Form 8-K - Current report
2024年1月8日 - 9:01PM
Edgar (US Regulatory)
false
0001429560
0001429560
2024-01-08
2024-01-08
iso4217:USD
xbrli:shares
iso4217:USD
xbrli:shares
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported):
January 8, 2024
TREVENA, INC.
(Exact name of registrant as specified in
its charter)
Delaware
(State or other jurisdiction of incorporation)
| 001-36193 |
|
26-1469215 |
(Commission
File No.) |
|
(IRS Employer
Identification No.) |
955 Chesterbrook Boulevard, Suite 110
Chesterbrook, PA 19087
(Address of principal executive offices and zip
code)
Registrant’s telephone number, including
area code: (610) 354-8840
Not applicable
(Former name or former address, if changed
since last report.)
Check the appropriate box below if the Form 8-K filing is intended
to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| ¨ | Written communications pursuant to Rule 425 under the
Securities Act (17 CFR 230.425) |
| ¨ | Soliciting material pursuant to Rule 14a-12 under the
Exchange Act (17 CFR 240.14a-12) |
| ¨ | Pre-commencement communications pursuant to Rule 14d-2(b)
under the Exchange Act (17 CFR 240.14d-2(b)) |
| ¨ | Pre-commencement communications pursuant to Rule 13e-4(c)
under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section
12(b) of the Act:
| Title of each class | |
Trading Symbol(s) | |
Name of each exchange on which registered |
| Common Stock, $0.001 par value | |
TRVN | |
The Nasdaq Stock Market LLC |
Indicate
by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405
of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company
¨
If
an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for
complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ¨
| Item 7.01 |
Regulation FD Disclosure |
On January 8, 2024,
Trevena, Inc. (the “Company”) updated its website to include an updated corporate presentation deck. A copy of the updated
corporate deck is attached hereto as Exhibit 99.1.
The information set forth
on this Item 7.01 and furnished hereto as Exhibits 99.1 shall not be deemed “filed” for purposes of Section 18 of the
Securities Exchange Act of 1934, as amended (the “Exchange Act”), and is not incorporated by reference into any of the Company’s
filings under the Securities Act of 1933, as amended (the “Securities Act”) or the Exchange Act, whether made before or after
the date hereof, except as shall be expressly set forth by specific reference in any such filing.
| Item 9.01. | Financial Statements and Exhibits. |
SIGNATURES
Pursuant to the requirements of the Exchange Act,
the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| |
TREVENA, INC. |
| |
|
|
| Date: January 8, 2024 |
By: |
/s/ Barry Shin |
| |
|
Barry Shin |
| |
|
Senior Vice President & Chief Financial
Officer |
Exhibit 99.1

| INNOVATING FOR PATIENTS
Nasdaq: TRVN I January 2024 |

| Forward-Looking Statements
To the extent that statements contained in this presentation are not descriptions of historical facts regarding Trevena, Inc. (the “Company” or “we”), they are forward-looking statements
reflecting management’s current beliefs and expectations. Forward-looking statements are subject to known and unknown risks, uncertainties, and other factors that may cause our or our
industry’s actual results, levels of activity, performance, or achievements to be materially different from those anticipated by such statements. You can identify forward-looking statements by
terminology such as “anticipate,” “believe,” “estimate,” “expect,” “intend,” “may,” “might,” “plan,” “objective,” “predict,” “project,” “suggest,” “target,” “potential,” “will,” “would,” “could,” “should,”
“continue,” “ongoing,” or the negative of these terms or similar expressions. Forward-looking statements contained in this presentation include, but are not limited to, (i) statements regarding
the timing of anticipated clinical trials for our product candidates; (ii) the timing of receipt of clinical data for our product candidates; (iii) our expectations regarding the potential safety, efficacy,
or clinical utility of our product candidates; (iv) the size of patient populations targeted by our product candidates and market adoption of our potential drugs by physicians and patients; (v) the
timing or likelihood of regulatory filings and approvals; and (vi) our cash needs.
Actual results may differ materially from those indicated by such forward-looking statements as a result of various important factors, including: the commercialization of any approved drug
product, the status, timing, costs, results and interpretation of our clinical trials or any future trials of any of our investigational drug candidates; the uncertainties inherent in conducting clinical
trials; expectations for regulatory interactions, submissions and approvals, including our assessment of the discussions with the FDA or other regulatory agencies about any and all of our
programs; uncertainties related to the commercialization of OLINVYK; available funding; uncertainties related to our intellectual property; other matters that could affect the availability or
commercial potential of our therapeutic candidates; and other factors discussed in the Risk Factors set forth in our Annual Report on Form 10-K and Quarterly Reports on Form 10-Q filed with
the Securities and Exchange Commission (SEC) and in other filings we make with the SEC from time to time. In addition, the forward-looking statements included in this presentation
represent our views only as of the date hereof. We anticipate that subsequent events and developments may cause our views to change. However, while we may elect to update these
forward-looking statements at some point in the future, we specifically disclaim any obligation to do so, except as may be required by law.
2 |

| BOARD OF DIRECTORS
Carrie L. Bourdow Jake R. Nunn
Scott Braunstein, M.D.
Lead Independent Director Anne M. Phillips, M.D.
Mark Corrigan, M.D. Barbara Yanni
Marvin H. Johnson, Jr.
SENIOR MANAGEMENT
Carrie L. Bourdow Chair, President & Chief Executive Officer
Mark A. Demitrack, M.D. SVP, Chief Medical Officer
Patricia Drake SVP, Chief Commercial Officer
Barry Shin SVP, Chief Financial Officer
Robert T. Yoder SVP, Chief Business Officer & Head
of Commercial Operations
Trevena’s Experienced Leadership Team
3 |
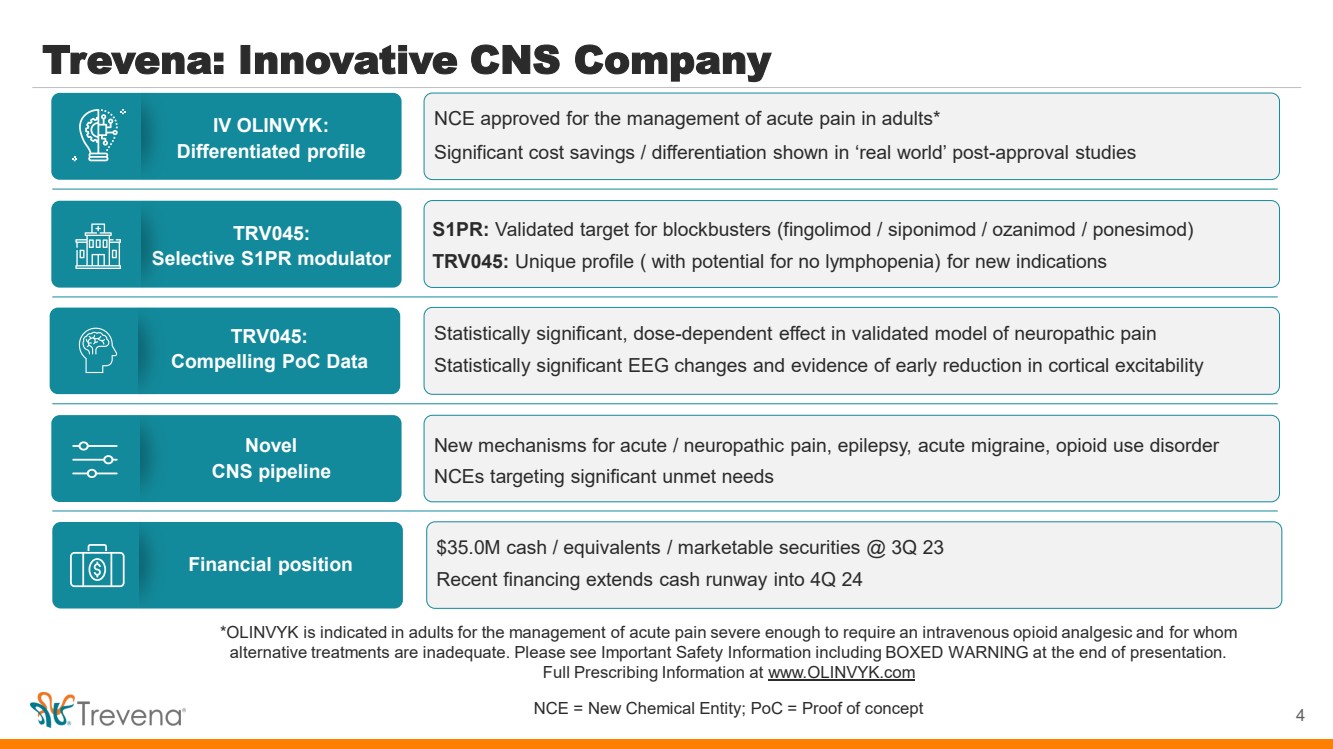
| *OLINVYK is indicated in adults for the management of acute pain severe enough to require an intravenous opioid analgesic and for whom
alternative treatments are inadequate. Please see Important Safety Information including BOXED WARNING at the end of presentation.
Full Prescribing Information at www.OLINVYK.com
NCE = New Chemical Entity; PoC = Proof of concept
Trevena: Innovative CNS Company
4
IV OLINVYK:
Differentiated profile
TRV045:
Selective S1PR modulator
Novel
CNS pipeline
TRV045:
Compelling PoC Data
Financial position
NCE approved for the management of acute pain in adults*
Significant cost savings / differentiation shown in ‘real world’ post-approval studies
S1PR: Validated target for blockbusters (fingolimod / siponimod / ozanimod / ponesimod)
TRV045: Unique profile ( with potential for no lymphopenia) for new indications
New mechanisms for acute / neuropathic pain, epilepsy, acute migraine, opioid use disorder
NCEs targeting significant unmet needs
Statistically significant, dose-dependent effect in validated model of neuropathic pain
Statistically significant EEG changes and evidence of early reduction in cortical excitability
$35.0M cash / equivalents / marketable securities @ 3Q 23
Recent financing extends cash runway into 4Q 24 |

| *OLINVYK is indicated in adults for the management of acute pain severe enough to require an intravenous opioid analgesic and for whom
alternative treatments are inadequate. Please see Important Safety Information including BOXED WARNING at the end of presentation.
Full Prescribing Information at www.OLINVYK.com
TRV045 and TRV734 are investigational products and are not approved by the FDA or any other regulatory agency
NDA = New Drug Application; PoC = Proof-of-Concept
PRE-CLIN PHASE 1 PHASE 2 PHASE 3 NDA POST-APPR Highlights
OLINVYK®
New chemical entity
(mu-opioid receptor)
TRV045
Selective S1P
receptor modulator
• Data reported
• Data reported
• Data expected 1Q 24
TRV734
G-protein selective agonist
(mu-opioid receptor)
Multiple Expected Catalysts
5
NIH / NIDA collab.
IV acute pain*
VOLITION clinical outcomes
Respiratory physiology
Cleveland Clinic / Wake Forest Baptist Health collab.
PoC – pain / target engagement
PoC - epilepsy
APPROVED
• Real world differentiation
• Data reported
• Commercial launch ongoing
• POC study ongoing
ARTEMIS clinical outcomes • $8.8k / 1.4 day savings
Opioid use disorder
Seiz. Prev NIH / ETSP investigating potential disease modifying role
Cognitive function
Cleveland Clinic / Wake Forest Baptist Health collab.
• Data reported |

