| PROSPECTUS |
|
Filed Pursuant to Rule 424(b)(3) |
| |
|
Registration No. 333-276588 |

Aditxt, Inc.
Up to 3,785,569 Shares of
Common Stock
Pursuant to this prospectus,
the selling stockholder identified herein (the “Selling Stockholder”) is offering on a resale basis an aggregate of 3,785,569
shares of common stock, par value $0.001 per share (the “Common Stock”) of Aditxt, Inc. (the “Company,” “we,”
“us” or “our”) consisting of (i) 1,237,114 shares of Common Stock that are issuable upon exercise of pre-funded
warrants (the “Pre-Funded Warrants”) issued pursuant to a securities purchase agreement entered into by and between us and
the Selling Stockholder dated December 29, 2023 (the “Purchase Agreement”), (ii) up to 2,474,228 shares of Common Stock issuable
upon exercise of warrants (the “Common Warrants”) issued pursuant to the Purchase Agreement, and (iii) up to 74,227 shares
of Common Stock issuable upon exercise of warrants (the “Placement Agent Warrants”, together with the Pre-Funded Warrants
and the Common Warrants, the “Warrants”) issued pursuant to the engagement letter dated as of December 3, 2023, as amended
on December 29, 2023, by and between the Company and H.C. Wainwright & Co., LLC (the “Placement Agent”).
We will not receive any of the
proceeds from the sale by the Selling Stockholders of the Common Stock. Upon any exercise of the Warrants by payment of cash, however,
we will receive the exercise price of the Warrants, which, if exercised in cash with respect to the 3,785,569 shares of Common Stock
offered hereby, would result in gross proceeds to us of approximately $12.3 million. However, we cannot predict when and in what amounts
or if the Warrants will be exercised by payments of cash and it is possible that the Warrants may expire and never be exercised, in which
case we would not receive any cash proceeds.
The Selling Stockholders may
sell or otherwise dispose of the Common Stock covered by this prospectus in a number of different ways and at varying prices. We provide
more information about how the Selling Stockholders may sell or otherwise dispose of the Common Stock covered by this prospectus in the
section entitled “Plan of Distribution” on page 106. Discounts, concessions, commissions and similar selling expenses attributable
to the sale of Common Stock covered by this prospectus will be borne by the Selling Stockholders. We will pay all expenses (other than
discounts, concessions, commissions and similar selling expenses) relating to the registration of the Common Stock with the Securities
and Exchange Commission (the “SEC”).
Our Common stock is listed
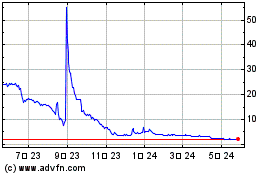
on The Nasdaq Capital Market under the symbol “ADTX”. On June 5, 2024, the closing price as reported on The Nasdaq Capital
Market was $1.99 per share. There is no established public trading market for the Pre-Funded Warrants and the Common Warrants, and we
do not expect a market to develop. Without an active trading market, the liquidity of the Pre-Funded Warrants and Warrants will be limited.
In addition, we do not intend to list the Pre-Funded Warrants or the Warrants on The Nasdaq Capital Market, any other national securities
exchange or any other trading system.
We are an “emerging growth
company” under the federal securities laws and, as such, are subject to reduced public company reporting requirements.
Investing in our Common Stock
involves a high degree of risk. See “Risk Factors” beginning on page 21 of this prospectus.
Neither the Securities and
Exchange Commission nor any state securities commission has approved or disapproved of these securities or determined if this prospectus
is truthful or complete. Any representation to the contrary is a criminal offense.
The date of this prospectus is June 13, 2024
TABLE OF CONTENTS
ABOUT
THIS PROSPECTUS
This prospectus relates to the
resale by the Selling Stockholders identified in this prospectus under the caption “Selling Stockholders,” from time to time,
of up to an aggregate of 3,785,569 shares of Common Stock. We are not selling any shares of Common Stock under this prospectus, and we
will not receive any proceeds from the sale of shares of Common Stock offered hereby by the Selling Stockholders, although we may receive
cash from the exercise of the Warrants.
You should rely only on the information
provided in this prospectus. We have not authorized anyone to provide you with any other information and we take no responsibility for,
and can provide no assurances as to the reliability of, any other information that others may give you. The information contained in
this prospectus speaks only as of the date set forth on the cover page and may not reflect subsequent changes in our business, financial
condition, results of operations and prospects.
We are not, and the Selling Stockholders
are not, making offers to sell these securities in any jurisdiction in which an offer or solicitation is not authorized or permitted
or in which the person making such offer or solicitation is not qualified to do so or to any person to whom it is unlawful to make such
an offer or solicitation. You should read this entire prospectus before making an investment decision.
CAUTIONARY
STATEMENT REGARDING FORWARD-LOOKING STATEMENTS
This prospectus contains forward-looking
statements, which reflect the views of our management with respect to future events and financial performance. These forward-looking
statements are subject to a number of uncertainties and other factors that could cause actual results to differ materially from such
statements. Forward-looking statements are identified by words such as “anticipates,” “believes,” “estimates,”
“expects,” “intends,” “plans,” “projects,” “targets,” and similar expressions.
Such forward-looking statements may be contained in the sections “Risk Factors,” and “Business,” among other
places in this prospectus. Readers are cautioned not to place undue reliance on these forward-looking statements, which are based on
the information available to management at this time and which speak only as of this date. We undertake no obligation to update or revise
any forward-looking statements, whether as a result of new information, future events or otherwise. For a discussion of some of the factors
that may cause actual results to differ materially from those suggested by the forward-looking statements, please read carefully the
information under “Risk Factors.”
The identification in this document
of factors that may affect future performance and the accuracy of forward-looking statements is meant to be illustrative and by no means
exhaustive. All forward-looking statements should be evaluated with the understanding of their inherent uncertainty. You may rely only
on the information contained in this prospectus.
We have not authorized anyone
to provide information different from that contained in this prospectus. Neither the delivery of this prospectus nor the sale of our
Common Stock means that information contained in this prospectus is correct after the date of this prospectus. This prospectus is not
an offer to sell or solicitation of an offer to buy these securities in any circumstances under which the offer or solicitation is unlawful.
PROSPECTUS
SUMMARY
This summary highlights
certain information about us, this offering and selected information contained elsewhere in this prospectus. This summary is not complete
and does not contain all the information that you should consider before deciding whether to invest in our securities. For a more complete
understanding of our company and this offering, we encourage you to read and consider carefully this entire prospectus, including the
information contained under the heading “Risk Factors,” “Management’s Discussion and Analysis of Financial condition
and Results of Operations,” and our financial statements and related notes included elsewhere in this prospectus. In
this prospectus, unless context requires otherwise, references to “we,” “us,” “our,” “ADTX,”
“Aditxt” or “the Company” refer to Aditxt, Inc.
Overview and Mission
We believe the world needs—and
deserves—a new approach to innovating that harnesses the power of large groups of stakeholders who work together to ensure that
the most promising innovations make it into the hands of people who need them most.
We were incorporated in the
State of Delaware on September 28, 2017, and our headquarters are in Mountain View, California. The company was founded with a mission
of bringing stakeholders together, to transform promising innovations into products and services that could address some of the most
challenging needs. The socialization of innovation through engaging stakeholders in every aspect of it, is key to transforming more innovations,
more rapidly, and more efficiently.
At inception, the first innovation
we took on was an immune modulation technology titled ADI/Adimune with a focus on prolonging life and enhancing life quality of patients
that have undergone organ transplants. Since then, we expanded our portfolio of innovations, and we continue to evaluate a variety of
promising health innovations.
Our Model
Aditxt is not about a single
idea or a single molecule. It is about making sure the right innovation is made possible. Our business model has three main components
as follows:
| |
(1) |
Securing an Innovation:
Our process begins with identifying and securing innovations through licensing or acquisition of an innovation asset. Assets come
from a variety of sources including research institutions, government agencies, and private organizations. |
| |
(2) |
Growing an Innovation:
Once an innovation is secured, we surround it with activation resources that take a systemized approach to bringing that idea to
life. Our activation resources include innovation, operations, commercialization, finance, content and engagement, personnel, and
administration. |
| |
(3) |
Monetizing an Innovation:
Our goal is for each innovation to become commercial-stage and financially and operationally self-sustainable, to create shareholder
value. |
We engage various stakeholders
for each of our programs on every level. This includes identifying researchers and research institution partners, such as Stanford University;
leading health institutions to get critical trials underway, such as Mayo Clinic; manufacturing partners who enable us to take innovations
from preclinical to clinical; municipalities and governments, such as the city of Richmond and the state of Virginia and public health
agencies who work with us to launch our program, Pearsanta’s laboratory; and thousands of shareholders around the globe. We seek
to enable promising innovation to become purposeful products that have the power to change lives.
Our Value Proposition
We believe that far too often,
promising treatment or technology does not reach commercialization due to lack of expertise, key resources, or efficiency. As a result,
potentially life-changing and lifesaving treatments are not available to the individuals who so desperately need them.
Aditxt seeks to bring the holistic
concept of an efficient, socialized ecosystem for advancing and accelerating innovations. Our process: We seek to license or acquire
promising innovations. We will then form and build out a subsidiary around each innovation and support the subsidiaries through innovation,
operation, commercialization, content and engagement, finance, personnel, and administration to thrive and grow as a successful, monetizable
business.
Since our inception, we have
built infrastructure consisting of innovation, operation, commercialization, content and engagement, finance, personnel, and administration,
to support the rapid transformation of untapped innovations. Each of the main components of our infrastructure has established global
access to partnerships with industry leaders, top-rated research and medical institutions, universities, manufacturing and distribution
companies, and critical infrastructure such as CLIA-certified state-of-the art labs and GMP manufacturing.
The Shifting Landscape of Innovation
Innovation
in general, and health innovations specifically, require significant resources. The convergence of biotech, high-tech, and media offers
new possibilities of accelerating breakthrough innovations faster and more efficiently. This approach reflects our mission of “Making
Promising Innovations Possible, Together”.
People deserve innovative solutions,
which have never been more within reach. We believe the best idea, best product and the best solution will come from creating an ecosystem
where all stakeholders, such as vendors, customers, municipalities, and shareholders contribute. When we disrupt the way we’re
innovating, through our collaborative model, we believe we can move faster and more efficiently to activate viable solutions that have
the potential to make a measurable impact.
Our Growth Strategy
We believe that the era of precision
and personalized medicine is here and that people around the globe would benefit from health diagnostics and treatments that more accurately
pinpoint the problems and more precisely treat the condition. In addition to our current programs, Adimune and Pearsanta, we look to
bring in future health innovations in the areas of software and AI, medical devices, therapeutics, and other technologies that take a
fundamentally different approach to health because they prioritize personalized precision medicine, timely disease root cause analysis,
and targeted treatments.
Year over year, we plan to continue
building our infrastructure and securing more personalized and precision health innovations that align with our mission. These opportunities
may come in different forms such as IP, an early-stage company, or a late-stage company. We will continue to scale our systemized approach
to the innovation process, making large-scale automation and enterprise systems available to our portfolio companies at every stage of
their growth. Specifically, certain subsidiaries will need to grow through further M&A activities, operational infrastructure implementation,
and development or acquisition of critical technologies.
Our Team
Aditxt is led by an entrepreneurial
team with passion for transforming promising innovations into successful businesses. Our leadership come from a variety of different
industries, with collective expertise in founding startup innovation companies, developing and marketing biopharmaceutical and diagnostic
products, designing clinical trials, manufacturing, and management of private and public companies. We have deep experience in identifying
and accessing promising health innovations and developing them into products and services with the ability to scale. We understand the
capital markets, both public and private, as well as M&A and facilitating complex IPOs.
The following are profiles of
three subsidiaries we have formed, including the terms of the intellectual property licenses that have been sublicensed from Aditxt to
help build each of the businesses.
THE ADITXT PROGRAMS
ADIMUNE, INC.
Formed in January 2023, Adimune™,
Inc. (“Adimune”) is focused on leading our immune modulation therapeutic programs. Adimune’s proprietary immune modulation
product Apoptotic DNA Immunotherapy™, or ADI-100™, utilizes a novel approach that mimics the way our bodies naturally induce
tolerance to our own tissues. It includes two DNA molecules designed to deliver signals to induce tolerance. ADI-100 has been successfully
tested in several preclinical models (e.g., skin grafting, psoriasis, type 1 diabetes, multiple sclerosis).
In May 2023, Adimune entered
into a clinical trial agreement with Mayo Clinic to advance clinical studies targeting autoimmune diseases of the central nervous system
(“CNS”) with the initial focus on the rare, but debilitating, autoimmune disease Stiff Person Syndrome (“SPS”).
According to the National Organization of Rare Diseases, the exact incidence and prevalence of SPS is unknown; however, one estimate
places the incidence at approximately one in one million individuals in the general population.
Pending approval by the International
Review Board, a human trial for SPS is expected get underway in the second half of 2023 or the first half of 2024 with enrollment of
10-15 patients, some of whom may also have type 1 diabetes. ADI-100 will initially be tested for safety and efficacy. ADI-100 is designed
to tolerize against an antigen known as glutamic acid decarboxylase (“GAD”), which is implicated in type-1 diabetes, psoriasis,
and in many autoimmune diseases of the CNS.
Background
The
discovery of immunosuppressive (anti-rejection and monoclonal) drugs over 40 years ago has made possible life-saving organ transplantation
procedures and blocking of unwanted immune responses in autoimmune diseases. However, immune suppression leads to significant undesirable
side effects, such as increased susceptibility to life-threatening infections and cancers, because it indiscriminately and broadly suppresses
immune function throughout the body. While the use of these drugs has been justifiable because they prevent or delay organ rejection,
their use for treatment of autoimmune diseases and allergies may not be acceptable because of the aforementioned side effects. Furthermore,
often transplanted organs ultimately fail despite the use of immune suppression, and about 40% of transplanted organs survive no more
than five years.
Through
Aditxt, Adimune has the right of use to the exclusive worldwide license for commercializing ADI nucleic acid-based technology (which
is currently at the pre-clinical stage) from Loma Linda University. ADI uses a novel approach that mimics the way the body naturally
induces tolerance to our own tissues (“therapeutically induced immune tolerance”). While immune suppression requires continuous
administration to prevent rejection of a transplanted organ, induction of tolerance has the potential to retrain the immune system to
accept the organ for longer periods of time. ADI may allow patients to live with transplanted organs with significantly reduced immune
suppression. ADI is a technology platform which we believe can be engineered to address a wide variety of indications.
Advantages
ADI™
is a nucleic acid-based technology (e.g., DNA-based), which we believe selectively suppresses only those immune cells involved
in attacking or rejecting self and transplanted tissues and organs. It does so by tapping into the body’s natural process of cell
turnover (i.e., apoptosis) to retrain the immune system to stop unwanted attacks on self or transplanted tissues. Apoptosis is a natural
process used by the body to clear dying cells and to allow recognition and tolerance to self-tissues. ADI triggers this process by enabling
the cells of the immune system to recognize the targeted tissues as “self.” Conceptually, it is designed to retrain the immune
system to accept the tissues, similar to how natural apoptosis reminds our immune system to be tolerant to our own “self”
tissues.
While various groups have promoted
tolerance through cell therapies and ex vivo manipulation of patient cells (i.e., takes place outside the body), to
our knowledge, we will be unique in our approach of using in-body induction of apoptosis to promote tolerance to specific tissues. In
addition, ADI treatment itself will not require additional hospitalization but only an injection of minute amounts of the therapeutic
drug into the skin.
Moreover,
preclinical studies have demonstrated that ADI treatment significantly and substantially prolongs graft survival, in addition to successfully
“reversing” other established immune-mediated inflammatory processes.
License Agreement with Loma Linda University (“LLU”)
On March 15, 2018, we entered
into a License Agreement with LLU, which was subsequently amended on July 1, 2020. Pursuant to the LLU License Agreement, we obtained
the exclusive royalty-bearing worldwide license to all intellectual property, including patents, technical information, trade secrets,
proprietary rights, technology, know-how, data, formulas, drawings, and specifications, owned or controlled by LLU and/or any of its
affiliates (the “LLU Patent and Technology Rights”) and related to therapy for immune-mediated inflammatory diseases (the
Adi™ technology). In consideration for the LLU License Agreement, we issued 625 shares of Common Stock to LLU.
PEARSANTA, INC.
Formed in January 2023, our
subsidiary Pearsanta™, Inc. (“Pearsanta”) seeks to take personalized medicine to a whole new level by delivering “Health
by the Numbers.” On November 22, 2023, Pearsanta entered into an assignment agreement with FirstVitals LLC, an entity controlled
by Pearsanta’s CEO, Ernie Lee (“FirstVitals”), pursuant to which FirstVitals assigned its rights in certain intellectual
property and website domain to Pearsanta in consideration of the issuance of 500,000 shares of Pearsanta common stock to FirstVitals.
On December 18, 2023, the board of directors of Pearsanta adopted the Pearsanta 2023 Omnibus Equity Incentive Plan (the “Pearsanta
Omnibus Incentive Plan”), pursuant to which it reserved 15 million shares of common stock of Pearsanta for future issuance under
the Pearsanta Omnibus Incentive Plan and the Pearsanta 2023 Parent Service Provider Equity Incentive Plan (the “Pearsanta Parent
Service Provider Plan”) and approved the issuance of 9.32 million shares of Pearsanta common stock under the Pearsanta Parent Service
Provider Plan.
Since its founding, Pearsanta
has been building the platform for enabling our vision of lab quality testing, anytime, anywhere. Our plan for Pearsanta’s platform
is for it to be the transactional backbone for sample collection, sample processing (on- and off-site), and reporting. This will require
the development and convergence of multiple components developed by Pearsanta, or through transactions with third parties, including
collection devices, “lab-on-a-chip” technologies, Lab Developed Test (LDT) assays, a data-driven analysis engine, and telemedicine.
According to a comprehensive research report by Market Research Future, the clinical and consumer diagnostic market is estimated to hit
$429.3 billion by 2030.
We believe that timely and personalized
testing enables far more informed treatment decisions. Pearsanta’s platform is being developed as a seamless digital healthcare
solution. This platform will integrate at-location sample collection, Point-of-Care (“POC”) and LDT assays, and an analytical
reporting engine, with telemedicine-enabled visits with licensed physicians to review test results and, if necessary, order a prescription.
Pearsanta’s goal of extending its platform to enable consumers to monitor their health more proactively as the goal is to provide
a more complete picture about someone’s dynamic health status, factoring in genetic makeup and their response to medication. The
POC component of Pearsanta would enable diagnostic testing at-home, at work, in pharmacies, and more to generate results quickly so that
an individual can access necessary treatment faster. With certain infections, prescribing the most effective treatment according to one’s
numbers can prevent hospital emergency room admissions and potentially life-threatening consequences.
Examples of indication-focused
tests for the Test2Treat platform will include the evaluation for advanced urinary tract infections (“UTIs”), COVID-19/flu/respiratory
syncytial virus, sexually transmitted infections, gut health, pharmacogenomics (i.e., how your genes affect the way your body responds
to certain therapeutics), and sepsis. We believe that these offerings are novel and needed as the current standard of care using broad
spectrum antibiotic treatment can be ineffective and potentially life-threatening. For example, improperly prescribed antibiotics may
approach 50% of outpatient cases. Further, according to an article published in Physician’s Weekly, only 1% of board-certified
critical care medicine physicians are trained in infectious disease.
Licensed Technologies – AditxtScoreTM
We intend
to sublicense to Pearsanta an exclusive worldwide sub-license for commercializing the AditxtScore™ technology which provides a
personalized comprehensive profile of the immune system. AditxtScore is intended to detect individual immune responses to viruses, bacteria,
peptides, drugs, supplements, bone marrow and solid organ transplants, and cancer. It has broad applicability to many other agents of
clinical interest impacting the immune system, including those not yet identified such as emerging infectious agents.
AditxtScore is being designed
to enable individuals and their healthcare providers to understand, manage and monitor their immune profiles and to stay informed about
attacks on or by their immune system. We believe AditxtScore can also assist the medical community and individuals by being able to anticipate
the immune system’s potential response to viruses, bacteria, allergens, and foreign tissues such as transplanted organs. This technology
may be able to serve as a warning signal, thereby allowing for more time to respond appropriately. Its advantages include the ability
to provide simple, rapid, accurate, high throughput assays that can be multiplexed to determine the immune status with respect to several
factors simultaneously, in approximately 3-16 hours. In addition, it can determine and differentiate between distinct types of cellular
and humoral immune responses (e.g., T and B cells and other cell types). It also provides for simultaneous monitoring of cell activation
and levels of cytokine release (i.e., cytokine storms).
We are
actively involved in the regulatory approval process for AditxtScore assays for clinical use and securing manufacturing, marketing, and
distribution partnerships for application in the various markets. To obtain regulatory approval to use AditxtScore as a clinical assay,
we have conducted validation studies to evaluate its performance in detection of antibodies and plan to continue conducting additional
validation studies for new applications in autoimmune diseases.
Advantages
The
sophistication of the AditxtScore technology includes the following:
| |
● |
greater sensitivity/specificity. |
| |
● |
20-fold higher dynamic
range, greatly reducing signal to noise compared to conventional assays. |
| |
● |
ability to customize assays
and multiplex a large number of analytes with speed and efficiency. |
| |
● |
ability to test for cellular
immune responses (i.e., T and B cells and cytokines). |
| |
● |
proprietary reporting algorithm. |
License Agreement with Leland Stanford Junior University (“Stanford”)
On February 3, 2020, we entered
into an exclusive license agreement (the “February 2020 License Agreement”) with Stanford with regard to a patent concerning
a method for detection and measurement of specific cellular responses. Pursuant to the February 2020 License Agreement, we received an
exclusive worldwide license to Stanford’s patent with regard to use, import, offer, and sale of Licensed Products (as defined in
the agreement). The license to the patented technology is exclusive, including the right to sublicense, beginning on the effective date
of the agreement, and ending when the patent expires. Under the exclusivity agreement, we acknowledged that Stanford had already granted
a non-exclusive license in the Nonexclusive Field of Use, under the Licensed Patents in the Licensed Field of Use in the Licensed Territory
(as those terms are defined in the “February 2020 License Agreement”). However, Stanford agreed not to grant further licenses
under the Licensed Patents in the Licensed Field of Use in the Licensed Territory. On December 29, 2021, we entered into an amendment
to the February 2020 License Agreement which extended our exclusive right to license the technology deployed in AditxtScoreTM and
securing worldwide exclusivity in all fields of use of the licensed technology.
