UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 6-K
REPORT OF FOREIGN PRIVATE ISSUER
PURSUANT TO RULE 13a-16 OR 15d-16 UNDER
THE SECURITIES EXCHANGE ACT OF 1934
For the Month of September 2023
Commission File Number: 001-38104
IMMURON LIMITED
(Name of Registrant)
Level 3, 62 Lygon Street, Carlton South,
Victoria, 3053, Australia
(Address of principal executive office)
Indicate by check mark whether the registrant files or will file annual
reports under cover of Form 20-F or Form 40-F.
Form 20-F ☒ Form
40-F ☐
IMMURON LIMITED
EXPLANATORY NOTE
Immuron Limited (the “Company”) published
one announcement (the “Public Notices”) to the Australian Securities Exchange on September 11, 2023 titled:
| - | “Immuron
CEO, Steven Lydeamore to present at H.C. Wainwright” |
A copy of the Public Notice is attached as an exhibit to this report
on Form 6-K.
This report on Form 6-K (including the exhibit
hereto) shall not be deemed to be “filed” for purposes of the Securities Exchange Act of 1934, as amended (the “Exchange
Act”) and shall not be incorporated by reference into any filing under the Securities Act of 1933, as amended, except as shall be
expressly set forth by specific reference in such filing.
EXHIBITS
SIGNATURE
Pursuant to the requirements
of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto
duly authorized.
| |
IMMURON LIMITED |
| |
|
|
| |
 |
| |
|
|
| Date: September 11, 2023 |
By: |
/s/ Phillip Hains |
| |
|
Phillip Hains |
| |
|
Company Secretary |
3
Exhibit
99.1

Immuron
CEO, Steven Lydeamore to present at H.C. Wainwright
Melbourne,
Australia, September 11, 2023: Immuron Limited (ASX: IMC; NASDAQ: IMRN), an Australian based and globally integrated biopharmaceutical
company is pleased to advise our Chief Executive Officer, Steven Lydeamore will be presenting virtually at the H.C. Wainwright 25th
Annual Global Investment Conference on September 11th.
The
webcast to this presentation will be available here1
at 7am Eastern Standard Time / 9pm Australian Eastern Standard time on September 11th.
A
copy of the presentation being made at the H.C. Wainwright 25th Annual Global Investment Conference is included below.
This
release has been authorised by the directors of Immuron Limited.
-
- - END - - -
COMPANY
CONTACT:
Steven
Lydeamore
Chief Executive Officer
Ph: +61 (0)3 9824 5254
info@immuron.com
About
Travelan®
Travelan®
is an orally administered passive immunotherapy that prophylactically reduces the likelihood of contracting traveller’s diarrhoea,
a digestive tract disorder that is commonly caused by pathogenic bacteria and the toxins they produce.
Travelan® is a highly purified tabletised preparation of hyperimmune bovine antibodies and other factors, which when taken with meals
bind to diarrhoea-causing bacteria and prevent colonisation and the pathology associated with traveller’s diarrhoea. In Australia,
Travelan® is a listed medicine on the Australian Register for Therapeutic Goods (AUST L 106709) and is indicated to reduce the risk
of Traveller’s Diarrhoea, reduce the risk of minor gastrointestinal disorders and is antimicrobial. In Canada, Travelan® is
a licensed natural health product (NPN 80046016) and is indicated to reduce the risk of Traveller’s Diarrhoea. In the U.S., Travelan®
is sold as a dietary supplement for digestive tract protection.
About
Traveller’s Diarrhoea
Traveller’s
Diarrhoea is a gastrointestinal infection with symptoms that include loose, watery (and occasionally bloody) stools, abdominal cramping,
bloating, and fever, Enteropathogenic bacteria are responsible for most cases, with enterotoxigenic Escherichia coli (ETEC) playing
a dominant causative role. Campylobacter spp. are also responsible for a significant proportion of cases. The more serious infections
with Salmonella spp. the bacillary dysentery organisms belonging to Shigella spp. and Vibrio spp. (the causative agent of cholera) are
often confused with Traveller’s Diarrhoea as they may be contracted while travelling and initial symptoms are often indistinguishable.
| 1 | https://journey.ct.events/view/03d18b80-9f8f-4626-82c9-df1db0dadeb9 |


