable patients were progres-sion-free with either stable disease or partial response, of which
6 were in the setanaxib arm and 1 was in the placebo arm. 6 of
the 7 patients were still on the study drug at the time of the data
readout, with the longest period on drug being reported as 21
weeks, related to a patient in the setanaxib arm. |

| 13 Calliditas Therapeutics | Interim Report Q3: January - September 2023
INTERVIEW WITH CMO, RICHARD PHILIPSON
Calliditas
Chief Medical Officer
Richard Philipson
The interim readout of the head and neck cancer
study means that we have now seen the first Calliditas-generated data on setanaxib. What stood out to you
most in these data?
We were really encouraged by the transcriptomic data from
tumor tissue that showed evidence that setanaxib is modulating
fibrosis pathways, which fits very well with the mechanism of
action of the drug. We also saw interesting numerical differences
in progression-free survival favoring patients treated with seta-naxib (on top of pembrolizumab), noting that we did not do any
formal statistical testing in this interim data review.
These data set the scene very nicely for the final analysis of data
that will come in the first half of 2024, where we would like
to see that, when given with pembrolizumab, setanaxib slows
tumor growth and improves progression-free survival, compared
to pembrolizumab alone.
The full NefIgArd data were recently published in
The Lancet. How important do you think this is for
nephrologists navigating the IgAN landscape?
The publication of the final analysis of the NefIgArd study is a
landmark for Nefecon and more generally for the field in IgA
nephropathy. We know that nephrologists are really keen to see
high-quality scientific and clinical data supporting the use of
Nefecon; the article in The Lancet, which was accepted through
a fast-track review process, provides a clear and comprehensive
description of kidney function outcomes over the 2-year study.
When looking at the outcomes of the study, what has particu-larly impressed me is the clearly beneficial effects of Nefecon
on kidney function (estimated glomerular filtration rate [eGFR])
achieved during 9 months of treatment, which is maintained
through the subsequent 15 months of observation off-treat-ment. This is accompanied by a sustained and durable improve-ment in proteinuria that is also maintained during the off-treat-ment period.
The open-label extension study for Nefecon is currently
ongoing with a readout expected in mid-2024. What
will you be looking for in this data set?
This is a very interesting and important study that will help us
understand the impact of re-treatment with Nefecon. In the
open-label extension study (OLE), all enrolled patients receive a
9-month treatment course of Nefecon. Some of these patients
will already have received a 9-month course of treatment with
Nefecon in the main NefIgArd study (followed by 15 months
of observation off-treatment), whereas the remaining patients
will be receiving Nefecon for the first time (since they received
placebo in the main study). We will be able to look at how the
second course of treatment impacts proteinuria and eGFR, and
therefore better understand the potential benefits of re-treat-ment with Nefecon. We hope to publish these outcomes shortly
after the final analysis of the study is completed.
You joined Calliditas in mid-2020. What has been
the most exciting development on the medical side
since you have been at the company?
In the three years since I joined the company, Calliditas has
grown substantially, and is a very different company compared to
the one I joined in July 2020. On the medical side, we have built
internal expertise in Clinical Operations, Biostatistics, Scientific
Communications and Pharmacovigilance, and I am particularly
excited by how we have been able to take compelling preclin-ical data in diseases such as head & neck cancer and Alport
syndrome, and translate these to clinical studies that will allow
us to evaluate the safety and clinical effects of setanaxib in these
diseases. We are absolutely committed to developing medicines
that make a difference for patients with rare diseases, and I am
very proud of the excellent network of relationships with experts
that we have built in nephrology, liver disease and head & neck
cancer. |

| 14 Calliditas Therapeutics | Interim Report Q3: January - September 2023
Calliditas develops its sustainability efforts
with its sights set on CSRD reporting
14 Calliditas Therapeutics | Interim Report Q3: January - September 2023
Calliditas is committed to focusing on
sustainability and is looking forward to
intensifying its efforts and becoming
increasingly transparent regarding sust-ainability issues. In the EU, the Corporate
Sustainability Reporting Directive (CSRD) will
introduce new reporting requirements and
provide both direction and a methodology
for our sustainability efforts.
Materiality assessment as the first step
As a first step in Calliditas’ intensified sustainability ambitions,
we have begun our work by carrying out a double materiality
assessment. This assessment will help to define the sustainability
issues that are most relevant to Calliditas. In line with the CSRD,
these issues are defined based on two perspectives. The impact
perspective is based on how people and the environment are
impacted by Calliditas’ operations, and the financial perspective
is based on how Calliditas is impacted financially by external
factors.
The defined material topics will then form the basis of Calliditas’
sustainability strategy, targets and operations. They will also
form the basis of Calliditas’ future CSRD-compliant sustainability
reporting.
SUSTAINABILITY
We have our sights set on the
2025 Annual and Sustainability Report
Calliditas will be subject to the CSRD as of the 2025 financial
year, for which the report will be published in 2026. To ensure
that Calliditas’ actual sustainability work is performed in a timely
“We have made solid strides in our work regarding
sustainability. It is important for Calliditas to carry
out our work in a socially and environmentally
ethical and responsible manner, and to be
compliant with the new CSRD directive.”
Åsa Hillsten
Head of IR & Sustainability
and thorough manner, the work on implementing the CSRD in
Calliditas’ sustainability efforts has already begun. As a first step,
the Board of Directors and the Management Team received
training on the topic. This training provided an overview of
the new rules and regulations, described how Calliditas will be
affected, and highlighted the increased responsibility of the
board and management team.
Sustainability issues have always been highly important to Callid-itas. Through our business model, we contribute to improving
the lives of patients with rare diseases with high unmet medical
needs. Considering the industry in which Calliditas operates,
ethical and responsible business practices are of the utmost
importance. It is also imperative to us at Calliditas to offer our
employees a safe, healthy and stimulating workplace. This
includes non-discrimination and a workplace culture that offers
equal opportunities for all. Calliditas also has major upstream
and downstream responsibility in its value chain, and therefore
has ambitions to reduce the Company’s environmental impact
and resource usage.
The CSRD entails stricter requirements on
quality as well as quantity in sustainability reporting
The CSRD is part of the EU’s package of measures to promote
fair and transparent sustainability reporting. This is part of a bigger
picture of the EU’s ambition to ensure that financing and capital
reach companies have sustainable investments and business
models, so that the EU meets the requirements of the Paris
Agreement and the 17 Sustainable Development Goals.
The requirements in the CSRD are more comprehensive than
previous sustainability reporting requirements and cover
numerous disclosures linked to Environmental, Social and Gover-nance (ESG) issues. Additional disclosures relate to the reporting
of sustainability governance in general and the development of
annual sustainability reports. Calliditas is looking forward to under-taking the work necessary to meet the CSRD requirements and
take strides in becoming a more sustainable business. |

| 15 Calliditas Therapeutics | Interim Report Q3: January - September 2023
January – September 2023
FINANCIAL OVERVIEW
Revenue
Net sales amounted to SEK 294.6 million and SEK 260.1 million
for the three months ended September 30, 2023 and 2022,
respectively. Net sales for the nine months ended September 30,
2023 and 2022 amounted to SEK 755.3 million and SEK 373.8
million, respectively. Net sales for the periods primarily originate
from net sales of TARPEYO® in the US, which amounted to
SEK 283.6 million and SEK 123.4 million for the three months
ended September 30, 2023 and 2022, respectively. For the nine
months ended September 30, 2023 and 2022, net sales from
TARPEYO amounted to SEK 728.5 million and SEK 205.0 million,
respectively. Royalty income from our partnership in Europe
amounted to SEK 9.6 million for the third quarter of 2023 and
SEK 22.8 million for the first nine months of 2023, regarding the
comparison periods 2022, no royalty income was recognized.
For the three and nine months ended September 30, 2023,
no milestones were recognized. For the three months ended
September 30, 2022, net sales amounted to SEK 135.0 million
and consisted of milestone fees from the commercialization of
Kinpeygo® in Europe. Further, regarding the nine months ended
September 30, 2022 the milestone fees amounted to SEK 163.8
million and included milestone fee regarding Everst Medicines.
For additional information see Note 4.
Cost of Sales
Cost of sales amounted to SEK 14.9 million and SEK 4.3 million
for the three months ended September 30, 2023 and 2022,
respectively. For the nine months ended September 30, 2023 and
2022 cost of sales amounted to SEK 38.2 million and SEK 7.3
million, respectively. The increase in the 2023 periods, was related
to the higher volume of product sales.
Total Operating Expenses
Total operating expenses amounted to SEK 439.2 million and SEK
292.0 million for the three months ended September 30, 2023
and 2022, respectively. For the nine months ended September
30, 2023 and 2022 total operating expenses amounted to SEK
1,132.0 million and SEK 821.0 million, respectively.
Research and Development Expenses
Research and development expenses amounted to SEK 179.9
million and SEK 102.9 million for the three months ended
September 30, 2023 and 2022, respectively. For the nine months
ended September 30, 2023 and 2022 research and development
expenses amounted to SEK 395.5 million and SEK 312.5 million,
respectively. The increase of SEK 77.0 million for the three
months ended September 30, 2023 and the increase of SEK
83.0 million for the nine months ended September 30, 2023, was
primarily due to increased clinical activities for the nox-platform,
including the ongoing setanaxib trials, and the one-time effect
from the impairment of SEK 32.1 million regarding Budenofalk,
an in-licensing that was established in 2019, compared to the
corresponding periods of the prior year.
Marketing and Selling Expenses
Marketing and selling expenses amounted to SEK 170.5 million
and SEK 116.1 million for the three months ended September
30, 2023 and 2022, respectively. For the nine months ended
September 30, 2023 and 2022 marketing and selling expenses
amounted SEK 529.2 million and SEK 323.3 million, respectively.
The increases of SEK 54.4 million for the three months ended
September 30, 2023, and SEK 205.9 million for the nine months
ended September 30, 2023, were primarily related to the costs
for sales and marketing of TARPEYO in the US, where marketing
activities have been intensified and the salesforce has been
increased, compared to the corresponding periods of the prior year.
Administrative Expenses
Administrative expenses amounted to SEK 88.6 million and SEK
71.0 million for the three months ended September 30, 2023 and
2022, respectively. For the nine months ended September 30,
2023 and 2022, administrative expenses amounted to SEK 238.4
million and SEK 178.4 million, respectively. The increases of SEK
17.6 million for the three months ended September 30, 2023, and
SEK 59.9 million for the nine months ended September 30, 2023,
were primarily related to increases due to a larger organization and
increased regulatory requirements.