ADIVIR, INC.
Formed in April of 2023, Adivir™,
Inc., is Aditxt’s most recently formed wholly owned subsidiary, dedicated to the clinical and commercial development efforts of
innovative antiviral products. These products have the potential to address a wide range of infectious diseases, including those that
currently lack viable treatment options.
Background
On April
18, 2023, we entered into an Asset Purchase Agreement (the “Asset Purchase Agreement”) with Cellvera Global Holdings LLC
(“Cellvera Global”), Cellvera Holdings Ltd. (“BVI Holdco”), Cellvera, Ltd. (“Cellvera Ltd.”), Cellvera
Development LLC (“Cellvera Development” and together with Cellvera Global, BVI Holdco, Cellvera Ltd. and Cellvera Development
(the “Sellers”), AiPharma Group Ltd. (“Seller Owner” and collectively with the Sellers, “Cellvera”),
and the legal representative of Cellvera, pursuant to which, the Company will purchase Cellvera’s 50% ownership interest in G Response
Aid FZE (“GRA”), certain other intellectual property and all goodwill related thereto (the “Acquired Assets”). Unless
expressly stated otherwise herein, capitalized terms used but not defined herein have the meanings ascribed to them in the Asset Purchase
Agreement. Pursuant to the Asset Purchase Agreement, the consideration for the Acquired Assets consists of (A) $24.5 million,
comprised of: (i) the forgiveness of the Company’s $14.5 million loan to Cellvera Global, and (ii) approximately $10 million in
cash, and (B) future revenue sharing payments for a term of seven years. GRA holds an exclusive, worldwide license for the antiviral
medication, Avigan® 200mg, excluding Japan, China and Russia. The other 50% interest in GRA is held by Agility, Inc. (“Agility”).
Additionally,
upon the closing, the Share Exchange Agreement previously entered into as of December 28, 2021, between Cellvera Global Holdings, LLC
f/k/a AiPharma Global Holdings, LLC (together with other affiliates and subsidiaries) and the Company, and all other related agreements
will be terminated.
The
obligations of the Company to consummate the Closing are subject to the satisfaction or waiver, at or prior to the Closing of certain
conditions, including but not limited to, the following:
| |
(i) |
Satisfactory completion
of due diligence; |
| |
(ii) |
Completion by the Company
of financing sufficient to consummate the transactions contemplated by the Asset Purchase Agreement; |
| |
(iii) |
Receipt by the Company
of all required Consents from Governmental Bodies for the Acquisition, including but not limited to, any consents required to complete
the transfer and assignment of Cellvera’s membership interests in GRA; |
| |
(iv) |
Receipt of executed payoff
letters reflecting the amount required to be fully pay all of each of Seller’s and Seller Owner’s Debt to be paid at
Closing; |
| |
(v) |
Receipt by the Company
of a release from Agility; |
| |
(vi) |
Execution of an agreement
acceptable to the Company with respect to the acquisition by the Company of certain intellectual property presently held by a third
party; |
| |
(vii) |
Execution of an amendment
to an asset purchase agreement previously entered into by Cellvera with a third party that effectively grants the Company the rights
to acquire the intellectual property from the third party under such agreement; |
| |
(viii) |
Receipt of a fairness opinion
by the Company with respect to the transactions contemplated by the Asset Purchase Agreement; and |
| |
(ix) |
Receipt by the Company
from the Seller Owner of written consent, whether through its official liquidator or the Board of Directors of Seller Owner, to the
sale and purchase of the Acquired Assets and Assumed Liabilities pursuant to the Assert Purchase Agreement. |
There
can be no assurance that the conditions to closing will be satisfied or that the proposed acquisition will be completed as proposed or
at all.
Our commitment to building our
antiviral portfolio is strategic and timely. We believe that there has never has there been a more important time to address the growing
global need to uncover new treatments or commercialize existing ones that treat life-threatening global viral infections.
Recent Developments
Promissory Note
On
April 10, 2024, Sixth Borough Capital Fund, LP (“Sixth Borough”) loaned $230,000 to Aditxt, Inc. The loan was evidenced
by an unsecured promissory note (the “Sixth Borough April Note”). Pursuant to the terms of the Sixth Borough April Note,
it will accrue interest at the Prime rate of eight and one-half percent (8.5%) per annum and is due on the earlier of April 19, 2024
or an event of default, as defined therein. On May 9, 2024, at which point the balance of the loan was $35,256, Sixth Borough loaned
an additional $20,000 to the Company bringing the balance of the loan to $55,256.03.
Lease Default
On
May 10, 2024, the Company received written notice (the “Lease Default Notice”) from LS Biotech Eight, LLC (the
“Landlord”) that the Company was in violation of its obligation to (i) pay Base Rent (as defined in the Lease) and
Additional Rent (as defined in the Lease) in the amount of $431,182.32 in the aggregate, together with administrative charges and interest,
as well as (ii) replenish the Security Deposit (as defined in the Lease) in the amount of $159,375.00, all as required under that certain
Lease Agreement dated as of May 4, 2021 by and between the Landlord and the Company (the “Lease”). Pursuant to the
Lease Default Notice, the Landlord has demanded that a payment of $590,557.31 plus administrative charges and interest, which
shall accrue at the Default Rate (as defined in the Lease) be made no later than May 17, 2024.
The
Company is working with the Landlord to come to an amicable resolution. However, no assurance can be given that the parties will reach
an amicable resolution on a timely basis, on favorable terms, or at all.
Equity Line of Credit
On
May 2, 2024, we entered into a Common Stock Purchase Agreement (the “ELOC Purchase Agreement”) with an equity line
investor (the “ELOC Investor”), pursuant to which the ELOC Investor has agreed to purchase from us, at our direction
from time to time, in its sole discretion, from and after the date effective date of the Registration Statement (as defined below) and
until the termination of the ELOC Purchase Agreement in accordance with the terms thereof, shares of our common stock having a total
maximum aggregate purchase price of $150,000,000 (the “ELOC Purchase Shares”), upon the terms and subject to the conditions
and limitations set forth in the ELOC Purchase Agreement.
In
connection with the ELOC Purchase Agreement, we also entered into a Registration Rights Agreement with the Investor (the “ELOC
Registration Rights Agreement”), pursuant to which the Company agreed to file a registration statement with the Securities
and Exchange Commission covering the resale of the shares of common stock issued to the Investor pursuant to the Purchase Agreement (the
“Registration Statement”) by the later of (i) the 30th calendar day following the closing date, and (ii)
the second business day following Stockholder Approval (defined below).
We may, from time to time
and at our sole discretion, direct the ELOC Investor to purchase shares of our common stock upon the satisfaction of certain
conditions set forth in the ELOC Purchase Agreement at a purchase price per share based on the market price of our common stock at the
time of sale as computed under the ELOC Purchase Agreement. There is no upper limit on the price per share that the ELOC Investor could
be obligated to pay for our common stock under the ELOC Purchase Agreement. We will control the timing and amount of any sales of our
common stock to the ELOC Investor, and the Investor has no right to require us to sell any shares to it under the ELOC
Purchase Agreement. Actual sales of shares of our common stock to the Investor under the ELOC Purchase Agreement will depend
on a variety of factors to be determined by us from time to time, including (among others) market conditions, the trading price of our
common stock and determinations by us as to available and appropriate sources of funding for the Company and our operations. The
ELOC Investor may not assign or transfer its rights and obligations under the ELOC Purchase Agreement.
Under the applicable Nasdaq
rules, in no event may we issue to the Investor under the ELOC Purchase Agreement more than 332,876 shares of common stock,
which number of shares is equal to 19.99% of the shares of the common stock outstanding immediately prior to the execution of the ELOC
Purchase Agreement (the “Exchange Cap”), unless (i) we obtain stockholder approval to issue shares of common stock
in excess of the Exchange Cap in accordance with applicable Nasdaq rules (“Stockholder Approval”), or (ii) the average
price per share paid by the Investor for all of the shares of common stock that we direct the Investor to purchase
from us pursuant to the ELOC Purchase Agreement, if any, equals or exceeds the official closing sale price on the Nasdaq Capital Market
immediately preceding the delivery of the applicable purchase notice to the Investor and (B) the average of the closing sale prices of
our common stock on the Nasdaq Capital Market for the five business days immediately preceding the delivery of such purchase notice.
In all cases, we may not
issue or sell any shares of our common stock to the ELOC Investor under the Purchase Agreement which, when aggregated with
all other shares of our common stock then beneficially owned by the ELOC Investor and its affiliates, would result in the
ELOC Investor beneficially owning more than 4.99% of the outstanding shares of the Company’s common stock.
The net proceeds under the
Purchase Agreement to us will depend on the frequency and prices at which we sell shares of its stock to the Investor. The Company
expects that any proceeds received by it from such sales to the Investor will be used for working capital and general corporate
purposes.
May 2024 Private Placement
On May 2, 2024, we entered
into a Securities Purchase Agreement (the “Purchase Agreement”) with certain accredited investors, pursuant to which
we agreed to issue and sell to such investors in a private placement (the “Private Placement”) (i) an aggregate of
4,186 shares of our Series C-1 Convertible Preferred Stock (the “Series C-1 Preferred Stock”), (ii) an aggregate of
4,186 shares of our Series D-1 Preferred Stock (the “Series D-1 Preferred Stock”), and (iii) warrants (the “Warrants”)
to purchase up to an aggregate of 1,613,092 shares of our common stock.
The Warrants are exercisable
commencing six months following the initial issuance date at an initial exercise price of $2.47 per share and expire five years from
the date of issuance.
On May 2, 2024, in connection
with the Purchase Agreement, we entered into a Registration Rights Agreement with the investors (the “Registration Rights Agreement”),
pursuant to which we agreed to prepare and file with the Securities and Exchange Commission (the
“SEC”) a registration statement on Form S-3 (the “Registration Statement”) covering the resale
of the shares of our common stock issuable upon conversion of the Series C-1 Preferred Stock (the “Conversion Shares”)
and upon exercise of the Warrants (the “Warrant Shares”) (i) on the later of (x) the 30th calendar
day after the closing date, or (y) the 2nd business day following the Stockholder Approval Date (as defined in the Purchase
Agreement), with respect to the initial registration statement and (ii) on the date on which we are required to file any additional Registration
Statement pursuant to the terms of the Registration Rights Agreement with respect to any additional Registration Statements that may
be required to be filed by us (the “Filing Deadline”). Pursuant to the Registration Rights Agreement, we are required
to have the initial Registration Statement declared effective by the SEC on the earlier of (x) the 60th calendar day
after the Filing Deadline (or the 90th calendar day after the Filing Deadline if subject to a full review by the SEC),
and (y) the 2nd business day after the date the Company is notified by the SEC that such Registration Statement will
not be reviewed. In the event that we fail to file the Registration Statement by the Filing Deadline, have it declared effective by the
Effectiveness Deadline, or the prospectus contained therein is not available for use or the investor is not otherwise able to sell its
Warrant Shares pursuant to Rule 144, we shall be required to pay the investor an amount equal to 2% of such investor’s Purchase
Price (as defined in the Purchase Agreement) on the date of such failure and on every thirty date anniversary until such failure is cured.
May 2024 Senior Note
On
May 20, 2024, we issued and sold a senior note (the “Senior Note”) to
an accredited investor (the “Holder”) in the original principal amount
of $93,918.75 for a purchase price of $75,135.00, reflecting an original issue discount of $18,783.75. Unless earlier redeemed, the Senior
Note will mature on August 18, 2024 (the “Maturity Date”), subject to
extension at the option of the Holder in certain circumstances as provided in the Senior Note. The Senior Note bears interest at a rate
of 8.5% per annum, which is compounded each calendar month and is payable in arrears on the Maturity Date. The Senior Note contains certain
standard events of default, as defined in the Note (each, an “Event of Default”).
Upon the occurrence of an Event of Default, the interest rate shall be increased to 18% per annum and the Holder may require the Company
to redeem the Senior Note, subject to an additional 5% redemption premium. In addition, if we sell any shares of our common stock pursuant
to any equity line of credit, we are required to redeem in cash a portion of the Senior Note equal to the lesser of (i) the outstanding
amount of the Senior Note, and (ii) 80% of 30% of such equity line proceeds, at a redemption price calculated based upon $1.20 for each
$1.00 of outstanding amount of the Senior Note. The Senior Note also contains an exchange right, which permits the Holder, in its discretion,
to exchange the Senior Note, in whole or in part, for securities to be sold by us in a subsequent placement, subject to certain exceptions
and an additional 20% premium of the amount of the Senior Note exchanged. The Senior Note is a senior, unsecured obligation of the Company,
ranking senior to all other unsecured indebtedness of the Company.
May
2024 Private Placement
On May 24, 2024, we entered
into a Securities Purchase Agreement (the “Securities Purchase Agreement”) with certain accredited investors pursuant
to which we issued and sold senior notes in the aggregate principal amount of $986,379.68 (the “Notes”) maturing on
August 22, 2024, which included the exchange of a previously issued Senior Note in the principal amount of $93,918.75. The Company received
cash proceeds of $775,000 from the sale of the Notes.
Upon an Event of Default
(as defined in the Note), the Note will bear interest at a rate of 14% per annum and the holder shall have the right to require the Company
to redeem the Note at a redemption premium of 125%. In addition, while the Note is outstanding, the Company is required to utilize 100%
of the proceeds from any offering of securities to redeem the Note. Pursuant to the Purchase Agreement, the Company agreed to use commercially
reasonable efforts, including the filing of a registration statement with the U.S. Securities and Exchange Commission (the “SEC”)
for a public offering, to pursue and consummate a financing transaction within 90 days of the closing date. In connection with the issuance
of the Notes, the Company issued an aggregate of 328,468 shares of its common stock (the “Commitment Shares”) as a
commitment fee to the investors. Pursuant to the Purchase Agreement, the Company also agreed to file a registration statement with the
SEC covering the resale of the Commitment Shares as soon as practicable following notice from an investor, and to cause such registration
statement to become effective within 60 days following the filing thereof.
Arrangement Agreement with Appili Therapeutics, Inc.
On April 1, 2024, we entered
into an Arrangement Agreement (the “Arrangement Agreement”) with Adivir, Inc., a wholly owned subsidiary of the Company (“Adivir”)
and Appili Therapetics, Inc. (“Appili”) pursuant to which Adivir will acquire all of
the issued and outstanding Class A common shares of Appili (the “Appili Shares”) on the terms and subject to the conditions
set forth therein. The acquisition of the Appili Shares (the “Arrangement”) will be completed by way of a statutory
plan of arrangement under the Canada Business Corporation Act (the “CBCA”).
At
the effective time of the Arrangement (the “Arrangement Effective Time”), each Appili Share outstanding immediately
prior to the Arrangement Effective Time (other than Appili Shares held by a registered holder of Appili Shares who has validly exercised
such holder’s dissent rights) will be deemed to be assigned and transferred by the holder thereof to Adivir in exchange for (i)
$0.0467 in cash consideration per share for an aggregate cash payment of $5,668,222 (the “Cash Consideration”) and
(ii) 0.002745004 of a share of our common stock or an aggregate of 332,876 shares (the “Consideration Shares” and
together with the Cash Consideration, the “Transaction Consideration”). In connection with the transaction, each outstanding
option and warrant of Appili will be cashed-out based on the implied in-the-money value of the Transaction Consideration, which is expected
to result in an additional aggregate cash payment of approximately $341,000 (based on the number of issued and outstanding options and
warrants and exchange rates as of the date of the Arrangement Agreement).
The respective obligations
of each of the Company, Adivir and Appili to consummate the closing of the Arrangement (the “Closing”) are subject
to the satisfaction or waiver, at or prior to the closing of the following conditions:
| |
(i) |
the Interim Order (as defined
in the Arrangement Agreement) shall have been granted on terms consistent with the Arrangement Agreement and the Interim Order shall
not have been set aside or modified in a manner unacceptable to either party; |
| |
|
|
| |
(ii) |
the Arrangement Resolution
(as defined in the Arrangement Agreement) shall have been adopted by the Appili shareholders at the Appili shareholders’ meeting
in accordance with the Interim Order; |
| |
|
|
| |
(iii) |
the Final Order (as defined
in the Arrangement Agreement) shall have been granted on terms consistent with the Arrangement Agreement and the Final Order shall
not have been set aside or modified in a manner unacceptable to either party; |
| |
|
|
| |
(iv) |
completion of an equity
or debt financing by the Company with minimum gross proceeds of at least $20 million (the “Financing”); |
| |
|
|
| |
(v) |
there shall not be any
law or order which prevents, prohibits or makes the consummation of the Arrangement illegal or otherwise prohibits the consummation
of the Arrangement or the other transaction contemplated by the Arrangement Agreement; |
| |
|
|
| |
(vi) |
the issuance of the Consideration
Shares will be exempt from the registration requirements of: (A) the Securities Act of 1933, as amended (the “Securities
Act”) pursuant to Section 3(a)(10) of the Securities Act; and (B) all applicable U.S. securities laws; |
| |
|
|
| |
(vii) |
the distribution of the
Consideration Shares pursuant to the Arrangement shall be exempt from the prospectus and registration requirements of applicable
securities laws ; and |
| |
|
|
| |
(viii) |
the Consideration Shares
to be issued pursuant to the Arrangement shall, subject to customary conditions, have been approved for listing on the NASDAQ. |
The Company and Adivir
The obligations of the Company
and Adivir to consummate the Closing are subject to the satisfaction or waiver, at or prior to the Closing of certain conditions, including
but not limited to, the following:
| |
(i) |
as qualified in the Arrangement
Agreement, the representations and warranties of Appili are true and correct as of the Effective Time as if made as at and as of
such time; |
| |
|
|
| |
(ii) |
Appili shall have fulfilled
or complied in all material respects with each of its obligations, covenants and agreements contained in the Arrangement Agreement;
|
| |
|
|
| |
(iii) |
dissent rights will not
have been exercised with respect to more than 10% of the issued and outstanding Appili Shares; |
| |
|
|
| |
(iv) |
the LZH Consent Agreement
(as defined in the Arrangement Agreement) continues to remain in full force and effect and enforceable against the parties thereto
and the parties thereto have performed their respective obligations thereunder required to be completed on or before the Closing; |
| |
|
|
| |
(v) |
there shall not have been
a Material Adverse Effect (as defined in the Arrangement Agreement) on Appili; and |
| |
|
|
| |
(vi) |
there is no action or proceeding
pending or threatened by any governmental entity that prohibits the Company from acquiring, holding or exercising full rights of
ownership of the Appili Shares, prevents or materially delays the consummation of the Arrangement, or prohibits or restricts in any
material respect the ownership or operation of the business of the Company or Appili. |
Appili
The obligations of Appili
to consummate the Closing are subject to the satisfaction or waiver, at or prior to the Closing of the following conditions:
| |
(i) |
as qualified in the Arrangement
Agreement, the representations and warranties of the Company and Adivir are true and correct as of the Effective Time as if made
as at and as of such time; |
| |
|
|
| |
(ii) |
the Company and Adivir
shall have fulfilled or complied in all material respects with each of its obligations, covenants and agreements contained in the
Arrangement Agreement; |
| |
|
|
| |
(iii) |
there shall not have been
a Material Adverse Effect on the Company or Adivir; |
| |
|
|
| |
(iv) |
the Third-Party Consents
(as defined in the Arrangement Agreement) shall have been obtained; |
| |
|
|
| |
(v) |
there is no action or proceeding
pending or threatened by any governmental entity that cease trades, enjoins or prohibits the Company’s ability to issue the
Consideration Shares, or prevents or material delays the consummation of the Arrangement; |
| |
|
|
| |
(vi) |
the Company shall have
taken steps to reconstitute the board of directors of Adivir such that it will consist of five directors as of the Effective Date,
one of whom will be the Company Nominee (as defined in the Arrangement Agreement); |
| |
(vii) |
the Company shall have
taken steps to have caused the Appointed Officers (as defined in the Arrangement Agreement) to be duly appointed officers of Adivir; |
| |
|
|
| |
(viii) |
the Company shall satisfy
on the Effective Date the payment in immediately available funds as directed in writing by Appili of not less than 50% of the outstanding
accounts payable of Appili, provided that in the event the Company raises gross proceeds of more than $25 million in the Financing,
any amounts raised by the Company in excess of $25 million shall be first applied by the Company to satisfy any unpaid Closing Company
Payables (as defined in the Arrangement Agreement); and |
| |
|
|
| |
(ix) |
the Company shall have
deposited with the Depositary (as defined in the Arrangement Agreement) the Consideration Shares and the Cash Consideration. |
The Arrangement Agreement
may be terminated at any time prior to the consummation of the Closing by mutual written consent of the Company and Appili.
As further set out in the
Arrangement Agreement, either the Company or Appili may also terminate the Arrangement Agreement prior to the Effective Time if (i) the
Appili shareholders’ meeting is duly convened and held and the Arrangement Resolution is voted on by the Appili shareholders and
not approved by the Appili shareholders as required by the Interim Order; (ii) if any Law (as defined in the Arrangement Agreement) is
enacted, made, issued, rendered, enforced or amended following the date of the Arrangement Agreement that prohibits the Arrangement and
such Law has become final and non-appealable; (iii) the Effective Time does not occur on or prior to July 31, 2024 (the “Outside
Date”); or (iv) the Financing is not completed on or before 5:00 p.m. (ET) on June 30, 2024 or such later date as the parties
may in writing agree.