About
Immuron
Immuron
Limited (ASX: IMC, NASDAQ: IMRN), is an Australian biopharmaceutical company focused on developing and commercialising orally delivered
targeted polyclonal antibodies for the treatment of infectious diseases.
For
more information visit: http://www.immuron.com
FORWARD-LOOKING
STATEMENTS:
This
press release may contain “forward-looking statements” within the meaning of Section 27A of the Securities Act of 1933 and
Section 21E of the Securities Exchange Act of 1934, each as amended. Such statements include, but are not limited to, any statements
relating to our growth strategy and product development programs and any other statements that are not historical facts. Forward-looking
statements are based on management’s current expectations and are subject to risks and uncertainties that could negatively affect
our business, operating results, financial condition and stock value. Factors that could cause actual results to differ materially from
those currently anticipated include: risks relating to our growth strategy; our ability to obtain, perform under and maintain financing
and strategic agreements and relationships; risks relating to the results of research and development activities; risks relating to the
timing of starting and completing clinical trials; uncertainties relating to preclinical and clinical testing; our dependence on third-party
suppliers; our ability to attract, integrate and retain key personnel; the early stage of products under development; our need for substantial
additional funds; government regulation; patent and intellectual property matters; competition; as well as other risks described in our
SEC filings. We expressly disclaim any obligation or undertaking to release publicly any updates or revisions to any forward-looking
statements contained herein to reflect any change in our expectations or any changes in events, conditions or circumstances on which
any such statement is based, except as required by law.


1 1 H.C. WAINWRIGHT & CO. VIRTUAL PRESENTATION 11 SEPTEMBER, 2023 S teven Lydeamore - CEO NASDAQ: IMRN ASX: IMC

Certain statements made in this presentation are forward - looking statements and are based on Immuron’s current expectations, estimates and projections. Words such as “anticipates,” “expects,” ” intends,” ” plans,” ” believes,” ” seeks,” “estimates,” “guidance” and s imilar expressions are intended to identify forward - looking statements. Although Immuron believes the forward - looking statements are based on reasonable assumptions, they are subject to certain r i sks and uncertainties, some of which are beyond Immuron’s control, including those r i sks or uncertainties inherent in the process of both developing and commercializing technology. As a result, actual results could materially differ f rom those expressed or forecasted in the forward - looking statements. The forward - looking statements made in this presentation relate only to events as of the date on which the statements are made. Immuron will not undertake any obligation to release publicly any revisions or updates to these forward - looking statements to reflect events, c i rcumstances or unanticipated events occurring after the date of this presentation except as required by law or by any appropriate regulatory authority. YTD FY 2023 results in this presentation are subject to audit review. 2 SAFE HARBOR STATEMENT

EXECUTIVE SUMMARY Company Overview Business Update Results & Outlook • Two commercially available oral immunotherapeutic products – Travelan® and Protectyn® • 4 clinical programs: Travelan®(NMRC: Phase 2 CHIM trial), Travelan®(USU: Phase 4 field study), CampETEC (Phase 2 CHIM trial), IMM - 529 (Protocol development phase, Phase 2 trial) • Flagship product Travelan® growing strongly as overseas travel rebounds • Travelan® (IMM - 124E) IND filed with and approved by FDA • Travelan® (IMM - 124E) Phase 2 clinical trial initiated • Travelan® Uniformed Health Services University (USU) P2TD IMM - 124E field clinical trial recruited 347 (~40% of target 868) • CampETEC IND approved (released from Clinical Hold) • FY23 sales of A$1.80 million up 136% on FY22 (subject to audit review) • Evaluating options to enter international markets through distributors • Evaluating options to add to marketed products portfolio in FY24 Immuron Ltd (NASDAQ:IMRN) (ASX:IMC) is a globally integrated biopharmaceutical company focused on developing, and commercialising, oral immunotherapeutics for the treatment of gut mediated diseases Financial Snapshot 227,798,346 Shares on Issue 14,568,559 Total Options IMC: A$0.075 Last Traded Price IMC: A$0.105/0.067 IMRN: $3.21/1.39 52 week High/Low IMC: A$17.085m Market Cap A$18.5m Cash & Cash Equivalents (as at 31 Dec 22) as of 21 August 2023 Major Shareholders 3 % of CSO Units Holder 34.7 % 78,973,505 BNY Mellon Asset Management 3.0 % 6,904,554 Management & Board 2.6 % 6,000,000 Authentics Australia Pty. Ltd. 1.7 % 3,846,712 Grandlodge