Other Operating Incomes/Expenses, net
Other operating income/(expenses), net amounted to (SEK
0.2 million) and (SEK 1.9 million) for the three months ended
September 30, 2023 and 2022, respectively. For the nine months
ended September 30, 2023 and 2022 other operating income/
(expenses), net amounted to SEK 31.1 million and (SEK 6.7 million),
respectively. The increase in both periods was primarily attributable
to movements in exchange rate related to operating receivables
and liabilities.
Net Financial Income and Expenses
Net financial income/(expenses) amounted to (SEK 4.5 million)
and SEK 20.3 million for the three months ended September
30, 2023 and 2022, respectively. For the nine months
ended September 30, 2023 and 2022, net financial income/
(expenses) amounted to (SEK 27.9 million) and SEK 35.0 million,
respectively. The decrease of SEK 24.8 million for the three
months ended September 30, 2023, and SEK 62.9 million for
the nine months ended September 30, 2023 were primarily
derived from interest expenses related to borrowing and
currency effects. |

| 16 Calliditas Therapeutics | Interim Report Q3: January - September 2023
FINANCIAL OVERVIEW
Tax
Total tax income/(expense) amounted to (SEK 4.3 million) and
SEK 6.9 million for the three months ended September 30, 2023
and 2022, respectively. For the nine months ended September
30, 2023 and 2022, total tax income/(expense) amounted
to (SEK 5.1 million) and SEK 10.9 million, respectively. The
increased tax expense for both periods was primarily explained
by taxable profit for the US subsidiaries. The Group’s tax losses
carried-forward have not been recognized as deferred tax assets,
other than to the extent such tax losses can be used to offset
temporary differences.
Result for the Period
For the three months ended September 30, 2023 and 2022,
loss for the period amounted to SEK 168.4 million and SEK 9.1
million, and the corresponding loss per share before and after
dilution amounted to SEK 3.14 and SEK 0.17, respectively. For
the nine months ended September 30, 2023 and 2022, loss
for the period amounted to SEK 447.8 million and SEK 408.6
million, and the corresponding loss per share before and after
dilution amounted to SEK 8.34 and SEK 7.72, respectively.
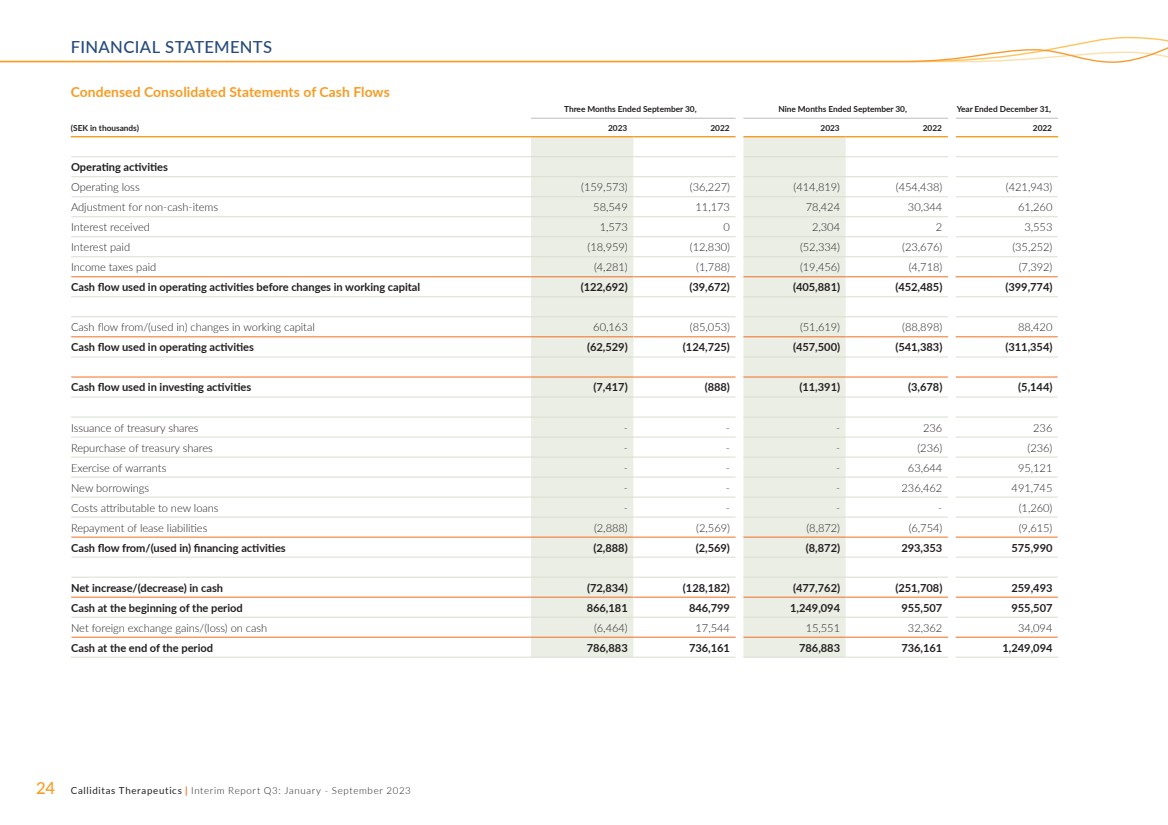
Cash Flow and Cash Position
Cash flow used in operating activities amounted to SEK 62.5
million and SEK 124.7 million for the three months ended
September 30, 2023 and 2022, respectively, and the improve-ment compared to previous year, is explained by the stronger
working capital. For the nine months ended September 30, 2023
and 2022, cash flow used in operating activities amounted to
SEK 457.5 million and SEK 541.4 million, respectively, and the
improvement is primarily explained by the increased operating
result driven by sales growth for TARPEYO® in the US, and
additional the stronger working capital had a positive impact.
Cash flow used in investing activities amounted to SEK
7.4 million and SEK 0.9 million for the three months ended
September 30, 2023 and 2022, respectively. For the nine
months ended September 30, 2023 and 2022, cash flow used
in investing activities amounted to SEK 11.4 million and SEK
3.7 million, respectively. The increase for the both periods was
primarily explained by acquisition of equipment.
Cash flow used in financing activities amounted to SEK
2.9 million and SEK 2.6 million for the three months ended
September 30, 2023 and 2022, respectively. For the nine
months ended September 30, 2023 and 2022, cash flow from
financing activities amounted to (SEK 8.9 million) and SEK 293.4
million, respectively. The decrease for the periods was mainly
attributable to previous years utilization of an external loan
facility of SEK 236.5 million.
Net decrease in cash amounted to SEK 72.8 million and
SEK 128.2 million for the three months ended September
30, 2023 and 2022, respectively. For the nine months ended
September 30, 2023 and 2022, net decrease in cash amounted
to SEK 477.8 million and SEK 251.7 million, respectively. Cash
amounted to SEK 786.9 million and SEK 736.2 million as of
September 30, 2023 and 2022, respectively.
Personnel
The average number of employees were 179 and 92 for the
three months ended September 30, 2023 and 2022, respec-tively and 174 and 81 employees for the nine months ended
September 30, 2023 and 2022, respectively.
Changes in Shareholders’
Equity and Number of Shares
Equity attributable to equity holders of the Parent Company
amounted to SEK 352.4 million and SEK 725.9 million as of
September 30, 2023 and 2022, respectively. The number of
registered shares amounted to 59,580,087 and 59,157,587 as
of September 30, 2023 and 2022, respectively.
Treasury Shares
As of September 30, 2023, Calliditas had 5,908,018 ordinary
shares held as treasury shares by the Parent Company. At the
Annual General Meeting in 2023, authorization was given that
Calliditas can transfer (sale) these ordinary shares with the
purpose to finance an acquisition of operations, to procure
capital to finance the development of projects, repayment of
loans or to commercialize Calliditas’ products. See Note 7 for
further information.
Incentive Programs
During the three months ended September 30, 2023, 965,000
options have been allocated for the ESOP 2023 Program. For
more information on incentive programs, see Note 9.
2023 Outlook
For 2023, Calliditas expects revenue growth in the US where:
Net sales from TARPEYO are estimated to be USD 100-120
million for the year ending December 31, 2023 (corresponding
to approx. SEK 1,047-1,257 million, using a SEK/USD average
exchange rate of 10.47).
Parent Company
Net sales for the Parent Company, Calliditas Therapeutics
AB, amounted to SEK 194.4 million and SEK 219.6 million
for the three months ended September 30, 2023 and 2022,
respectively. For the nine months ended September 30, 2023
and 2022 net sales amounted to SEK 468.4 million and SEK
251.8 million, respectively. The decrease for the three months
ended September 30, 2023, compared to corresponding period
prior year refers to milestone fees from the commercialization
of Kinpeygo in Europe, which amounted to SEK 135.0 million.
Further, for the nine months ended September 30, 2022, the
milestone fees amounted to SEK 163.8 million.
The increase in net sale for the nine months ended September
30, 2023, was primarily derived from sales of TARPEYO compared
to the corresponding period of the prior year. Operating loss
amounted to SEK 104.1 million and SEK 47.4 million for the three
months ended September 30, 2023 and 2022, respectively. For
the nine months ended September 30, 2023 and 2022 oper-ating loss amounted to SEK 268.9 million and SEK 302.1 million,
respectively. |

| 17 Calliditas Therapeutics | Interim Report Q3: January - September 2023
Significant Events
FINANCIAL COMMENTS
Significant Events During the Period
July 1 – September 30, 2023
• On July 13 Calliditas announced supportive interim data from
Phase 2 head and neck cancer trial with lead NOX inhibitor
candidate, setanaxib.
• On August 15 Calliditas announced full results from the NefIgArd
Phase 3 trial published in The Lancet.
• On August 18 FDA granted priority review for full approval of
TARPEYO® for the treatment of IgA Nephropathy.
• On September 27 Calliditas granted orphan drug designation by
the FDA for the treatment of Alport syndrome with setanaxib.
• On September 28 STADA and Calliditas announced the filing for
full marketing authorization of Kinpeygo® in the EU.
• On September 29 Calliditas presented Data from the NefIgArd
Phase 3 trial at the 17th International Symposium on IgA
Nephropathy (IIgANN) Tokyo 2023.
Significant Events After the Period
• On October 3 Calliditas announced filing with UK MHRA for
Kinpeygo in IgA nephropathy.