The Company may terminate
the Arrangement Agreement if prior to the Effective Time: (i) a breach of any representation or warranty or failure to perform any covenant
or agreement on the part of Appili that would cause any Company or Adivir conditions to closing not to be satisfied; (ii) prior to the
approval of the Arrangement Resolution by the Appili shareholders the Appili board of directors makes a Company Change in Recommendation
or enters into a written agreement to effect a Company Acquisition Proposal; (iii) there has been a Material Adverse Effect in respect
of Appili which is incapable of being cured on or before the Outside Date; (iv) Appili is in material breach of any of the non-solicitation
or match provisions of the Arrangement Agreement; or (v) any of the conditions to closing for the mutual benefit of the parties or for
the benefit of the Company or Adivir cannot be satisfied prior to the Outside Date and such breach or failure is incapable of being cured,
provided that such breach or failure did not result from any action of the Company or Adivir.
Appili may terminate the
Arrangement Agreement if prior to the Effective Time (i) a breach of any representation or warranty or failure to perform any covenant
or agreement on the part of the Company that would cause any Appili conditions to closing not to be satisfied; (ii) prior to the approval
of the Arrangement Resolution by the Appili shareholders if the Appili board of directors makes a Company Change in Recommendation or
enters into a written agreement to effect a Company Superior Proposal; (iii) the Company does not provide the Depositary with sufficient
Consideration to complete the transactions contemplated by the Arrangement Agreement; (iv) there has occurred a Material Adverse Effect
in respect of the Company which is incapable of being cured prior to the Outside Date; (v) any of the conditions to closing for the mutual
benefit of the parties or for the benefit of Appili cannot be satisfied prior to the Outside Date and such breach or failure is incapable
of being cured, provided that such breach or failure did not result from any action of Appili; or (vi) the Company does not take all
actions within its control that are necessary to remedy a default on or prior to 5:00 p.m. (ET) on the day that is fifteen (15) business
days from the date of the Arrangement Agreement.
Termination Fee
The Company shall be entitled
to a termination fee of CDN$1,250,000 (the “Termination Fee”) in the event that:
| |
(i) |
the Arrangement Agreement
is terminated by Appili or the Company as a result of a Company Change in Recommendation / to enter into a Company Superior Proposal;
or |
| |
(ii) |
the Arrangement Agreement
is terminated by the Company as a result of breach of: (A) a representation, warranty or covenant by Appili (including Appili’s
non-solicitation covenants); (B) failure of the Appili shareholder’s to approve the Arrangement resolution; or (C), the Arrangement
Agreement is terminated by the Company or Appili if the Effective Time is not prior to the Outside Date, provided that prior to the
termination of the Arrangement Agreement, a Company Acquisition Proposal has been made to Appili or announced and within 12 months
following the date of such termination: (1) a Company Acquisition Proposal made, publicly announced or otherwise communicated to
the Appili board prior to the termination of the Arrangement Agreement is consummated; or (2) Appili enters into a definitive agreement
in respect of a Company Acquisition Proposal made, publicly announced or otherwise communicated to the Appili board prior to the
termination of the Arrangement Agreement and at any time thereafter such Company Acquisition Proposal is consummated. |
Appili shall be entitled
to the Termination Fee in the event that:
| |
(i) |
the Arrangement Agreement
is terminated by the Company or Appili as a result of the failure to complete the Financing on or before June 30, 2024; |
| |
(ii) |
the Arrangement Agreement
is terminated by as a result of the failure to take all actions within its control that are necessary to remedy a default of the
Parent on or prior to 5:00 p.m. (ET) on the day that is fifteen (15) business days from the date of the Arrangement Agreement; |
| |
(iii) |
the Arrangement Agreement
is terminated by Appili as a result of a breach of a representation, warranty or covenant by the Company or the Arrangement Agreement
is terminated by the Company or Appili if the Effective Time is not prior to the Outside Date, provided that prior to the termination
of the Arrangement Agreement, a Parent Acquisition Proposal (as defined in the Arrangement Agreement) has been made to the Company
or announced and within 12 months following the date of such termination: (1) a Parent Acquisition Proposal made, publicly announced
or otherwise communicated to the Company board prior to the termination of the Arrangement Agreement is consummated; or (2) the Company
enters into a definitive agreement in respect of a Parent Acquisition Proposal made, publicly announced or otherwise communicated
to the Company board prior to the termination of the Arrangement Agreement and at any time thereafter such Parent Acquisition Proposal
is consummated. |
Merger Agreement with Evofem
Biosciences, Inc.
On December 11, 2023, we entered
into an Agreement and Plan of Merger (the “Merger Agreement”) with Adifem f/k/a Adicure, Inc., a majority owned subsidiary
of the Company (“Merger Sub”) and Evofem, pursuant to which, Merger Sub will be merged
into and with Evofem (the “Merger”), with Evofem surviving the Merger as a wholly
owned subsidiary of the Company. Evofem is a commercial-stage women’s health company with a strong focus on innovation. Evofem
is the creator of an FDA-approved hormone-fee contraceptive gel, Phexxi®.
At
the effective time of the Merger (the “Merger Effective Time”), (i) all issued and outstanding shares of common stock of
Evofem (“Evofem Common Stock”), other than any shares of Evofem Common Stock held
by the Company or Merger Sub immediately prior to the Merger Effective Time, will be converted into the right to receive an aggregate
of 610,000 shares of our Common Stock; and (ii) all issued and outstanding shares of Series E-1 Preferred Stock of Evofem (the “Evofem
Unconverted Preferred Stock”), other than any shares of Evofem Unconverted Preferred Stock held by the Company or Merger
Sub immediately prior to the Merger Effective Time, will be converted into the right to receive an aggregate of 2,327 shares of our Series
A-1 Convertible Preferred Stock, having such rights, powers, and preferences set forth in the form of Certificate of Designation of Series
A-1 Convertible Preferred Stock, the form of which is attached as Exhibit C to the Merger Agreement. For additional information regarding
the rights, powers and preferences of the Series A-1 Convertible Preferred Stock, see “Description of Capital Stock—Series
A-1 Convertible Preferred Stock”.
The
closing of the Merger is subject to the satisfaction or waiver of a number of conditions. Including but not limited to: (i) approval
by our shareholders and the Evofem shareholders of the transactions contemplated by the Merger Agreement; (ii) the registration statement
on Form S-4 pursuant to which the shares of our Common Stock issuable in the Merger having been declared effective by the SEC; (iii)
all preferred stock of Evofem subject to certain exceptions shall have been converted to Evofem common stock; (iv) Evofem shall have
received agreements from all holders of Evofem warrants which provide: (a) waivers with respect to any fundamental transaction, change
in control or other similar rights that such warrant holder may have under any such Evofem warrants, and (b) an agreement to such Evofem
warrants to exchange such warrants for not more than an aggregate (for all holders of Evofem warrants) of 551 shares of our preferred
stock of Evofem; (v) the Company shall have obtained agreements from the holders of certain convertible notes and purchase rights
of Evofem to exchange such convertible notes and purchase rights for not more than an aggregate (for all holders of Evofem convertible
notes) of 86,153 shares of our preferred stock; and (vi) we shall have received waivers form the holders of certain of our securities
which contain prohibitions on variable rate transactions.
The
Merger Agreement may be terminated at any time prior to the consummation of the Closing by mutual written consent of us and Evofem. The
Merger Agreement may also be terminated by us or Evofem if (i) the Merger shall not have been consummated on or before 5:00 p.m. Eastern
Time on May 8, 2024; (ii) if any judgment, law or order prohibiting the Merger or the transactions contemplated in connection therewith
has become final and non-appealable; (iii) the required vote of Evofem stockholders was not obtained; or (iv) in the event of any
Terminable Breach (as defined in the Merger Agreement). We may also terminate the Merger Agreement if (i) prior to approval by the required
vote of Evofem’s shareholders if the Evofem board of directors shall have effected a change in recommendation with respect to the
Merger; or (ii) in the event that we determine, in our reasonable discretion, that the acquisition of Evofem could result in a material
adverse amount of cancellation of indebtedness income to us. Evofem may terminate the Merger Agreement if (i) prior to approval by the
required vote of Evofem’s shareholders if the Evofem board of directors determines to terminate the Merger Agreement in connection
with a superior proposal in order to enter into a definitive agreement for such superior proposal provided that Evofem has paid the termination
fee of $4 million; (ii) our Common Stock is no longer listed for trading on Nasdaq; or (iii) we have not made a loan to Evofem of no
less than $3 million prior to January 31, 2024.
In
connection with the Merger Agreement, we entered into an Assignment Agreement dated December 11, 2023 (the “Assignment Agreement”),
with Evofem and the holders (the “Holders”) of certain senior indebtedness of Evofem (the “Notes”), pursuant
to which the Holders assigned the Notes to us in consideration for the issuance by us of (i) an aggregate principal amount of $5 million
in our secured notes due on January 2, 2024 (the “January 2024 Secured Notes”), (ii) an aggregate principal amount of $8
million in secured notes of the Company due on September 30, 2024 (the “September 2024 Secured Notes”), (iii) an aggregate
principal amount of $5 million in ten-year unsecured notes (the “Unsecured Notes”), and (iv) payment of $154,480 in respect
of net sales of Phexxi in respect of the calendar quarter ended September 30, 2023, which amount is due and payable on December 14, 2023.
The January 2024 Secured Notes are secured by certain intellectual property assets of the Company and its subsidiaries pursuant to an
Intellectual Property Security Agreement entered into in connection with the Assignment Agreement. The September 2024 Secured Notes are
secured by the Notes and certain associated security documents pursuant to a Security Agreement entered into in connection with the Assignment
Agreement.
On
January 2, 2024, we entered into amendments to the January 2024 Secured Notes (“Amendment No. 1 to January 2024 Secured Notes”)
with the Holders, pursuant to which the maturity date of the January 2024 Notes was extended to January 5, 2024. On January 5, 2024,
we entered into amendments to the January 2024 Secured Notes (“Amendment No. 2 to January 2024 Secured Notes”) and amendments
to the September 2024 Secured Notes (“Amendment No. 1 to September 2024 Secured Notes”) with the Holders, pursuant to which
the Company and the Holders agreed that in consideration of a principal payment in the aggregate amount of $1 million on the January
2024 Secured Notes and in increase in the aggregate principal balance of $250,000 on the September 2024 Secured Notes, that the maturity
date of the January 2024 Secured Notes would be further extended to January 31, 2024.
On
January 31, 2024, the Company and the Holders entered into amendments to the January 2024 Secured Notes (“Amendment No. 3 to January
2024 Secured Notes”), pursuant to which the maturity date of the January 2024 Notes was extended to February 29, 2024. In addition,
on January 31, 2024, the Company and the Holders entered into amendments to the September 2024 Secured Notes (“Amendment No. 2
to September 2024 Secured Notes”), pursuant to which the Company and the Holders agreed that in consideration of a principal payment
in the aggregate amount of $1.25 million not later than February 9, 2024 on the January 2024 Secured Notes and in increase in the aggregate
principal balance of $300,000 on the September 2024 Secured Notes.
On
February 26, 2024, the Company and the Holders entered into an Assignment Agreement (the “February Assignment Agreement”),
pursuant to which the Company assigned all remaining amounts due under the January 2024 Secured Notes, the September 2024 Secured Notes
and the Unsecured Notes (collectively, the “Notes”) back to the Holders. In connection with the February Assignment
Agreement, the Company and the Holders entered into a payoff letter (the “Payoff Letter”) and amendments to the January
2024 Secured Notes (“Amendment No. 4 to January 2024 Secured Notes”), pursuant to which the maturity date of the January
2024 Secured Notes was extended to March 31, 2024 and the outstanding balance under the Notes, after giving effect to the transactions
contemplated by the February Assignment Agreement as applied pursuant to the Payoff Letter, was adjusted to $250,000. On April 15, 2024
the $250,000 was paid.
In
connection with the Merger Agreement and the transactions contemplated thereby, on December 22, 2023, we entered into an Exchange Agreement
(the “Exchange Agreement”) with the holders of an aggregate of 22,280 shares of Series F-1 Convertible Preferred Stock of
Evofem, pursuant to which such holders agreed to exchange their respective shares of Evofem Series F-1 Preferred Stock for an aggregate
of 22,280 shares of a new series of our convertible preferred stock of the Company designated as Series A-1 Convertible Preferred Stock.
On December 26, 2023, in connection with the Exchange Agreement, we entered into a Registration Rights Agreement with the holders, pursuant
to which we agreed to prepare and file with the SEC covering the resale of the shares of our Common Stock issuable upon conversion of
the Series A-1 Convertible Preferred Stock (i) on the later of (x) the 15th calendar day after the closing date, or (y)
the 2nd business day following the Stockholder Approval Date (as defined in the Exchange Agreement”), with respect
to the initial registration statement and (ii) on the date on which the Company is required to file any additional Registration Statement
pursuant to the terms of the Registration Rights Agreement with respect to any additional Registration Statements that may be required
to be filed by the Company (the “Filing Deadline”). Pursuant to the Registration Rights Agreement, we are required (i) to
have the initial Registration Statement declared effective by the SEC on the earlier of (x) the 60th calendar day after the
Filing Deadline (or the 90th calendar day after the Filing Deadline if subject to a full review by the SEC), and (y)
the 2nd business day after the date we are notified by the SEC that such Registration Statement will not be reviewed,
and (ii) with respect to any additional Registration Statements that may be required to be filed, the earlier of (x) the 60th calendar
day following the date on which we were required to file such additional Registration Statement (or the 90th calendar
day if subject to a full review by the SEC), and (y) the 2nd business day after the date we are notified by the SEC that
such Registration Statement will not be reviewed. In the event that we fail to file the Registration Statement by the Filing Deadline,
have it declared effective by the Effectiveness Deadline, or the prospectus contained therein is not available for use or the investor
is not otherwise able to sell its Warrant Shares pursuant to Rule 144, we will be required to pay the investor an amount equal to 2%
of the stated value of such Holder’s Series A-1 Preferred Stock on the date of such failure and on every thirty date anniversary
until such failure is cured. For additional information regarding the rights, powers and preferences of the Series A-1 Convertible
Preferred Stock, see “Description of Capital Stock—Series A-1 Convertible Preferred Stock”.
On
January 8, 2024, the Company, Adicure and Evofem entered into the First Amendment to the Merger Agreement (the “First Amendment
to Merger Agreement”), pursuant to which the parties agreed to extend the date by which the joint proxy statement would be filed
with the SEC until February 14, 2024. On January 30, 2024, the Company, Adicure and Evofem entered into the Second Amendment to the Merger
Agreement (the “Second Amendment to Merger Agreement”) to amend (i) the date of the Parent Loan (as defined in the Merger
Agreement) to Evofem to be February 29, 2024, (ii) to change the date by which Evofem may terminate the Merger Agreement for failure
to receive the Parent Loan to be February 29, 2024, and (iii) to change the filing date for the Joint Proxy Statement (as defined in
the Merger Agreement) to April 1, 2024.
On
February 29, 2024, the Company, Adicure and Evofem entered into the Third Amendment to the Merger Agreement (the “Third
Amendment to Merger Agreement”) in order to (i) make certain conforming changes to the Merger Agreement regarding the Notes,
(ii) extend the date by which the Company and Evofem will file the joint proxy statement until April 30, 2024, and (iii) remove the requirement
that the Company make the Parent Loan (as defined in the Merger Agreement) by February 29, 2024 and replace it with the requirement that
the Company make an equity investment into Evofem consisting of (a) a purchase of 2,000 shares of Evofem Series F-1 Preferred Stock for
an aggregate purchase price of $2.0 million on or prior to April 1, 2024 (the “Initial Parent Equity Investment”), and (b)
a purchase of 1,500 shares of Evofem Series F-1 Preferred Stock for an aggregate purchase price of $1.5 million on or prior to April
30, 2024.
On
April 26, 2024, the Company received notice from Evofem (the “Termination Notice”)
that Evofem was exercising its right to terminate the Merger Agreement as a result of the Company’s failure to provide the Initial
Parent Equity Investment by April 1, 2024.
On
May 2, 2024, the Company, Adifem, Inc. f/k/a Adicure, Inc. and Evofem Biosciences, Inc. (“Evofem”) entered into the Reinstatement
and Fourth Amendment to the Merger Agreement (the “Fourth Amendment”) in order
to waive and amend, among other things, the several provisions listed below.
Amendments to Article
VI: Covenants and Agreement
Article VI of the Merger
Agreement is amended to:
| |
● |
reinstate
the Merger Agreement, as amended by the Fourth Amendment, as if never terminated; |
| |
|
|
| |
● |
reflect
the Company’s payment to Evofem, in the amount of $1,000,000 (the “Initial Payment”),
via wire initiated by May 2, 2024; |
| |
|
|
| |
● |
delete
Section 6.3, which effectively eliminates the “no shop” provision, and the several defined terms used therein; |
| |
|
|
| |
● |
add a
new defined term “Company Change of Recommendation;” and |
| |
|
|
| |
● |
revise
section 6.10 of the Merger Agreement such that, after the Initial Payment, and upon the closing of each subsequent capital raise
by the Company (each a “Parent Subsequent Capital Raise”), the Company shall
purchase that number of shares of Evofem’s Series F-1 Preferred Stock, par value $0.0001 per share (the “Series
F-1 Preferred Stock”), equal to forty percent (40%) of the gross proceeds of such Parent Subsequent Capital Raise divided
by 1,000, up to a maximum aggregate amount of $2,500,000 or 2,500 shares of Series F-1 Preferred Stock. A maximum of $1,500,000 shall
be raised prior to June 17, 2024 and $1,000,000 prior to July 1, 2024 (the “Parent Capital
Raise”). |
Amendments to Article
VIII: Termination
Article VIII of the
Merger Agreement is amended to:
| |
● |
extend
the date after which either party may terminate from May 8, 2024 to July 15, 2024; |
| |
|
|
| |
● |
revise
Section 8.1(d) in its entirety to allow Company to terminate at any time after there has been a Company Change of Recommendation,
provided that Aditxt must receive ten day written notice and have the opportunity to negotiate a competing offer in good faith; and |
| |
|
|
| |
● |
amend
and restate Section 8.1(f) in its entirety, granting the Company the right to terminate the agreement if (a) the full $1,000,000
Initial Payment required by the Fourth Amendment has not been paid in full by May 3, 2024 (b) $1,500,000 of the Parent Capital Raise
Amount has not been paid to the Company by June 17, 2024, (c) $1,000,000 of the Parent Capital Raise Amount has not been paid to
the Company by July 1, 2024, or (d) Aditxt does not pay any portion of the Parent Equity Investment within five calendar days after
each closing of a Parent Subsequent Capital Raise. |
Asset Purchase Agreement
with MDNA Life Sciences, Inc.
On December
17, 2023, the Company entered into an Asset Purchase Agreement (the “Purchase Agreement”) with
Pearsanta, Inc., our majority owned subsidiary (“Pearsanta”) and MDNA Life Sciences, Inc. (“MDNA”), pursuant
to which Pearsanta agreed to acquire certain intellectual property and other specified assets relating to MDNA’s early cancer detection
platform (the “Acquired Assets”). MDNA’s Mitomic™ technology provides a tool for identifying biomarkers associated
with various diseases that lead to mtDNA mutations. The Acquired Assets include, but are not limited to, the following:
| ● | The
Mitomic Endometriosis Test (MET™) is in development as a blood-based assay for diagnosis
of endometriosis. This test aims to provide early diagnostic insights, potentially reducing
delays in diagnosing endometriosis. |
| ● | The
Mitomic Prostate Test (MPT™) is currently under development as a blood-based assay
for diagnosis of prostate cancer. We believe that this test holds the potential to provide
more specific and clinically informative data especially in the prostate-specific antigen
(PSA) grey zone. It aims to address the challenges of over-diagnosis and mitigate risks associated
with low-grade cancers. |
Pursuant
to the Purchase Agreement, the consideration for the transaction was to consist of: (i) an upfront working capital payment of $500,000
(the “Upfront Working Capital Payment”), which is payable upon the satisfaction of certain conditions set forth in the Purchase
Agreement, (ii) a working capital payment at closing of $500,000, (iii) 50,000 shares of
our Common Stock, (iv) a warrant to purchase 50,000 shares of our Common Stock exercisable for a term of 5 years at an exercise price
equal to the opening price per share of our Common Stock as of the Closing Date (as defined below), and (v) 5,000 shares of Pearsanta
Series A Preferred Stock, par value $0.001 per share (the “Pearsanta Preferred Stock”), provided, however, that if the value
of such Pearsanta Preferred Stock, on an as-converted basis, at the time of the pricing of the Pearsanta common stock in connection with
the sale of shares of Pearsanta common stock to the public in a firm-commitment underwritten public offering pursuant to an effective
registration statement under the Securities Act of 1933, as amended does not equal $25,000,000, an additional amount of Pearsanta Preferred
Stock (“Additional Pearsanta Preferred Stock”) so that the sum of the value of the Pearsanta Preferred Stock plus the Additional
Pearsanta Preferred Stock (if any) shall equal $25,000,000. The Pearsanta Preferred Stock shall have such rights, powers, and preferences
as set forth in the form of Certificate of Designation of Series A Preferred Stock, the form of which is attached as Exhibit D to the
Purchase Agreement.
On January
4, 2024, we entered into a First Amendment to Asset Purchase Agreement with Pearsanta and MDNA, pursuant to which the parties agreed
to: (i) the removal of the Upfront Working Capital Payment, (ii) the removal of the Closing Working Capital Payment (as defined in the
Purchase Agreement”), and (iii) to increase the maximum amount of payments to be made by us under the Transition Services Agreement
(as defined below) from $2.2 million to $3.2 million.
On January
4, 2024, Pearsanta and MDNA entered into a Transition Services Agreement (the “Transition Services Agreement”), pursuant
to which MDNA agreed that it would perform, or cause certain of its affiliates or third parties to perform, certain services as described
in the Transition Services Agreement for a term of three months in consideration for the payment by Pearsanta of certain fees as provided
in the Transition Services Agreement, in an amount not to exceed $3.2 million.
On January 4,
2024, we completed its acquisition of the Acquired Assets and issued to MDNA 50,000 shares of our Common Stock, a warrant to purchase
50,000 shares of our Common Stock, and the Pearsanta Preferred Stock.