4 ADDRESSABLE MARKET & INDUSTRY OVERVIEW Industry tailwinds Travel picking up significantly following COVID lockdowns Frequent Symptom 30% - 70% of travelers experience traveller’s diarrhoea** $83m Based on US annual travel numbers and a penetration rate of 15%, the market potential is estimated at $83m* $50m Based on EU travel numbers and a penetration rate of 15%, the market potential is estimated at $50m* $1.7b Clostridioides difficile infections (CDIs) to grow to almost $1.7 billion by 2026, according to GlobalData Billion Dollar Market Traveller’s diarrhoea treatment market is large and growing at a CAGR of ~7% * IMC Company Report - Travelan Market Analysis 2019 ** Centers for Disease Control and Prevention Yellow Book USA Market amazon.com shopfront launched 1QFY24 Re - entry into retail pharmacies will be explored in FY24 Evaluating options: • for entry into international markets • to add marketed products to portfolio in FY24 Chief Commercial Officer has 20+ year’s experience with local and global (Asia, UK) commercial leadership roles with GSK and P&G

TECHNOLOGY PLATFORM Bovine colostrum is the first milk of cows after calving. It is rich in immunoglobulins, lactoferrin, lysozyme, lactoperoxidase, growth factors and bioactive peptides. Colostrum has higher levels of protein, fat, vitamins, and minerals when compared to milk. This enables full development of the newborn calf in addition to immunity against several pathogens. * Immuron’s proprietary technology platform combines the natural human nutrition & health benefits of bovine colostrum with a novel class of specifically targeted oral polyclonal antibodies that offer delivery within the gastrointestinal (“GI”) tract and can be used to target viruses or bacteria and neutralize the toxins they produce at mucosal surfaces. STEP 1 Development of Highly Specific Vaccines STEP 2 Isolation of Hyperimmune antibody - rich bovine colostrum STEP 3 Oral Antimicrobial therapeutics without drawbacks of antibiotics FINAL PRODUCT Toxin Neutralization + Clearance of targeted gut pathogens x Reduce occurrence and reduce/relieve diarrhoea x Reduce/relieve abdominal cramping x Reduce/relieve gastrointestinal pain x Assists repair of gastrointestinal/gut wall lining x Enhance/promote immune defence x Enhance/promote health liver function Australian Permitted indications; these statements have not been evaluated by the Food and Drug Administration (FDA) 5 * Gomes et. al., NFS Journal, Volume 25, November 2021, pages 1 - 11, https://doi.org/10.1016/j.nfs.2021.10.001