| OLINVYK Overview |
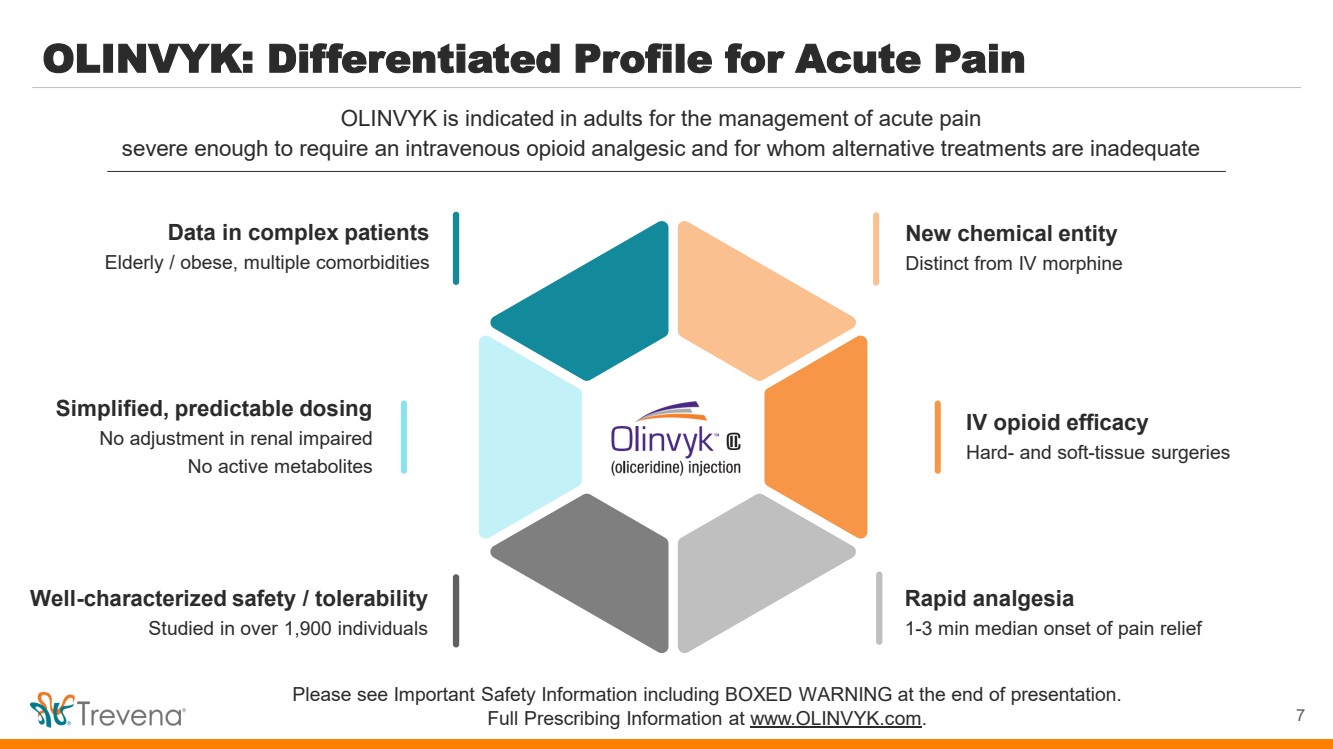
| OLINVYK: Differentiated Profile for Acute Pain
7
OLINVYK is indicated in adults for the management of acute pain
severe enough to require an intravenous opioid analgesic and for whom alternative treatments are inadequate
Please see Important Safety Information including BOXED WARNING at the end of presentation.
Full Prescribing Information at www.OLINVYK.com.
New chemical entity
Distinct from IV morphine
IV opioid efficacy
Hard- and soft-tissue surgeries
Rapid analgesia
1-3 min median onset of pain relief
Simplified, predictable dosing
No adjustment in renal impaired
No active metabolites
Data in complex patients
Elderly / obese, multiple comorbidities
Well-characterized safety / tolerability
Studied in over 1,900 individuals |
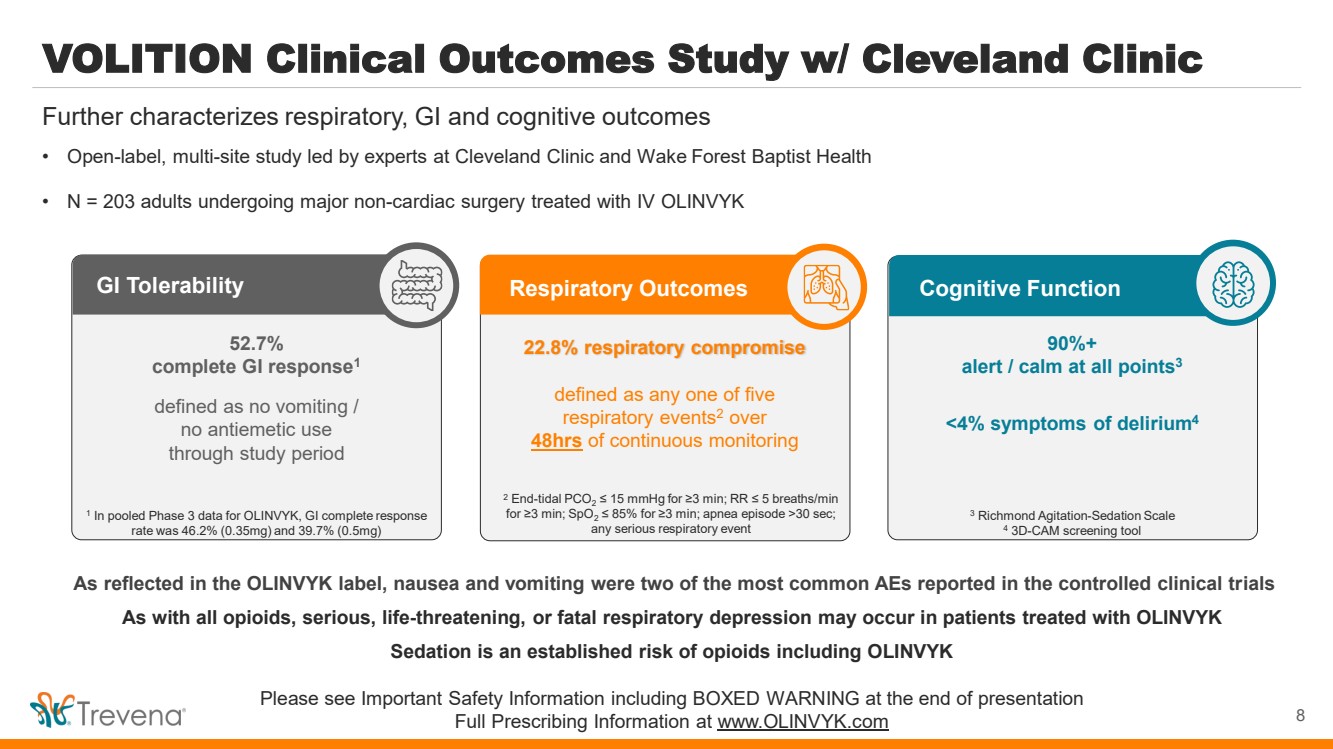
| VOLITION Clinical Outcomes Study w/ Cleveland Clinic
• Open-label, multi-site study led by experts at Cleveland Clinic and Wake Forest Baptist Health
• N = 203 adults undergoing major non-cardiac surgery treated with IV OLINVYK
Further characterizes respiratory, GI and cognitive outcomes
Cognitive Function
90%+
alert / calm at all points3
<4% symptoms of delirium4
3 Richmond Agitation-Sedation Scale
4 3D-CAM screening tool
As reflected in the OLINVYK label, nausea and vomiting were two of the most common AEs reported in the controlled clinical trials
As with all opioids, serious, life-threatening, or fatal respiratory depression may occur in patients treated with OLINVYK
Sedation is an established risk of opioids including OLINVYK
Please see Important Safety Information including BOXED WARNING at the end of presentation
Full Prescribing Information at www.OLINVYK.com
GI Tolerability
52.7%
complete GI response1
defined as no vomiting /
no antiemetic use
through study period
1
In pooled Phase 3 data for OLINVYK, GI complete response
rate was 46.2% (0.35mg) and 39.7% (0.5mg)
Respiratory Outcomes
22.8% respiratory compromise
defined as any one of five
respiratory events2 over
48hrs of continuous monitoring
2 End-tidal PCO2 ≤ 15 mmHg for ≥3 min; RR ≤ 5 breaths/min
for ≥3 min; SpO2 ≤ 85% for ≥3 min; apnea episode >30 sec;
any serious respiratory event
8 |
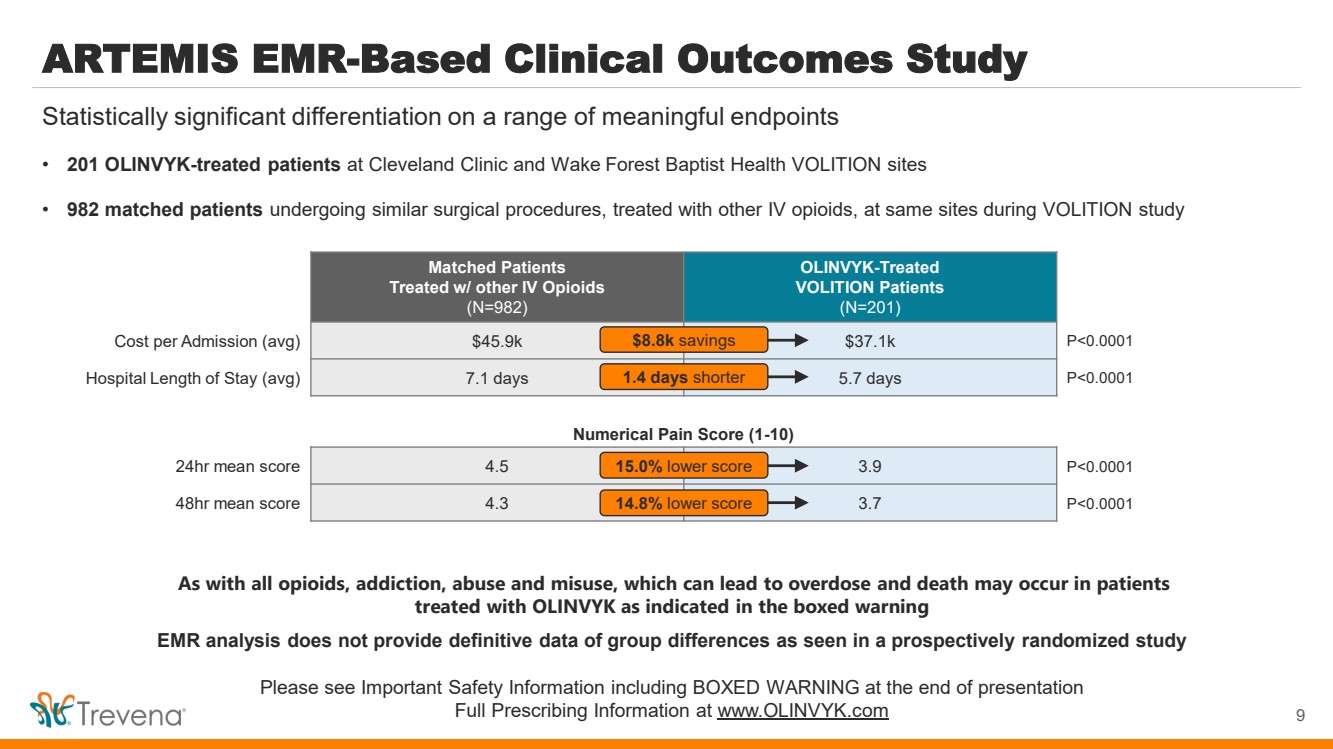
| ARTEMIS EMR-Based Clinical Outcomes Study
• 201 OLINVYK-treated patients at Cleveland Clinic and Wake Forest Baptist Health VOLITION sites
• 982 matched patients undergoing similar surgical procedures, treated with other IV opioids, at same sites during VOLITION study
9
Statistically significant differentiation on a range of meaningful endpoints
Matched Patients
Treated w/ other IV Opioids
(N=982)
OLINVYK-Treated
VOLITION Patients
(N=201)
Cost per Admission (avg) $45.9k $37.1k P<0.0001
Hospital Length of Stay (avg) 7.1 days 5.7 days P<0.0001
$8.8k savings
1.4 days shorter
24hr mean score 4.5 3.9 P<0.0001
48hr mean score 4.3 3.7 P<0.0001
15.0% lower score
14.8% lower score
Numerical Pain Score (1-10)
As with all opioids, addiction, abuse and misuse, which can lead to overdose and death may occur in patients
treated with OLINVYK as indicated in the boxed warning
EMR analysis does not provide definitive data of group differences as seen in a prospectively randomized study
Please see Important Safety Information including BOXED WARNING at the end of presentation
Full Prescribing Information at www.OLINVYK.com |
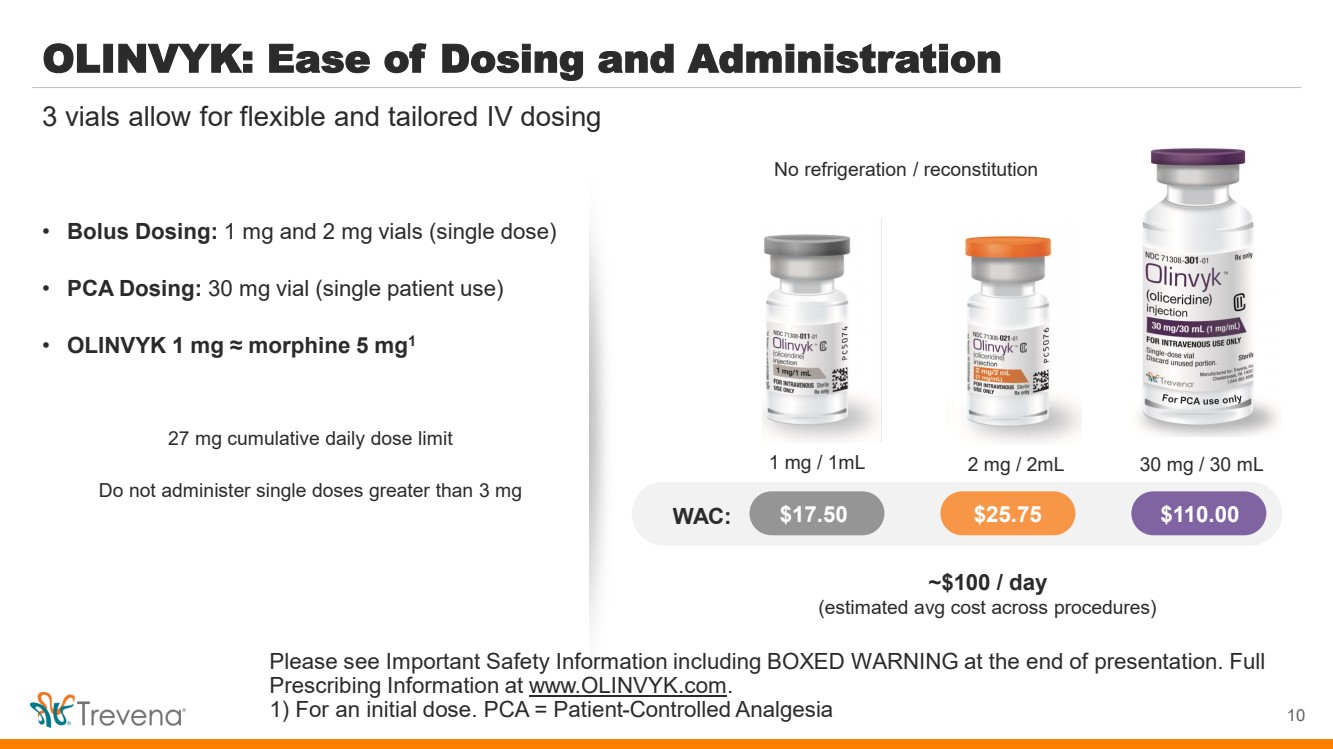
| OLINVYK: Ease of Dosing and Administration
• Bolus Dosing: 1 mg and 2 mg vials (single dose)
• PCA Dosing: 30 mg vial (single patient use)
• OLINVYK 1 mg ≈ morphine 5 mg1
27 mg cumulative daily dose limit
Do not administer single doses greater than 3 mg
10
3 vials allow for flexible and tailored IV dosing
No refrigeration / reconstitution
For PCA use only
WAC: $17.50 $25.75 $110.00
1 mg / 1mL 2 mg / 2mL 30 mg / 30 mL
~$100 / day
(estimated avg cost across procedures)
Please see Important Safety Information including BOXED WARNING at the end of presentation. Full
Prescribing Information at www.OLINVYK.com.
1) For an initial dose. PCA = Patient-Controlled Analgesia |