• On October 5 European Medicines Agency Committee
for Orphan Medicinal Products provided positive opinion on
Calliditas’ application for setanaxib in Alport syndrome.
• On October 23 Calliditas announced the Nomination Committee
for the AGM 2024 was appointed. The Nomination Committee
consists of Patrick Sobocki, appointed by Stiftelsen Industri-fonden, Karl Tobieson, appointed by Linc AB, Spike Loy, appointed
by BVF and Elmar Schnee (chairman of the board of directors).
• 27 October Calliditas announced that its commercial partner
Everest Medicines received approval from the Pharmaceutical
Administration Bureau of the Macau Special Administrative
Region, China.
Executive Management
The Executive Management of Calliditas Therapeutics
AB consists of: CEO Renée Aguiar-Lucander, CFO Fredrik
Johansson, CMO Richard Philipson, Group General Counsel
Jonathan Schur, President North America Andrew Udell, Vice
President Regulatory Affairs Frank Bringstrup and Head of
Human Resource Sandra Frithiof.
Dividend
Any future dividend and the size thereof, will be determined
based on long-term growth, earnings trends and capital require-ments of Calliditas. The Board of Directors does not intend to
propose any dividend before the company generates long-term
sustainable profits and positive cash flow. There was no dividend
paid for the 2022 financial year. |

| 18 Calliditas Therapeutics | Interim Report Q3: January - September 2023
FINANCIAL COMMENTS
Review
This interim report has not been subject to review by the
company’s auditors.
Stockholm 7 November, 2023
Renée Aguiar-Lucander
Ten largest shareholders as of September, 2023 %
BVF Partners LP 10.51
Linc AB 10.01
Stiftelsen Industrifonden 5.28
Avanza Pension 3.81
Handelsbanken Fonder 3.47
Fjärde AP-fonden 3.19
Unionen 3.02
Polar Capital 2.94
Sofinnova Partners 2.36
The Invus Group 1.79
Subtotal, 10 largest shareholders 46.38
Treasury shares 9.92
Other shareholders 43.70
Total 100.00
Annual General Meeting 2024
The 2024 Annual General Meeting will be held in May, 2024 in
Stockholm, Sweden. All documentation from previous AGMs
are published on the companys´ website at https://www.callid-itas.se/en/governance/general-meeting/general-meetings/
Nomination Committee AGM 2024 appointed
On October 2023 Calliditas announced that the companys´
major owners have appointed a nomination committee for the
AGM 2024. The nomination committee, which is appointed in
accordance with the principles adopted by the extraordinary
general meeting in 2017, consists of: Patrick Sobocki, appointed
by Stiftelsen Industrifonden, Karl Tobieson, appointed by Linc
AB, Spike Loy, appointed by BVF, and Elmar Schnee (chairman
of the board of directors). Shareholders who wish to submit
proposals to the nomination committee for the annual general
meeting 2024, can do so by e-mail to finance@calliditas.com.
Proposals should be submitted to the nomination committee
before March 15, 2024.
The Share
As of September 30, 2023, the number of shares amounted to
59,580,087 ordinary shares, of which, 5,908,018 are held as
treasury shares by the Parent Company. As of September 29,
2023, the closing price for the Calliditas Therapeutics share
CALTX was SEK 97.5. The total number of shareholders as of
September 30, 2023 was approximately 15,000.
Shareholder Structure |

| 19 Calliditas Therapeutics | Interim Report Q3: January - September 2023
Presentation to Investors,
Analysts and Press
For further information
please contact
Upcoming Events
• Calliditas invites investors, analysts and press to a presentation of the
Quarterly Report 2023 at 14:30 pm. on November 7. The report will
be published at 7:00 am the same day.
• Calliditas’ CEO Renée Aguiar-Lucander will present the report together
with CFO Fredrik Johansson, CMO Richard Philipson and President
North America Andrew Udell. The presentations will be given in
English.
• Time: Tuesday 14:30 pm CET. on November 7.
• Link to webcast https://financialhearings.com/event/46414
• To participate via conference call register via this link:
https://conference.financialhearings.com/teleconference/?id=2001082
After registration, you will receive a phone number and a conference
ID to log in to the conference call. Via the telephone conference, there
is an opportunity to ask oral questions.
FEBRUARY 22, 2024
Year-end report, Q4
January – December 2023
ANNUAL REPORT 2023
Will be published digitally
in April, 2024
MAY 16, 2024
Interim Report, Q1
January – March 2024
Renée Aguiar-Lucander / CEO
+46 (0)8 411 30 05
renee.lucander@calliditas.com
Åsa Hillsten / Head of IR & Sustainability
+46 (0) 764 03 35 43
asa.hillsten@calliditas.com
Supplemental Information
This interim report has not been reviewed or audited by the Company’s auditors.
The information in the Interim Report is information that Calliditas is obliged to make public pursuant to
the EU Market Abuse Regulation. The information was sent for publication, through the agency of the
contact persons set out above, on November 7, 2023, at 7:00 a.m. CET.
Registred Office
Calliditas Therapeutics AB
Kungsbron 1
SE 111 22 Stockholm, Sweden
calliditas.com / ir@calliditas.com
Forward Looking Statements
This Interim Report contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, as amended,
including, without limitation, statements regarding Calliditas’ strategy, business plans, revenue and other financial projections, and focus. The words “may,”
“will,” “could,” “would,” “should,” “expect,” “plan,” “anticipate,” “intend,” “believe,” “estimate,” “predict,” “project,” “potential,” “continue,” “target” and similar
expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words.
Any forward-looking statements in this Interim Report are based on management’s current expectations and beliefs and are subject to a number of risks,