Set forth below is a summary
of the rights, powers and preferences of the Pearsanta Series A Convertible Preferred Stock:
Series A Convertible Preferred Stock
On
January 2, 2024, we filed a Certificate of Designations for the Pearsanta Series A Preferred Stock with the Secretary of State of Delaware
(the “Series A Certificate of Designations”). The following is only a summary of the Series A Certificate of Designations,
and is qualified in its entirety by reference to the full text of the Series A Certificate of Designations, a copy of which is filed
as an exhibit to the Purchase Agreement.
Designation,
Amount, and Par Value. The number of Series A Preferred Stock designated is 5,000 shares. The shares of Series A Preferred
Stock have a par value of $0.001 per share and a stated value of $5,000 per share.
a)
Conversion. each outstanding share of Series A Preferred Stock shall be mandatorily and automatically converted, with no
further action on the part of the holders thereof, into 1,000 fully paid and nonassessable shares of Common Stock (1:1,000) (the “Conversion
Ratio”) upon the consummation of a firm underwritten initial public offering of the Common Stock for cash effected pursuant
to a registration statement or similar document filed by or on behalf of the Company under the Securities Act (a “Qualifying
IPO”). If the product of (i) the number of shares of Common Stock issued upon conversion based on the Conversion Ratio and
(ii) the initial trading price upon a Qualifying IPO (“IPO Price”), is less than $25,000,000 then the Conversion Ratio
shall be adjusted to 1:X (such ratio, the “Adjusted Conversion Ratio”) where:
X = (25,000,000/ IPO Price) / 5000
and
the number of shares of Common Stock issued or issuable upon conversion of the Series A Preferred Stock shall be determined based on
such Adjusted Conversion Ratio.
Dividends.
Holders of Series A Preferred Stock shall be entitled to receive, and the Corporation shall pay, dividends on shares of Series
A Preferred Stock equal (on an as-if-converted-to-Common-Stock basis) to and in the same form as dividends actually paid on shares of
the Common Stock
Liquidation. In
the event of a liquidation, the holders the Series A Preferred Stock shall be entitled to receive in cash out of the assets of the Company,
the same amount that a holder of Common Stock would receive if the Series A Preferred Stock were fully converted (disregarding for such
purposes any conversion limitations hereunder) to Common Stock which amounts shall be paid pari passu with all holders of Common
Stock.
Company
Redemption. At any time prior to a Qualified IPO, the Company may redeem all, or any portion, of the Series A Preferred
Stock for cash, at a price per share of Series A Preferred Stock equal to the Stated Value per share.
Voting
Rights. The holders of the Series A Preferred Stock shall have no voting power and no right to vote on any matter at any
time, either as a separate series or class or together with any other series or class of share of capital stock, and shall not be entitled
to call a meeting of such holders for any purpose nor shall they be entitled to participate in any meeting of the holders of Common Stock,
except as expressly provided in the Series A Certificate of Designations and where required by the DGCL.
December 2023 PIPE
As previously reported in a Current
Report on Form 8-K, on December 29, 2023, we entered into a Securities Purchase Agreement (the “Purchase Agreement”) with
the Selling Stockholder for the issuance and sale in a private placement of (i) pre-funded warrants
(the “Pre-Funded Warrants”) to purchase up to 1,237,114 shares of our Common Stock at an exercise price of $0.001 per share,
and (ii) warrants (the “Common Warrants”) to purchase up to 2,474,228 shares of our Common Stock, at a purchase price of
$4.85 per share. The Common Warrants are exercisable immediately upon issuance at an exercise price of $4.60 per share and have a term
of exercise equal to three years from the date of issuance. The Pre-Funded Warrants are exercisable immediately and may be exercised
at any time until the Pre-Funded Warrants are exercised in full. A holder of Pre-Funded Warrants or Warrants (together with its affiliates)
may not exercise any portion of a warrant to the extent that the holder would own more than 4.99% (or, at the election of the holder
9.99%) of the Company’s outstanding common stock immediately after exercise.
Pursuant
to the Purchase Agreement, we also agreed to reduce the exercise price of 106,594 outstanding warrants to purchase Common Stock of the
Company (“Outstanding Warrants”) held by the Purchaser to $4.60 per share in consideration for the cash payment by the Purchaser
of $0.125 per share of Common Stock underlying the Outstanding Warrants, effective immediately. As of the date of this prospectus, none
of the repriced Outstanding Warrants have been exercised.
In connection
with the Private Placement, we entered into a registration rights agreement (the “Registration Rights Agreement”), dated
as of December 29, 2023, with the Selling Stockholder, pursuant to which we agreed to prepare and file a registration statement with
the Securities and Exchange Commission (the “SEC”) registering the resale of the shares of Common Stock underlying
the Pre-Funded Warrants and the Common Warrants no later than 10 trading days after the date of the Registration Rights Agreement, and
to use best efforts to have the registration statement declared effective as promptly as practical thereafter, and in any event no later
than 45 days following the date of the Registration Rights Agreement (or 75 days following the date of the Registration Rights Agreement
in the event of a “full review” by the SEC).
On January
3, 2024, we entered into a settlement agreement and general release with the Selling Stockholder, pursuant to which we agreed to settle
an action filed in the United States District Court in the Southern District of New York by the Selling Stockholder against the Company
(the “Action”) in consideration of the issuance by us of shares of our Common Stock (the “Settlement Shares”),
which will be issued within two business days following court approval of a joint motion to be filed by the Selling Stockholder and the
Company. The number of Settlement Shares to be issued will be equal to $1.6 million divided by the closing price of our Common Stock
on the day prior to court approval of the joint motion. On January 17, 2024, we issued 296,296 Settlement Shares. Following the issuance
of the Settlement Shares, the Selling Stockholder will file a dismissal stipulation in the Action.
The
private placement closed on January 4, 2024. The net proceeds to us from the private placement were approximately $5.5 million, after
deducting placement agent fees and expenses and estimated offering expenses payable by us. The Company used a portion of the proceeds
from the December 2023 PIPE to repay outstanding loans from our Chief Executive Officer, including accrued interest. In addition, the
Company also used a portion of the proceeds to satisfy outstanding obligations under the August Loan Agreement and the October MCA Agreement.
As of the date of this prospectus, we are one payment in arrears on the August Loan Agreement.
Note Exchange
On
December 29, 2023, we entered into an Exchange Agreement with the holder of our secured promissory note in the principal amount of $2.625
million (the “Note”), pursuant to which the holder agreed, subject to the terms and conditions set forth therein, to exchange
the Note, including all accrued but unpaid interest thereon, for an aggregate of 2,625 shares of a new series of convertible preferred
stock of the Company, designated as Series B-2 Convertible Preferred Stock. For additional information regarding the rights, powers
and preferences of the Series B-2 Convertible Preferred Stock, see “Description of Capital Stock—Series B-2 Convertible
Preferred Stock”.
Nasdaq Compliance
As
previously reported in a Current Report on Form 8-K, on September 29, 2023, we received written notice from The Nasdaq Capital Market
(“Nasdaq”) that the Hearing Panel had granted us an exception through December
26, 2023 to allow us to complete its plan to demonstrate compliance with Nasdaq Listing Rule 5550(b)(1) (the “Stockholders’
Equity Rule”) and Nasdaq Listing Rule 5550(a)(4) (the “Public Float Rule”). On November 21, 2023, we received written
notice from Nasdaq that we had regained compliance with the Public Float Rule. On December 29, 2023, we received written notice from
Nasdaq that we had regained compliance with the Stockholders’ Equity Rule, but will be subject to a Mandatory Panel Monitor for
a period of one year.
Acquisition of Certain Assets of Brain Scientific,
Inc.
On
January 24, 2024, we entered into an Assignment and Assumption Agreement (the “Assignment Agreement”) with the agent (the
“Agent”) of certain secured creditors (the “Brain Creditors”) of Brain Scientific, Inc., a Nevada corporation
(“Brain Scientific”) and Philip J. von Kahle (the “Seller”), as assignee of Brain Scientific and certain affiliated
entities (collectively, the “Brain Companies”) under an assignment for the benefit of creditors pursuant to Chapter 727 of
the Florida Statutes. Pursuant to the Assignment Agreement, the Agent assigned its rights under that certain Asset Purchase and Settlement
Agreement dated October 31, 2023 between the Seller and the Agent (the “Asset Purchase Agreement”) to the Company in consideration
for the issuance by the Company of an aggregate of 6,000 shares of a new series of convertible
preferred stock of the Company, designated as Series B-1 Convertible Preferred Stock, $0.001 par value (the “Series B-1 Preferred
Stock”). The shares of Series B-1 Preferred Stock were issued pursuant to a Securities Purchase Agreement entered into by and between
the Company and each of the purchasers signatory thereto (the “Purchase Agreement”). For additional information regarding
the rights, powers and preferences of the Series B-1 Convertible Preferred Stock, see “Description of Capital Stock—Series
B-1 Convertible Preferred Stock”.
The
assets acquired through the assignment for the benefit of creditors include 16 patents covering a portfolio of EEG brain monitoring technologies
and devices.
In
connection with the Assignment Agreement, on January 24, 2024, we entered into a Patent Assignment with the Seller (the “Patent
Assignment”), pursuant to which the Seller assigned all of its rights, titles and interests in certain patents and patent applications
that were previously held by the Brain Companies to the Company.
In
connection with the Purchase Agreement, the Company and certain of its officers and directors entered into a Voting Agreement (the “Voting
Agreement”) with the purchasers, pursuant to which such officers and directors agreed to vote any and all shares of Common Stock
or other capital stock of the Company which they own, directly or indirectly, in favor of any proposal to issue the purchasers a number
of shares of Common Stock that would require approval of the Company’s stockholders in order for such issuance to be in compliance
with Nasdaq Rule 5635(a)(1) or 5635(d), and any other applicable rules and regulations of The Nasdaq Stock Market.
November 2023 Business Loan
On
November 7, 2023, we entered into a Business Loan and Security Agreement (the “November Loan Agreement”) with a commercial
funding source (the “November Lender”), pursuant to which the Company obtained a loan from the November Lender in the
principal amount of $2,100,000 (the “November Loan”). Pursuant to the November Loan Agreement, the Company granted the November
Lender a continuing secondary security interest in certain collateral (as defined in the November Loan Agreement). The total amount of
interest and fees payable by us to the Lender under the November Loan will be $3,129,000, which will be repaid in 34 weekly installments
ranging from $69,000 - $99,000. As of the date of this prospectus, we are approximately 4 weeks in arrears on payments under the
November Loan Agreement. In addition, as a result of the default in required principal payment on the January 2024 Secured Notes as described
above, the November Loan is also now in default and the Lender may, at any time, exercise the rights and remedies available to it under
the January Loan Agreement, including but not limited to, declaring the full amount outstanding under the November Loan Agreement immediately
due and payable.
January 2024 Business Loan
On
January 24, 2024, we entered into a Business Loan and Security Agreement (the “January Loan Agreement”) with a commercial
funding source (the “Lender”), pursuant to which we obtained a loan from the Lender in the principal amount of $3,600,000,
which includes origination fees of $252,000 (the “January Loan”). Pursuant to the January Loan Agreement, we granted the
Lender a continuing secondary security interest in certain collateral (as defined in the January Loan Agreement). The total amount of
interest and fees payable by the Company to the Lender under the January Loan will be $5,364,000, which will be repayable by the Company
in 30 weekly installments of $178,800. The Company received net proceeds from the January Loan of $814,900 following repayment of the
outstanding balance on the October Purchased Amount of $2,533,100. As of the date of this prospectus, we are approximately 1 week in
arrears on payments under the January Loan Agreement. In addition As a result of the default in required principal payment on the January
2024 Secured Notes as described above, the January Loan is also now in default and the Lender may, at any time, exercise the rights and
remedies available to it under the January Loan Agreement, including but not limited to, declaring the full amount outstanding under
the January Loan Agreement immediately due and payable.
February
2024 Unsecured Loan
On
February 7, 2024, Amro Albanna, the Chief Executive Officer of the Company loaned $30,000 to the Company. The loan was evidenced by an
unsecured promissory note (the “February Note”). Pursuant to the terms of the February Note, it will accrue interest at the
Prime rate of eight and one-half percent (8.5%) per annum and is due on the earlier of August 7, 2024 or an event of default, as defined
therein.
Corporate Information
We were incorporated as a
Delaware corporation on September 28, 2017. Our principal executive offices are located at 2569 Wyandotte St., Suite 101, Mountain View,
CA 94043, and our telephone number is (650) 870-1200.
Our Common Stock trades on The
Nasdaq Capital Market under the symbol “ADTX.”
THE
OFFERING
| Common
Stock to be offered by the Selling Stockholder |
|
Up
to 3,785,569 shares
of Common Stock |
| |
|
|
| Common
stock outstanding prior to this offering |
|
1,665,214 shares of Common Stock. |
| |
|
|
| Common
stock to be outstanding after this offering |
|
5,154,487 shares of Common
Stock, assuming the exercise of all of the Warrants |
| |
|
|
| Use
of proceeds |
|
We
will not receive any proceeds from the sale of the shares of Common Stock by the Selling Stockholders, except for the Warrant exercise
price paid for the Common Stock offered hereby and issuable upon the exercise of the Warrants. See “Use of Proceeds”
on page 48 of this prospectus. |
| |
|
|
| Risk
Factors |
|
See “Risk Factors”
beginning on page 21 of this prospectus, as well as other information included in this prospectus, for a discussion of factors you
should read and consider carefully before investing in our securities. |
| |
|
|
| Nasdaq
Capital Markets symbol |
|
Our common stock is listed
on The Nasdaq Capital Markets under the symbol “ADTX”. There is no established trading market for the Common Warrants
or the Pre-Funded Warrants, and we do not expect a trading market to develop. We do not intend to list the Common Warrants or the
Pre-Funded Warrants on any securities exchange or other trading market. Without a trading market, the liquidity of the Common Warrants
and Pre-Funded Warrants will be extremely limited. |
The number of shares of our
Common Stock to be outstanding after this offering as shown above is based on 1,993,682shares outstanding as of June 4, 2024 and excludes
as of that date:
| |
● |
6,791,215 shares of our
Common Stock issuable upon exercise of warrants, subject to vesting having a weighted average exercise
price of $10.85 per share;
|
| |
● |
45,572 shares of our Common
Stock issuable upon exercise of outstanding options under our 2017 Equity Incentive Plan and 2021 Equity Incentive Plan or the 2017
Plan and the 2021 plan, respectively, subject to vesting; |
| |
● |
5,018,019 shares of our Common Stock
issuable upon conversion of outstanding Series A-1 Convertible Preferred Stock; |
| |
● |
1,477,833
shares of our Common Stock issuable upon conversion of outstanding Series B-1 Convertible Preferred Stock; |
| |
● |
557,325
shares of our Common Stock issuable upon conversion of outstanding Series B-2 Convertible Preferred Stock; and |
| |
● |
1,613,092
shares of our Common Stock issuable upon conversion of outstanding Series C-1 Convertible Preferred Stock. |
RISK
FACTORS
An investment in our securities
involves a high degree of risk. This prospectus contains a discussion of the risks applicable to an investment in our securities. Prior
to deciding about investing in our securities, you should carefully consider the specific factors discussed within this prospectus. The
risks and uncertainties we have described are not the only ones we face. Additional risks and uncertainties not presently known to us
or that we currently deem immaterial may also affect our operations. The occurrence of any of these known or unknown risks might cause
you to lose all or part of your investment in the offered securities.
Risks Related to Our Financial Position and Need
for Capital
Our financial situation creates doubt whether
we will continue as a going concern.
We were incorporated in September
2017 and have a limited operating history and our business is subject to all the risks inherent in the establishment of a new business
enterprise. Our likelihood of success must be considered in light of the problems, expenses, difficulties, complications and delays frequently
encountered in connection with development and expansion of a new business enterprise. Since inception, we have incurred losses and expect
to continue to operate at a net loss for at least the next several years as we commence our research and development efforts, conduct
clinical trials, and develop manufacturing, sales, marketing, and distribution capabilities. Our net loss attributable to common stockholders
for the years ended December 31, 2023 and 2022 was $32,700,710 and $27,687,553, respectively, and our accumulated deficit as of December
31, 2023 was $127,741,072. There can be no assurance that the products under development by us will be approved for sale in the U.S.
or elsewhere or that we will successfully close any of our pending M&A transactions. Furthermore, there can be no assurance that
if such products are approved, they will be successfully commercialized, and the extent of our future losses and the timing of our profitability
are highly uncertain. In addition, we may not be able to successfully integrate any businesses that we acquire. If we are unable to achieve
profitability, we may be unable to continue our operations. There can be no assurances that we will be able to achieve a level of revenues
adequate to generate sufficient cash flow from operations or additional financing through private placements, public offerings and/or
bank financing necessary to support our working capital requirements. To the extent that funds generated from any private placements,
public offerings and/or bank financing are insufficient, we will have to raise additional working capital. No assurance can be given
that additional financing will be available, or if available, will be on acceptable terms. These conditions raise substantial doubt about
our ability to continue as a going concern. If adequate working capital is not available, we may be forced to discontinue operations,
which would cause investors to lose their entire investment.
If we fail to obtain the capital necessary
to fund our operations, we will be unable to continue or complete our product development and you will likely lose your entire investment.
We will need to continue to seek
capital from time to time to continue development of our lead drug candidate beyond our initial combined Phase I/IIa clinical trial and
to acquire and develop other product candidates. Once approved for commercialization, we cannot provide any assurances that any revenues
it may generate in the future will be sufficient to fund our ongoing operations.
Our business or operations may
change in a manner that would consume available funds more rapidly than anticipated and substantial additional funding may be required
to maintain operations, fund expansion, develop new or enhance products, acquire complementary products, business or technologies, or
otherwise respond to competitive pressures and opportunities, such as a change in the regulatory environment or a change in preferred
treatment modalities. In addition, we may need to accelerate the growth of our sales capabilities and distribution beyond what is currently
envisioned, and this would require additional capital. However, we may not be able to secure funding when we need it or on favorable
terms. We may not be able to raise sufficient funds to commercialize the product candidates we intend to develop.
If we cannot raise adequate funds
to satisfy our capital requirements, we will have to delay, scale back or eliminate our research and development activities, clinical
studies, or future operations. We may also be required to obtain funds through arrangements with collaborators, which arrangements may
require us to relinquish rights to certain technologies or products that we otherwise would not consider relinquishing, including rights
to future product candidates or certain major geographic markets. This could result in sharing revenues which we might otherwise retain
for ourselves. Any of these actions may harm our business, financial condition, and results of operations.
The amount of capital we may
need depends on many factors, including the progress, timing and scope of our product development programs; the progress, timing and
scope of our preclinical studies and clinical trials; the time and cost necessary to obtain regulatory approvals; the time and cost necessary
to further develop manufacturing processes and arrange for contract manufacturing; our ability to enter into and maintain collaborative,
licensing and other commercial relationships; and our partners’ commitment of time and resources to the development and commercialization
of our products.
Our obligations
to certain of our creditors are secured by security interests in our assets. If we default on our obligations under these agreements,
our creditors may foreclose on some or all of our assets at any time.
Our
obligations to certain of our creditors are secured by security interests in our assets.
As of the date of this prospectus, approximately $7.9 million was owed to such secured creditors.
Under such agreements, we are required to pay $277,800
on a weekly basis to such creditors. If we default on our obligations under these
agreements, our secured creditors could foreclose on their security interests and liquidate
some or all of these assets, which would harm our financial condition and results of operations
and would require us to reduce or cease operations and possibly seek Bankruptcy Protection.
In the event we pursue
Bankruptcy Protection, we will be subject to the risks and uncertainties associated with such proceedings.
In the event we file for relief
under the United States Bankruptcy Code, our operations, our ability to develop and execute our business plan and our continuation as
a going concern will be subject to the risks and uncertainties associated with bankruptcy proceedings, including, among others: our ability
to execute, confirm and consummate a plan of reorganization; the additional, significant costs of bankruptcy proceedings and related
fees; our ability to obtain sufficient financing to allow us to emerge from bankruptcy and execute our business plan post-emergence,
and our ability to comply with terms and conditions of that financing; our ability to continue our operations in the ordinary course;
our ability to maintain our relationships with our consumers, business partners, counterparties, employees and other third parties; our
ability to obtain, maintain or renew contracts that are critical to our operations on reasonably acceptable terms and conditions; our
ability to attract, motivate and retain key employees; the ability of third parties to use certain limited safe harbor provisions of
the United States Bankruptcy Code to terminate contracts without first seeking Bankruptcy Court approval; the ability of third parties
to force us to into Chapter 7 proceedings rather than Chapter 11 proceedings and the actions and decisions of our stakeholders and other
third parties who have interests in our bankruptcy proceedings that may be inconsistent with our operational and strategic plans. Any
delays in our bankruptcy proceedings would increase the risks of our being unable to reorganize our business and emerge from bankruptcy
proceedings and may increase our costs associated with the bankruptcy process or result in prolonged operational disruption for us. Also,
we would need the prior approval of the bankruptcy court for transactions outside the ordinary course of business during the course of
any bankruptcy proceedings, which may limit our ability to respond timely to certain events or take advantage of certain opportunities.
Because of the risks and uncertainties associated with any bankruptcy proceedings, we cannot accurately predict or quantify the ultimate
impact of events that could occur during any such proceedings. There can be no guarantees that if we seek Bankruptcy Protection we will
emerge from Bankruptcy Protection as a going concern or that holders of our Common Stock will receive any recovery from any bankruptcy
proceedings.
In the event we are
unable to pursue Bankruptcy Protection under Chapter 11 of the United States Bankruptcy Code, or, if pursued, successfully emerge from
such proceedings, it may be necessary to pursue Bankruptcy Protection under Chapter 7 of the United States Bankruptcy Code for all or
a part of our businesses.
In the event we are unable to
pursue Bankruptcy Protection under Chapter 11 of the United States Bankruptcy Code, or, if pursued, successfully emerge from such proceedings,
it may be necessary for us to pursue Bankruptcy Protection under Chapter 7 of the United States Bankruptcy Code for all or a part of
our businesses. In such event, a Chapter 7 trustee would be appointed or elected to liquidate our assets for distribution in accordance
with the priorities established by the United States Bankruptcy Code. We believe that liquidation under Chapter 7 would result in significantly
smaller distributions being made to our stakeholders than those we might obtain under Chapter 11 primarily because of the likelihood
that the assets would have to be sold or otherwise disposed of in a distressed fashion over a short period of time rather than in a controlled
manner and as a going concern.