| TRV045
S1P Receptor Modulator
Novel MOA for Diabetic Neuropathic Pain |
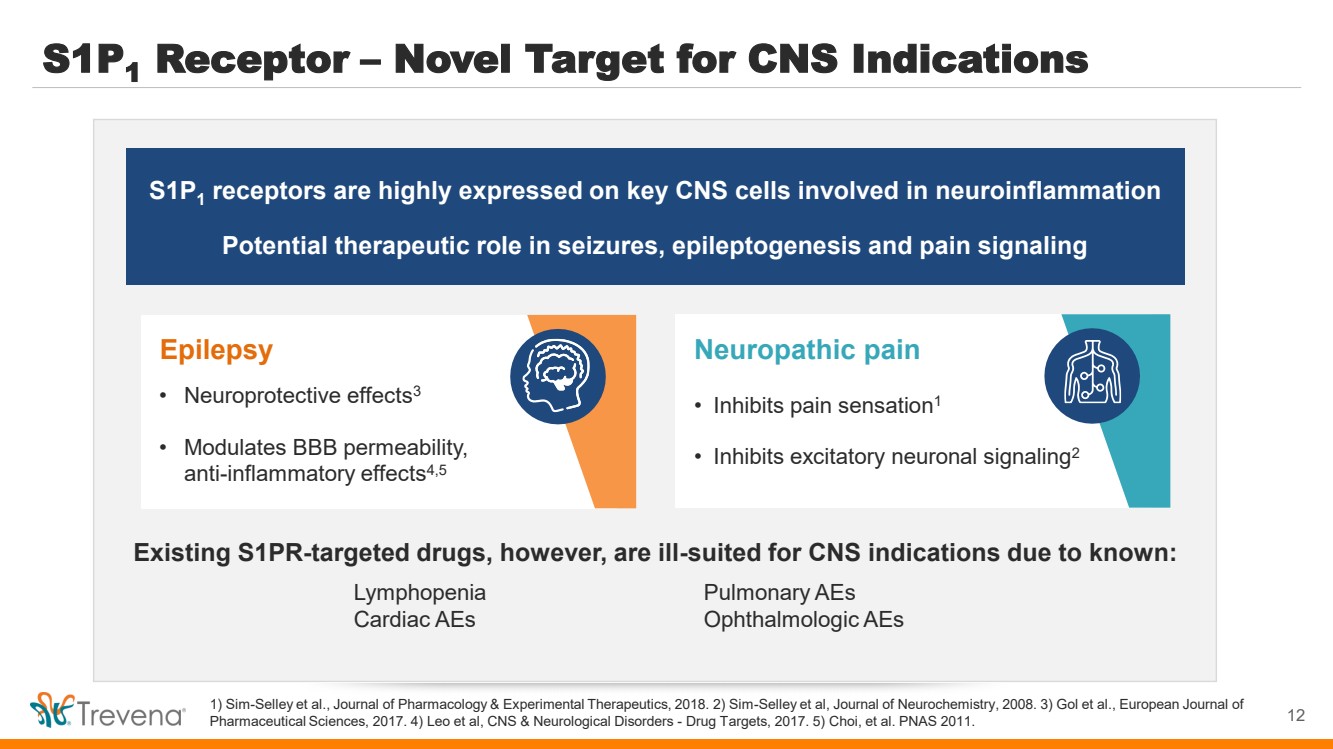
| S1P1 Receptor – Novel Target for CNS Indications
1) Sim-Selley et al., Journal of Pharmacology & Experimental Therapeutics, 2018. 2) Sim-Selley et al, Journal of Neurochemistry, 2008. 3) Gol et al., European Journal of
Pharmaceutical Sciences, 2017. 4) Leo et al, CNS & Neurological Disorders - Drug Targets, 2017. 5) Choi, et al. PNAS 2011.
S1P1
receptors are highly expressed on key CNS cells involved in neuroinflammation
Potential therapeutic role in seizures, epileptogenesis and pain signaling
Neuropathic pain
• Inhibits pain sensation1
• Inhibits excitatory neuronal signaling2
Existing S1PR-targeted drugs, however, are ill-suited for CNS indications due to known:
Lymphopenia Pulmonary AEs
Cardiac AEs Ophthalmologic AEs
Epilepsy
• Neuroprotective effects3
• Modulates BBB permeability,
anti-inflammatory effects4,5
12 |
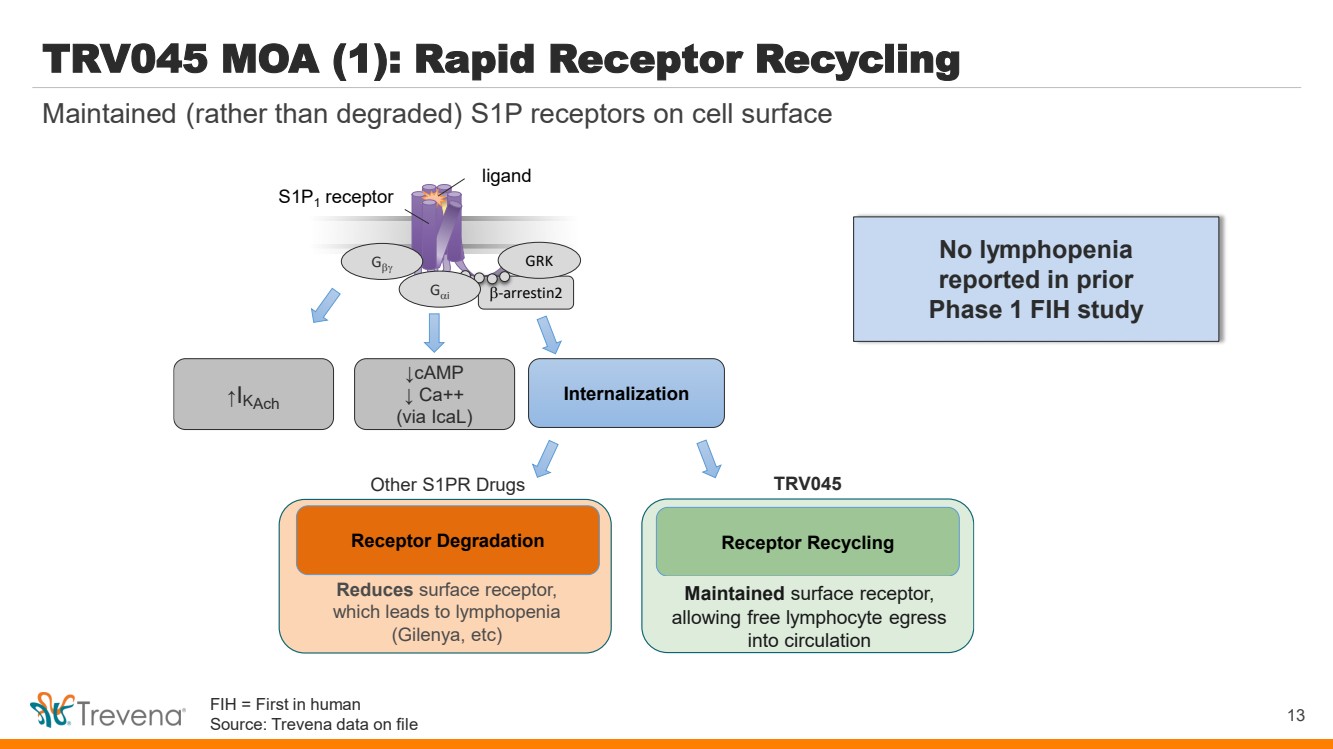
| TRV045 MOA (1): Rapid Receptor Recycling
b-arrestin2
S1P1
receptor
Gb GRK
↑I Internalization KAch
ligand
Gai
↓cAMP
↓ Ca++
(via IcaL)
Reduces surface receptor,
which leads to lymphopenia
(Gilenya, etc)
Receptor Recycling
Maintains surface receptor and
avoids lymphopenia, allowing free
lymphocyte egress into circulation
Receptor Degradation
Other S1PR Drugs TRV045 13
Maintained (rather than degraded) S1P receptors on cell surface
No lymphopenia
reported in prior
Phase 1 FIH study
Maintained surface receptor,
allowing free lymphocyte egress
into circulation
FIH = First in human
Source: Trevena data on file |
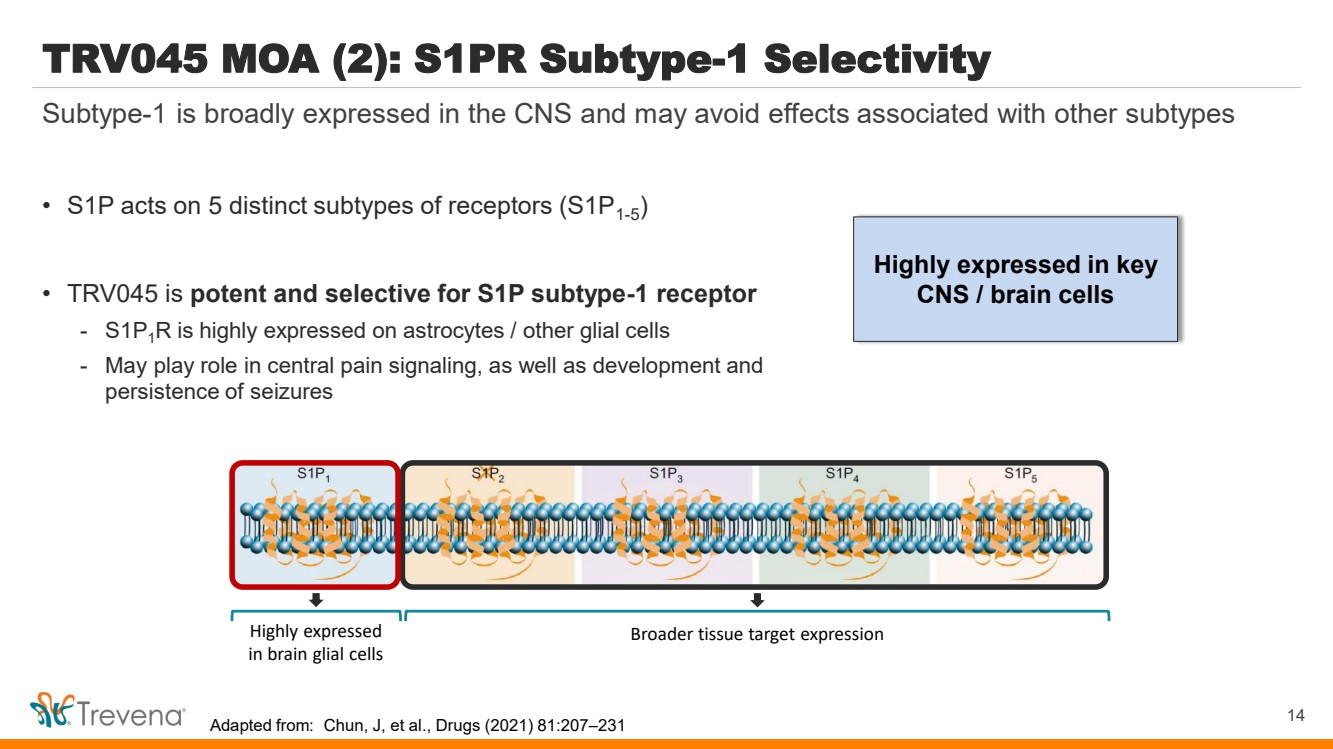
| Highly expressed in key
CNS / brain cells
TRV045 MOA (2): S1PR Subtype-1 Selectivity
• S1P acts on 5 distinct subtypes of receptors (S1P1-5
)
• TRV045 is potent and selective for S1P subtype-1 receptor
- S1P1R is highly expressed on astrocytes / other glial cells
- May play role in central pain signaling, as well as development and
persistence of seizures
14
Subtype-1 is broadly expressed in the CNS and may avoid effects associated with other subtypes
Highly expressed
in brain glial cells
Broader tissue target expression
Adapted from: Chun, J, et al., Drugs (2021) 81:207–231 |
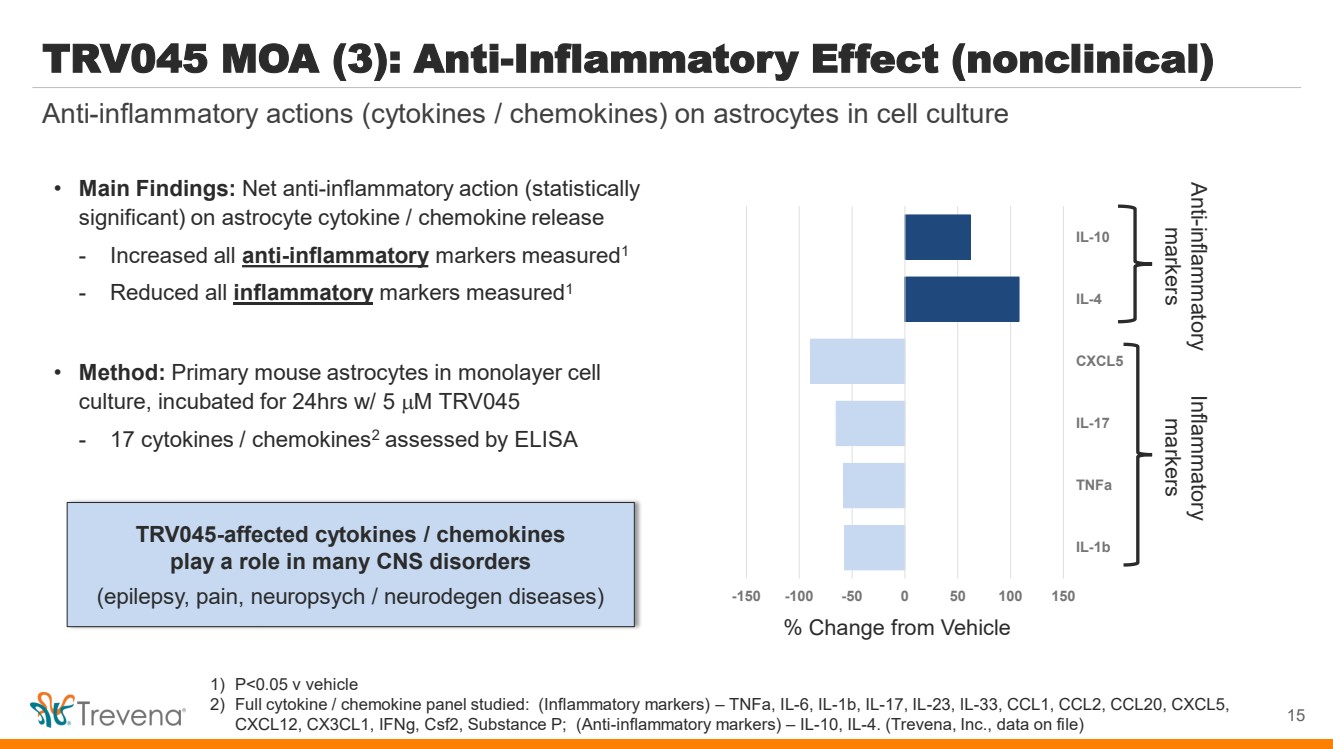
| 1) P<0.05 v vehicle
2) Full cytokine / chemokine panel studied: (Inflammatory markers) – TNFa, IL-6, IL-1b, IL-17, IL-23, IL-33, CCL1, CCL2, CCL20, CXCL5,
CXCL12, CX3CL1, IFNg, Csf2, Substance P; (Anti-inflammatory markers) – IL-10, IL-4. (Trevena, Inc., data on file)
TRV045 MOA (3): Anti-Inflammatory Effect (nonclinical)
• Main Findings: Net anti-inflammatory action (statistically
significant) on astrocyte cytokine / chemokine release
- Increased all anti-inflammatory markers measured1
- Reduced all inflammatory markers measured1
• Method: Primary mouse astrocytes in monolayer cell
culture, incubated for 24hrs w/ 5 M TRV045
- 17 cytokines / chemokines2 assessed by ELISA
TRV045-affected cytokines / chemokines
play a role in many CNS disorders
(epilepsy, pain, neuropsych / neurodegen diseases)
Anti-inflammatory actions (cytokines / chemokines) on astrocytes in cell culture
15
-150 -100 -50 0 50 100 150
IL-1b
TNFa
IL-17
CXCL5
IL-4
IL-10
% Change from Vehicle
Anti-markers inflammatory Inflammatory markers |

