Nemaura Medical Announces Appointment of Mr Nikolai Rozanov as Strategic Adviser in Artificial Intelligence, to Assist in Development of its AI Health Coach, and Strengthen Outcomes in its CGM Based Metabolic and Type 2 Diabetes Programs.
2023年9月26日 - 10:50PM

Nemaura Medical, Inc.(NASDAQ: NMRD) (“Nemaura” or the “Company”), a
medical technology company focused on developing and
commercialising non-invasive wearable diagnostic devices and
supporting personalized lifestyle coaching programs, today
announced the appointment of Nikolai Rozanov as strategic adviser
in AI development.
Mr Rozanov is a specialist in the field of
artificial intelligence and was selected for inclusion by Forbes
magazine in their ’30 Under 30’ list in the field of technology, a
prestigious honour accorded to the best up-and-coming talent in the
sector. The appointment of Mr Rozanov solidifies the Company’s
expertise in the growing AI sector, at a time when AI developments
in healthcare are gathering pace. The Company is now well placed to
further integrate data from its CGM to create tailored programs
that can make tailored personalised recommendations.
Dr. Faz Chowdhury, Chief Executive Officer,
stated, “We are excited to be working with Nikolai to leverage our
CGM data to give better insights to members. The use of AI based
models is becoming more widespread, and we believe they have a
vital role to play in the healthcare and lifestyle sector. Deeper
personalised insights better engage users, and the capability of AI
means this can be scaled efficiently”.
About Nemaura Medical,
Inc.
Nemaura Medical Inc. is a medical technology
company developing and commercialising non-invasive wearable
diagnostic devices. The Company is currently also commercialising
sugarBEAT® and proBEAT™. sugarBEAT®, a CE mark approved Class
IIb medical device, is a non-invasive and flexible continuous
glucose monitor (CGM) providing actionable insights derived from
real time glucose measurements and daily glucose trend data, which
may help people with diabetes and prediabetes to better manage,
reverse, and prevent the onset of diabetes. Nemaura has submitted a
PMA (Premarket Approval Application) for sugarBEAT® to the
U.S. Food and Drug Administration (the “FDA”).
proBEAT™ combines non-invasive glucose data processed using
artificial intelligence as part of a lifestyle program that is
being refined for commercial launch.
The Company sits at the intersection of the
global Type 2 diabetes market that is expected to reach nearly $59
billion by 2025, the $50+ billion pre-diabetic market, and the
wearable health-tech sector for weight loss and wellness
applications that is estimated to reach $60 billion by 2023.
For more information, please
visit www.NemauraMedical.com.
Cautionary Statement Regarding Forward-Looking
Statements:
The statements in this press release that are
not historical facts may constitute forward-looking statements that
are based on current expectations and are subject to risks and
uncertainties that could cause actual future results to differ
materially from those expressed or implied by such statements.
Those risks and uncertainties include, but are not limited to, the
launch of proBEAT™ in the U.S., risks related to regulatory
status and the failure of future development and preliminary
marketing efforts, Nemaura’s ability to secure additional
commercial partnering arrangements, risks and uncertainties
relating to Nemaura’s and its partners’ ability to develop, market
and sell proBEAT™, the availability of substantial additional
equity or debt capital to support its research, development and
product commercialization activities, and the success of its
research, development, regulatory approval, marketing and
distribution plans and strategies, including those plans and
strategies related to both proBEAT™ digital health, and
sugarBEAT®. There can be no assurance that the Company will be able
to reach a part of or any of the global market for CGM with its
products/services. The FDA reserves the right to re-evaluate its
decision that proBEAT™ qualifies as a general wellness product
should it become aware of any issues such as skin irritation or
other adverse events from the device, as well as any misuse
impacting patient safety, and any other reason as the FDA may see
fit at its discretion to determine the product does not fit the
definition of a general wellness product. These and other risks and
uncertainties are identified and described in more detail in
Nemaura’s filings with the United States Securities and Exchange
Commission, including, without limitation, its Annual Report on
Form 10-K for the most recently completed fiscal year, its
Quarterly Reports on Form 10-Q, and its Current Reports on Form
8-K, as the same may be amended from time to time. Nemaura
undertakes no obligation to publicly update or revise any
forward-looking statements.
Investor Relations:
IR@NemauraMedical.Com
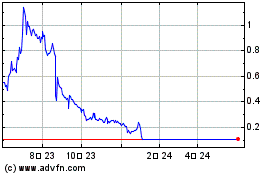
Nemaura Medical (NASDAQ:NMRD)
過去 株価チャート
から 4 2024 まで 5 2024
Nemaura Medical (NASDAQ:NMRD)
過去 株価チャート
から 5 2023 まで 5 2024