We will need to raise substantial additional
capital, which may not be available on acceptable terms, or at all. Failure to obtain this necessary capital when needed may force us
to delay, limit or terminate our product development efforts or cease operations.
We do not expect that our current
cash position will be sufficient to fund our current operations for the next 12 months. We also do not presently have sufficient cash
to fund certain obligations under our Merger Agreement with Evofem. In addition, we are required to complete an equity or debt financing
with minimum gross proceeds of at least $20 million in order to close the transactions contemplated under our Arrangement Agreement with
Appili. Our operating plan may change because of many factors currently unknown to us, and we may need to seek additional funds sooner
than planned, through public or private equity or debt financings, government or other third-party funding, marketing and distribution
arrangements and other collaborations, strategic alliances and licensing arrangements or a combination of these approaches. In any event,
we will require additional capital to obtain regulatory approval for, and to commercialize, our product candidates. Raising funds in
the current economic environment may present additional challenges. Even if we believe we have sufficient funds for our current or future
operating plans, we may seek additional capital if market conditions are favorable or if we have specific strategic considerations.
Any additional fundraising efforts
may divert our management from their day-to-day activities, which may adversely affect our ability to develop and commercialize our product
candidates. In addition, we cannot guarantee that future financing will be available in sufficient amounts or on terms acceptable to
us, if at all. Moreover, the terms of any financing may adversely affect the holdings or the rights of our stockholders and the issuance
of additional securities, whether equity or debt, by us, or the possibility of such issuance, may cause the market price of our shares
to decline. The sale of additional equity or convertible securities may dilute our existing stockholders. The incurrence of indebtedness
would result in increased fixed payment obligations, and we may be required to agree to certain restrictive covenants, such as limitations
on our ability to incur additional debt, limitations on our ability to acquire, sell or license intellectual property rights and other
operating restrictions that could adversely impact our ability to conduct our business. We could also be required to seek funds through
arrangements with collaborative partners or otherwise at an earlier stage than otherwise would be desirable and we may be required to
relinquish rights to some of our technologies or product candidates or otherwise agree to terms unfavorable to us, any of which may have
a material adverse effect on our business, operating results and prospects.
If we are unable to obtain funding
on a timely basis, we may be required to significantly curtail, delay, or discontinue one or more of our research or development programs
or the commercialization of any product candidate or be unable to expand our operations, execute on strategic M&A initiatives, or
otherwise capitalize on our business opportunities, as desired, which could materially affect our business, financial condition and results
of operations.
Even if we can raise additional funding, we
may be required to do so on terms that are dilutive to you.
The capital markets have been
unpredictable in the past for unprofitable companies such as ours. In addition, it is generally difficult for development stage companies
to raise capital under current market conditions. The amount of capital that a company such as ours is able to raise often depends on
variables that are beyond our control. As a result, we may not be able to secure financing on terms attractive to us, or at all. If we
can consummate a financing arrangement, the amount raised may not be sufficient to meet our future needs. If adequate funds are not available
on acceptable terms, or at all, our business, including our results of operations, financial condition and our continued viability will
be materially adversely affected.
Risks Related to Product Development, Regulatory
Approval, Manufacturing and Commercialization
The regulatory approval process is expensive,
time-consuming, and uncertain and may prevent us from obtaining approvals for the commercialization of our future product candidates,
if any.
We will not be permitted to market
our product candidates in the United States until we receive approval from the FDA, or in any foreign countries until we receive the
requisite approval from corresponding agencies in such countries. The testing, manufacturing, labeling, approval, selling, marketing
and distribution of health and life science-related products are subject to extensive regulation, which regulations differ from country
to country.
Successfully completing our clinical
program and obtaining approval of a Biologics License Application (“BLA”) is a complex, lengthy, expensive and uncertain
process, and the FDA or other applicable foreign regulator may delay, limit or deny approval of our product candidates for many reasons,
including, among others, because:
| |
● |
we may not be able to demonstrate
that our product candidates are safe and effective in treating patients to the satisfaction of the FDA or foreign regulator; |
| |
● |
the results of our clinical
trials may not meet the level of statistical or clinical significance required by the FDA or foreign regulator for marketing approval; |
| |
● |
the FDA or foreign regulator
may disagree with the number, design, size, conduct or implementation of our clinical trials; |
| |
● |
the FDA or foreign regulator
may require that we conduct additional clinical trials; |
| |
● |
the FDA or foreign regulator
may not approve the formulation, labeling or specifications of our product candidates; |
| |
● |
the contract research organizations
(CROs) and other contractors that we may retain to conduct our clinical trials may take actions outside of our control that materially
adversely impact our clinical trials; |
| |
● |
the FDA or foreign regulator
may find the data from preclinical studies and clinical trials insufficient to demonstrate that our product candidate(s) are safe
and effective for their proposed indications; |
| |
● |
the FDA or foreign regulator
may disagree with our interpretation of data from our preclinical studies and clinical trials; |
| |
● |
the FDA or foreign regulator
may not accept data generated at our clinical trial sites or may disagree with us over whether to accept efficacy results from clinical
trial sites outside the United States or outside the EU, as applicable, where the standard of care is potentially different from
that in the United States or in the EU, as applicable; |
| |
● |
if and when our BLAs or
foreign equivalents are submitted to the applicable regulatory authorities, such agencies may have difficulties scheduling the necessary
review meetings in a timely manner, may recommend against approval of our application or may recommend or require, as a condition
of approval, additional preclinical studies or clinical trials, limitations on approved labeling or distribution and use restrictions; |
| |
● |
the FDA or foreign regulator
may require development of a Risk Evaluation and Mitigation Strategy (REMS), which would use risk minimization strategies to ensure
that the benefits of certain prescription drugs outweigh their risks, as a condition of approval or post-approval; |
| |
● |
the FDA or other applicable
foreign regulatory agencies may not approve the manufacturing processes or facilities of third-party manufacturers with which we
contract; or |
| |
● |
the FDA or the other applicable
foreign regulatory agencies may change their approval policies or adopt new regulations. |
We may encounter substantial delays in completing
our clinical studies which in turn will require additional costs, or we may fail to demonstrate adequate safety and efficacy to the satisfaction
of applicable regulatory authorities.
It is difficult to predict if
or when any of our product candidates, will prove safe or effective in humans or will receive regulatory approval. Before obtaining
marketing approval from regulatory authorities for the sale of our product candidates, we must conduct extensive clinical studies to
demonstrate the safety and efficacy of the product candidates in humans. Clinical testing is expensive, time-consuming, and uncertain
as to outcome. We cannot guarantee that any clinical studies will be conducted as planned or completed on schedule, if at all. A failure
of one or more clinical studies can occur at any stage of testing. Events that may prevent successful or timely completion of clinical
development include:
| |
● |
delays in reaching, or
failing to reach, a consensus with regulatory agencies on study design; |
| |
● |
delays in reaching, or
failing to reach, agreement on acceptable terms with a sufficient number of prospective contract research organizations (“CROs”)
and clinical study sites, the terms of which can be subject to extensive negotiation and may vary significantly among different CROs
and trial sites; |
| |
|
|
| |
● |
delays in obtaining required
Institutional Review Board (“IRB”) or Ethics Committee (“EC”) approval at each clinical study site; |
| |
|
|
| |
● |
delays in recruiting a
sufficient number of suitable patients to participate in our clinical studies including, but not limited to, recruitment challenges
due to COVID-19; |
| |
|
|
| |
● |
imposition of a clinical
hold by regulatory agencies, after an inspection of our clinical study operations or study sites; |
| |
|
|
| |
● |
failure by our CROs,
other third parties or us to adhere to the clinical study, regulatory or legal requirements; |
| |
|
|
| |
● |
failure to perform in accordance
with the FDA’s good clinical practices (“GCP”) or applicable regulatory guidelines in other countries; |
| |
|
|
| |
● |
delays in the testing,
validation, manufacturing, and delivery of sufficient quantities of our product candidates to the clinical sites; |
| |
|
|
| |
● |
delays in having patients’
complete participation in a study or return for post-treatment follow-up; |
| |
|
|
| |
● |
clinical study sites or
patients dropping out of a study; |
| |
|
|
| |
● |
delay or failure to address
any patient safety concerns that arise during the course of a trial; |
| |
|
|
| |
● |
unanticipated costs or
increases in costs of clinical trials of our product candidates; |
| |
|
|
| |
● |
occurrence of serious adverse
events associated with the product candidates that are viewed to outweigh their potential benefits; or |
| |
|
|
| |
● |
changes in regulatory requirements
and guidance that require amending or submitting new clinical protocols. |
We could also encounter delays
if a clinical trial is suspended or terminated by us, by the IRBs or ECs of the institutions in which such trials are being conducted,
by an independent Safety Review Board (“SRB”) for such trial or by the FDA, European Medicines Agency (“EMA”),
or other regulatory authorities. Such authorities may suspend or terminate a clinical trial due to a number of factors, including
failure to conduct the clinical trial in accordance with regulatory requirements or our clinical protocols, inspection of the clinical
trial operations or trial site by the FDA, EMA, or other regulatory authorities resulting in the imposition of a clinical hold, unforeseen
safety issues or adverse side effects, failure to demonstrate a benefit from using a drug, changes in governmental regulations or administrative
actions or lack of adequate funding to continue the clinical trial.
Any inability to successfully
complete preclinical and clinical development could result in additional costs to us or impair our ability to generate revenues from
product sales, regulatory and commercialization milestones, and royalties. In addition, if we make manufacturing or formulation changes
to our product candidates, we may need to conduct additional studies to bridge our modified product candidates to earlier versions.
Clinical study delays could also
shorten any periods during which we may have the exclusive right to commercialize our product candidates or allow our competitors to
bring products to market before we do, which could impair our ability to successfully commercialize our product candidates. In addition,
any delays in completing our clinical trials will increase our costs, slow down our product candidate development and approval
process and jeopardize our ability to commence product sales and generate revenues. Any of these occurrences may significantly harm our
business, financial condition, and prospects. In addition, many of the factors that cause, or lead to, a delay in the commencement or
completion of clinical trials may also ultimately lead to the denial of regulatory approval of our product candidates.
The outcome of preclinical studies
and early clinical trials may not be predictive of the success of later clinical trials, and interim results of a clinical trial do not
necessarily predict final results. Further, preclinical and clinical data are often susceptible to various interpretations and analyses,
and many companies that have believed their product candidates performed satisfactorily in preclinical studies and clinical trials have,
nonetheless, failed to obtain marketing approval. If the results of our clinical studies are inconclusive or if there are safety
concerns or adverse events associated with our other product candidates, we may:
| |
● |
be delayed in obtaining
marketing approval for our product candidates, if approved at all; |
| |
|
|
| |
● |
obtain approval for indications
or patient populations that are not as broad as intended or desired; |
| |
|
|
| |
● |
obtain approval with labeling
that includes significant use or distribution restrictions or safety warnings; |
| |
|
|
| |
● |
be required to change the
way the product is administered; |
| |
|
|
| |
● |
be required to perform
additional clinical studies to support approval or be subject to additional post-marketing testing requirements; |
| |
|
|
| |
● |
have regulatory authorities
withdraw their approval of a product or impose restrictions on its distribution in the form of a modified risk evaluation and mitigation
strategy; |
| |
|
|
| |
● |
be sued; or |
| |
|
|
| |
● |
experience damage to our reputation. |
If we, ours collaborators, or
our contract manufacturing organizations (“CMOs”) fail to comply with applicable regulatory requirements at any stage during
the regulatory process, such noncompliance could result in, among other things delays in the approval of applications or supplements
to approved applications; refusal of a regulatory authority, including the FDA, to review pending market approval applications or supplements
to approved applications; warning letters; fines; import and/or export restrictions; product recalls or seizures; injunctions; total
or partial suspension of production; civil penalties; withdrawals of previously approved marketing applications or licenses; recommendations
by the FDA or other regulatory authorities against governmental contracts; and/or criminal prosecutions.
Additionally, our product candidates
could potentially cause other adverse events that have not yet been predicted. The inclusion of ill patients in our clinical studies
may result in deaths or other adverse medical events due to other therapies or medications that such patients may be using. As described
above, any of these events could prevent us from achieving or maintaining market acceptance of our product candidates and impair our
ability to commercialize our products.
We may not be able to meet requirements for
the chemistry, manufacturing, and control of our drug product candidates.
To receive approval of our products
by the FDA and comparable foreign regulatory authorities, we must show that we and our contract manufacturing partners are able to characterize,
control and manufacture our drug products safely and in accordance with regulatory requirements. This includes synthesizing the active
ingredient, developing an acceptable formulation, performing tests to adequately characterize the formulated product, documenting a repeatable
manufacturing process, and demonstrating that our drug products meet stability requirements. Meeting these chemistry, manufacturing and
control (“CMC”) requirements is a complex task that requires specialized expertise. If we are not able to meet the CMC requirements,
we may not be successful in getting our products approved.
Enrollment and retention
of patients in clinical trials is an expensive and time-consuming process and could be made more difficult or rendered impossible by
multiple factors outside our control.
We may encounter delays or difficulties
in enrolling, or be unable to enroll, a sufficient number of patients to complete any of our clinical trials on its current timelines,
or at all, and even once enrolled we may be unable to retain a sufficient number of patients to complete any of our trials. Enrollment
in our clinical trials may be slower than we anticipate, leading to delays in our development timelines. For example, we may face difficulty
enrolling or maintaining a sufficient number of patients in our clinical trials due to the existing alternative treatments approved for
any of our targeted indications as patients may decline to enroll or decide to withdraw from our clinical trials due to the risk of receiving
placebo. Patient enrollment and retention in clinical trials depends on many factors, including the size of the patient population, the
nature of the trial protocol, our ability to recruit clinical trial investigators with the appropriate competencies and experience, the
existing body of safety and efficacy data with respect to the study drug, the number and nature of competing treatments and ongoing clinical
trials of competing drugs for the same indication, the proximity of patients to clinical sites, the eligibility criteria for the trial
and the proportion of patients screened that meets those criteria, our ability to obtain and maintain patient consents, and our ability
to successfully complete prerequisite studies before enrolling certain patient populations.
Furthermore, any negative results
or new safety signals we may report in clinical trials of our product candidates may make it difficult or impossible to recruit and retain
patients in other clinical trials. Similarly, negative results reported by our competitors about their drug candidates may negatively
affect patient recruitment in our clinical trials. Also, marketing authorization of competitors in this same class of drugs may impair
our ability to enroll patients into our clinical trials, delaying or potentially preventing it from completing recruitment of one or
more of our trials.
Delays or failures in planned
patient enrollment or retention may result in increased costs, program delays or both, which could have a harmful effect on our ability
to develop our product candidates or could render further development impossible. In addition, we expect to rely on CROs and clinical
trial sites to ensure proper and timely conduct of our future clinical trials, and, while we intend to enter into agreements governing
their services, we will be limited in our ability to compel their actual performance.
If our future pre-clinical development or future
clinical Phase I/II studies are unsuccessful, we may be unable to obtain regulatory approval of, or commercialize, our product candidates
on a timely basis or at all.
The successful completion of
pre-clinical development and multiple clinical trials is critical to the success of our future products. If the pre-clinical development
and clinical trials are unsuccessful or produce inconsistent results or unanticipated adverse side effects, or if we are unable to collect
reliable data, regulatory approval of our products could be delayed or not given and as a result we may be unable to commercialize our
products. Generally, we expect to engage third parties such as consultants, universities or other collaboration partners to conduct clinical
trials on our behalf. Incompatible practices or misapplication of our products by these third parties could impair the success of our
clinical trials.
Even if we receive regulatory approval for
any of our product candidates, we may not be able to successfully commercialize the product and the revenue that we generate from their
sales, if any, may be limited.
If approved for marketing, the
commercial success of our product candidates will depend upon each product’s acceptance by the medical community, including physicians,
patients, and health care payors. The degree of market acceptance for any of our product candidates will depend on a number of factors,
including:
| |
● |
demonstration of clinical
safety and efficacy; |
| |
|
|
| |
● |
relative convenience, dosing
burden and ease of administration; |
| |
|
|
| |
● |
the prevalence and severity
of any adverse effects; |
| |
|
|
| |
● |
the willingness of physicians
to prescribe our product candidates, and the target patient population to try new therapies; |
| |
● |
efficacy of our product
candidates compared to competing products; |
| |
|
|
| |
● |
the introduction of any
new products that may in the future become available targeting indications for which our product candidates may be approved; |
| |
|
|
| |
● |
new procedures or therapies
that may reduce the incidences of any of the indications in which our product candidates may show utility; |
| |
● |
pricing and cost-effectiveness; |
| |
|
|
| |
● |
the inclusion or omission
of our product candidates in applicable therapeutic and vaccine guidelines; |
| |
|
|
| |
● |
the effectiveness of our
own or any future collaborators’ sales and marketing strategies; |
| |
|
|
| |
● |
limitations or warnings
contained in approved labeling from regulatory authorities; |
| |
|
|
| |
● |
our ability to obtain and
maintain sufficient third-party coverage or reimbursement from government health care programs, including Medicare and Medicaid,
private health insurers and other third-party payors or to receive the necessary pricing approvals from government bodies regulating
the pricing and usage of therapeutics; and |
| |
|
|
| |
● |
the willingness of patients
to pay out-of-pocket in the absence of third-party coverage or reimbursement or government pricing approvals. |
If any of our product candidates
are approved, but do not achieve an adequate level of acceptance by physicians, health care payors, and patients, we may not generate
sufficient revenues and we may not be able to achieve or sustain profitability. Our efforts to educate the medical community and third-party
payors on the benefits of our product candidates may require significant resources and may never be successful.
In addition, even if we obtain
regulatory approvals, the timing or scope of any approvals may prohibit or reduce our ability to commercialize our product candidates
successfully. For example, if the approval process takes too long, we may miss market opportunities and give other companies the ability
to develop competing products or establish market dominance. Any regulatory approval we ultimately obtain may be limited or subject to
restrictions or post-approval commitments that render our product candidates not commercially viable. For example, regulatory authorities
may approve any of our product candidates for fewer or more limited indications than we request, may grant approval contingent on the
performance of costly post-marketing clinical trials, or may approve any of our product candidates with a label that does not include
the labeling claims necessary or desirable for the successful commercialization for that indication. Further, the FDA or comparable foreign
regulatory authorities may place conditions on approvals or require risk management plans or a Risk Evaluation and Mitigation Strategy
(“REMS”) to assure the safe use of the drug. If the FDA or applicable foreign regulatory agency concludes a REMS is needed,
the sponsor of the BLA must submit a proposed REMS; the regulatory agencies will not approve the BLA without an approved REMS, if required.
A REMS could include medication guides, physician communication plans, or elements to assure safe use, such as restricted distribution
methods, patient registries and other risk minimization tools. The regulatory agencies may also require a REMS for approved product when
new safety information emerges. Any of these limitations on approval or marketing could restrict the commercial promotion, distribution,
prescription or dispensing of our product candidates. Moreover, product approvals may be withdrawn for non-compliance with regulatory
standards or if problems occur following the initial marketing of the product. Any of the foregoing scenarios could materially harm the
commercial success of our product candidates.
Adverse events involving our products may lead
the FDA or applicable foreign regulatory agency to delay or deny clearance for our products or result in product recalls that could harm
our reputation, business and financial results.
Once a product receives regulatory
clearance or approval, the agency has the authority to require the recall of commercialized products in the event of adverse side effects,
material deficiencies or defects in design or manufacture. The authority to require a recall must be based on a regulatory finding that
there is a reasonable probability that the product would cause serious injury or death. Manufacturers may, under their own initiative,
recall a product if any material deficiency in a product is found. A government-mandated or voluntary recall by us or one of our distributors
could occur because of adverse side effects, impurities or other product contamination, manufacturing errors, design or labeling defects
or other deficiencies and issues. Recalls of any of our products would divert managerial and financial resources and have an adverse
effect on our financial condition and results of operations. The regulatory agencies require that certain classifications of recalls
be reported to them within ten (10) working days after the recall is initiated. Companies are required to maintain certain records of
recalls, even if they are not reportable to the regulatory agency. We may initiate voluntary recalls involving our products in the future
that we determine do not require notification of the regulatory agencies. If the regulatory agency disagrees with our determinations,
they could require us to report those actions as recalls. A future recall announcement could harm our reputation with customers and negatively
affect our sales. In addition, the regulatory agency could take enforcement action for failing to report the recalls when they were conducted.
The in-licensing of technologies and the successful
testing and early development of technologies in the laboratory may not be indicative of future results and may not result in commercially
viable technologies or products. Further, our future products may have to be modified from their originally conceived versions in order
to reach or be successful in the market.
Positive results from laboratory
testing and early developmental successes, may not be predictive of future successful development, commercialization and sales results
and should not be relied upon as evidence that products developed from our technologies will become commercially viable and successful.
Further, the products we plan to develop in the future may have to be significantly modified from their originally conceived versions
in order for us to control costs, compete with similar products, receive market acceptance, meet specific development and commercialization
timeframes, avoid potential infringement of the proprietary rights of others, or otherwise succeed in developing our business and earning
ongoing revenues. This can be a costly and resource draining activity. What appear to be promising technologies when we license them
may not lead to viable technologies or products, or to commercial success.
Complying with numerous regulations pertaining
to our business is an expensive and time-consuming process, and any failure to comply could result in substantial penalties.
We are subject to the Clinical
Laboratory Improvement Amendment of 1988, or CLIA, which is a federal law regulating clinical laboratories that perform testing on specimens
derived from humans for the purpose of providing information for the diagnosis, prevention, or treatment of disease. Our clinical laboratory
is in Richmond, Virginia and must be certified under CLIA for us to perform testing on human specimens. CLIA is intended to ensure the
quality and reliability of clinical laboratories in the United States by mandating specific standards in the areas of personnel qualifications,
administration, and participation in proficiency testing, patient test management, quality control, quality assurance and inspections.