| TRV045 Proof-of-Concept Study Program – Highlights
• Target Engagement. Demonstrated CNS penetration and target engagement
• Neuropathic Pain. Statistically significant, dose-dependent effect in validated model of neuropathic pain
• EEG Spectral Power. Statistically significant increases in brain waves (alpha, beta, gamma) associated
with arousal, alertness, cognitive processing, learning and memory
Statistically significant decrease in delta brain waves, and no significant change in
theta brain waves, both of which are associated with sedation / sleep
• Cortical Excitability. Directional evidence of early reduction in cortical excitability
Preliminary data*
Not all of the results were statistically significant;
details of the findings are presented on the following slides
Studies were conducted outside the United States and not under the IND for TRV045 16 |
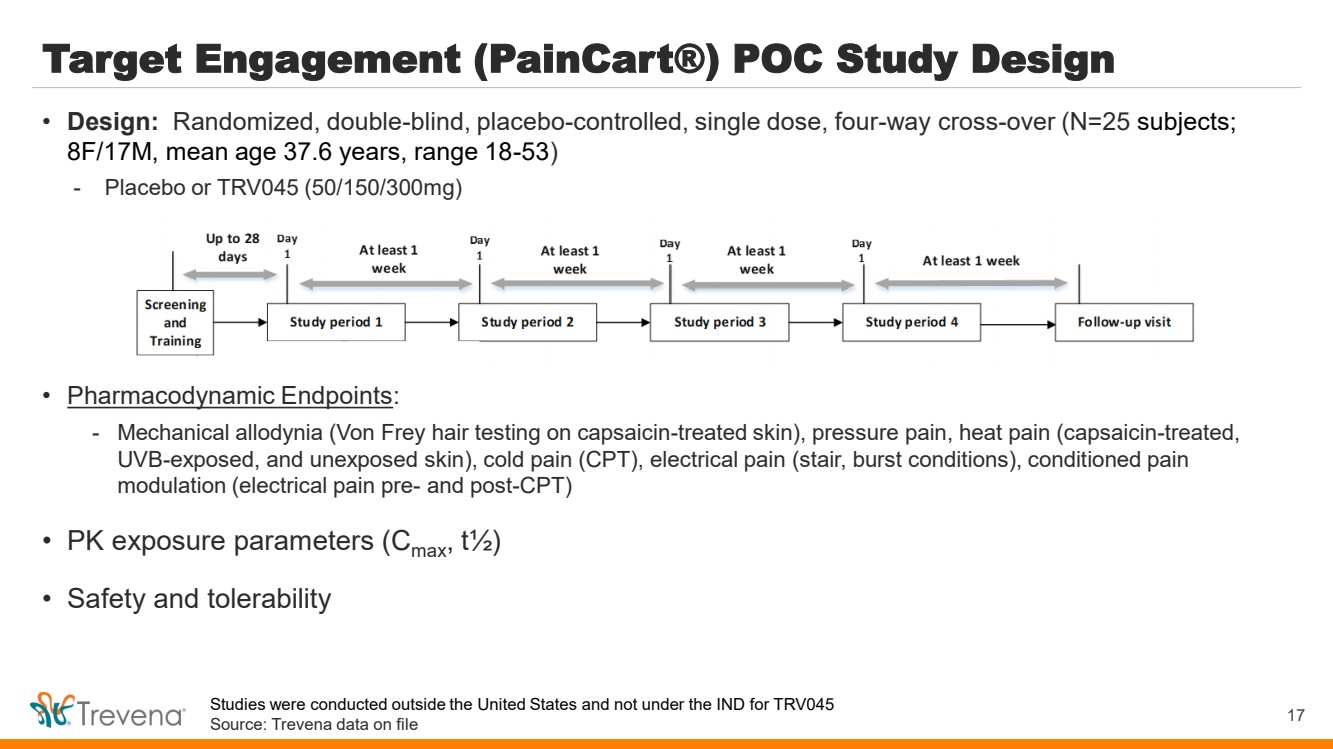
| Target Engagement (PainCart®) POC Study Design
• Pharmacodynamic Endpoints:
- Mechanical allodynia (Von Frey hair testing on capsaicin-treated skin), pressure pain, heat pain (capsaicin-treated,
UVB-exposed, and unexposed skin), cold pain (CPT), electrical pain (stair, burst conditions), conditioned pain
modulation (electrical pain pre- and post-CPT)
• PK exposure parameters (Cmax, t½)
• Safety and tolerability
17
• Design: Randomized, double-blind, placebo-controlled, single dose, four-way cross-over (N=25 subjects;
8F/17M, mean age 37.6 years, range 18-53)
- Placebo or TRV045 (50/150/300mg)
Studies were conducted outside the United States and not under the IND for TRV045
Source: Trevena data on file |
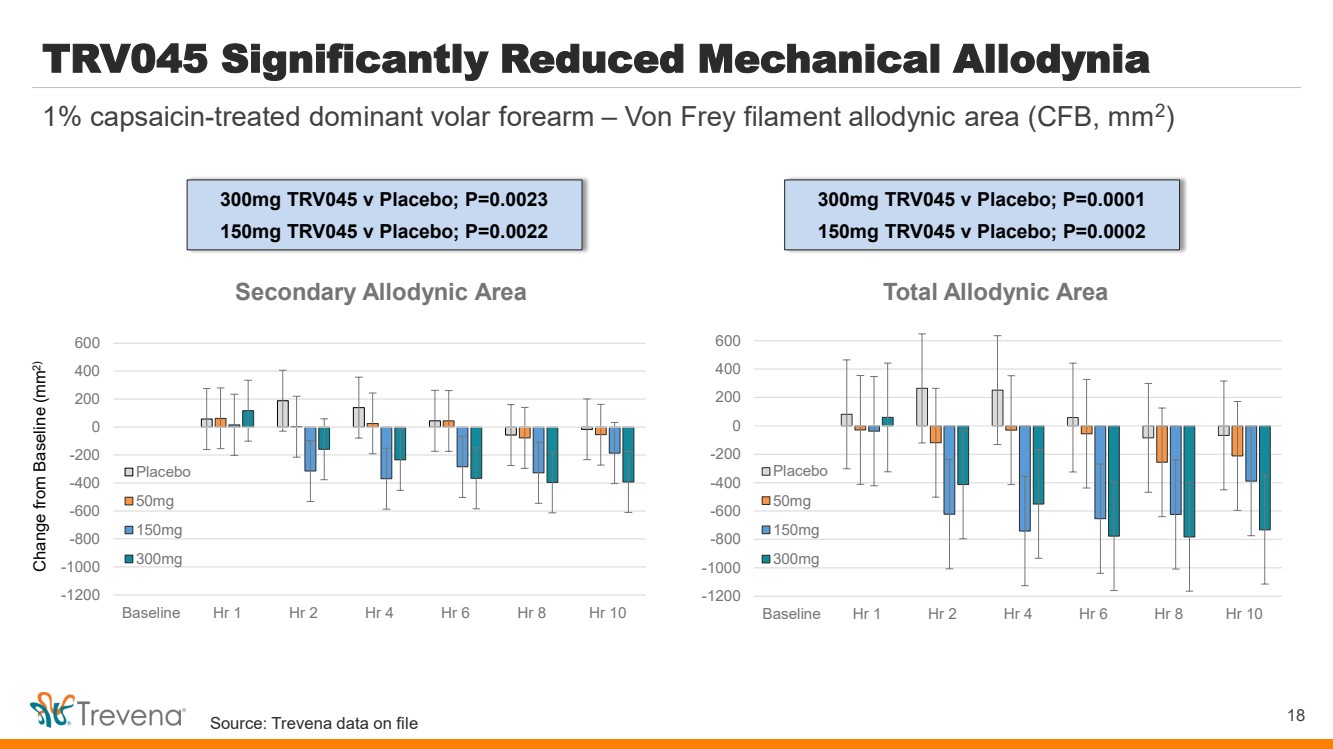
| TRV045 Significantly Reduced Mechanical Allodynia
18
1% capsaicin-treated dominant volar forearm – Von Frey filament allodynic area (CFB, mm2
)
Change from Baseline (mm2)
-1200
-1000
-800
-600
-400
-200
0
200
400
600
Baseline Hr 1 Hr 2 Hr 4 Hr 6 Hr 8 Hr 10
Total Allodynic Area
Placebo
50mg
150mg
300mg
-1200
-1000
-800
-600
-400
-200
0
200
400
600
Baseline Hr 1 Hr 2 Hr 4 Hr 6 Hr 8 Hr 10
Secondary Allodynic Area
Placebo
50mg
150mg
300mg
300mg TRV045 v Placebo; P=0.0023
150mg TRV045 v Placebo; P=0.0022
300mg TRV045 v Placebo; P=0.0001
150mg TRV045 v Placebo; P=0.0002
Source: Trevena data on file |
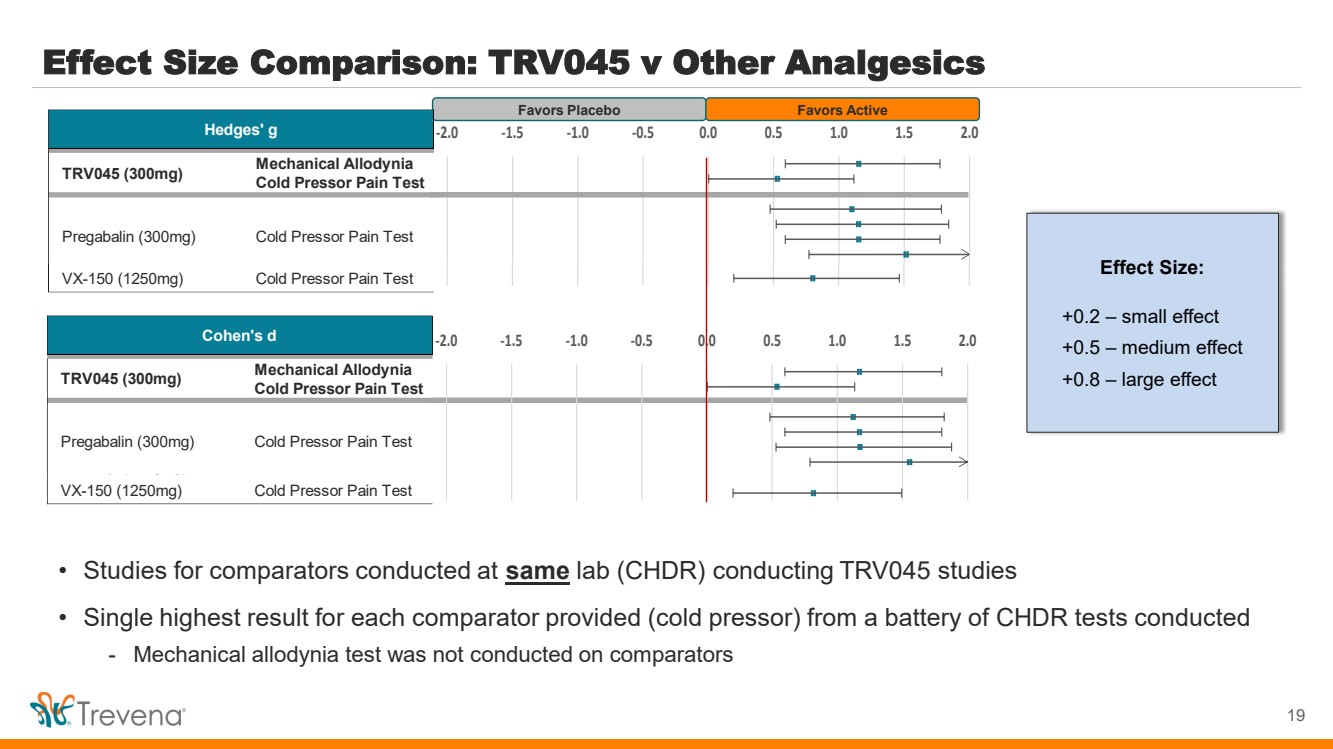
| Effect Size Comparison: TRV045 v Other Analgesics
19
Favors Placebo Favors Active
• Studies for comparators conducted at same lab (CHDR) conducting TRV045 studies
• Single highest result for each comparator provided (cold pressor) from a battery of CHDR tests conducted
- Mechanical allodynia test was not conducted on comparators
Effect Size:
+0.2 – small effect
+0.5 – medium effect
+0.8 – large effect
Mechanical Allodynia
Cold Pressor Pain Test
Fentanyl (30µg/kg) Cold Pressor Pain Test
VX-150 (1250mg) Cold Pressor Pain Test
Hedges' g
Cold Pressor Pain Test
TRV045 (300mg)
Pregabalin (300mg)
-2.0 -1.5 -1.0 -0.5 0.0 0.5 1.0 1.5 2.0
Mechanical Allodynia
Cold Pressor Pain Test
Fentanyl (30µg/kg) Cold Pressor Pain Test
VX-150 (1250mg) Cold Pressor Pain Test
Hedges' g
Cold Pressor Pain Test
TRV045 (300mg)
Pregabalin (300mg)
-2.0 -1.5 -1.0 -0.5 0.0 0.5 1.0 1.5 2.0
Mechanical Allodynia
Cold Pressor Pain Test
Fentanyl (30µg/kg) Cold Pressor Pain Test
VX-150 (1250mg) Cold Pressor Pain Test
Cohen's d
Cold Pressor Pain Test
TRV045 (300mg)
Pregabalin (300mg)
-2.0 -1.5 -1.0 -0.5 0.0 0.5 1.0 1.5 2.0
Mechanical Allodynia
Cold Pressor Pain Test
Fentanyl (30µg/kg) Cold Pressor Pain Test
VX-150 (1250mg) Cold Pressor Pain Test
Cohen's d
Cold Pressor Pain Test
TRV045 (300mg)
Pregabalin (300mg)
-2.0 -1.5 -1.0 -0.5 0.0 0.5 1.0 1.5 2.0 |