uncertainties and important factors that may cause actual events or results to differ materially from those expressed or implied by any forward-looking
statements contained in this Interim Report, including, without limitation, any related to Calliditas’ business, operations, commercialization of TARPEYO
and Kinpeygo, clinical trials, supply chain, strategy, goals and anticipated timelines for development and potential approvals, competition from other
biopharmaceutical companies, revenue and product sales projections or forecasts, including 2023 net sales of TARPEYO guidance and cash runway, and
other risks identified in the section entitled “Risk Factors” in Calliditas’ reports filed with the Securities and Exchange Commission.
Calliditas cautions you not to place undue reliance on any forward-looking statements, which speak only as of the date they are made. Calliditas disclaims
any obligation to publicly update or revise any such statements to reflect any change in expectations or in events, conditions or circumstances on which
any such statements may be based, or that may affect the likelihood that actual results will differ from those set forth in the forward-looking statements.
Any forward-looking statements contained in this Interim Report represent Calliditas’ views only as of the date hereof and should not be relied upon as
representing its views as of any subsequent date.
This Interim Report has been prepared in a Swedish original and has been translated into English. In case of differences between the two, the Swedish
version shall apply. |
| 20 Calliditas Therapeutics | Interim Report Q3: January - September 2023
FINANCIAL STATEMENTS
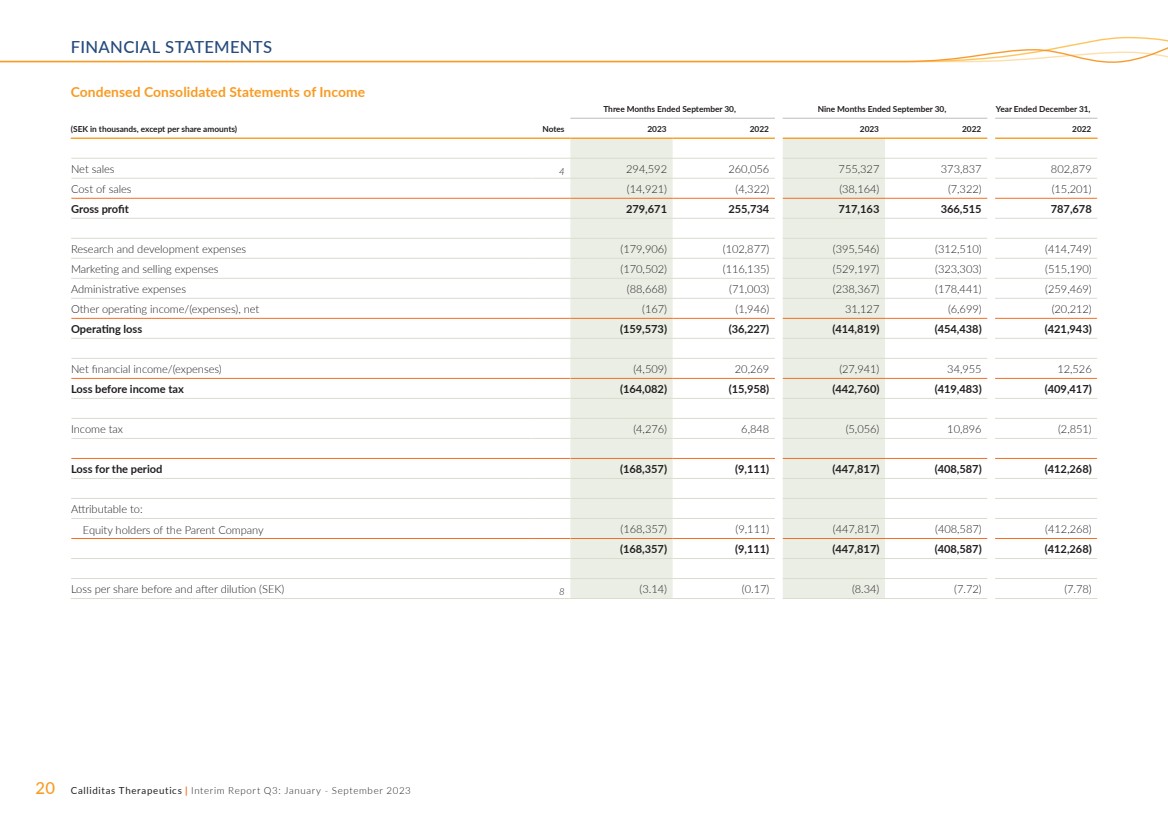
Condensed Consolidated Statements of Income
Three Months Ended September 30, Nine Months Ended September 30, Year Ended December 31,
(SEK in thousands, except per share amounts) Notes 2023 2022 2023 2022 2022
Net sales 4 294,592 260,056 755,327 373,837 802,879
Cost of sales (14,921) (4,322) (38,164) (7,322) (15,201)
Gross profit 279,671 255,734 717,163 366,515 787,678
Research and development expenses (179,906) (102,877) (395,546) (312,510) (414,749)
Marketing and selling expenses (170,502) (116,135) (529,197) (323,303) (515,190)
Administrative expenses (88,668) (71,003) (238,367) (178,441) (259,469)
Other operating income/(expenses), net (167) (1,946) 31,127 (6,699) (20,212)
Operating loss (159,573) (36,227) (414,819) (454,438) (421,943)
Net financial income/(expenses) (4,509) 20,269 (27,941) 34,955 12,526
Loss before income tax (164,082) (15,958) (442,760) (419,483) (409,417)
Income tax (4,276) 6,848 (5,056) 10,896 (2,851)
Loss for the period (168,357) (9,111) (447,817) (408,587) (412,268)
Attributable to:
Equity holders of the Parent Company (168,357) (9,111) (447,817) (408,587) (412,268)
(168,357) (9,111) (447,817) (408,587) (412,268)
Loss per share before and after dilution (SEK) 8 (3.14) (0.17) (8.34) (7.72) (7.78) |
| 21 Calliditas Therapeutics | Interim Report Q3: January - September 2023
FINANCIAL STATEMENTS
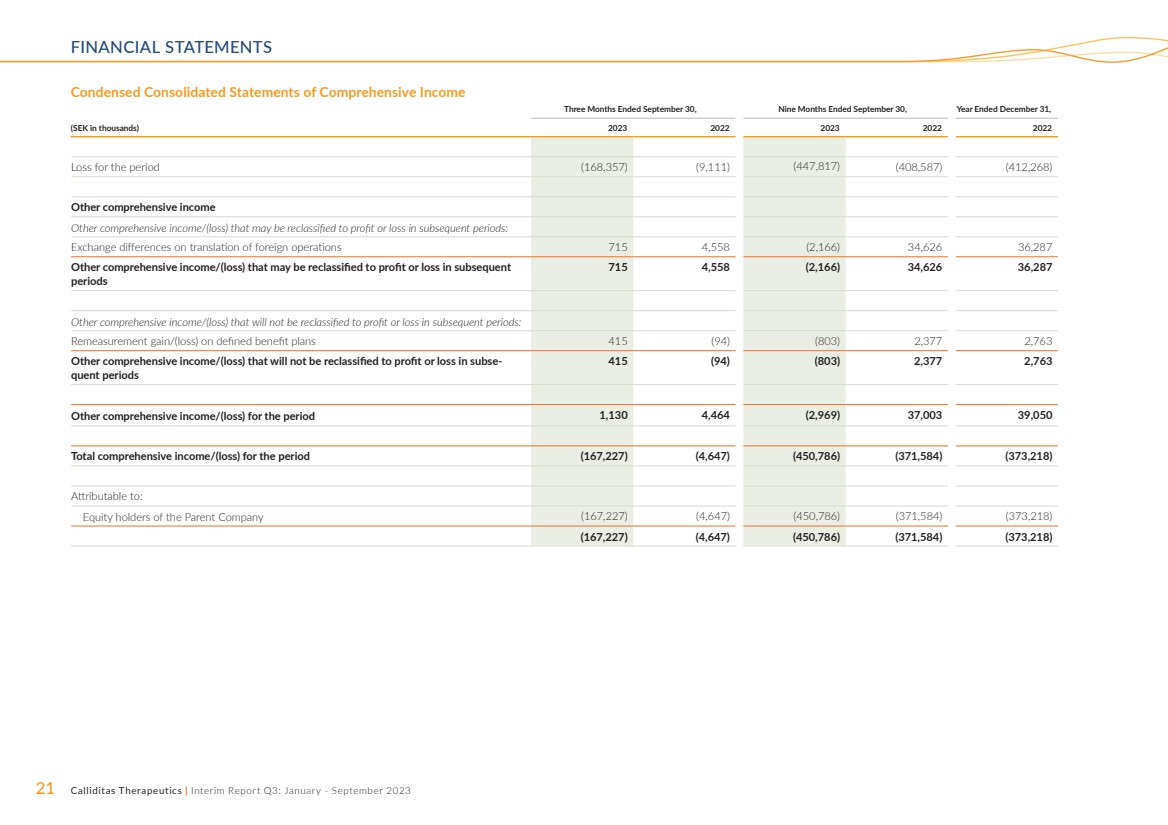
Condensed Consolidated Statements of Comprehensive Income
Three Months Ended September 30, Nine Months Ended September 30, Year Ended December 31,
(SEK in thousands) 2023 2022 2023 2022 2022
Loss for the period (168,357) (9,111) (447,817) (408,587) (412,268)
Other comprehensive income
Other comprehensive income/(loss) that may be reclassified to profit or loss in subsequent periods:
Exchange differences on translation of foreign operations 715 4,558 (2,166) 34,626 36,287
Other comprehensive income/(loss) that may be reclassified to profit or loss in subsequent
periods
715 4,558 (2,166) 34,626 36,287
Other comprehensive income/(loss) that will not be reclassified to profit or loss in subsequent periods:
Remeasurement gain/(loss) on defined benefit plans 415 (94) (803) 2,377 2,763
Other comprehensive income/(loss) that will not be reclassified to profit or loss in subse-quent periods
415 (94) (803) 2,377 2,763
Other comprehensive income/(loss) for the period 1,130 4,464 (2,969) 37,003 39,050
Total comprehensive income/(loss) for the period (167,227) (4,647) (450,786) (371,584) (373,218)
Attributable to:
Equity holders of the Parent Company (167,227) (4,647) (450,786) (371,584) (373,218)
(167,227) (4,647) (450,786) (371,584) (373,218) |
| 22 Calliditas Therapeutics | Interim Report Q3: January - September 2023
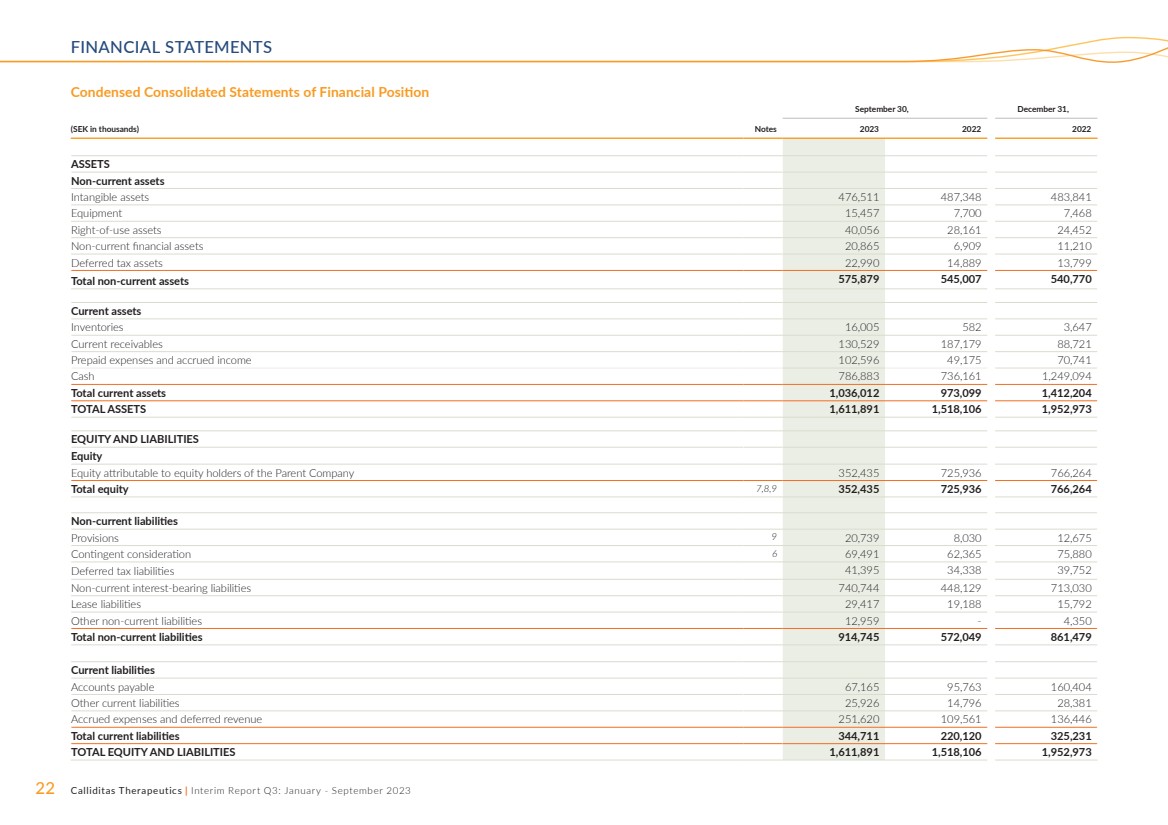
FINANCIAL STATEMENTS
September 30, December 31,
(SEK in thousands) Notes 2023 2022 2022
ASSETS
Non-current assets
Intangible assets 476,511 487,348 483,841
Equipment 15,457 7,700 7,468
Right-of-use assets 40,056 28,161 24,452
Non-current financial assets 20,865 6,909 11,210
Deferred tax assets 22,990 14,889 13,799
Total non-current assets 575,879 545,007 540,770
Current assets
Inventories 16,005 582 3,647
Current receivables 130,529 187,179 88,721
Prepaid expenses and accrued income 102,596 49,175 70,741
Cash 786,883 736,161 1,249,094
Total current assets 1,036,012 973,099 1,412,204
TOTAL ASSETS 1,611,891 1,518,106 1,952,973
EQUITY AND LIABILITIES
Equity
Equity attributable to equity holders of the Parent Company 352,435 725,936 766,264
Total equity 7,8,9 352,435 725,936 766,264
Non-current liabilities
Provisions 9 20,739 8,030 12,675
Contingent consideration 6 69,491 62,365 75,880
Deferred tax liabilities 41,395 34,338 39,752
Non-current interest-bearing liabilities 740,744 448,129 713,030
Lease liabilities 29,417 19,188 15,792
Other non-current liabilities 12,959 - 4,350
Total non-current liabilities 914,745 572,049 861,479
Current liabilities
Accounts payable 67,165 95,763 160,404
Other current liabilities 25,926 14,796 28,381
Accrued expenses and deferred revenue 251,620 109,561 136,446
Total current liabilities 344,711 220,120 325,231
TOTAL EQUITY AND LIABILITIES 1,611,891 1,518,106 1,952,973