We currently hold a CLIA certificate to perform high-complexity testing. Laboratories performing high complexity testing are required
to meet more stringent requirements than laboratories performing fewer complex tests. CLIA regulations require clinical laboratories
like ours to comply with various operational, personnel, facilities administration, quality, and proficiency testing requirements intended
to ensure that testing services are accurate, reliable, and timely. CLIA certification is a prerequisite for reimbursement eligibility
for services provided to state and federal health care program beneficiaries. CLIA is user-fee funded. Therefore, all costs of administering
the program must be covered by the regulated facilities, including certification and survey costs. To renew this certificate, we are
subject to survey and inspection every two years. Moreover, CLIA inspectors may make periodic inspections of our clinical laboratory
outside of the renewal process. The failure to comply with CLIA requirements can result in enforcement actions, including the revocation,
suspension, or limitation of our CLIA certificate of compliance, as well as a directed plan of correction, state on-site monitoring,
civil money penalties, civil injunctive suit and/or criminal penalties. We must maintain CLIA compliance and certification to be eligible
to bill for assays provided to Medicare beneficiaries. If we were to be found out of compliance with CLIA program requirements and subjected
to sanctions, our business and reputation could be harmed. Even if it were possible for us to bring our laboratory back into compliance,
we could incur significant expenses and potentially lose revenue in doing so.
Additionally, certain states
require laboratory licenses to test specimens from patients in those states or received from ordering physicians in those states. We
may also be subject to regulation in foreign jurisdictions if we seek to expand international distribution of our assays outside the
United States.
If we were to lose our CLIA certification
or state laboratory licenses, whether because of a revocation, suspension or limitation, we would no longer be able to offer our assays
(including our AditxtScore™ platform), which would limit our revenues and harm our business. If we were to lose, or fail to obtain,
a license in any other state where we are required to hold a license, we would not be able to test specimens from those states.
Our AditxtScore™ tests are currently
being offered as a LDTs. Should the FDA disagree that AditxtScore™ tests are LDTs, if our LDTs do not receive the required emergency
use authorizations, or if the FDA’s regulatory approach to LDTs should change in the future, our commercialization strategy may
be adversely affected, which would negatively affect our results of operations and financial condition.
The FDA has historically asserted
its authority to regulate Laboratory Developed Tests (LDTs) as medical devices under the Federal Food, Drug, and Cosmetic Act (the “FDCA”),
but it has generally exercised enforcement discretion regarding LDTs. This means that even though the FDA believes it can impose regulatory
requirements on LDTs, such as requirements to obtain premarket approval, de novo classification, or clearance of LDTs,
it has generally chosen not to enforce those requirements. The FDA has, on occasion, sent warning letters to laboratories offering LDTs
that the agency believed were not eligible for enforcement discretion because of how they were developed, validated, performed or marketed
and consequent risks to the public.
The FDA considers an LDT to be
a test that is developed, validated, and performed within a single laboratory. We are providing AditxtScore™ as a service as a
Laboratory Developed Test (LDT) to assess immunity status to COVID-19. Our AditxtScore™ tests are currently manufactured
in our Mountain View, CA facility and performed in our Richmond, VA facility. If the FDA believes that the AditxtScore™ is not
regulated as an LDT, we may be forced to stop performing AditxtScore™ while we worked to obtain the appropriate FDA authorizations
which could negative affect our business, results of operations and financial condition.
On November 15, 2021, FDA revised
its guidance document titled “Policy for Coronavirus Disease-2019 Tests During the Public Health Emergency (Revised)” (“FDA
COVID-19 Testing Guidance”) to require all COVID-19 diagnostic assays conducted as LDTs to apply for EUA authorization within a
60-day period from the revised guidance’s issuance date. The FDA COVID-19 Testing Guidance states that FDA does not intend
to object to continued offering of LDTs that are the subject of submitted EUA requests while FDA reviews the EUA requests. The FDA COVID-19
Testing Guidance further states that if FDA declines to issue an EUA or otherwise decides not to authorize a test for any reason, including
a determination that there is a lack of adequate data to support authorization, FDA generally expects developers to cease marketing and
offering their test within 15 calendar days. Moreover, if FDA identifies a significant problem or concern with a test, based either on
the provided information or external reports, FDA generally expects the developer to take appropriate steps to address such problems,
which could include a recall of the test and/or notification concerning corrected test reports indicating prior test results may not
be accurate. We have submitted EUA requests for our SARS-CoV-2 LDTs, and the applications are pending before FDA. There can
be no assurance that the EUA requests that we submitted for our SARS-CoV-2 LDTs will be granted on a timely basis or at all.
If FDA declines to issue a EUAs for our SARS-CoV-2 LDTs, we may be required to cease marketing the tests and our business, results
of operations and financial condition could be negatively affected. Regardless of if our EUA applications are granted by FDA, we may
recall, replace, or make corrections to our LDTs if we become aware of a product concern, which could negatively impact manufacturing,
supply, and customer relationships, and may result in adverse regulatory action, including revision or revocation of an EUA.
In addition, there have been
numerous legislative proposals to clarify the FDA’s regulatory authority over medical devices. These include two bills reintroduced
in 2021: the VALID Act, which would expressly grant the FDA authority to regulate LDTs under a risk-based framework; and the VITAL Act,
which would assign LDTs to regulation solely under CLIA and would direct CMS to update its CLIA regulations. We cannot predict if either
of these bills will be enacted in their current (or any other) form and cannot quantify the effect of these bills on our business. In
the meantime, the regulation by the FDA of LDTs remains uncertain. If FDA premarket review, classification or approval is required for
AditxtScore™, our laboratory could be forced to stop performing AditxtScore™ while we worked to obtain the appropriate
FDA authorizations which could negative affect our business, results of operations and financial condition.
We are subject to
various governmental regulations relating to the labeling, marketing and sale of our products.
Both before and after a product
is commercially released, we have ongoing responsibilities under regulations promulgated by the FDA, the Federal Trade Commission, and
similar U.S. and foreign regulations governing product labeling and advertising, distribution, sale, and marketing of our products.
Manufacturers of medical devices
are permitted to promote products solely for the uses and indications set forth in the device’s authorization. A number of enforcement
actions have been taken against manufacturers that promote products for “off-label” uses (i.e., uses that are not
described in the device’s authorization), including actions alleging that claims submitted to government healthcare programs for
reimbursement of products that were promoted for “off-label” uses are fraudulent in violation of the Federal False
Claims Act or other federal and state statutes and that the submission of those claims was caused by off-label promotion. The
failure to comply with prohibitions on “off-label” promotion can result in significant monetary penalties, revocation
or suspension of a company’s business license, suspension of sales of certain products, product recalls, civil or criminal sanctions,
exclusion from participating in federal healthcare programs, or other enforcement actions. In the United States, allegations of such
wrongful conduct could also result in a corporate integrity agreement with the U.S. government that imposes significant administrative
obligations and costs.
We and our employees and contractors are subject,
directly, or indirectly, to federal, state and foreign healthcare fraud and abuse laws, including false claims laws. If we are unable
to comply, or have not fully complied, with such laws, we could face substantial penalties.
Our operations are subject to
various federal, state, and foreign fraud and abuse laws. These laws may constrain our operations, including the financial arrangements
and relationships through which we market, sell and distribute our products.
U.S. federal and state laws that
affect our ability to operate include, but are not limited to:
| |
● |
the federal Anti-Kickback
Statute, which prohibits, among other things, persons or entities from knowingly and willfully soliciting, receiving, offering or
paying any remuneration (including any kickback, bribe, or rebate), directly or indirectly, overtly or covertly, in cash or in kind
in return for, the purchase, recommendation, leasing or furnishing of an item or service reimbursable under a federal healthcare
program, such as the Medicare and Medicaid programs; |
| |
● |
federal physician self-referral
law, which prohibits a physician from referring a patient to an entity with which the physician (or an immediate family member) has
a financial relationship, for the furnishing of certain designated health services for which payment may be made by Medicare or Medicaid,
unless an exception applies; |
| |
● |
federal civil and criminal
false claims laws and civil monetary penalty laws, which prohibit, among other things, individuals, or entities from knowingly presenting,
or causing to be presented, claims for payment or approval from Medicare, Medicaid, or other government payers that are false or
fraudulent; |
| |
● |
Section 242 of HIPAA codified
at 18 U.S.C. § 1347, which created new federal criminal statutes that prohibit a person from knowingly and willfully executing
a scheme or from making false or fraudulent statements to defraud any healthcare benefit program (i.e., public or private); |
| |
● |
federal transparency laws,
including the Physician Payments Sunshine Act which requires the tracking and disclosure to the federal government by pharmaceutical
and medical device manufacturers of payments and other transfers of value to physicians and teaching hospitals as well as ownership
and investment interests that are held by physicians and their immediate family members; and |
| |
● |
state law equivalents of
each of these federal laws, such as anti-kickback and false claims laws that may apply to items or services reimbursed by any third-party
payer, including commercial insurers; state laws that require pharmaceutical and medical device companies to comply with their industry’s
voluntary compliance guidelines and the applicable compliance guidance promulgated by the federal government or otherwise restrict
certain payments that may be made to healthcare providers and other potential referral sources; state laws that require drug and
medical device manufacturers to report information related to payments and other transfers of value to physicians and other healthcare
providers or marketing expenditures; state laws that prohibit giving gifts to licensed healthcare professionals; and state laws governing
the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways
and may not have the same effect, thus complicating compliance efforts in certain circumstances, such as specific disease states.
|
In particular, activities and
arrangements in the healthcare industry are subject to extensive laws and regulations intended to prevent fraud, waste and other abusive
practices. These laws and regulations may restrict or prohibit a wide range of activities or other arrangements related to the development,
marketing or promotion of products, including pricing and discounting of products, provision of customer incentives, provision of reimbursement
support, other customer support services, provision of sales commissions or other incentives to employees and independent contractors
and other interactions with healthcare practitioners, other healthcare providers and patients.
Because of the breadth of these
laws and the narrow scope of the statutory or regulatory exceptions and safe harbors available, our business activities could be challenged
under one or more of these laws.
Government expectations and industry
best practices for compliance continue to evolve and past activities may not always be consistent with current industry best practices.
Further, there is a lack of government guidance as to whether various industry practices comply with these laws, and government interpretations
of these laws continue to evolve, all of which create compliance uncertainties. Any non-compliance could result in regulatory sanctions,
criminal or civil liability and serious harm to our reputation. It is not always possible to identify and deter misconduct concerning
applicable laws, regulations, guidelines, policies and standards, and the precautions we take to detect and prevent this activity may
not be effective in preventing such conduct, mitigating risks, or reducing the chance of governmental investigations or other actions
or lawsuits stemming from a failure to comply with these laws or regulations.
If a government entity opens
an investigation into possible violations of any of these laws (which may include the issuance of subpoenas or civil investigative demands),
we would have to expend significant resources to defend ourselves against the allegations. Allegations that we, our officers, or our
employees violated any one of these laws can be made by individuals called “whistleblowers” who may be our employees, customers,
competitors or other parties. Government policy is to encourage individuals to become whistleblowers and file a complaint in federal
court alleging wrongful conduct. The government is required to investigate all of these complaints and decide whether to intervene. If
the government intervenes and we are required to pay money back to the government, the whistleblower, as a reward, is awarded a percentage
of the collection. If the government declines to intervene, the whistleblower may proceed on their own and, if they are successful, they
will receive a percentage of any judgment or settlement amount the company is required to pay. The government may also initiate an investigation
on its own. Such actions could have a significant impact on our business, including the imposition of significant fines, and other sanctions
that may materially impair our ability to run a profitable business. In particular, if our operations are found to be in violation of
any of the laws described above or if we agree to settle with the government without admitting to any wrongful conduct or if we are found
to be in violation of any other governmental regulations that apply to us, we, our officers and employees may be subject to sanctions,
including civil and criminal penalties, damages, fines, exclusion from participation in government health care programs, such as Medicare
and Medicaid, imprisonment, the curtailment or restructuring of our operations and the imposition of a corporate integrity agreement,
any of which could adversely affect our business, results of operations and financial condition.
Risks Related to our Company and our Business
Our technology is subject to licenses from
LLU and Stanford, each of which are revocable in certain circumstances, including in the event we do not achieve certain payments and
milestone deadlines. Without these licenses, we may not be able to continue to develop our product candidates.
The LLU License Agreement may
be terminated by LLU in the event of a breach by us of any non-payment provision (including the provision that requires us to meet certain
deadlines for milestone events (each, a “Milestone Deadline”)) not cured within 90 days after delivery of written notice
by LLU. Additional Milestone Deadlines include: (i) the requirement to have regulatory approval of an IND application to initiate first-in-human
clinical trials on or before March 31, 2022, (ii) the completion of first-in-human (phase I/II) clinical trials by March 31, 2024, (iii)
the completion of Phase III clinical trials by March 31, 2026 and (iv) biologic licensing approval (BLA) by the FDA by March 31, 2027.
If the LLU License Agreement were to be terminated by LLU, we would lose our most significant asset and may no longer be able to develop
our product candidates, which would have a material adverse effect on our operations.
The February 2020 License Agreement
with Stanford may be terminated by Stanford if we (i) are delinquent on any report or payments; (ii) are not diligently developing and
commercializing Licensed Product (as defined in the February 2020 License Agreement); (iii) miss a milestone described in the agreement;
(iv) are in breach of any other provision of the agreement; or (v) if we provide a false report to Stanford. The Termination discussed
above will take effect only upon 30 days written notice by Stanford unless we remedy the breach within a 30-day cure period. If the February
2020 License Agreement were to be terminated by Stanford, we would lose a significant asset and may no longer be able to develop our
product candidates, which would have a material adverse effect on our operations.
Our results of operations will be affected
by the level of royalty and milestone payments that we are required to pay to third parties.
The LLU License Agreement and
February 2020 License Agreement with Stanford each require us to remit royalty payments and meet certain performance milestones related
to in-licensed intellectual property. Any failure on our part to pay royalties owed or meet milestones could lead to us losing rights
under our licenses and could thereby adversely affect our business. As our product sales increase, we may, from time-to-time, disagree
with our third-party collaborators as to the appropriate royalties owed and the resolution of such disputes may be costly and may consume
management’s time. Furthermore, we may enter into additional license agreements in the future, which may also include royalty payments.
We face substantial competition, which may
result in others discovering, developing or commercializing products before or more successfully than we do.
The development and commercialization
of drugs is highly competitive. We compete with a variety of multinational pharmaceutical companies and specialized biotechnology companies,
as well as products and processes being developed at universities and other research institutions. Our competitors have developed, are
developing or will develop product candidates and processes competitive with our product candidates. Competitive therapeutic treatments
include those that have already been approved and accepted by the medical community and any new treatments that may enter the market.
We believe that a significant number of products are currently available, under development, and may become commercially available in
the future, for the treatment of indications for which we may try to develop product candidates.
More established companies may
have a competitive advantage over us due to their greater size, cash flows and institutional experience. Compared to us, many of our
competitors may have significantly greater financial, technical and human resources. As a result of these factors, our competitors may
have an advantage in marketing their approved products and may obtain regulatory approval of their product candidates before we are able
to, which may limit our ability to develop or commercialize our product candidates. Our competitors may also develop drugs that are safer,
more effective, more widely used and less expensive than ours, and may also be more successful than us in manufacturing and marketing
their products.
Mergers and acquisitions in the
pharmaceutical and biotechnology industries may result in even more resources being concentrated among a smaller number of our competitors.
Smaller and other early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements
with large and established companies. These companies compete with us in recruiting and retaining qualified scientific, management and
commercial personnel, establishing clinical trial sites and subject registration for clinical trials, as well as in acquiring technologies
complementary to, or necessary for, our programs.
Our technologies and products under development,
and our business, may fail if we are not able to successfully commercialize them and ultimately generate significant revenues as a result.
Successful development of technologies
and our product candidates will require significant additional investment, including costs associated with additional development, completing
trials and obtaining regulatory approval, as well as the ability to manufacture or have others manufacture our products in sufficient
quantities at acceptable costs while also preserving product quality. Difficulties often encountered in scaling up production include
problems involving production yields, quality control and assurance, shortage of qualified personnel, production costs and process controls.
In addition, we are subject to inherent risks associated with new technologies and products. These risks include the possibility that
any of our technologies or future products may:
| |
● |
be ineffective or less effective than anticipated; |
| |
● |
fail to receive necessary regulatory approvals; |
| |
● |
be difficult to competitively price relative to alternative
solutions; |
| |
● |
be harmful to consumers or the environment; |
| |
● |
be difficult to manufacture on an economically viable
scale; |
| |
● |
be subject to supply chain constraints for raw materials; |
| |
● |
fail to be developed and
accepted by the market prior to the successful marketing of alternative products by competitors; |
| |
● |
be difficult to market
because of infringement on the proprietary rights of third parties; or |
| |
● |
be too expensive for commercial
use. |
Furthermore, we may be faced
with lengthy market partner or distributor evaluation and approval processes. Consequently, we may incur substantial expenses and devote
significant management effort to customize products for market partner or distributor acceptance, though there can be no assurance of
such acceptance. As a result, we cannot accurately predict the volume or timing of any future sales.
Customers may not adopt our products quickly,
or at all.
Customers in the sector in which
we operate can be generally cautious in their adoption of new products and technologies. In addition, given the relative novelty of our
future planned products (including our AditxtScore™ platform), customers of those products may require education regarding their
utility and use, which may delay their adoption. There can be no assurance that customers will adopt our products quickly, or at all.
The significant level of competition in the
markets for our products developed in the future may result in pricing pressure, reduced margins or the inability of our future products
to achieve market acceptance.
The markets for our future products
are intensely competitive and rapidly changing. We may be unable to compete successfully, which may result in price reductions, reduced
margins and the inability to achieve market acceptance for our products.
Our competitors may have longer
operating histories, significantly greater resources, greater brand recognition and large customer bases than we do. As a result, they
may be able to devote greater resources to the manufacture, promotion or sale of their products, receive greater resources and support
from market partners and independent distributors, initiate or withstand substantial price competition or more readily take advantage
of acquisition or other opportunities.
We rely on third parties for the distribution
of our current and future products, including our AditxtScore™ platform. If these parties do not distribute our products in
a satisfactory or timely manner, in sufficient quantities or at an acceptable cost, our sales and development efforts could be delayed
or otherwise negatively affected.
We rely on third parties for
the distribution of our current and future products, including our AditxtScore™ platform. Our reliance on third parties to distribute
products may present significant risks to us, including the risk that should any of these third parties fail to adequately distribute
our products and services to end consumers and other market participants, our business may be materially harmed. Additionally, if we
need to enter into agreements for the distribution of our future products with other third parties, there can be no assurance we will
be able to do so on favorable terms, if at all.
We may rely on third parties to produce our
future products. If these parties do not produce our products at a satisfactory quality, in a timely manner, in sufficient quantities
or at an acceptable cost, our sales and development efforts could be delayed or otherwise negatively affected.
We may rely on third parties
for the manufacture of our future products. Our reliance on third parties to manufacture our future products may present significant
risks to us, including the following:
| |
● |
reduced control over delivery
schedules, yields and product reliability; |
| |
● |
manufacturing deviations
from internal and regulatory specifications; |
| |
● |
the failure of a key manufacturer
to perform as we require for technical, market or other reasons; |
| |
● |
difficulties in establishing
additional manufacturer relationships if we are presented with the need to transfer our manufacturing process technologies to them; |
| |
● |
misappropriation of our
intellectual property; and |
| |
● |
other risks in potentially
meeting our product development schedule or satisfying the requirements of our market partners, distributors, direct customers and
end users. |
If we need to enter into agreements
for the manufacturing of our future products, there can be no assurance we will be able to do so on favorable terms, if at all.
If we are unable to establish successful relations
with third-party market partners or distributors, or these market partners or distributors do not focus adequate resources on selling
our products or are otherwise unsuccessful in selling them, sales of our products may not develop.
We anticipate relying on independent
market partners and distributors to distribute and assist us with the marketing and sale of our products. Our future revenue generation
and growth will depend in large part on our success in establishing and maintaining this sales and distribution channel. If our market
partners and distributors are unable to sell our products, or receive negative feedback from end users, they may not continue to purchase
or market our products. In addition, there can be no assurance that our market partners and distributors will focus adequate resources
on selling our products to end users or will be successful in selling them. Many of our potential market partners and distributors are
in the business of distributing and sometimes manufacturing other, possibly competing, products. As a result, these market partners
and distributors may perceive our products as a threat to various product lines currently being distributed or manufactured by them.
In addition, these market partners and distributors may earn higher margins by selling competing products or combinations of competing
products. If we are unable to establish successful relationships with independent market partners and distributors, we will need to further
develop our own sales and distribution capabilities, which would be expensive and time-consuming and might not be successful.
If we are not able to attract and retain highly
skilled employees and contractors, we may not be able to implement our business model successfully.
We will rely upon employees and
third-party consultant/contractors to effectively establish, manage and grow our business. Consequently, we believe that our future viability
will depend largely on our ability to attract and retain highly skilled personnel. In order to do so, we may need to pay higher
compensation, fees, and/or other incentives to our employees or consultants than we currently expect, and such higher compensation payments
would have a negative effect on our operating results. Competition for experienced, high-quality employees, consultants and contractors
is intense and we cannot assure that we will be able to recruit and retain such personnel. We may not be able to hire or retain the necessary
personnel to implement our business strategy. Our failure to hire and retain such personnel could impair our ability to develop new products
and manage our business effectively.
The loss of our management team or other key
personnel would have an adverse impact on our future development and impair our ability to succeed.
In the early stages of development,
our business will be significantly dependent on the Company’s management team and other key personnel. Our success will be particularly
dependent upon Mr. Amro Albanna and Dr. Shahrokh Shabahang. The loss of any one of these individuals or any other future key personnel
could have a material adverse effect on the Company and our ability to further execute our intended business.
The use of our products may be limited by regulations,
and we may be exposed to product liability and remediation claims.