| Source: Trevena data on file
Target Engagement (PainCart®) Study
• Statistically significant, dose-dependent, treatment effect in model of capsaicin-induced mechanical allodynia
provides evidence of the therapeutic potential of TRV045 in neuropathic pain
- Recognized and validated study index of central pain processing (‘central sensitization’)
- Note that effect appeared similar with both 150mg and 300mg dose, potentially guiding future decisions on dose range for
use in Phase 2
• Supportive evidence of analgesic action in select other nociceptive models
- Other endpoints did not show trend / statistically significant results (pressure pain / conditioned pain modulation)
• Overall, strong support for CNS penetrance and engagement of pain signaling pathways in the brain
- PK exposure consistent with parameters observed at comparable doses studied under fed conditions in Phase 1 FIH study
• No SAEs, no drug-related study discontinuations
20
PainCart observations |
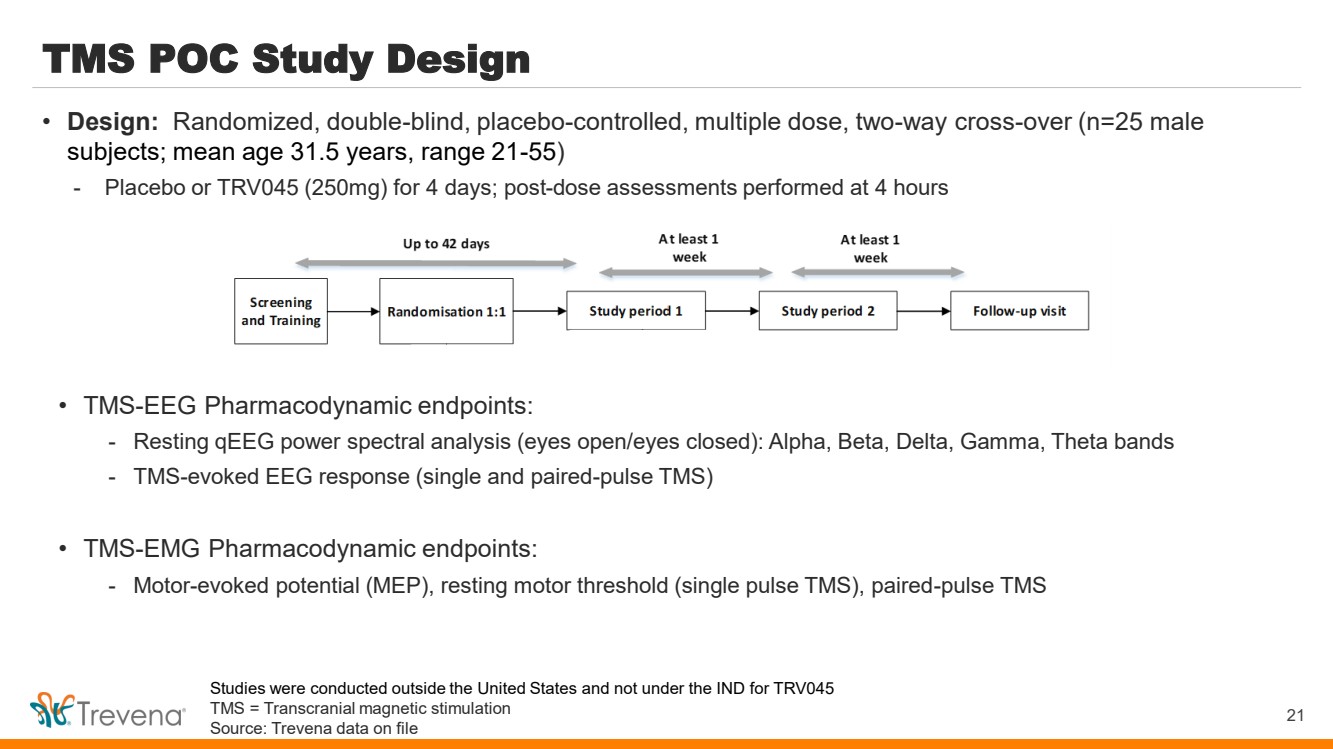
| TMS POC Study Design
• TMS-EEG Pharmacodynamic endpoints:
- Resting qEEG power spectral analysis (eyes open/eyes closed): Alpha, Beta, Delta, Gamma, Theta bands
- TMS-evoked EEG response (single and paired-pulse TMS)
• TMS-EMG Pharmacodynamic endpoints:
- Motor-evoked potential (MEP), resting motor threshold (single pulse TMS), paired-pulse TMS
21
• Design: Randomized, double-blind, placebo-controlled, multiple dose, two-way cross-over (n=25 male
subjects; mean age 31.5 years, range 21-55)
- Placebo or TRV045 (250mg) for 4 days; post-dose assessments performed at 4 hours
Studies were conducted outside the United States and not under the IND for TRV045
TMS = Transcranial magnetic stimulation
Source: Trevena data on file |
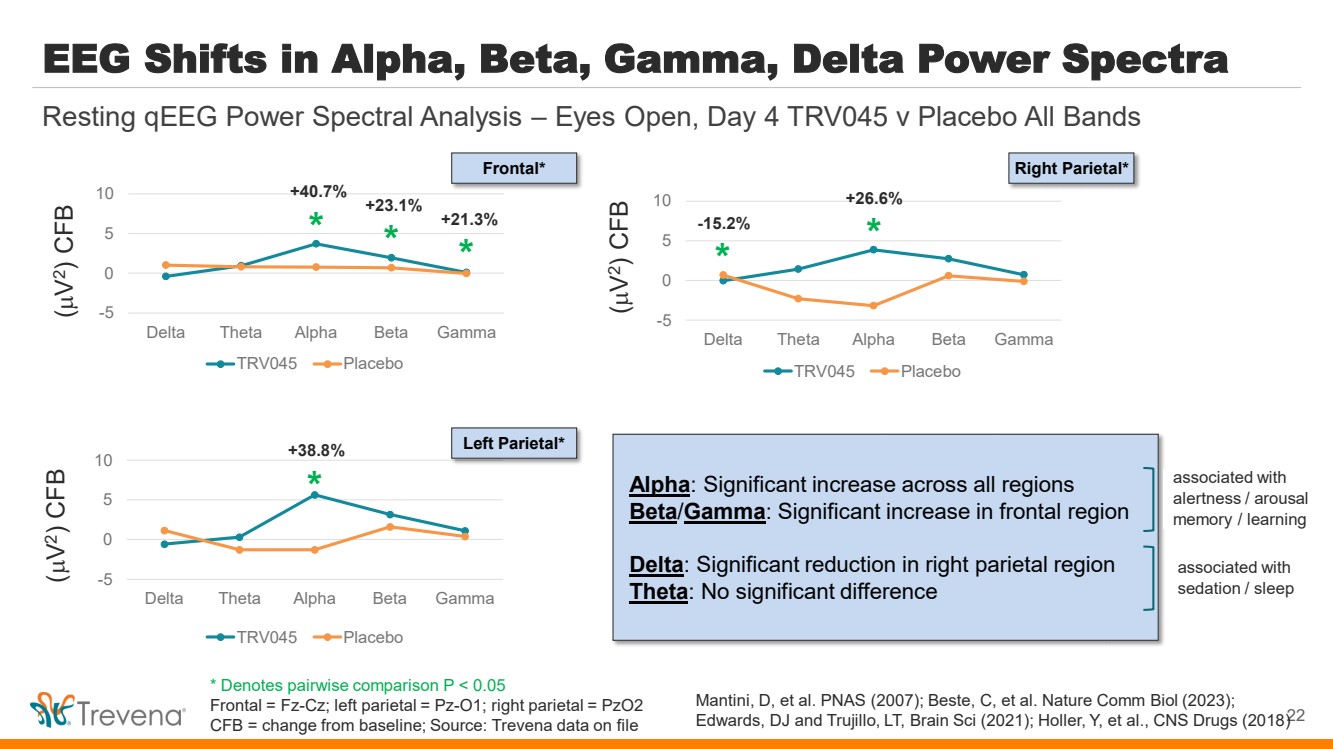
| EEG Shifts in Alpha, Beta, Gamma, Delta Power Spectra
-5
0
5
10
Delta Theta Alpha Beta Gamma
TRV045 Placebo
22
Resting qEEG Power Spectral Analysis – Eyes Open, Day 4 TRV045 v Placebo All Bands
-5
0
5
10
Delta Theta Alpha Beta Gamma
TRV045 Placebo
-5
0
5
10
Delta Theta Alpha Beta Gamma
TRV045 Placebo
*
*
*
* *
*
+21.3% -15.2%
+23.1%
+40.7%
+38.8%
+26.6%
(
V
2) CFB (
V
2) CFB
(
V
2) CFB
Frontal*
Left Parietal*
Right Parietal*
Alpha: Significant increase across all regions
Beta/Gamma: Significant increase in frontal region
Delta: Significant reduction in right parietal region
Theta: No significant difference
associated with
alertness / arousal
memory / learning
associated with
sedation / sleep
Mantini, D, et al. PNAS (2007); Beste, C, et al. Nature Comm Biol (2023);
Edwards, DJ and Trujillo, LT, Brain Sci (2021); Holler, Y, et al., CNS Drugs (2018)
* Denotes pairwise comparison P < 0.05
Frontal = Fz-Cz; left parietal = Pz-O1; right parietal = PzO2
CFB = change from baseline; Source: Trevena data on file |
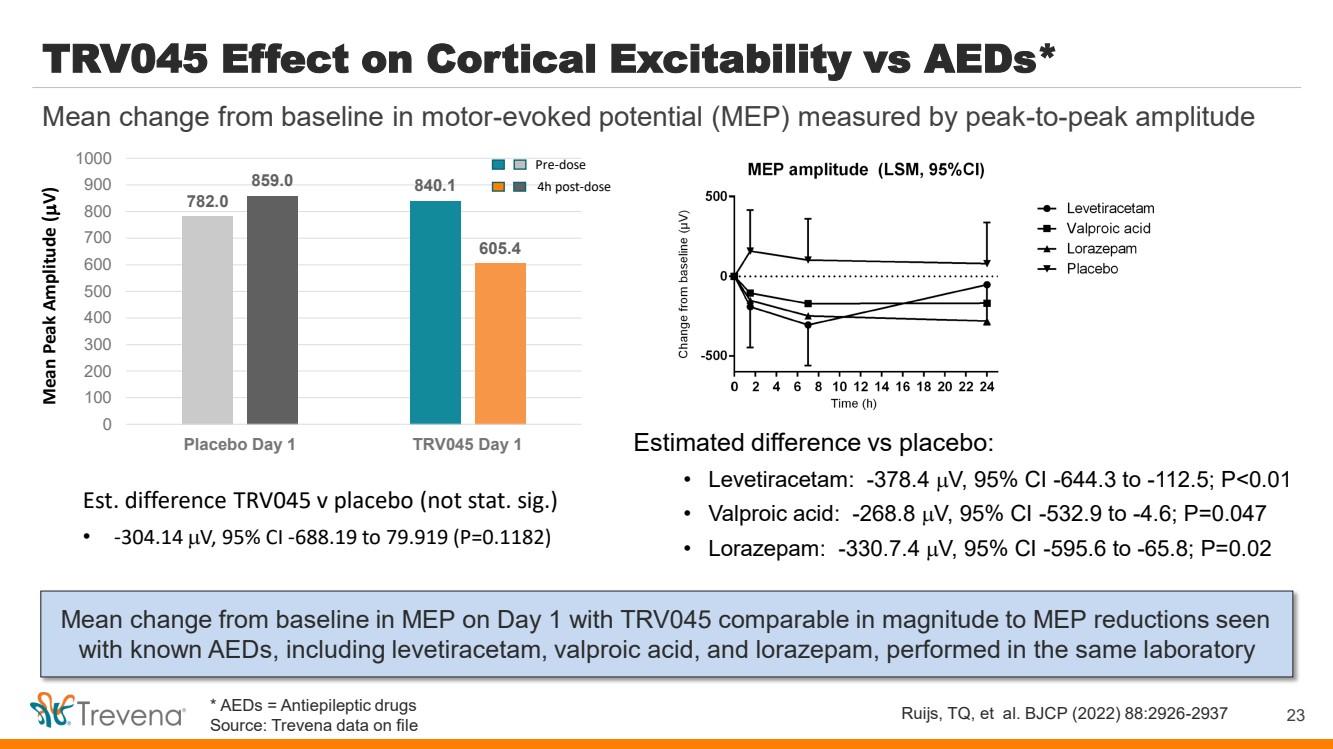
| TRV045 Effect on Cortical Excitability vs AEDs*
Mean change from baseline in MEP on Day 1 with TRV045 comparable in magnitude to MEP reductions seen
with known AEDs, including levetiracetam, valproic acid, and lorazepam, performed in the same laboratory
23
Mean change from baseline in motor-evoked potential (MEP) measured by peak-to-peak amplitude
Ruijs, TQ, et al. BJCP (2022) 88:2926-2937
Estimated difference vs placebo:
• Levetiracetam: -378.4 V, 95% CI -644.3 to -112.5; P<0.01
• Valproic acid: -268.8 V, 95% CI -532.9 to -4.6; P=0.047
• Lorazepam: -330.7.4 V, 95% CI -595.6 to -65.8; P=0.02
782.0
840.1 859.0
605.4
0
100
200
300
400
500
600
700
800
900
1000
Placebo Day 1 TRV045 Day 1
Mean Peak Amplitude (
V)
Pre-dose
4h post-dose
Est. difference TRV045 v placebo (not stat. sig.)
• -304.14 V, 95% CI -688.19 to 79.919 (P=0.1182)
* AEDs = Antiepileptic drugs
Source: Trevena data on file |