STATUS OF PRODUCT PORTFOLIO AND KEY MILESTONES US Department of Defense grant of US$4.45 million to examine a dosing regimen for Travelan® more suited for use by the military b • Market evaluation by Lumanity confirms the Traveler’s Diarrhea market opportunity for IMM - 124E (Travelan®) c • IMM - 124E (Travelan®) IND was approved by the FDA in 2H2022 a • Clinical trial initiated May 2023 g • First patients enrolled in July 2023 I • Anticipated clinical study report – 1H 2024 Market evaluation by Lumanity confirms the Clostridioides difficile market opportunity for IMM - 529 d • 600mg solid dose active formulation development completed • Anticipated manufacture of cGMP IMM - 529 – 31 December 2023 h • Anticipated pre - IND submission to the FDA – 30 June 2024 h Travelan® - Uniformed Services University has recruited ~40% of participants in a randomized clinical trial with Travelan® to evaluate the effectiveness for prophylaxis during deployment or travel to a high traveler’s diarrhea risk region e, i • Anticipated completion of enrollment – 31 March 2024 • Anticipated completion of in - patient phase – 30 June 2024 Naval Medical Research Center Clinical Trials of CampETEC in campylobacter and enterotoxigenic E.coli (ETEC) • Animal ethics approval for Toxicology study – 22 November 2022 • Immuron sponsored Toxicology study - completed 20 December 2022 • FDA Removed Clinical Hold on Campylobacter ETEC – 8 May 2023 f a ,b,c,d,e,f g,h,i ASX Release links in appendix Market Phase III Phase II Phase I Indication Compound or brand name Traveler’s Diarrhea ETEC challenge IMM - 124E Travelan® Clostridioides difficile Infection & Recurrence IMM - 529 Market Phase III Phase II Phase I Partner Compound or brand name rvices University Uniformed Se Travelan® al Naval Medic CampETEC Research Center Immuron’s Clinical Programs Our Partners’ Clinical Programs *Further information on the clinical programs can be found on slide 7 6