The use of our planned products
may be regulated by various local, state, federal and foreign regulators. Even if we are able to comply with all such regulations
and obtain all necessary registrations, we cannot provide assurance that our future products will not cause injury to the environment,
people, or animals and/or otherwise have unintended adverse consequences, under all circumstances. For example, our products may be improperly
combined with other chemicals or, even when properly combined, our products may be blamed for damage caused by those other chemicals.
The costs of remediation or products liability could materially adversely affect our results, financial condition and operations.
We may be held liable for, or incur costs to
settle, liability and remediation claims if any products we develop, or any products that use or incorporate any of our technologies,
cause injury or are found unsuitable during product testing, manufacturing, marketing, sale or use. These risks exist even with respect
to products that have received, or may in the future receive, regulatory approval, registration or clearance for commercial use. We cannot
guarantee that we will be able to avoid product liability exposure.
At the stage customary to do
so, we expect to maintain product liability insurance at levels we believe are sufficient and consistent with industry standards for
like companies and products. However, we cannot guarantee that our product liability insurance will be sufficient to help us avoid product
liability-related losses. In the future, it is possible that meaningful insurance coverage may not be available on commercially reasonable
terms or at all. In addition, a product liability claim could result in liability to us greater than our assets or insurance coverage.
Moreover, even if we have adequate insurance coverage, product liability claims or recalls could result in negative publicity or force
us to devote significant time and attention to these matters, which could harm our business.
There may be limitations on the effectiveness
of our internal controls, and a failure of our control systems to prevent error or fraud may materially harm our Company.
We do not expect that internal
control over financial accounting and disclosure, even if timely and well established, will prevent all error and all fraud. A control
system, no matter how well designed and operated, can provide only reasonable, not absolute, assurance that the control system’s
objectives will be met. Further, the design of a control system must reflect the fact that there are resource constraints, and the benefits
of controls must be considered relative to their costs. Because of the inherent limitations in all control systems, no evaluation of
controls can provide absolute assurance that all control issues and instances of fraud, if any, have been detected. Failure of our control
systems to prevent error or fraud could materially adversely affect our business.
COVID-19 may impact our operations.
On January 30, 2020, the World
Health Organization declared the COVID-19 coronavirus outbreak a “Public Health Emergency of International Concern” and on
March 10, 2020, declared it to be a pandemic. Actions taken around the world to help mitigate the spread of the coronavirus include restrictions
on travel, and quarantines in certain areas, and forced closures for certain types of public places and businesses. The COVID-19 coronavirus
and actions taken to mitigate it have had and are expected to continue to have an adverse impact on the economies and financial markets
of many countries, including the geographical area in which the Company operates. While it is unknown how long these conditions will
last and what the complete financial effect will be to the Company, capital raise efforts and additional development of our technologies
may be negatively affected.
Risks Related to Our Acquisition Strategy
Our acquisition strategy exposes us to substantial
risk.
Our acquisition of companies
is subject to substantial risk, including but not limited to the failure to identify material problems during due diligence (for which
we may not be indemnified post-closing), the risk of over-paying for assets (or not making acquisitions on an accretive basis), the ability
to obtain or retain customers and the risks of entering markets where we have limited experience. While we perform due diligence on prospective
acquisitions, we may not be able to discover all potential operational deficiencies in such entities.
Our
acquisition targets may not perform as expected or the returns from such businesses may not support the financing utilized to acquire
them or maintain them. Furthermore, integration and consolidation of acquired businesses requires substantial human, financial and other
resources and may divert management’s attention from our existing business concerns, disrupt our ongoing business or not be successfully
integrated. Even if we consummate businesses that we believe will be accretive, those businesses may in fact result in a decrease in
revenues as a result of incorrect assumptions in our evaluation of such businesses, unforeseen consequences, or other external events
beyond our control. Furthermore, if we consummate any future acquisitions, our capitalization and results of operations may change significantly,
and stockholders will generally not have the opportunity to evaluate the economic, financial, and other relevant information that we
will consider in determining the application of these funds and other resources. As a result, the consummation of acquisitions may have
a material adverse effect on our business, financial condition, results of operations and cash flows.
We may experience difficulty as we evaluate,
acquire and integrate businesses that we may acquire, which could result in drains on our resources, including the attention of our management,
and disruptions of our on-going business.
We acquire small to mid-sized
businesses in various industry segments. Generally, because such businesses are privately held, we may experience difficulty in evaluating
potential target businesses as much of the information concerning these businesses is not publicly available. Therefore, our estimates
and assumptions used to evaluate the operations, management and market risks with respect to potential target businesses may be subject
to various risks and uncertainties. Further, the time and costs associated with identifying and evaluating potential target businesses
may cause a substantial drain on our resources and may divert our management team’s attention away from the operations of our businesses
for significant periods of time.
In addition, we may have difficulty
effectively integrating and managing acquisitions. The management or improvement of businesses we acquire may be hindered by a number
of factors, including limitations in the standards, controls, procedures and policies implemented in connection with such acquisitions.
Further, the management of an acquired business may involve a substantial reorganization of the business’ operations resulting
in the loss of employees and customers or the disruption of our ongoing businesses. We may experience greater than expected costs or
difficulties relating to an acquisition, in which case, we might not achieve the anticipated returns from any particular acquisition.
We may not be able to effectively integrate
the businesses that we acquire.
Our ability to realize the anticipated
benefits of acquisitions will depend on our ability to integrate those businesses with our own. The combination of multiple independent
businesses is a complex, costly and time-consuming process and there can be no assurance that we will be able to successfully integrate
businesses into our business, or if such integration is successfully accomplished, that such integration will not be costlier or take
longer than presently contemplated. Integration of future acquisitions may include various risks and uncertainties, including the factors
discussed in the paragraph below. If we cannot successfully integrate and manage the businesses within a reasonable time, we may not
be able to realize the potential and anticipated benefits of such acquisitions, which could have a material adverse effect on our stock
price, business, cash flows, results of operations and financial position.
We will consider acquisitions
that we believe will complement, strengthen and enhance our growth. We evaluate opportunities on a preliminary basis from time to time,
but these transactions may not advance beyond the preliminary stages or be completed. Such acquisitions are subject to various risks
and uncertainties, including:
| |
● |
the inability to integrate
effectively the operations, products, technologies and personnel of the acquired companies (some of which are in diverse geographic
regions) and achieve expected synergies; |
| |
● |
the potential disruption
of existing business and diversion of management’s attention from day-to-day operations; |
| |
● |
the inability to maintain
uniform standards, controls, procedures and policies; |
| |
● |
the need or obligation
to divest portions of the acquired companies; |
| |
● |
the potential failure to
identify material problems and liabilities during due diligence review of acquisition targets; |
| |
● |
the potential failure to
obtain sufficient indemnification rights to fully offset possible liabilities associated with acquired businesses; and |
| |
● |
the challenges associated
with operating in new geographic regions. |
The integration of
our acquisitions may result in significant accounting charges that adversely affect the announced results of our Company.
The financial results of our
Company may be adversely affected by cash expenses and non-cash accounting charges incurred in connection with our recent acquisitions.
In addition to the anticipated cash charges, costs associated with the amortization of intangible assets are expected. The price of our
Common Stock could decline to the extent our financial results are materially affected by the foregoing charges or if the foregoing charges
are larger than anticipated.
Our planned acquisitions
may result in unexpected consequences to our business and results of operations.
Although we believe that our
planned acquisitions will generally be subject to risks similar to those to which we are subject to in our existing operations, we may
not have discovered all risks applicable to these businesses during the due diligence process. Some of these risks could produce unexpected
and unwanted consequences for us. Undiscovered risks may result in us incurring financial liabilities, which could be material and have
a negative impact on our business operations.
Failure to manage our growing and changing
business could have a material adverse effect on our business, prospects, financial condition, and results of operations.
As we grow, we expect to encounter
additional challenges to our internal processes, capital commitment process, and acquisition funding and financing capabilities. Our
existing operations, personnel, systems, and internal control may not be adequate to support our growth and expansion and may require
us to make additional unanticipated investments in our infrastructure. To manage the future growth of our operations, we will be required
to improve our administrative, operational, and financial systems, procedures, and controls, and maintain, expand, train, and manage
our growing employee base. If we are unable to manage our growth effectively, we may not be able to take advantage of market opportunities,
execute our business strategies successfully or respond to competitive pressures. As a result, our business, prospects, financial condition,
and results of operations could be materially and adversely affected.
We face competition for businesses that fit
our acquisition strategy and, therefore, we may have to acquire targets at sub-optimal prices or, alternatively, forego certain acquisition
opportunities.
Our acquisition strategy is focused
on the acquisition of small to mid-sized businesses. In pursuing such acquisitions, we expect to face strong competition from a wide
range of other potential purchasers. Although the pool of potential purchasers for such businesses is typically smaller than for larger
businesses, those potential purchasers can be aggressive in their approach to acquiring such businesses. Furthermore, we expect that
we will need to use third-party financing in order to fund some or all of these potential acquisitions, thereby increasing our acquisition
costs. To the extent that other potential purchasers do not need to obtain third-party financing or are able to obtain such financing
on more favorable terms, they may be in a position to be more aggressive with their acquisition proposals. As a result, in order to be
competitive, our acquisition proposals may need to be aggressively priced, including at price levels that exceed what we originally determined
to be fair or appropriate. Alternatively, we may determine that we cannot pursue on a cost-effective basis what would otherwise be an
attractive acquisition opportunity.
We may not be able to successfully fund acquisitions
due to the unavailability of equity or debt financing on acceptable terms, which could impede the implementation of our acquisition strategy.
We intend to finance acquisitions
primarily through additional debt and equity financings. Because the timing and size of acquisitions cannot be readily predicted, we
may need to be able to obtain funding on short notice to benefit fully from attractive acquisition opportunities. The sale of additional
shares of any class of equity will be subject to market conditions and investor demand for such shares at prices that may not be in the
best interest of our stockholders. The sale of additional equity securities could also result in dilution to our stockholders. The incurrence
of indebtedness would result in increased debt service obligations and could require us to agree to operating and financial covenants
that would restrict our operations. Financing may not be available in amounts or on terms acceptable to us, if at all. These risks may
materially adversely affect our ability to pursue our acquisition strategy.
We may change our management and acquisition
strategies without the consent of our stockholders, which may result in a determination by us to pursue riskier business activities.
We may change our strategy at
any time without the consent of our stockholders, which may result in our acquiring businesses or assets that are different from, and
possibly riskier than, the strategy described in this prospectus. A change in our strategy may increase our exposure to interest rate
and currency fluctuations, subject us to regulation under the Investment Company Act or subject us to other risks and uncertainties
that affect our operations and profitability.
In the future, we may seek to enter into credit
facilities to help fund our acquisition capital and working capital needs. These credit facilities may expose us to additional risks
associated with leverage and may inhibit our operating flexibility.
We may seek to enter into credit
facilities with third-party lenders to help fund our acquisitions. Such credit facilities will likely require us to pay a commitment
fee on the undrawn amount and will likely contain a number of affirmative and restrictive covenants. If we violate any such covenants,
our lenders could accelerate the maturity of any debt outstanding. Such debt may be secured by our assets, including the stock we may
own in businesses that we acquire. Our ability to meet our debt service obligations may be affected by events beyond our control and
will depend primarily upon cash produced by businesses that we currently manage and may acquire in the future and distributed or paid
to us. Any failure to comply with the terms of our indebtedness may have a material adverse effect on our financial condition.
In addition, we expect that such
credit facilities will bear interest at floating rates which will generally change as interest rates change. We will bear the risk that
the rates that we are charged by our lenders will increase faster than we can grow the cash flow from our businesses or businesses that
we may acquire in the future, which could reduce profitability, materially adversely affect our ability to service our debt, cause us
to breach covenants contained in our third-party credit facilities and reduce cash flow available for distribution.
If, in the future, we cease to control and
operate our businesses or other businesses that we acquire in the future or engage in certain other activities, we may be deemed to be
an investment company under the Investment Company Act.
We have the ability to make investments
in businesses that we will not operate or control. If we make significant investments in businesses that we do not operate or control,
or that we cease to operate or control, or if we commence certain investment-related activities, we may be deemed to be an investment
company under the Investment Company Act. Our decision to sell a business will be based upon financial, operating and other considerations
rather than a plan to complete a sale of a business within any specific time frame. If we were deemed to be an investment company, we
would either have to register as an investment company under the Investment Company Act, obtain exemptive relief from the Securities
and Exchange Commission, or the SEC, or modify our investments or organizational structure or our contract rights to fall outside the
definition of an investment company. Registering as an investment company could, among other things, materially adversely affect our
financial condition, business and results of operations, materially limit our ability to borrow funds or engage in other transactions
involving leverage and require us to add directors who are independent of us and otherwise will subject us to additional regulation that
will be costly and time-consuming.
If intangible assets
and goodwill that we recorded in connection with our acquisitions become impaired, we may have to take significant charges against earnings.
In connection
with the accounting for our completed acquisitions, we may be required to record a significant amount of intangible assets, including
developed technology, in-process research and development, and customer relationships relating to the acquired product lines, and goodwill.
Under generally accepted accounting principles in the United States, we must assess, at least annually and potentially more frequently,
whether the value of indefinite-lived intangible assets and goodwill have been impaired. Intangible assets and goodwill are assessed
for impairment in the event of an impairment indicator. Any reduction or impairment of the value of intangible assets and goodwill will
result in a charge against earnings, which could materially adversely affect our results of operations and shareholders’ equity
in future periods.
While we have entered into a Merger Agreement
with Evofem, we cannot assure you that the transactions contemplated by the Merger Agreement will be consummated or, that if such
transactions are consummated, they will be accretive to stockholder value.
The
Merger Agreement may be terminated by us or Evofem if the merger has not been consummated by May 8, 2023. We can provide no assurance
that the conditions to the closing of the merger, including but not limited to, the approval of the transaction by our stockholders,
will be completed in the time frame or in the manner currently anticipated, or that we will recognize the anticipated benefits of the
transaction.
Risks Relating to Our Intellectual Property Rights
The failure to obtain or maintain patents,
licensing agreements and other intellectual property could materially impact our ability to compete effectively.
In order for our business to
be viable and to compete effectively, we need to develop and maintain, and we will heavily rely on, a proprietary position with respect
to our technologies and intellectual property. However, there are significant risks associated with our actual or proposed intellectual
property. The risks and uncertainties that we face with respect to our rights principally include the following:
| |
● |
pending patent applications,
we have filed or will file may not result in issued patents or may take longer than we expect to result in issued patents; |
| |
● |
we may be subject to interference
proceedings; |
| |
● |
we may be subject to reexamination
proceedings; |
| |
● |
we may be subject to post
grant review proceedings; |
| |
● |
we may be subject to inter
partes review proceedings; |
| |
● |
we may be subject to derivation
proceedings; |
| |
● |
we may be subject to opposition
proceedings in the U.S. or in foreign countries; |
| |
● |
any patents that are issued
to us may not provide meaningful protection; |
| |
● |
we may not be able to develop
additional proprietary technologies that are patentable; |
| |
● |
other companies may challenge
patents licensed or issued to us; |
| |
● |
other companies may have
independently developed and patented (or may in the future independently develop and patent) similar or alternative technologies,
or duplicate our technologies; |
| |
● |
other companies may design
around technologies we have licensed or developed; |
| |
● |
enforcement of patents
is complex, uncertain, and very expensive and we may not be able to secure, enforce and defend our patents; and |
| |
● |
if we were to ever seek
to enforce our patents in ligation, there is some risk that they could be deemed invalid, not infringed, or unenforceable. |
We cannot be certain that any
patents will be issued because of any pending or future applications, or that any patents, once issued, will provide us with adequate
protection from competing products. For example, issued patents may be circumvented or challenged, declared invalid or unenforceable,
or narrowed in scope. In addition, since publication of discoveries in scientific or patent literature often lags actual discoveries,
we cannot be certain that we or our licensors were the first to invent or to file patent applications covering them.
It is also possible that others
may have or may obtain issued patents that could prevent us from commercializing our products or require us to obtain licenses requiring
the payment of significant fees or royalties to enable us to conduct our business. There is no guarantee that such licenses will be available
based on commercially reasonable terms. As to those patents that we have licensed, our rights depend on maintaining our obligations to
the licensor under the applicable license agreement, and we may be unable to do so.
If we are unable to obtain and maintain patent
protection for our products, or if the scope of the patent protection obtained is not sufficiently broad, competitors could develop and
commercialize products similar or identical to ours, and our ability to successfully commercialize our products could be impaired.
The patent prosecution process
is expensive and time-consuming, and we may not be able to file and prosecute all necessary or desirable patent applications at a reasonable
cost, in a timely manner, or in all jurisdictions. It is also possible that we will fail to identify patentable aspects of our development
output before it is too late to obtain patent protection.
The patent position of life science
companies generally is highly uncertain, involves complex legal and factual questions and has in past years been the subject of much
litigation. In addition, the laws of foreign countries may not protect our rights to the same extent as the laws of the United States
and we may fail to seek or obtain patent protection in all major markets. For example, unlike the U.S., European patent law restricts
the patentability of methods of treatment of the human body. Our pending and future patent applications may not result in patents being
issued which protect our technology or products, in whole or in part, or which effectively prevent others from commercializing competitive
technologies and products. Changes in either the patent laws or interpretation of the patent laws in the United States and other countries
may diminish the value of our patents or narrow the scope of our patent protection, even post-grant.
Recent patent reform legislation
has increased the uncertainties and costs surrounding the prosecution of patent applications and the enforcement or defense of issued
patents. On September 16, 2011, the Leahy-Smith America Invents Act, or the Leahy-Smith Act, was signed into law. The Leahy-Smith Act
includes a number of significant changes to United States patent law. These include provisions that affect the way patent applications
are prosecuted and may also affect patent litigation. The U.S. Patent and Trademark Office, or USPTO, recently developed new regulations
and procedures to govern administration of the Leahy-Smith Act, and many of the substantive changes to patent law associated with the
Leahy-Smith Act, and in particular, the first to file provisions, only became effective on March 16, 2013. Accordingly, it is not clear
what, if any, impact the Leahy-Smith Act will have on the operation of our business. However, the Leahy-Smith Act and its implementation
could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our
issued patents, all of which could have a material adverse effect on our business and financial condition.
Moreover, we may be subject to
a third-party pre-issuance submission of prior art to the USPTO, or become involved in opposition, derivation, reexamination, inter
partes review, post-grant review or interference proceedings challenging our patent rights (whether licensed or otherwise held)
or the patent rights of others. An adverse determination in any such submission, proceeding or litigation could reduce the scope of,
or invalidate, our patent rights (whether licensed or otherwise held), allow third parties to commercialize our technology or products
and compete directly with us, without payment to us, or result in our inability to manufacture or commercialize products without infringing
third-party patent rights. In addition, if the breadth or strength of protection provided by our patents and patent applications (whether
licensed or otherwise held) is threatened, it could dissuade companies from collaborating with us to license, develop or commercialize
current or future product candidates.
Even if our patent applications
(whether licensed or otherwise held) result in the issuance of patents, they may not issue in a form that will provide us with any meaningful
protection, prevent competitors from competing with us or otherwise provide us with any competitive advantage. Our competitors may be
able to circumvent our owned or licensed patents by developing similar or alternative technologies or products in a non-infringing manner.
The issuance of a patent is not
conclusive as to its inventorship, scope, validity or enforceability, and our licensed or owned patents may be challenged in the courts
or patent offices in the United States and abroad. Such challenges may result in loss of exclusivity or freedom to operate or in patent
claims being narrowed, invalidated, or held unenforceable, in whole or in part, which could limit our ability to stop others from using
or commercializing similar or identical products, or limit the duration of the patent protection of our products. Given the amount of
time required for the development, testing and regulatory review of new life science product candidates, patents protecting such candidates
might expire before or shortly after such candidates are commercialized. As a result, our intellectual property rights portfolio may
not provide us with sufficient rights to exclude others from commercializing products similar or identical to ours.
We may become involved in lawsuits to protect
or enforce our intellectual property rights, which could be expensive, time-consuming, and ultimately unsuccessful.
Competitors may infringe our
intellectual property. To counter infringement or unauthorized use, we may be required to file infringement claims, which can be expensive
and time-consuming. Any claims we assert against perceived infringers could provoke these parties to assert counterclaims against us
alleging that we infringe their intellectual property or that our intellectual property is invalid or unenforceable. In addition, in
a patent infringement proceeding, a court may decide that a licensed or owned patent of ours is invalid or unenforceable, in whole or
in part, construe the patent’s claims narrowly or refuse to stop the other party from using the technology at issue on the grounds
that our patents do not cover that technology. Moreover, lawsuits to protect or enforce our intellectual property rights could be expensive,
time-consuming, and ultimately unsuccessful.
Third parties may initiate legal proceedings
alleging that we are infringing their intellectual property rights, the outcome of which would be uncertain.
Our commercial success depends
upon our ability to develop, manufacture, market and sell our product candidates without infringing the proprietary rights of third parties.
There is considerable intellectual property litigation in the life sciences industry. We cannot guarantee that our product candidates
will not infringe third-party patents or other proprietary rights. We may become party to, or threatened with, future adversarial proceedings
or litigation regarding intellectual property rights with respect to our products and technology, including inter partes review,
interference, or derivation proceedings before the USPTO and similar bodies in other countries. Third parties may assert infringement
claims against us based on existing intellectual property rights and intellectual property rights that may be granted in the future.
If we are found to infringe a
third party’s intellectual property rights, we could be required to obtain a license from such third party to continue developing
and marketing our products. However, we may not be able to obtain any required license on commercially reasonable terms or at all. Even
if we were able to obtain a license, it could be non-exclusive, thereby giving our competitors access to the same technologies licensed
to us. We could be forced, including by court order, to cease commercializing the infringing technology or product. In addition, we could
be found liable for monetary damages, including treble damages and attorneys’ fees if we are found to have willfully infringed
a patent. A finding of infringement could prevent us from commercializing our product candidates or force us to cease some of our business
operations, which could materially harm our business. Claims that we have misappropriated the confidential information or trade secrets
of third parties could have a similar negative impact on our business.