Condensed Consolidated Statements of Financial Position |
| 23 Calliditas Therapeutics | Interim Report Q3: January - September 2023
FINANCIAL STATEMENTS
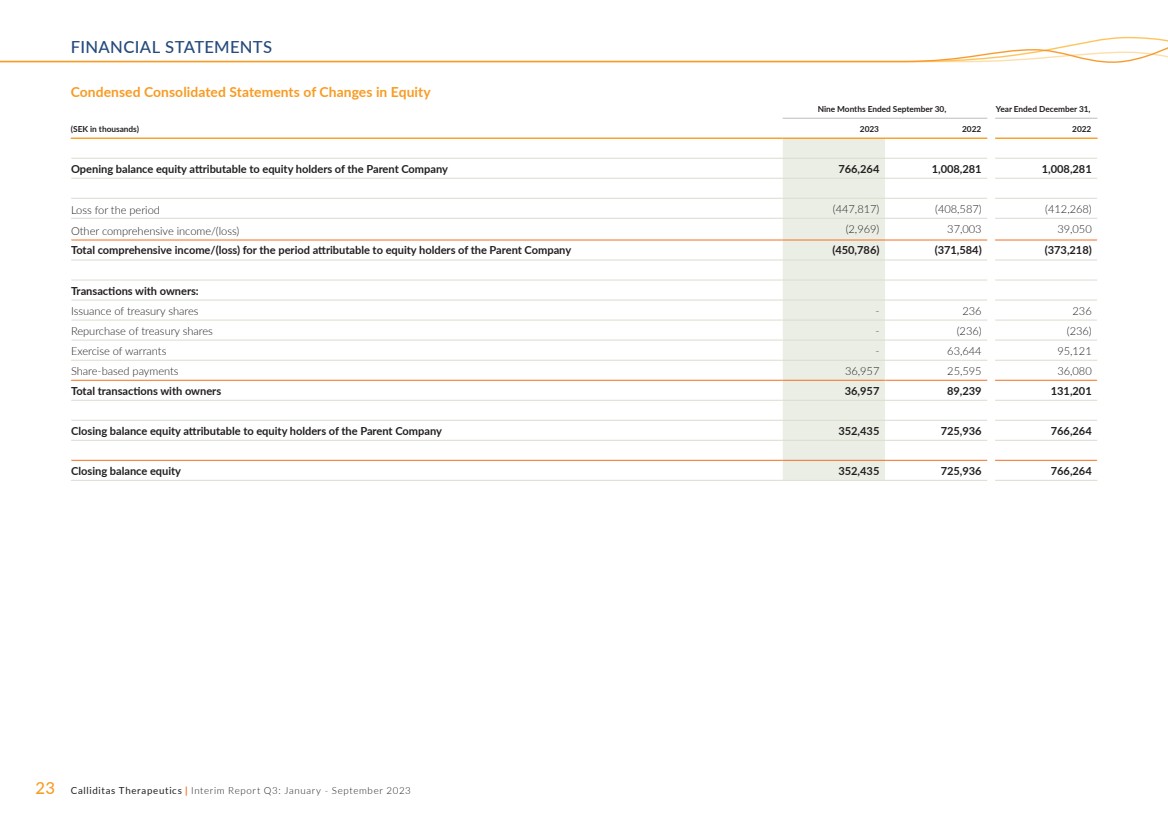
Condensed Consolidated Statements of Changes in Equity
Nine Months Ended September 30, Year Ended December 31,
(SEK in thousands) 2023 2022 2022
Opening balance equity attributable to equity holders of the Parent Company 766,264 1,008,281 1,008,281
Loss for the period (447,817) (408,587) (412,268)
Other comprehensive income/(loss) (2,969) 37,003 39,050
Total comprehensive income/(loss) for the period attributable to equity holders of the Parent Company (450,786) (371,584) (373,218)
Transactions with owners:
Issuance of treasury shares - 236 236
Repurchase of treasury shares - (236) (236)
Exercise of warrants - 63,644 95,121
Share-based payments 36,957 25,595 36,080
Total transactions with owners 36,957 89,239 131,201
Closing balance equity attributable to equity holders of the Parent Company 352,435 725,936 766,264
Closing balance equity 352,435 725,936 766,264 |
| 24 Calliditas Therapeutics | Interim Report Q3: January - September 2023
FINANCIAL STATEMENTS
Three Months Ended September 30, Nine Months Ended September 30, Year Ended December 31,
(SEK in thousands) 2023 2022 2023 2022 2022
Operating activities
Operating loss (159,573) (36,227) (414,819) (454,438) (421,943)
Adjustment for non-cash-items 58,549 11,173 78,424 30,344 61,260
Interest received 1,573 0 2,304 2 3,553
Interest paid (18,959) (12,830) (52,334) (23,676) (35,252)
Income taxes paid (4,281) (1,788) (19,456) (4,718) (7,392)
Cash flow used in operating activities before changes in working capital (122,692) (39,672) (405,881) (452,485) (399,774)
Cash flow from/(used in) changes in working capital 60,163 (85,053) (51,619) (88,898) 88,420
Cash flow used in operating activities (62,529) (124,725) (457,500) (541,383) (311,354)
Cash flow used in investing activities (7,417) (888) (11,391) (3,678) (5,144)
Issuance of treasury shares - - - 236 236
Repurchase of treasury shares - - - (236) (236)
Exercise of warrants - - - 63,644 95,121
New borrowings - - - 236,462 491,745
Costs attributable to new loans - - - - (1,260)
Repayment of lease liabilities (2,888) (2,569) (8,872) (6,754) (9,615)
Cash flow from/(used in) financing activities (2,888) (2,569) (8,872) 293,353 575,990
Net increase/(decrease) in cash (72,834) (128,182) (477,762) (251,708) 259,493
Cash at the beginning of the period 866,181 846,799 1,249,094 955,507 955,507
Net foreign exchange gains/(loss) on cash (6,464) 17,544 15,551 32,362 34,094
Cash at the end of the period 786,883 736,161 786,883 736,161 1,249,094
Condensed Consolidated Statements of Cash Flows |
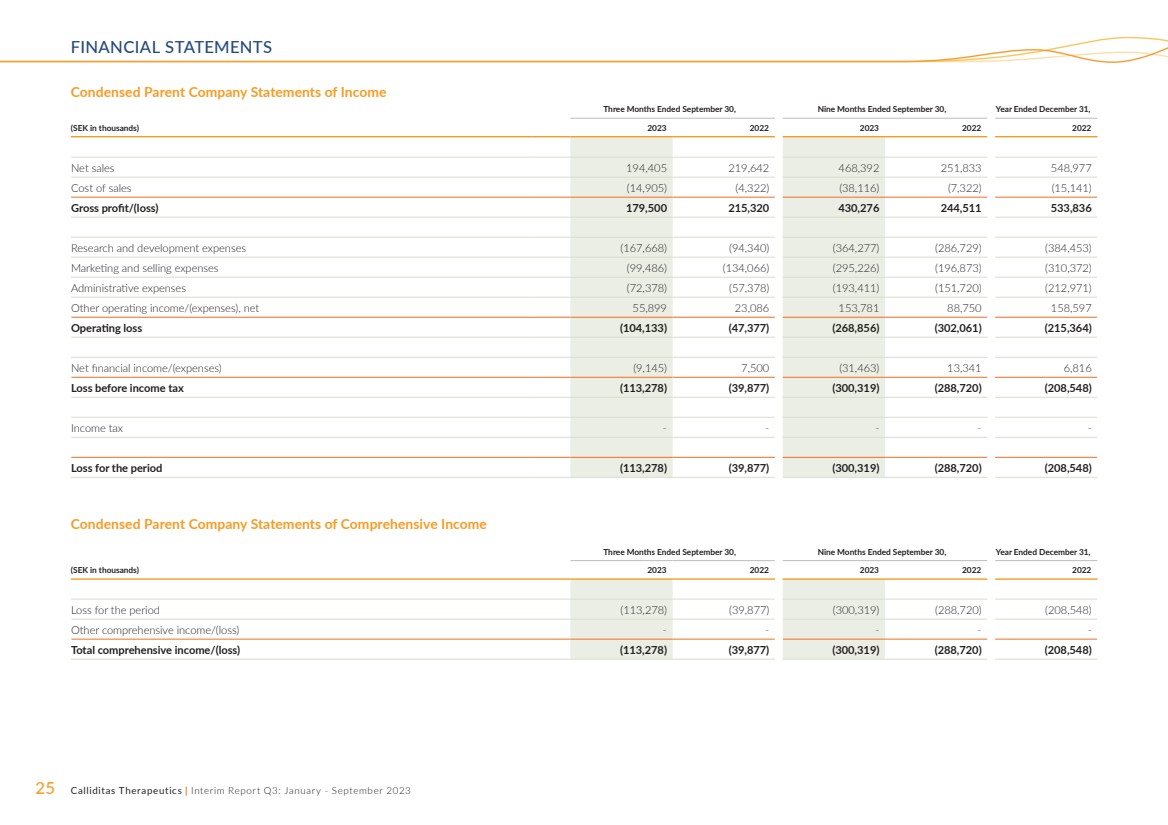
| 25 Calliditas Therapeutics | Interim Report Q3: January - September 2023
Condensed Parent Company Statements of Income
FINANCIAL STATEMENTS
Three Months Ended September 30, Nine Months Ended September 30, Year Ended December 31,
(SEK in thousands) 2023 2022 2023 2022 2022
Net sales 194,405 219,642 468,392 251,833 548,977
Cost of sales (14,905) (4,322) (38,116) (7,322) (15,141)
Gross profit/(loss) 179,500 215,320 430,276 244,511 533,836
Research and development expenses (167,668) (94,340) (364,277) (286,729) (384,453)
Marketing and selling expenses (99,486) (134,066) (295,226) (196,873) (310,372)
Administrative expenses (72,378) (57,378) (193,411) (151,720) (212,971)
Other operating income/(expenses), net 55,899 23,086 153,781 88,750 158,597
Operating loss (104,133) (47,377) (268,856) (302,061) (215,364)
Net financial income/(expenses) (9,145) 7,500 (31,463) 13,341 6,816
Loss before income tax (113,278) (39,877) (300,319) (288,720) (208,548)
Income tax - - - - -
Loss for the period (113,278) (39,877) (300,319) (288,720) (208,548)
Three Months Ended September 30, Nine Months Ended September 30, Year Ended December 31,
(SEK in thousands) 2023 2022 2023 2022 2022
Loss for the period (113,278) (39,877) (300,319) (288,720) (208,548)
Other comprehensive income/(loss) - - - - -
Total comprehensive income/(loss) (113,278) (39,877) (300,319) (288,720) (208,548)
Condensed Parent Company Statements of Comprehensive Income |
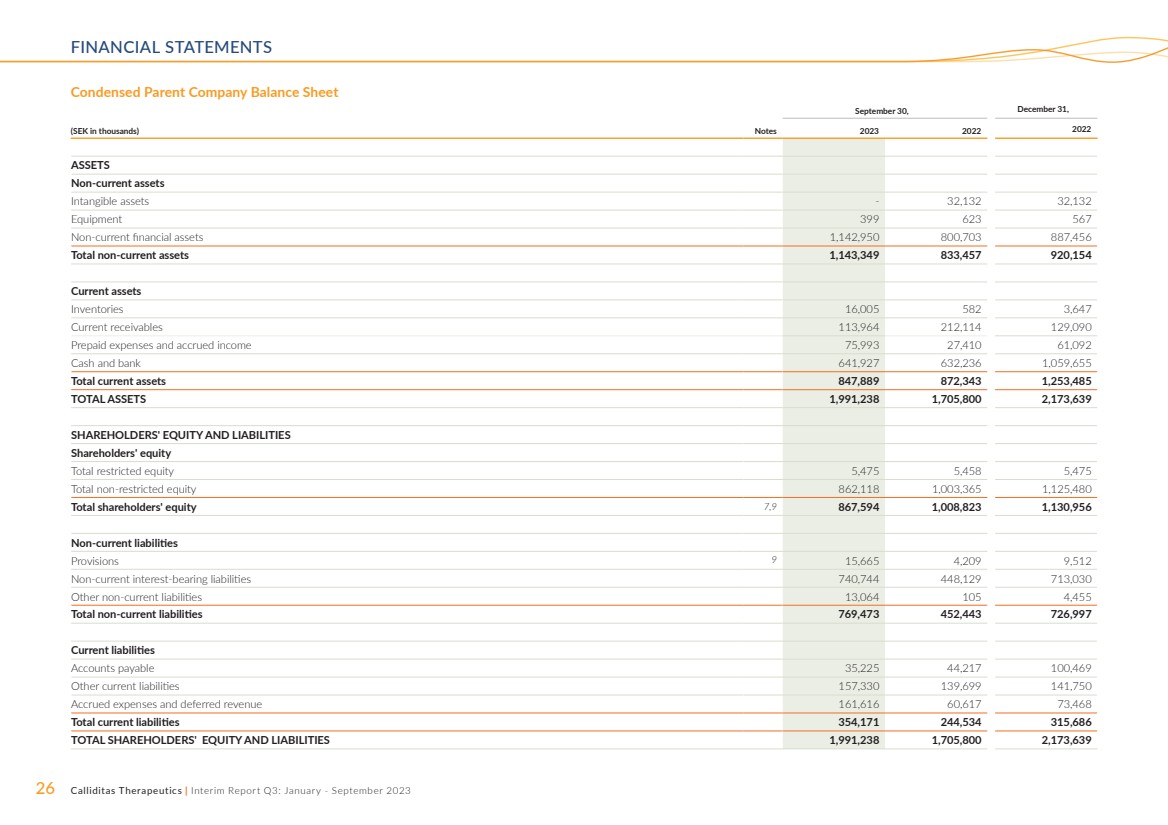
| 26 Calliditas Therapeutics | Interim Report Q3: January - September 2023
FINANCIAL STATEMENTS
Condensed Parent Company Balance Sheet
September 30, December 31,
(SEK in thousands) Notes 2023 2022 2022
ASSETS
Non-current assets
Intangible assets - 32,132 32,132
Equipment 399 623 567
Non-current financial assets 1,142,950 800,703 887,456
Total non-current assets 1,143,349 833,457 920,154
Current assets
Inventories 16,005 582 3,647
Current receivables 113,964 212,114 129,090
Prepaid expenses and accrued income 75,993 27,410 61,092
Cash and bank 641,927 632,236 1,059,655
Total current assets 847,889 872,343 1,253,485
TOTAL ASSETS 1,991,238 1,705,800 2,173,639
SHAREHOLDERS' EQUITY AND LIABILITIES
Shareholders' equity
Total restricted equity 5,475 5,458 5,475
Total non-restricted equity 862,118 1,003,365 1,125,480
Total shareholders' equity 7,9 867,594 1,008,823 1,130,956
Non-current liabilities
Provisions 9 15,665 4,209 9,512
Non-current interest-bearing liabilities 740,744 448,129 713,030
Other non-current liabilities 13,064 105 4,455
Total non-current liabilities 769,473 452,443 726,997
Current liabilities
Accounts payable 35,225 44,217 100,469
Other current liabilities 157,330 139,699 141,750
Accrued expenses and deferred revenue 161,616 60,617 73,468
Total current liabilities 354,171 244,534 315,686
TOTAL SHAREHOLDERS' EQUITY AND LIABILITIES 1,991,238 1,705,800 2,173,639 |

| 27 Calliditas Therapeutics | Interim Report Q3: January - September 2023
NOTES
Note 1 - Description of Business
Calliditas Therapeutics AB (publ) (“Calliditas” or the “Parent Company”), with corporate registration