| Safety and Tolerability Summary
• No AEs assessed as probably / definitely related to study drug; no AEs reported as severe; no SAEs
- 98% of AEs (102 of 104) in PainCart® study reported as mild
- 99% of AEs (79 of 80) in TMS study reported as mild
• Screening / follow-up physical exams (including ophthalmologic exams) with no clinically significant findings
• Lab results showed no drug-related: Reduction in total lymphocyte count
Changes in heart rate or blood pressure
Changes in ECG interval measures
(no prolongation of PR or QTcF intervals)
24
POC data generally consistent with FIH study |
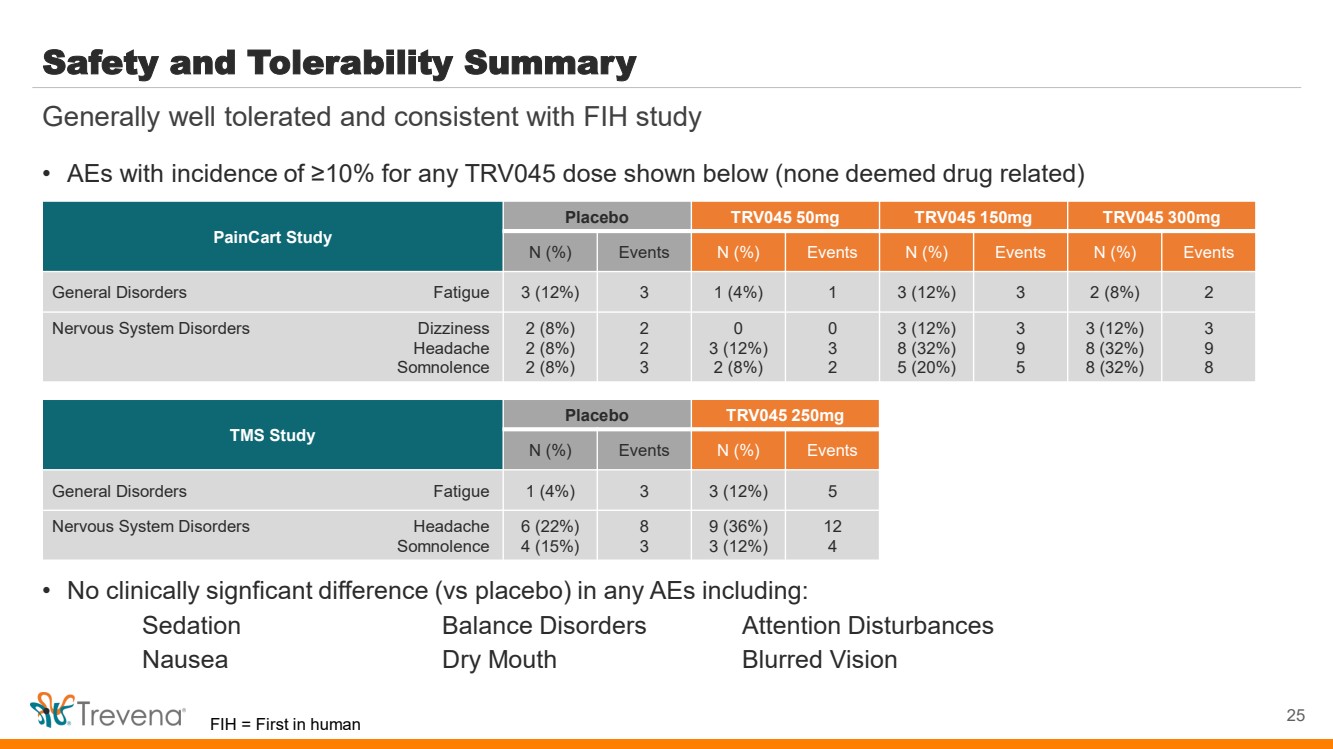
| Safety and Tolerability Summary
• AEs with incidence of ≥10% for any TRV045 dose shown below (none deemed drug related)
• No clinically signficant difference (vs placebo) in any AEs including:
Sedation Balance Disorders Attention Disturbances
Nausea Dry Mouth Blurred Vision
• 25
Generally well tolerated and consistent with FIH study
PainCart Study
Placebo TRV045 50mg TRV045 150mg TRV045 300mg
N (%) Events N (%) Events N (%) Events N (%) Events
General Disorders Fatigue 3 (12%) 3 1 (4%) 1 3 (12%) 3 2 (8%) 2
Nervous System Disorders Dizziness
Headache
Somnolence
2 (8%)
2 (8%)
2 (8%)
2
2
3
0
3 (12%)
2 (8%)
0
3
2
3 (12%)
8 (32%)
5 (20%)
3
9
5
3 (12%)
8 (32%)
8 (32%)
3
9
8
TMS Study
Placebo TRV045 250mg
N (%) Events N (%) Events
General Disorders Fatigue 1 (4%) 3 3 (12%) 5
Nervous System Disorders Headache
Somnolence
6 (22%)
4 (15%)
8
3
9 (36%)
3 (12%)
12
4
FIH = First in human |