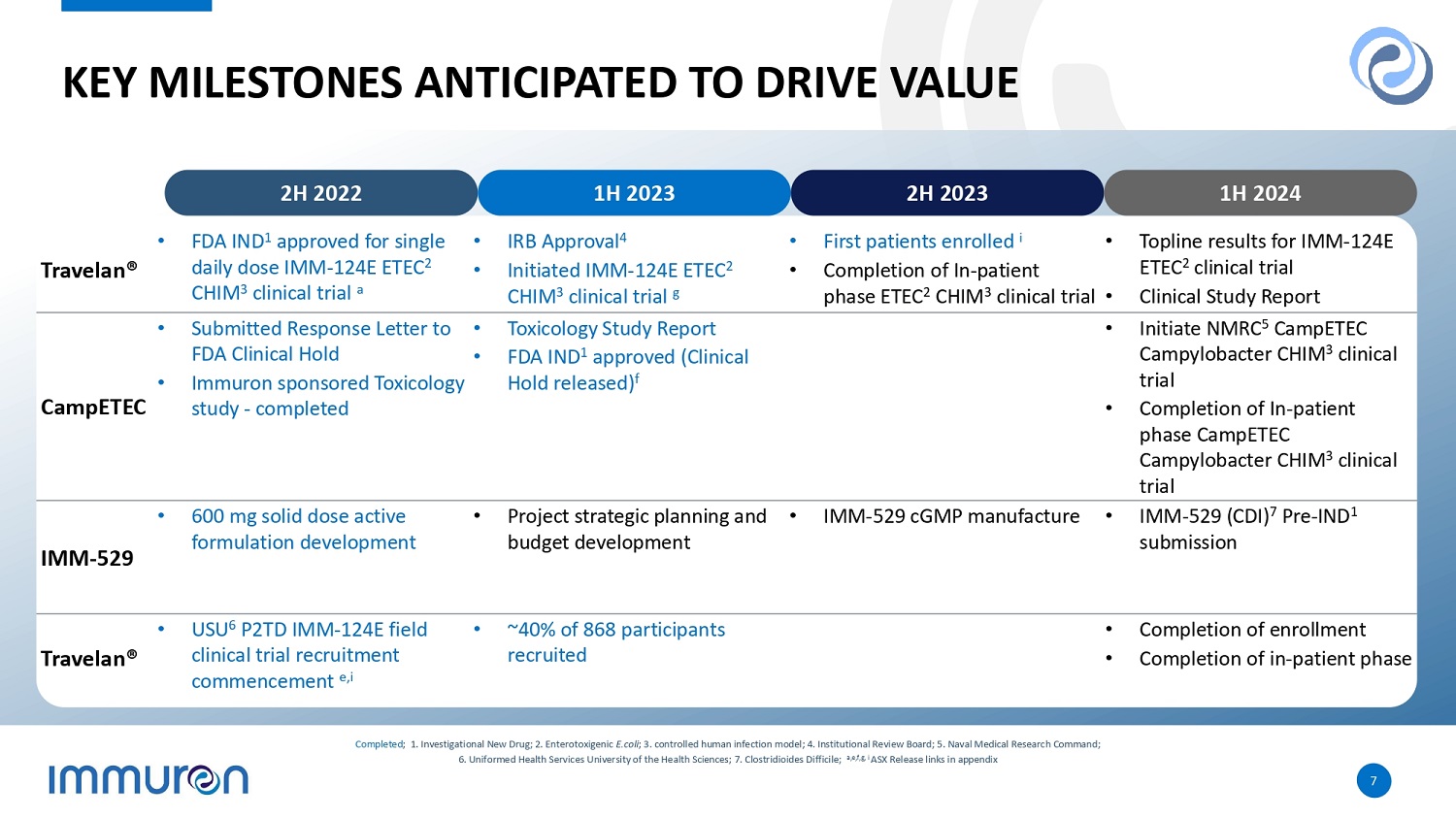
KEY MILESTONES ANTICIPATED TO DRIVE VALUE • Topline results for IMM - 124E ETEC 2 clinical trial • Clinical Study Report • First patients enrolled i • Completion of In - patient phase ETEC 2 CHIM 3 clinical trial • IRB Approval 4 • Initiated IMM - 124E ETEC 2 CHIM 3 clinical trial g • FDA IND 1 approved for single daily dose IMM - 124E ETEC 2 CHIM 3 clinical trial a Travelan® • Initiate NMRC 5 CampETEC Campylobacter CHIM 3 clinical trial • Completion of In - patient phase CampETEC Campylobacter CHIM 3 clinical trial • Toxicology Study Report • FDA IND 1 approved (Clinical Hold released) f • Submitted Response Letter to FDA Clinical Hold • Immuron sponsored Toxicology study - completed CampETEC • IMM - 529 (CDI) 7 Pre - IND 1 submission • IMM - 529 cGMP manufacture • Project strategic planning and budget development • 600 mg solid dose active formulation development IMM - 529 • Completion of enrollment • Completion of in - patient phase • ~40% of 868 participants recruited • USU 6 P2TD IMM - 124E field clinical trial recruitment commencement e,i Travelan® 2H 2022 1H 2023 2H 2023 1H 2024 7 Completed ; 1. Investigational New Drug; 2. Enterotoxigenic E.coli ; 3. controlled human infection model; 4. Institutional Review Board; 5. Naval Medical Research Command; 6. Uniformed Health Services University of the Health Sciences; 7. Clostridioides Difficile; a,e,f,g, i ASX Release links in appendix