number 556659-9766, and its subsidiaries (collectively, the “Group”) conducts commercial and
development activities in pharmaceuticals. These interim condensed consolidated financial state-ments encompass the Group, domiciled in Stockholm, Sweden, and its subsidiaries for the nine
months ended September 30, 2023 and 2022.
Calliditas is a Swedish public limited company registered in and with its registered office in
Stockholm. The registered address of the corporate headquarters is Kungsbron 1, D5, Stockholm,
Sweden. Calliditas is listed at Nasdaq Stockholm in the Mid Cap segment with ticker “CALTX” and,
in the form of ADSs, on the Nasdaq Global Select Market in the United States with the ticker “CALT”.
These interim condensed consolidated financial statements were approved by the Board of
Directors (the “Board”) for publication on November 7, 2023.
This report may include forward-looking statements. Actual outcomes may deviate from what has
been stated. Internal factors such as successful management of research projects, and intellectual
property rights may affect future results. There are also external conditions, (e.g. the economic
climate, political changes, and competing research projects) that may affect the Group’s results.
Note 2 - Accounting Policies
These interim condensed consolidated financial statements have been prepared in accordance
with International Accounting Standard No. 34 (IAS 34), “Interim Financial Reporting”. The Parent
Company applies the Swedish Financial Reporting Board recommendation RFR2, Accounting
for legal entities. The accounting policies adopted in the preparation of the interim condensed
consolidated financial statements are consistent with those followed in the preparation of the
Annual Report for 2022. None of the new or amended standards and interpretations that became
effective January 1, 2023, have had a significant impact on the Group’s financial reporting.
Significant accounting principles can be found on pages 49-54 of the Annual Report for 2022.
The ESMA (European Securities and Markets Authority) guidelines on alternative key perform-ance ratios are applied, which means disclosure requirements regarding financial measures that
are not defined in accordance with IFRS. For key ratios not defined by IFRS, see the Definitions
and reconciliations of alternative performance measures on page 31.
Note 3 - Risks and Uncertainties in the Group and the Parent Company
Operational Risks
Research and drug development up to approved registration is subject to considerable risk and
is a capital-intensive process. The majority of all initiated projects will never reach market regist-ration due to the technological risks, such as a failure to demonstrate efficacy or a favorable risk/
benefit profile, or manufacturing problems. Competing pharmaceuticals can capture market share
or reach the market faster, or if competing research projects achieve better product profiles, the
future value of the product portfolio may be lower than expected. The operations may also be
impacted negatively by regulatory decisions, such as lack of approvals and price changes.
Calliditas has a commercialized product, which has been approved under accelerated approval in
the US. under the brand name TARPEYO and has received conditional marketing authorization
in the EU and the UK under the brand name Kinpeygo. There is a risk that commercialization will
not go according to plan or that the uptake of prescribing physicians will be worse than planned or
that the drug will not have sufficient effect or show unwanted side effects, which may affect the
sales negatively. The impact on the financial statements is described in the Financial overview in
the section Research and Development Expenses.
Financial Risks
Calliditas’ financial policy governing the management of financial risks has been designed by the
Board of Directors and represents the framework of guidelines and rules in the form of risk
mandated and limits for financial activities. The Group is primarily affected by foreign exchange
risk, since the development costs for Nefecon and setanaxib are mainly paid in USD and EUR.
Further, the Group holds account receivables in USD and cash in USD and EUR to meet future
expected costs in USD and EUR in connection with commercialization of TARPEYO in the US and
the clinical development programs. Regarding the Group and the Parent Company’s financial risk
management, the risks are essentially unchanged compared with the description in the Annual
Report for 2022.
For more information and full disclosure regarding the operational and financial risks, reference
is made to the Annual Report for 2022 and the Annual Report on Form 20-F, filed with the SEC
in April 2023.
Notes to Condensed Consolidated Financial Statements |

| 28 Calliditas Therapeutics | Interim Report Q3: January - September 2023
NOTES
Note 4 - Revenue from Contracts with Customers
Three Months Ended
September 30,
Nine Months Ended
September 30,
Year Ended
December 31,
(SEK in thousands) 2023 2022 2023 2022 2022
Type of goods or services
Product sales 284,992 125,045 732,563 206,634 375,515
Outlicensing of product - 135,011 - 163,816 421,689
Royalty income 9,600 - 22,764 - 2,287
Performance of certain regulatory
services
- - - 3,387 3,387
Total 294,592 260,056 755,327 373,837 802,879
Geographical markets
USA 283,591 123,400 728,522 204,989 372,247
Europe 11,001 136,656 26,805 140,044 143,955
Asia - - - 28,804 286,677
Total 294,592 260,056 755,327 373,837 802,879
Net sales for the periods primarily originate from net sales of TARPEYO in the US, which amounted
to SEK 283.6 million and SEK 123.4 million for the three months ended September 30, 2023 and
2022, respectively. For the nine months ended September 30, 2023 and 2022, net sales from
TARPEYO amounted to SEK 728.5 million and SEK 205.0 million, respectively.