| Overall Conclusions
• Target Engagement. Demonstrated CNS penetration and target engagement
• Neuropathic Pain. Statistically significant, dose-dependent effect in validated model of neuropathic pain
• Epilepsy. Promising evidence of early reduction in cortical excitability
Statistically significant increases in brain waves (alpha, beta, gamma) associated
with arousal, alertness, cognitive processing, learning and memory
Statistically significant decrease in delta brain waves, and no significant change in theta brain
waves, which are both associated with sedation / sleep
• Differentiated Profile. Novel MOA; expected once-daily oral dosing; potentially effective with favorable
safety / tolerability
TRV045 Proof-of-Concept Study Program
Taken together, these two POC studies provide
strong support and direction for future development of TRV045
Studies were conducted outside the United States and not under the IND for TRV045 26 |

| TRV045: Broad Potential Applicability
Potential fields for development may include: Seizure treatment (anticonvulsant)
Prevention of seizure (epileptogenesis)
Pain (DPN, CIPN)
Autoimmune (MS, RA, UC, Crohn’s Disease)
Neuropsychiatric / neurodegenerative (MDD, schizophrenia, AD, PD)
Potent and selective S1P1R target engagement
anti-inflammatory and nociceptive effects
No lymphopenia (in FIH study)
potentially limits other S1PR modulators
May avoid AEs associated with approved S1PR drugs
cardiac / pulmonary / ophthalmologic
27
Unique MOA Produces Compelling Profile
potential disease-modifying MOA
DPN: diabetic neuropathic pain; CIPN: chemotherapy-induced peripheral neuropathy; MS: multiple sclerosis; RA: rheumatoid arthritis; MDD:
major depressive disorder; AD: Alzheimer’s disease; PD: Parkinson’s disease |
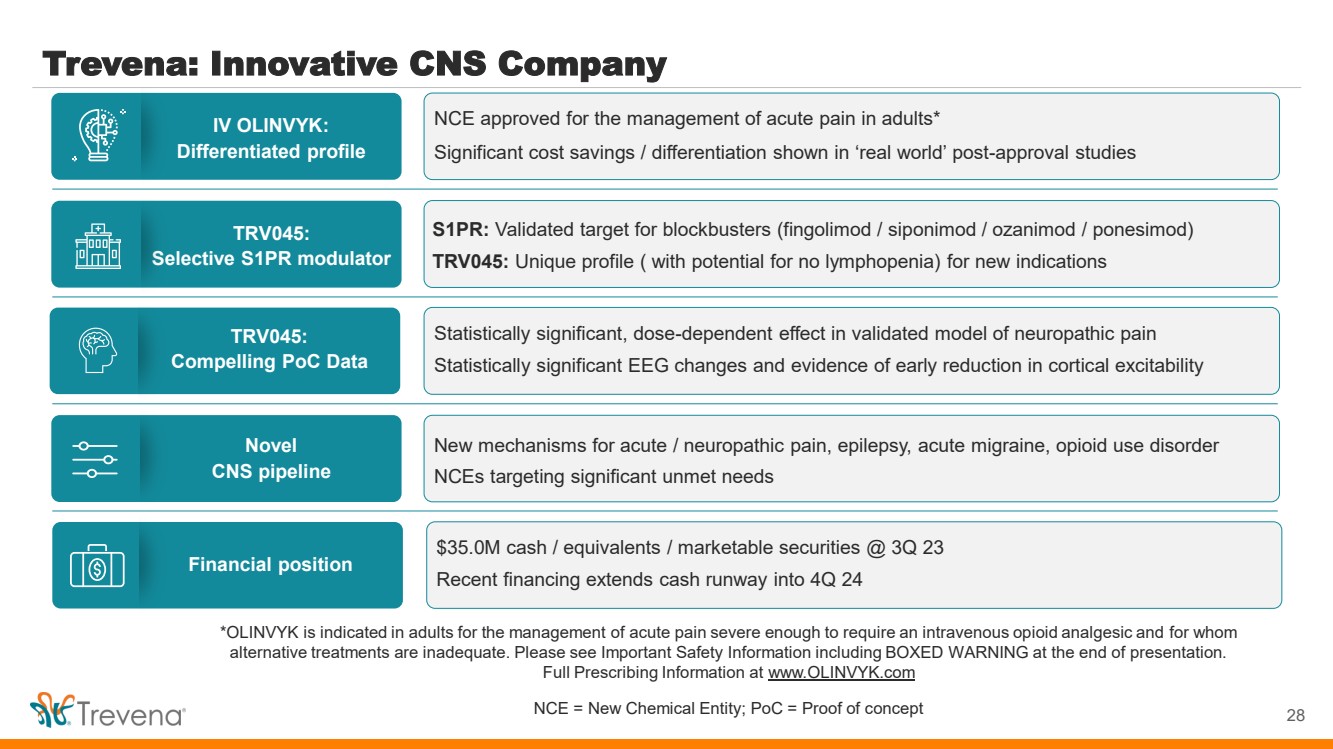
| *OLINVYK is indicated in adults for the management of acute pain severe enough to require an intravenous opioid analgesic and for whom
alternative treatments are inadequate. Please see Important Safety Information including BOXED WARNING at the end of presentation.
Full Prescribing Information at www.OLINVYK.com
NCE = New Chemical Entity; PoC = Proof of concept
Trevena: Innovative CNS Company
28
IV OLINVYK:
Differentiated profile
TRV045:
Selective S1PR modulator
Novel
CNS pipeline
TRV045:
Compelling PoC Data
Financial position
NCE approved for the management of acute pain in adults*
Significant cost savings / differentiation shown in ‘real world’ post-approval studies
S1PR: Validated target for blockbusters (fingolimod / siponimod / ozanimod / ponesimod)
TRV045: Unique profile ( with potential for no lymphopenia) for new indications
New mechanisms for acute / neuropathic pain, epilepsy, acute migraine, opioid use disorder
NCEs targeting significant unmet needs
Statistically significant, dose-dependent effect in validated model of neuropathic pain
Statistically significant EEG changes and evidence of early reduction in cortical excitability
$35.0M cash / equivalents / marketable securities @ 3Q 23
Recent financing extends cash runway into 4Q 24 |