IMMURON’S CLINICAL PROGRAMS – OPPORTUNITY ASSESSMENT › Immuron’s development of IMM - 124E (hyperimmune bovine colostrum) as a prescription medication has the potential to address this unmet need › Primary care physicians (PCP)s impressed with clinical efficacy endpoint targets demonstrating > 80% protection against the development of diarrhea. › If base case efficacy targets are reached, IMM - 124E would mostly be used by travelers going to the highest risk areas (e.g., rural Central America/Asia/Africa). › Based on the estimated market size and pricing, the base case yearly revenue in USA for IMM - 124E is projected at US$102M . › Reaching higher efficacy goals could broaden use. L u C m o a r n p i o t y r a * t e Op p ortuni t y Assessment for IMM - 124E › Infectious disease experts reacted favorably to the IMM - 529 MOA, and its unique ability to target three elements of the rCDI infection – the spores, vegetative cells, and Toxin B › Non - microbiome approaches (such as IMM - 529) are still appealing to experts, who noted that clinical trial efficacy (reduction in relapse rate) and cost/access will be the key drivers of clinical use in recurrent patients, not mechanism of action › Based on the estimated market size, anticipated payer restrictions, pricing, and competition, base case yearly revenue in USA for IMM - 529 is conservatively projected at US$93M for the target patient population (limited to 2nd recurrence and later based on trial design and payer coverage) › Positioning IMM - 529 earlier than second recurrence and/or efficacy targets could lead to higher uptake. Lu m C o a r n p i t o y r a O t p e po r tu ni t y Assessment for IMM - 529 Market Phase III Phase II Phase I Indication Compound or brand name Traveler’s Diarrhea ETEC challenge IMM - 124E - Travelan® Clostridioides difficile Infection & Recurrence IMM - 529 Lumanity, a leading lifescience consulting company: https://lumanity.com/company/our - story/ 8
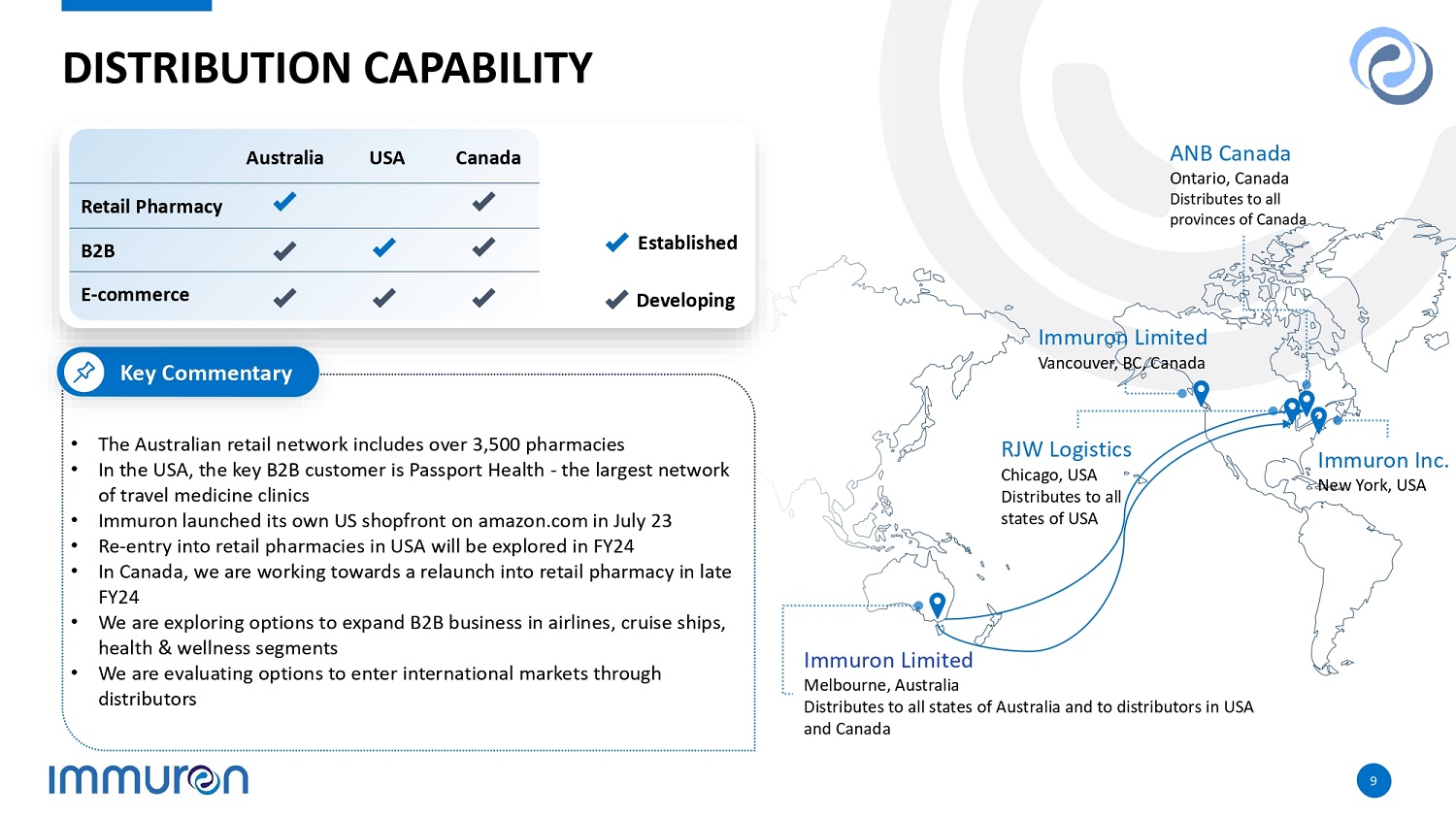
DISTRIBUTION CAPABILITY • The Australian retail network includes over 3,500 pharmacies • In the USA, the key B2B customer is Passport Health - the largest network of travel medicine clinics • Immuron launched its own US shopfront on amazon.com in July 23 • Re - entry into retail pharmacies in USA will be explored in FY24 • In Canada, we are working towards a relaunch into retail pharmacy in late FY24 • We are exploring options to expand B2B business in airlines, cruise ships, health & wellness segments • We are evaluating options to enter international markets through distributors Key Commentary ANB Canada Ontario, Canada Distributes to all provinces of Canada Immuron Limited Vancouver, BC, Canada Immuron Inc. New York, USA RJW Logistics Chicago, USA Distributes to all states of USA Immuron Limited Melbourne, Australia Distributes to all states of Australia and to distributors in USA and Canada Established Developing Canada USA Australia Retail Pharmacy B2B E - commerce 9