Royalty income from our partnership in Europe amounted to SEK 9.6 (-) million for the third
quarter of 2023 and SEK 22.8 (-) million for the first nine months of 2023. For the three and nine
months ended September 30, 2023, no milestones were recognized. For the three months ended
September 30, 2022, net sales amounted to SEK 135.0 million and consisted of milestone fees
from the commercialization of Kinpeygo in Europe. Further, for the nine months ended September
30, 2022 the milestone fee amounted to SEK 163.8 million and included milestone fee regarding
Everst Medicines.
The total liability for expected returns and rebates amounts to SEK 44.1 million and SEK 17.2
million as of September 30, 2023 and 2022, respectively, which are recognized in other current
liabilities and accrued expenses and deferred revenue.
Note 5 - Related-Party Transactions
During the reporting period, no significant related-party transactions have occurred.
For information about incentive programs please see Note 9.
Note 6 - Financial Instruments
The Group’s financial assets comprise of non-current financial assets, current receivables and cash,
which are recognized at amortized cost. The Group’s financial liabilities comprise of contingent
consideration, non-current interest-bearing liabilities, other non-current liabilities, lease liabilities,
accounts payable, other current liabilities, and accrued expenses, all of which except contingent
consideration, are recognized at amortized cost. The carrying amount is an approximation of the
fair value.
Contingent consideration are recognized at fair value, measured at Level 3 of the IFRS value
hierarchy. The contingent consideration has been computed in accordance with the present value
method and the probability has been taken into account if and when the various milestones will
occur. The calculations are based on a discount rate of 12.0 percent. The most significant input
affecting the valuation of the contingent consideration is the Group’s estimate of the probability
of the milestones being reached. For the three months ended September 30, 2023 and 2022, the
profit/(loss) for the period amounted to (SEK 1.8 million) and (SEK 1.4 million), respectively and for
the nine months ended September 30, 2023 and 2022, the affecting profit/(loss) for the period
amounted to SEK 8.8 million and (SEK 4.1 million), respectively, which are recognized in other
operating income/(expenses), net. This was attributable to the change of study design for the PBC
project within the setanaxib platform. For more information see the Annual Report for 2022.
Note 7 - Treasury Shares
As of September 30, 2023, Calliditas had 5,908,018 ordinary shares held as treasury shares by
the Parent Company. At the Annual General Meeting 2023, authorization was given that Calliditas
can transfer (sale) these ordinary shares with the purpose to finance an acquisition of operations,
to procure capital to finance the development of projects, repayment of loans or to commercialize
Calliditas’ products. No transfer (sale) of treasury shares have occurred as of September 30, 2023.
The total number of issued shares as of September 30, 2023, is presented in Note 8. |
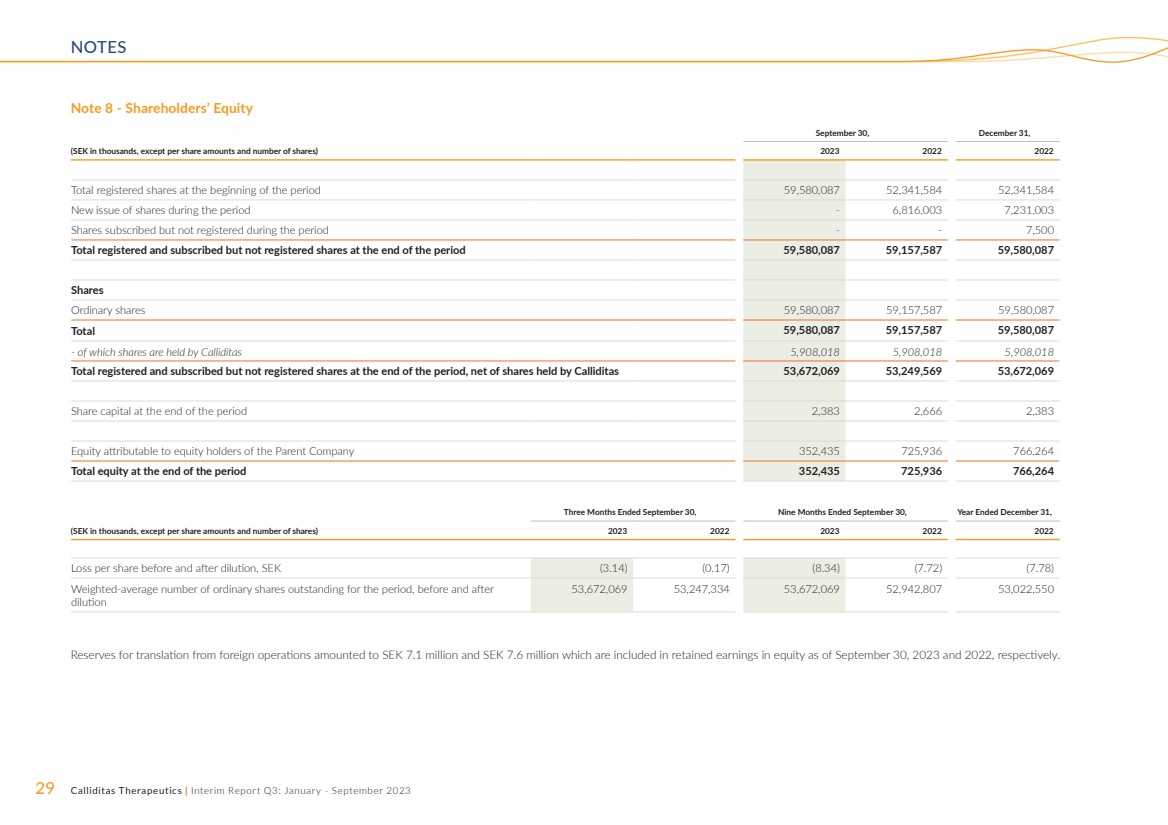
| 29 Calliditas Therapeutics | Interim Report Q3: January - September 2023
NOTES
Note 8 - Shareholders’ Equity
September 30, December 31,
(SEK in thousands, except per share amounts and number of shares) 2023 2022 2022
Total registered shares at the beginning of the period 59,580,087 52,341,584 52,341,584
New issue of shares during the period - 6,816,003 7,231,003
Shares subscribed but not registered during the period - - 7,500
Total registered and subscribed but not registered shares at the end of the period 59,580,087 59,157,587 59,580,087
Shares
Ordinary shares 59,580,087 59,157,587 59,580,087
Total 59,580,087 59,157,587 59,580,087
- of which shares are held by Calliditas 5,908,018 5,908,018 5,908,018
Total registered and subscribed but not registered shares at the end of the period, net of shares held by Calliditas 53,672,069 53,249,569 53,672,069
Share capital at the end of the period 2,383 2,666 2,383
Equity attributable to equity holders of the Parent Company 352,435 725,936 766,264
Total equity at the end of the period 352,435 725,936 766,264
Three Months Ended September 30, Nine Months Ended September 30, Year Ended December 31,
(SEK in thousands, except per share amounts and number of shares) 2023 2022 2023 2022 2022
Loss per share before and after dilution, SEK (3.14) (0.17) (8.34) (7.72) (7.78)
Weighted-average number of ordinary shares outstanding for the period, before and after
dilution
53,672,069 53,247,334 53,672,069 52,942,807 53,022,550
Reserves for translation from foreign operations amounted to SEK 7.1 million and SEK 7.6 million which are included in retained earnings in equity as of September 30, 2023 and 2022, respectively. |
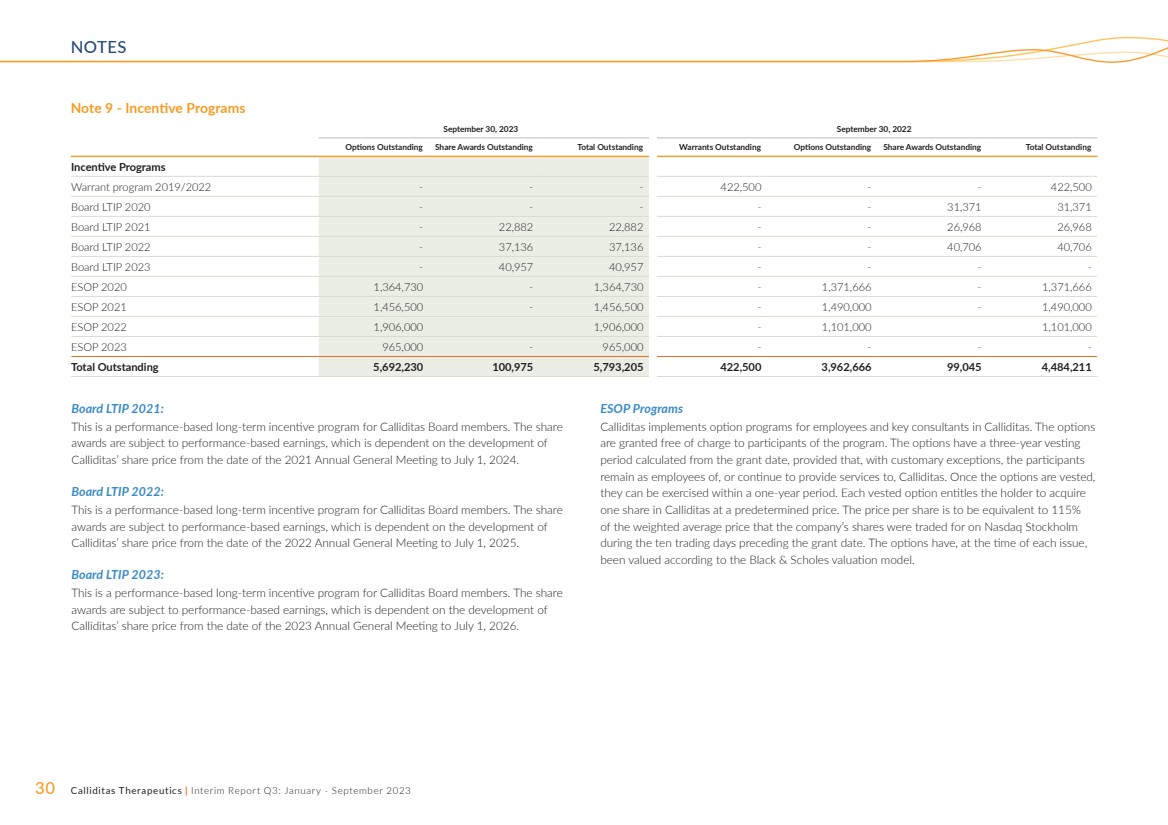
| 30 Calliditas Therapeutics | Interim Report Q3: January - September 2023
NOTES
Note 9 - Incentive Programs
Board LTIP 2021:
This is a performance-based long-term incentive program for Calliditas Board members. The share
awards are subject to performance-based earnings, which is dependent on the development of
Calliditas’ share price from the date of the 2021 Annual General Meeting to July 1, 2024.