| 29
IMPORTANT SAFETY INFORMATION |

| WARNING: ADDICTION, ABUSE, AND MISUSE; LIFE-THREATENING RESPIRATORY
DEPRESSION; NEONATAL OPIOID WITHDRAWAL SYNDROME; and RISKS FROM
CONCOMITANT USE WITH BENZODIAZEPINES OR OTHER CENTRAL NERVOUS
SYSTEM (CNS) DEPRESSANTS
Addiction, Abuse, and Misuse
OLINVYK exposes patients and other users to the risks of opioid addiction, abuse, and
misuse, which can lead to overdose and death. Assess each patient’s risk before
prescribing OLINVYK, and monitor all patients regularly for the development of
behaviors or conditions.
Life-Threatening Respiratory Depression
Serious, life-threatening, or fatal respiratory depression may occur with use of OLINVYK.
Monitor for respiratory depression, especially during initiation of OLINVYK or following a dose
increase.
Neonatal Opioid Withdrawal Syndrome
Prolonged use of OLINVYK during pregnancy can result in neonatal opioid withdrawal
syndrome, which may be life-threatening if not recognized and treated, and requires
management according to protocols developed by neonatology experts. If opioid use is required
for a prolonged period in a pregnant woman, advise the patient of the risk of neonatal opioid
withdrawal syndrome and ensure that appropriate treatment will be available.
Risk From Concomitant Use With Benzodiazepines or Other CNS Depressants
Concomitant use of opioids with benzodiazepines or other CNS depressants, including alcohol,
may result in profound sedation, respiratory depression, coma, and death. Reserve concomitant
prescribing for use in patients for whom alternative treatment options are inadequate; limit
dosages and durations to the minimum required; and follow patients for signs and symptoms of
respiratory depression and sedation.
Limitations of Use
Because of the risks of addiction, abuse, and misuse with opioids, even at recommended doses, reserve OLINVYK
for use in patients for whom alternative treatment options [e.g., non-opioid analgesics or opioid combination
products]:
• Have not been tolerated, or are not expected to be tolerated
• Have not provided adequate analgesia, or are not expected to provide adequate analgesia.
The cumulative total daily dose should not exceed 27 mg, as total daily doses greater than 27 mg may increase the
risk for QTc interval prolongation.
CONTRAINDICATIONS
OLINVYK is contraindicated in patients with:
• Significant respiratory depression
• Acute or severe bronchial asthma in an unmonitored setting or in the absence of resuscitative equipment
• Known or suspected gastrointestinal obstruction, including paralytic ileus
• Known hypersensitivity to oliceridine (e.g., anaphylaxis)
WARNINGS AND PRECAUTIONS
• OLINVYK contains oliceridine, a Schedule II controlled substance, that exposes users to the risks of addiction,
abuse, and misuse. Although the risk of addiction in any individual is unknown, it can occur in patients
appropriately prescribed OLINVYK. Assess risk, counsel, and monitor all patients receiving opioids.
• Serious, life-threatening respiratory depression has been reported with the use of opioids, even when used as
recommended, especially in patients with chronic pulmonary disease, or in elderly, cachectic and debilitated
patients. The risk is greatest during initiation of OLINVYK therapy, following a dose increase, or when used
with other drugs that depress respiration. Proper dosing of OLINVYK is essential, especially when converting
patients from another opioid product to avoid overdose. Management of respiratory depression may include
close observation, supportive measures, and use of opioid antagonists, depending on the patient’s clinical
status.
• Opioids can cause sleep-related breathing disorders including central sleep apnea (CSA) and sleep-related
hypoxemia with risk increasing in a dose-dependent fashion. In patients who present with CSA, consider
decreasing the dose of opioid using best practices for opioid taper.
INDICATIONS AND USAGE
OLINVYK is a new chemical entity indicated in adults for the management of acute pain severe enough to
require an intravenous opioid analgesic and for whom alternative treatments are inadequate.
30 |

| WARNINGS AND PRECAUTIONS
• Prolonged use of opioids during pregnancy can result in withdrawal in the neonate that may be
life-threatening. Observe newborns for signs of neonatal opioid withdrawal syndrome and manage
accordingly. Advise pregnant women using OLINVYK for a prolonged period of the risk of neonatal
opioid withdrawal syndrome and ensure that appropriate treatment will be available.
• Profound sedation, respiratory depression, coma, and death may result from the concomitant use of
OLINVYK with benzodiazepines or other CNS depressants (e.g., non-benzodiazepine sedatives/hypnotics,
anxiolytics, tranquilizers, muscle relaxants, general anesthetics, antipsychotics, other opioids, or alcohol).
Because of these risks, reserve concomitant prescribing of these drugs for use in patients for whom
alternative treatment options are inadequate, prescribe the lowest effective dose, and minimize the duration.
• OLINVYK was shown to have mild QTc interval prolongation in thorough QT studies where patients were
dosed up to 27 mg. Total cumulative daily doses exceeding 27 mg per day were not studied and may
increase the risk for QTc interval prolongation. Therefore, the cumulative total daily dose of OLINVYK
should not exceed 27 mg.
• Increased plasma concentrations of OLINVYK may occur in patients with decreased Cytochrome P450
(CYP) 2D6 function or normal metabolizers taking moderate or strong CYP2D6 inhibitors; also in patients
taking a moderate or strong CYP3A4 inhibitor, in patients with decreased CYP2D6 function who are also
receiving a moderate or strong CYP3A4 inhibitor, or with discontinuation of a CYP3A4 inducer. These
patients may require less frequent dosing and should be closely monitored for respiratory depression and
sedation at frequent intervals. Concomitant use of OLINVYK with CYP3A4 inducers or discontinuation of
a moderate or strong CYP3A4 inhibitor can lower the expected concentration, which may decrease
efficacy, and may require supplemental doses.
• Cases of adrenal insufficiency have been reported with opioid use (usually greater than one month).
Presentation and symptoms may be nonspecific and include nausea, vomiting, anorexia, fatigue, weakness,
dizziness, and low blood pressure. If confirmed, treat with physiologic replacement doses of corticosteroids
and wean patient from the opioid.
• OLINVYK may cause severe hypotension, including orthostatic hypotension and syncope in ambulatory
patients.
• There is increased risk in patients whose ability to maintain blood pressure has already been compromised
by a reduced blood volume or concurrent administrationof certain CNS depressant drugs (e.g.,
phenothiazines or general anesthetics). Monitor these patientsfor signs of hypotension. In patients with
circulatory shock, avoid the use of OLINVYK as it may cause vasodilation that can further reduce cardiac
output and blood pressure.
• Avoid the use of OLINVYK in patients with impaired consciousness or coma. OLINVYK should be used
with caution in patients who may be susceptible to the intracranial effects of CO2
retention,such as those
with evidence of increased intracranial pressure or brain tumors, as a reduction in respiratory drive and the
resultantCO2
retention can further increase intracranial pressure. Monitor such patientsfor signs of
sedation and respiratory depression, particularlywhen initiating therapy.
• As with all opioids, OLINVYK may cause spasm of the sphincter of Oddi, and may cause increases in
serum amylase. Monitor patients with biliary tract disease, including acute pancreatitis,for worsening
symptoms.
• There is increased risk in patients whose ability to maintain blood pressure has already been compromised
by a reduced blood volume or concurrent administrationof certain CNS depressant drugs (e.g.,
phenothiazines or general anesthetics). Monitor these patientsfor signs of hypotension. In patients with
circulatory shock, avoid the use of OLINVYK as it may cause vasodilation that can further reduce cardiac
output and blood pressure.
• Avoid the use of OLINVYK in patients with impaired consciousness or coma. OLINVYK should be used
with caution in patients who may be susceptible to the intracranial effects of CO2
retention,such as those
with evidence of increased intracranial pressure or brain tumors, as a reduction in respiratory drive and the
resultantCO2
retention can further increase intracranial pressure. Monitor such patientsfor signs of
sedation and respiratory depression, particularlywhen initiating therapy.
• As with all opioids, OLINVYK may cause spasm of the sphincter of Oddi, and may cause increases in
serum amylase. Monitor patients with biliary tract disease, including acute pancreatitis,for worsening
symptoms.
• OLINVYK may increase the frequency of seizures in patients with seizure disorders and may increase the
risk of seizuresin vulnerable patients. Monitor patients with a history of seizure disorders for worsened
seizure control.
• Do not abruptly discontinue OLINVYK in a patient physically dependent on opioids. Gradually taper the
dosage to avoid a withdrawal syndrome and return of pain. Avoid the use of mixed agonist/antagonist (e.g.,
pentazocine, nalbuphine, and butorphanol) or partial agonist (e.g., buprenorphine) analgesics in patients
who are receiving OLINVYK, as they may reduce the analgesic effect and/or precipitate withdrawal
symptoms.
• OLINVYK may impair the mental or physical abilities needed to perform potentially hazardous activities
such as driving a car or operating machinery.
• Although self-administration of opioids by patient-controlled analgesia (PCA) may allow each patient to
individually titrate to an acceptable level of analgesia, PCA administration has resulted in adverse
outcomes and episodes of respiratory depression. Health care providers and family members monitoring
patients receiving PCA analgesia should be instructed in the need for appropriate monitoring for excessive
sedation, respiratory depression, or other adverse effects of opioid medications.
ADVERSE REACTIONS
Adverse reactions are described in greater detail in the Prescribing Information.
The most common (incidence ≥10%) adverse reactions in Phase 3 controlled clinical trials were nausea,
vomiting, dizziness, headache, constipation, pruritus, and hypoxia.
PLEASE see www.OLNVYK.com for full prescribing information including BOXED warning and important safety
information 31 |
Cover
|
Jan. 08, 2024 |
| Cover [Abstract] |
|
| Document Type |
8-K
|
| Amendment Flag |
false
|
| Document Period End Date |
Jan. 08, 2024
|
| Entity File Number |
001-36193
|
| Entity Registrant Name |
TREVENA, INC.
|
| Entity Central Index Key |
0001429560
|
| Entity Tax Identification Number |
26-1469215
|
| Entity Incorporation, State or Country Code |
DE
|
| Entity Address, Address Line One |
955 Chesterbrook Boulevard
|
| Entity Address, Address Line Two |
Suite 110
|
| Entity Address, City or Town |
Chesterbrook
|
| Entity Address, State or Province |
PA
|
| Entity Address, Postal Zip Code |
19087
|
| City Area Code |
610
|
| Local Phone Number |
354-8840
|
| Written Communications |
false
|
| Soliciting Material |
false
|
| Pre-commencement Tender Offer |
false
|
| Pre-commencement Issuer Tender Offer |
false
|
| Title of 12(b) Security |
Common Stock, $0.001 par value
|
| Trading Symbol |
TRVN
|
| Security Exchange Name |
NASDAQ
|
| Entity Emerging Growth Company |
false
|
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 2 such as Street or Suite number
| Name: |
dei_EntityAddressAddressLine2 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the state or province.
| Name: |
dei_EntityAddressStateOrProvince |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:stateOrProvinceItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 13e
-Subsection 4c
| Name: |
dei_PreCommencementIssuerTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14d
-Subsection 2b
| Name: |
dei_PreCommencementTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Section 14a
-Number 240
-Subsection 12
| Name: |
dei_SolicitingMaterial |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Securities Act
-Number 230
-Section 425
| Name: |
dei_WrittenCommunications |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
Trevena (NASDAQ:TRVN)
過去 株価チャート
から 5 2024 まで 6 2024

Trevena (NASDAQ:TRVN)
過去 株価チャート
から 6 2023 まで 6 2024