BUSINESS POSITIONED FOR ORGANIC GROWTH AND NEW M&A STRATEGY 1. Travelan®: • Sales expansion across target geographies • Growth in distribution network and sales & marketing initiatives • Product development (new formulations including once daily dosing) e.g. FDA approval 2. Protectyn®: • Sales expansion across target geographies • Growth in distribution network and sales & marketing initiatives • Product development and broader applications 02 04 05 Organic Growth Strategy Focus on commercialised products and near - term development extensions, including: M&A Strategy By pursuing growth through M&A of a fragmented market, IMC believes that it will be able to increase market geographies, sales channels and penetration driving revenue growth and ultimately shareholder value Our M&A Key Criteria focusses on: 01 Expand market verticals & product offering Expand existing customer base 03 Cost & Earnings Synergies Strength of IP and Management Distribution network and sales & marketing by each product 10

EMAIL: STEVE@IMMURON.COM PHONE: AUSTRALIA: +613 8892 4854 11 STEVEN LYDEAMORE CHIEF EXECUTIVE OFFICER IMMURON LIMITED CONTACT INFORMATION:

SCIENTIFIC REFERENCES Travelan® (IMM - 124E) Scandinavian Journal of Gastroenterology, 46:7 - 8, 862 - 868, DOI: 10.3109/00365521.2011.574726 Travelan® has been shown to reduce both the incidence and severity of ETEC - induced diarrhea in up to 90% of volunteers Immuron Limited, 29 April, 2011 Travelan as a broad Spectrum anti - bacterial US Department of Defense, Armed Forces Research Institute of Medical Sciences (AFRIM), 4 September, 2019 Travelan® demonstrates broad reactivity to Vibrio cholera strains from Southeast Asia indicating broad potential for prevention of traveler’s diarrhea US Department of Defense, Armed Forces Research Institute of Medical Sciences (AFRIM), 5 September, 2018 Travelan® prevented clinical shigellosis (bacillary dysentery) in 75% of Travelan® treated animals compared to placebo and demonstrated a significant clinical benefit US Department of Defense, Armed Forces Research Institute of Medical Sciences (AFRIM), 30 January, 2017 Travelan® able to bind and was reactive to 60 clinical isolates of each bacteria, Campylobacter, ETEC, and Shigella Islam et al., 2020. Submitted to mSphere, American Society for Microbiology Efficacy of hyperimmune bovine colostrum against shigellosis in rhesus macaque (Macaca mulatta), and bioactivity of HBC against common enteric pathogens Clin Vaccine Immunol 24:e00186 - 16. https://doi.org/10.1128/CVI.00186 - 16 Bioactive Immune Components of Travelan® Rachele Gore, Mitra Mohsenipour, Jennifer L Wood, Gayathri K Balasuriya, Elisa L Hill - Yardin, Ashley E Franks Hyperimmune bovine colostrum containing lipopolysaccharide antibodies (IMM - 124E) has a non - detrimental effect on gut microbial communities in unchallenged mice Journal of Crohn’s and Colitis, Volume 13, Issue 6, June 2019, Pages 785 – 797, https://doi.org/10.1093/ecco - jcc/jjy213 Administration of the Hyper - immune Bovine Colostrum Extract IMM - 124E Ameliorates Experimental Murine Colitis IMM - 529 Sci Rep 7, 3665 (2017). https://doi.org/10.1038/s41598 - 017 - 03982 - 5 Bovine antibodies targeting primary and recurrent Clostridium difficile disease are a potent antibiotic alternative 12

ASX RELEASES REFERENCED ASX Release Slide statement Ref Immuron Receives FDA Approval for Travelan IND Application Travelan® (IMM - 124E) IND filed with and approved by FDA a Immuron awarded A$6.2 million US DoD funding for Travelan US Department of Defense grant of US$4.45 million to examine a dosing regimen for Travelan® more suited for use by the military b AGM Presentation Market evaluation by Lumanity confirms the Traveler’s Diarrhea market opportunity for IMM - 124E (Travelan®) c AGM Presentation Market evaluation by Lumanity confirms the Clostridioides difficile market opportunity for IMM - 529 d US DOD Travelan Clinical Recruitment Milestone Travelan® - Uniformed Services University has recruited more than 20% of participants in a randomized clinical trial with Travelan® to evaluate the effectiveness for prophylaxis during deployment or travel to a high traveler’s diarrhea risk region e FDA Removed Clinical Hold on Campylobacter ETEC Therapeutic Immuron Announces FDA Removed Clinical Hold on New Campylobacter ETEC Therapeutic Paves Way for Clinical Trial Initiation f Immuron Initiates Recruitment of Travelan Clinical Study Immuron Initiates Recruitment of Travelan® Clinical Study g Letter to Shareholders Immuron Board approves IMM - 529 cGMP manufacturing and FDA pre - IND submission h Immuron enrols patients in Travelan clinical study Immuron Announces First Patients Enrolled in Travelan® Clinical Study i 13
Immuron (NASDAQ:IMRN)
過去 株価チャート
から 4 2024 まで 5 2024

Immuron (NASDAQ:IMRN)
過去 株価チャート
から 5 2023 まで 5 2024