Board LTIP 2022:
This is a performance-based long-term incentive program for Calliditas Board members. The share
awards are subject to performance-based earnings, which is dependent on the development of
Calliditas’ share price from the date of the 2022 Annual General Meeting to July 1, 2025.
Board LTIP 2023:
This is a performance-based long-term incentive program for Calliditas Board members. The share
awards are subject to performance-based earnings, which is dependent on the development of
Calliditas’ share price from the date of the 2023 Annual General Meeting to July 1, 2026.
ESOP Programs
Calliditas implements option programs for employees and key consultants in Calliditas. The options
are granted free of charge to participants of the program. The options have a three-year vesting
period calculated from the grant date, provided that, with customary exceptions, the participants
remain as employees of, or continue to provide services to, Calliditas. Once the options are vested,
they can be exercised within a one-year period. Each vested option entitles the holder to acquire
one share in Calliditas at a predetermined price. The price per share is to be equivalent to 115%
of the weighted average price that the company’s shares were traded for on Nasdaq Stockholm
during the ten trading days preceding the grant date. The options have, at the time of each issue,
been valued according to the Black & Scholes valuation model.
September 30, 2023 September 30, 2022
Options Outstanding Share Awards Outstanding Total Outstanding Warrants Outstanding Options Outstanding Share Awards Outstanding Total Outstanding
Incentive Programs
Warrant program 2019/2022 - - - 422,500 - - 422,500
Board LTIP 2020 - - - - - 31,371 31,371
Board LTIP 2021 - 22,882 22,882 - - 26,968 26,968
Board LTIP 2022 - 37,136 37,136 - - 40,706 40,706
Board LTIP 2023 - 40,957 40,957 - - - -
ESOP 2020 1,364,730 - 1,364,730 - 1,371,666 - 1,371,666
ESOP 2021 1,456,500 - 1,456,500 - 1,490,000 - 1,490,000
ESOP 2022 1,906,000 1,906,000 - 1,101,000 1,101,000
ESOP 2023 965,000 - 965,000 - - - -
Total Outstanding 5,692,230 100,975 5,793,205 422,500 3,962,666 99,045 4,484,211 |

| 31 Calliditas Therapeutics | Interim Report Q3: January - September 2023
NOTES
Definitions and Reconciliations of Alternative Performance Measures
Definitions of Alternative Performance Measures
Alternative Key Performance Indicator Definitions Reason for Inclusion
Equity ratio at the end of the period in % The ratio at the end of respective period is calculated by dividing
total shareholders’ equity by total assets.
The equity ratio measures the proportion of the total assets that are
financed by shareholders.
Reconciliations of Alternative Performance Measures
September 30, December 31,
(SEK in thousands or otherwise indicated) 2023 2022 2022
Equity ratio at the end of the period in %
Total shareholders' equity at the end of the period 352,435 725,936 766,264
Total assets at the end of the period 1,611,891 1,518,106 1,952,973
Equity ratio at the end of the period in % 22% 48% 39% |
Exhibit 99.2

| Stockholm, Sweden | |
November 7, 2023 |
Calliditas Interim Report January –
September 2023
The Lancet publication of full Phase
3 data set
In August we
were excited to see the full data set from our Phase 3 clinical trial, NefIgArd, published in The Lancet and we are looking forward to
a potential full approval of TARPEYO for treatment of IgA Nephropathy, for which the PDUFA date is December 20, 2023.
Financial Summary of Q3 2023
July 1 – September 30, 2023
| - | Net sales amounted to SEK 294.6 million,
of which TARPEYO® net sales amounted to SEK 283.6 million, for the three months
ended September 30, 2023. For the three months ended September 30, 2022, net sales
amounted to SEK 260.1 million, of which TARPEYO® net sales amounted to SEK
123.4 million. |
| - | Operating loss amounted to SEK 159.6 million and SEK 36.2 million for the three months ended September 30,
2023, and 2022, respectively. |
| - | Loss per share before and after dilution amounted to SEK 3.14 and SEK 0.17 for the three months ended
September 30, 2023, and 2022, respectively. |
| - | Cash amounted to SEK 786.9 million and SEK 736.2 million as of September 30, 2023, and 2022, respectively. |
January 1 - September 30, 2023
| - | Net sales amounted to SEK 755.3 million, of which TARPEYO net sales amounted to SEK 728.5 million, for
the nine months ended September 30, 2023. For the nine months ended September 30, 2022, net sales amounted to SEK 373.8 million,
of which TARPEYO net sales amounted to SEK 205.0 million. |
| - | Operating loss amounted to SEK 414.8 million and SEK 454.4 million for the nine months ended September 30,
2023, and 2022, respectively. |
| - | Loss per share before and after dilution amounted to SEK 8.34 and SEK 7.72 for the nine months ended September 30,
2023, and 2022, respectively. |
“The recent ASN meeting in
Philadelphia provided us with numerous opportunities to meet and engage with nephrologists across the country, further cementing our
belief that the strong long-term data from our Phase 3 trial, in combination with a potential full approval, will significantly impact
how nephrologists view and use the product going forward.
We reconfirm
our guidance for 2023 of USD 100 – 120m of net sales from TARPEYO and look forward to an exciting 2024.” Renee
Aguiar-Lucander, CEO

Significant Events in Q3 2023, in Summary
| · | On July 13 Calliditas announced supportive interim data from Phase 2 head and neck cancer trial with
lead NOX inhibitor candidate, setanaxib. |
| · | On August 15 Calliditas announced full results from the NefIgArd Phase 3 trial published in The Lancet. |
| · | On
August 18 FDA granted priority review for full approval of TARPEYO® for
the treatment of IgA Nephropathy. |
| · | On September 27 Calliditas granted orphan drug designation by the FDA for the treatment of Alport
syndrome with setanaxib. |
| · | On
September 28 STADA and Calliditas announced the filing for full marketing authorization
of Kinpeygo® in the EU. |
| · | On September 29 Calliditas presented Data from the NefIgArd Phase 3 trial at the 17th International
Symposium on IgA Nephropathy (IIgANN) Tokyo 2023. |
Investor Presentation
Calliditas invites investors, analysts and press
to a presentation of the Quarterly Report 2023 at 14:30 pm. on November 7. Calliditas’ CEO Renee Aguiar-Lucander will present
the report together with CFO Fredrik Johansson, CMO Richard Philipson and President North America Andrew Udell. The presentation will
be given in English.
| · | Time: Tuesday 14:30 pm CET. on November 7 |
| · | Link
to webcast https://financialhearings.com/event/46414 |
| · | To participate via conference call register via this link: https://conference.financialhearings.com/teleconference/?id=2001082 |
After registration, you will receive a phone number and a conference
ID to log in to the conference call. Via the telephone conference, there is an opportunity to ask oral questions.
For further information, please contact:
Åsa Hillsten, Head of Investor Relations & Sustainability,
Calliditas
Phone.: +46 764 03 35 43, E mail:
asa.hillsten@calliditas.com
The information in the press release is information
that Calliditas is obliged to make public pursuant to the EU Market Abuse Regulation. The information was sent for publication, through
the agency of the contact person set out above, on November 7, 2023, at 7:00 a.m. CET.
About Calliditas
Calliditas Therapeutics is a commercial
stage biopharma company based in Stockholm, Sweden focused on identifying, developing, and commercializing novel treatments in
orphan indications, with an initial focus on renal and hepatic diseases with significant unmet medical needs. Calliditas’ lead
product, developed under the name Nefecon, has been granted accelerated approval by the FDA under the trade name
TARPEYO® and conditional marketing authorization by the European Commission under the trade name
Kinpeygo®. Kinpeygo is being commercialized in the European Union Member States by Calliditas’ partner, STADA
Arzneimittel AG. Additionally, Calliditas is conducting a Phase 2b clinical trial in primary biliary cholangitis and a Phase 2
proof-of-concept trial in head and neck cancer with its NOX inhibitor product candidate, setanaxib. Calliditas’ common shares
are listed on Nasdaq Stockholm (ticker: CALTX) and its American Depositary Shares are listed on the Nasdaq Global Select Market
(ticker: CALT).

Forward-Looking Statements
This press release contains forward-looking
statements within the meaning of the Private Securities Litigation Reform Act of 1995, as amended, including, without limitation,
statements regarding Calliditas’ strategy, commercialization efforts, business plans, regulatory submissions, clinical
development plans, revenue and product sales projections or forecasts and focus. The words “may,” “will,”
“could,” “would,” “should,” “expect,” “plan,” “anticipate,”
“intend,” “believe,” “estimate,” “predict,” “project,”
“potential,” “continue,” “target,” and similar expressions are intended to identify
forward-looking statements, although not all forward-looking statements contain these identifying words. Any forward-looking
statements in this press release are based on management’s current expectations and beliefs and are subject to a number of
risks, uncertainties, and important factors that may cause actual events or results to differ materially from those expressed or
implied by any forward-looking statements contained in this press release, including, without limitation, any related to
Calliditas’ business, operations, continued and additional regulatory approvals for TARPEYO and Kinpeygo, market acceptance of
TARPEYO and Kinpeygo, clinical trials, supply chain, strategy, goals and anticipated timelines, competition from other
biopharmaceutical companies, revenue and product sales projections or forecasts, including 2023 revenue guidance, and other risks
identified in the section entitled “Risk Factors” in Calliditas’ reports filed with the Securities and Exchange
Commission. Calliditas cautions you not to place undue reliance on any forward-looking statements, which speak only as of the date
they are made. Calliditas disclaims any obligation to publicly update or revise any such statements to reflect any change in
expectations or in events, conditions, or circumstances on which any such statements may be based, or that may affect the likelihood
that actual results will differ from those set forth in the forward-looking statements. Any forward-looking statements contained in
this press release represent Calliditas’ views only as of the date hereof and should not be relied upon as representing its
views as of any subsequent date.
Calliditas Therapeutics AB (PK) (USOTC:CLTEF)
過去 株価チャート
から 6 2024 まで 7 2024

Calliditas Therapeutics AB (PK) (USOTC:CLTEF)
過去 株価チャート
から 7 2023 まで 7 2024