Obtaining and maintaining our
patent protection depends on compliance with various procedural, document submission, fee payment and other requirements imposed by governmental
patent agencies, and our own patent protection could be reduced or eliminated for noncompliance with these requirements.
Periodic maintenance fees and
annuities on any issued patent are due to be paid to the USPTO and foreign patent agencies in several stages over the lifetime of the
patent. The USPTO and various foreign governmental patent agencies require compliance with a number of procedural, documentary, fee payment
and other similar provisions during the patent application process. While an inadvertent lapse can in many cases be cured by payment
of a late fee or by other means in accordance with the applicable rules, there are situations in which noncompliance can result in abandonment
or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. Noncompliance
events that could result in abandonment or lapse of a patent or patent application include, but are not limited to, failure to respond
to official actions within prescribed time limits, non-payment of fees and failure to properly legalize and submit formal documents.
In such an event, our competitors might be able to enter our markets, which could have a material adverse effect on our business.
We may be subject to claims by third parties
asserting that our employees or we have misappropriated their intellectual property or claiming ownership of what we regard as our own
intellectual property.
Certain employees and contractors
were previously employed at universities or other companies, including potential competitors. Although we try to ensure that our employees
and contractors do not use the proprietary information or know-how of others in their work for us, we may be subject to claims that these
employees or we have used or disclosed intellectual property, including trade secrets or other proprietary information, of any such employee’s
former employer. Litigation may be necessary to defend against these claims, and any such litigation could have an unfavorable outcome.
In addition, while it is our
policy to require our employees and contractors who may be involved in the development of intellectual property to execute agreements
assigning such intellectual property to us, we may be unsuccessful in executing such an agreement with each party who in fact develops
intellectual property that we regard as our own. Our and their assignment agreements may not be self-executing or may be breached, and
we may be forced to bring claims against third parties, or defend claims they may bring against us, to determine the ownership of what
we regard as our intellectual property.
If we fail in prosecuting or
defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights or personnel. Even
if we are successful in prosecuting or defending against such claims, litigation could result in substantial costs and adverse results,
and be a distraction to management.
Some intellectual property which we own or
have licensed may have been discovered through government funded programs such as, for example, the government funded programs referenced
in intellectual property licensed under the LLU License Agreement, and thus may be subject to federal regulations such as “march-in”
rights, certain reporting requirements, and a preference for United States industry. Compliance with such regulations may limit our exclusive
rights, subject us to expenditure of resources with respect to reporting requirements and limit our ability to contract with non-U.S.
manufacturers.
Some of the intellectual property
rights we own or have licensed have been generated using United States government funding and may therefore be subject to certain federal
regulations. As a result, the United States government may have certain rights to intellectual property embodied in our current or future
products and product candidates pursuant to the Bayh-Dole Act of 1980. These United States government rights in certain inventions developed
under a government-funded program include a non-exclusive, non-transferable, irrevocable worldwide license to use inventions for any
governmental purpose. In addition, the United States government has the right to require us to grant exclusive, partially exclusive,
or non-exclusive licenses to any of these inventions to a third party if it determines that: (i) adequate steps have not been taken
to commercialize the invention; (ii) government action is necessary to meet public health or safety needs; or (iii) government
action is necessary to meet requirements for public use under federal regulations (also referred to as “march-in rights”).
The United States government also has the right to take title to these inventions if we fail to disclose the invention to the government
and fail to file an application to register the intellectual property within specified time limits. In addition, the United States government
may acquire title to these inventions in any country in which a patent application is not filed within specified time limits. Intellectual
property generated under a government funded program is also subject to certain reporting requirements, compliance with which may require
us to expend substantial resources. In addition, the United States government requires that any products embodying the subject invention
or produced using the subject invention be manufactured substantially in the United States. The manufacturing preference requirement
can be waived if the owner of the intellectual property can show that reasonable but unsuccessful efforts have been made to grant licenses
on similar terms to potential licensees that would be likely to manufacture substantially in the United States or that under the circumstances
domestic manufacture is not commercially feasible. This preference for United States manufacturers may limit our ability to contract
with non-U.S. product manufacturers for products covered by such intellectual property. Any exercise by the government of any of the
foregoing rights could harm our competitive position, business, financial condition, results of operations and prospects.
Intellectual property litigation could cause
us to spend substantial resources and distract our personnel from their normal responsibilities.
Even if resolved in our favor,
litigation or other legal proceedings relating to intellectual property claims may cause us to incur significant expenses and could distract
our technical and management personnel from their normal responsibilities. In addition, there could be public announcements of the results
of hearings, motions or other interim proceedings or developments and if securities analysts or investors perceive these results to be
negative, it could have an adverse effect on the price of our Common Stock. Such litigation or proceedings could increase our operating
losses and reduce the resources available for development activities or any future sales, marketing or distribution activities. We may
not have sufficient financial or other resources to conduct such litigation or proceedings adequately. Some of our competitors may be
able to sustain the costs of such litigation or proceedings more effectively than we can because of their greater financial resources.
Uncertainties resulting from the initiation and continuation of patent litigation or other proceedings could compromise our ability to
compete in the marketplace.
We may spend considerable resources developing
and maintaining patents, licensing agreements and other intellectual property that may later be abandoned or may otherwise never result
in products brought to market.
Not all technologies and candidate
products that initially show potential as the basis for future products ultimately meet the rigors of our development process and as
a result may be abandoned and/or never otherwise result in products brought to market. In some cases, prior to abandonment we may
be required to incur significant costs developing and maintaining intellectual property and/or maintaining license agreements and our
business could be harmed by such costs.
We rely on information technology, and if we
are unable to protect against service interruptions, data corruption, cyber-based attacks or network security breaches, our operations
could be disrupted, and our business could be negatively affected.
We rely on information technology
networks and systems to process, transmit and store electronic and financial information; to coordinate our business; and to communicate
within our Company and with customers, suppliers, partners and other third parties. These information technology systems may be susceptible
to damage, disruptions or shutdowns, hardware or software failures, power outages, computer viruses, cyber-attacks, telecommunication
failures, user errors or catastrophic events. If our information technology systems suffer severe damage, disruption or shutdown, and
our business continuity plans do not effectively resolve the issues in a timely manner, our operations could be disrupted, and our business
could be negatively affected. In addition, cyber-attacks could lead to potential unauthorized access and disclosure of confidential information,
and data loss and corruption. There is no assurance that we will not experience these service interruptions or cyber-attacks in the future.
Risks Related to Our Common Stock
We received a written notice from Nasdaq that
we regained compliance with the filing requirement in Listing Rule 5550(b)(1) (the “Equity Rule”), however, we will remain
subject to a Mandatory Panel Monitor for a period of one year ending on December 29, 2024. If, within that one-year monitoring period,
we are unable to maintain compliance with the Equity Rule, it may result in our Common Stock being delisted from the Nasdaq Stock Market.
On
September 29, 2023, we received written notice from Nasdaq that the Hearing Panel had granted the Company an exception through December
26, 2023 to allow the Company to complete its plan to demonstrate compliance with the Equity Rule and Nasdaq Listing Rule 5550(a)(4)
(the “Public Float Rule”). On November 21, 2023, we received written notice from Nasdaq that we had regained compliance with
the Public Float Rule. On December 29, 2023, we received written notice from Nasdaq that we had regained compliance with the Stockholders’
Equity Rule, but will be subject to a Mandatory Panel Monitor for a period of one year. If, within that one-year monitoring period,
we are unable to maintain compliance with the Equity Rule, it may result in our Common Stock being delisted from the Nasdaq Stock Market.
We do not expect to pay dividends in the foreseeable
future.
We do not intend to declare dividends
for the foreseeable future, as we anticipate that we will reinvest any and all future earnings in the development and growth of our business.
Therefore, investors will not receive any funds unless they sell their securities, and holders may be unable to sell their securities
on favorable terms or at all. We cannot assure you of a positive return on your investment or that you will not lose the entire amount
of your investment.
Limitation of Liability and Indemnification
of Management.
The Delaware General Corporation
Law and the Company’s Amended and Restated Certificate of Incorporation provide for the limitation of the liability of directors
for monetary damages. Such provisions may discourage shareholders from bringing a lawsuit against directors for breaches of fiduciary
duty and may also have the effect of reducing the likelihood of derivative litigation against directors and officers even though such
action, if successful, might otherwise be a benefit to the Company’s shareholders. In addition, a shareholder’s investment
in the Company may be adversely affected to the extent that costs of settlement and damage awards against the Company’s officers
or directors are paid by the Company pursuant to such provisions. Additionally, in accordance with Delaware law and the Company’s
Amended and Restated Certificate of Incorporation, the Company shall indemnify, hold harmless and provide advancement of expenses, to
the fullest extent permitted by applicable law, directors, officers, employees, and agents that are made a party or threatened to be
made a party to legal proceedings by reason of the fact that such parties were working at the request of the Company. We direct
you to the Company’s Amended and Restated Certificate of Incorporation for more information.
Anti-takeover provisions under Delaware law
could discourage, delay or prevent a change in control of our Company and could affect the trading price of our securities.
We are a Delaware corporation,
and the anti-takeover provisions of the Delaware General Corporation Law may discourage, delay or prevent a change in control by prohibiting
us from engaging in a business combination with an interested stockholder for a period of three years after the person becomes an interested
stockholder, even if a change in control would be beneficial to our existing stockholders.
Our management team is required to devote substantial
time to public company compliance initiatives.
As a publicly reporting company,
we incur significant legal, accounting, and other expenses. Our management and other personnel devote a substantial amount of time to
comply with our reporting obligations. Moreover, these reporting obligations increase our legal and financial compliance costs and make
some activities more time-consuming and costly.
Failure to develop our internal controls over
financial reporting as we grow could have an adverse impact on us.
As our Company matures, we will
need to develop our current internal control systems and procedures to manage our growth. We are required to establish and maintain appropriate
internal controls over financial reporting. Failure to establish appropriate controls, or any failure of those controls once established,
could adversely impact our public disclosures regarding our business, financial condition, or results of operations. In addition, management’s
assessment of internal controls over financial reporting may identify weaknesses and conditions that need to be addressed in our internal
controls over financial reporting or other matters that may raise concerns for investors. Any actual or perceived weaknesses and conditions
that need to be addressed in our internal control over financial reporting, disclosure of management’s assessment of our internal
controls over financial reporting or disclosure of our public accounting firm’s attestation to or report on management’s
assessment of our internal controls over financial reporting may have an adverse impact on the price of our Common Stock.
We could issue “blank check” preferred
stock without stockholder approval with the effect of diluting interests of then-current stockholders and impairing their voting
rights, and provisions in our charter documents and under Delaware law could discourage a takeover that stockholders may consider favorable.
Our Amended and Restated Certificate
of Incorporation provides for the authorization to issue up to 3,000,000 shares of “blank check” preferred stock with
designations, rights and preferences as may be determined from time to time by our board of directors. Our board of directors is empowered,
without stockholder approval, to issue one or more series of preferred stock with dividend, liquidation, conversion, voting or other
rights which could dilute the interest of, or impair the voting power of, our common stockholders. The issuance of a series of preferred
stock could be used as a method of discouraging, delaying, or preventing a change in control. For example, it would be possible for our
board of directors to issue preferred stock with voting or other rights or preferences that could impede the success of any attempt to
change control of our company. In addition, advanced notice is required prior to stockholder proposals, which might further delay a change
of control.
Our Amended and Restated Certificate of Incorporation
provides that the Court of Chancery of the State of Delaware will be the sole and exclusive forum for substantially all disputes
between the Company and its stockholders, which could limit stockholders’ ability to obtain a favorable judicial forum for disputes
with the Company or its directors, officers or employees.
Our Amended and Restated Certificate
of Incorporation provides that unless the Company consents in writing to the selection of an alternative forum, the State of Delaware
is the sole and exclusive forum for: (i) any derivative action or proceeding brought on behalf of the Company, (ii) any action asserting
a claim of breach of a fiduciary duty owed by any director, officer or other employee of the Company to the Company or the Company’s
stockholders, (iii) any action asserting a claim against the Company, its directors, officers or employees arising pursuant to any provision
of the Delaware General Corporation Law (the “DGCL”) or our Amended and Restated Certificate of Incorporation or
the Company’s Amended and Restated Bylaws, or (iv) any action asserting a claim against the Company, its directors, officers, employees
or agents governed by the internal affairs doctrine, except for, as to each of (i) through (iv) above, any claim as to which the Court
of Chancery determines that there is an indispensable party not subject to the jurisdiction of the Court of Chancery (and the indispensable
party does not consent to the personal jurisdiction of the Court of Chancery within ten days following such determination), which is
vested in the exclusive jurisdiction of a court or forum other than the Court of Chancery, or for which the Court of Chancery does not
have subject matter jurisdiction. This exclusive forum provision would not apply to suits brought to enforce any liability or duty created
by the Securities Act or the Exchange Act or any other claim for which the federal courts have exclusive jurisdiction. To the extent
that any such claims may be based upon federal law claims, Section 27 of the Exchange Act creates exclusive federal jurisdiction over
all suits brought to enforce any duty or liability created by the Exchange Act or the rules and regulations thereunder.
Section 22 of the Securities
Act creates concurrent jurisdiction for federal and state courts over all suits brought to enforce any duty or liability created by the
Securities Act or the rules and regulations thereunder. However, our Amended and Restated Bylaws contain a federal forum provision which
provides that unless the Company consents in writing to the selection of an alternative forum, the federal district courts of the United
States of America will be the exclusive forum for the resolution of any complaint asserting a cause of action arising under the Securities
Act. Any person or entity purchasing or otherwise acquiring any interest in shares of capital stock of the Corporation are deemed to
have notice of and consented to this provision. The Supreme Court of Delaware has held that this type of exclusive federal forum provision
is enforceable. There may be uncertainty, however, as to whether courts of other jurisdictions would enforce such a provision, if applicable.
These choice of forum provisions
may limit a stockholder’s ability to bring a claim in a judicial forum that it finds favorable for disputes with the Company or
its directors, officers or other employees, which may discourage such lawsuits against us and our directors, officers and other employees.
Alternatively, if a court were to find our choice of forum provisions contained in either our Amended and Restated Certificate of Incorporation
or Amended and Restated Bylaws to be inapplicable or unenforceable in an action, we may incur additional costs associated with resolving
such action in other jurisdictions, which could harm our business, results of operations, and financial condition.
We are an “emerging growth company”
and will be able to avail ourselves of reduced disclosure requirements applicable to emerging growth companies, which could make our
Common Stock less attractive to investors.
We are an “emerging growth
company,” as defined in the Jumpstart Our Business Startups Act of 2012 (the “JOBS Act”), and we intend to take advantage
of certain exemptions from various reporting requirements that are applicable to other public companies that are not “emerging
growth companies” including not being required to comply with the auditor attestation requirements of Section 404(b) of the
Sarbanes-Oxley Act, reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements, and
exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and shareholder approval of any golden
parachute payments not previously approved. In addition, pursuant to Section 107 of the JOBS Act, as an “emerging growth company”
we intend to take advantage of the extended transition period provided in Section 7(a)(2)(B) of the Securities Act, for complying
with new or revised accounting standards. In other words, an “emerging growth company” can delay the adoption of certain
accounting standards until those standards would otherwise apply to private companies. As a result, our financial statements may not
be comparable to those of companies that comply with public company effective dates for complying with new or revised accounting standards.
We cannot predict if investors
will find our Common Stock less attractive because we may rely on these exemptions. If some investors find our Common Stock less attractive
as a result, there may be a less active trading market for our Common Stock and our stock price may be more volatile. We may take advantage
of these reporting exemptions until we are no longer an “emerging growth company.” We will remain an “emerging growth
company” until the earliest of (i) the last day of the fiscal year in which we have total annual gross revenues of $1.07 billion
or more; (ii) the last day of our fiscal year following the fifth anniversary of the date of the completion of our initial public
offering; (iii) the date on which we have issued more than $1 billion in nonconvertible debt during the previous three years;
or (iv) the date on which we are deemed to be a large accelerated filer under the rules of the SEC.
USE OF PROCEEDS
We will not receive any of the
proceeds from the sale by the Selling Stockholders of the Common Stock. Upon any exercise of the Warrants by payment of cash, however,
we will receive the exercise price of the Warrants, which, if exercised in cash with respect to the 3,785,569 shares of Common Stock
offered hereby, would result in gross proceeds to us of approximately $12.3 million. However, we cannot predict when and in what amounts
or if the Warrants will be exercised by payments of cash and it is possible that the Warrants may expire and never be exercised, in which
case we would not receive any cash proceeds.
DIVIDEND
POLICY
We have
never paid cash dividends on our Common Stock and we do not anticipate paying cash dividends in the foreseeable future, but intend to
retain our capital resources for reinvestment in our business. Any future determination to pay cash dividends on our Common Stock will
be at the discretion of our board of directors and will be dependent upon our financial condition, results of operations, capital requirements
and other factors as the board of directors deems relevant.
DETERMINATION
OF OFFERING PRICE
The
prices at which the shares of Common Stock covered by this prospectus may actually be sold will be determined by the prevailing public
market price for shares of our Common Stock or by negotiations between the Selling Stockholders and buyers of our Common Stock in private
transactions or as otherwise described in “Plan of Distribution.”
Management’s
Discussion and Analysis of Financial Condition and Results of Operations
The
following discussion and analysis of our financial condition and results of operations should be read together with the unaudited condensed
consolidated financial statements and related notes appearing elsewhere in this prospectus and the audited financial statements and related
notes for the year ended December 31, 2022 included in this prospectus. In addition to historical information, this discussion and analysis
contains forward-looking statements that involve risks, uncertainties, and assumptions. Our actual results may differ materially from
those anticipated in these forward-looking statements as a result of certain factors. We discuss factors that we believe could cause
or contribute to these differences below and elsewhere in this prospectus, including those factors set forth in the section entitled
“Cautionary Note Regarding Forward-Looking Statements” and in the section entitled “Risk Factors”.
Overview and Mission
We
believe the world needs—and deserves—a new approach to innovating that harnesses the power of large groups of stakeholders
who work together to ensure that the most promising innovations make it into the hands of people who need them most.
We
were incorporated in the State of Delaware on September 28, 2017, and our headquarters are in Mountain View, California. The company
was founded with a mission of bringing stakeholders together, to transform promising innovations into products and services that could
address some of the most challenging needs. The socialization of innovation through engaging stakeholders in every aspect of it, is key
to transforming more innovations, more rapidly, and more efficiently.
At
inception, the first innovation we took on was an immune modulation technology titled ADI/Adimune with a focus on prolonging life and
enhancing life quality of patients that have undergone organ transplants. Since then, we expanded our portfolio of innovations, and we
continue to evaluate a variety of promising health innovations.
ADIMUNE, INC.
Formed
in January 2023, Adimune™, Inc. (“Adimune”) is focused on leading our immune modulation therapeutic programs. Adimune’s
proprietary immune modulation product candidate, ADI-100™, based on the Apoptotic DNA Immunotherapy™ platform technology,
utilizes a novel approach that mimics the way our bodies naturally induce tolerance to our own tissues. It includes two DNA molecules
designed to deliver signals to induce tolerance. ADI-100 has been successfully tested in several preclinical models (e.g., skin grafting,
psoriasis, type 1 diabetes, multiple sclerosis).
In
May 2023, Adimune entered into a clinical trial agreement with Mayo Clinic to advance clinical studies targeting autoimmune diseases
of the central nervous system (“CNS”) with the initial focus on the rare, but debilitating, autoimmune disease Stiff Person
Syndrome (“SPS”). According to the National Organization of Rare Diseases, the exact incidence and prevalence of SPS is unknown;
however, one estimate places the incidence at approximately one in one million individuals in the general population.
Pending
approval by the International Review Board, a human trial for SPS is expected get underway in the first half of 2024 with enrollment
of up to 20 patients, some of whom may also have type 1 diabetes. ADI-100 will initially be tested for safety and efficacy. ADI-100 is
designed to tolerize against an antigen known as glutamic acid decarboxylase (“GAD”), which is implicated in type-1 diabetes,
psoriasis, stiff person syndrome, and in many autoimmune diseases of the CNS.
Background
The
discovery of immunosuppressive (anti-rejection and monoclonal) drugs over 40 years ago has made possible life-saving organ transplantation
procedures and blocking of unwanted immune responses in autoimmune diseases. However, immune suppression leads to significant undesirable
side effects, such as increased susceptibility to life-threatening infections and cancers, because it indiscriminately and broadly suppresses
immune function throughout the body. While the use of these drugs has been justifiable because they prevent or delay organ rejection,
their use for treatment of autoimmune diseases and allergies may not be acceptable because of the aforementioned side effects. Furthermore,
often transplanted organs ultimately fail despite the use of immune suppression, and about 40% of transplanted organs survive no more
than five years.
Through
Aditxt, Adimune has the right of use to the exclusive worldwide license for commercializing ADI nucleic acid-based technology (which
is currently at the pre-clinical stage) from Loma Linda University. ADI uses a novel approach that mimics the way the body naturally
induces tolerance to our own tissues (“therapeutically induced immune tolerance”). While immune suppression requires continuous
administration to prevent rejection of a transplanted organ, induction of tolerance has the potential to retrain the immune system to
accept the organ for longer periods of time. ADI may allow patients to live with transplanted organs with significantly reduced immune
suppression. ADI is a technology platform which we believe can be engineered to address a wide variety of indications.
Advantages
ADI™
is a nucleic acid-based technology (e.g., DNA-based), which we believe selectively suppresses only those immune cells involved
in attacking or rejecting self and transplanted tissues and organs. It does so by tapping into the body’s natural process of cell
turnover (i.e., apoptosis) to retrain the immune system to stop unwanted attacks on self or transplanted tissues. Apoptosis is a natural
process used by the body to clear dying cells and to allow recognition and tolerance to self-tissues. ADI triggers this process by enabling
the cells of the immune system to recognize the targeted tissues as “self.” Conceptually, it is designed to retrain the immune
system to accept the tissues, similar to how natural apoptosis reminds our immune system to be tolerant to our own “self”
tissues.
While
various groups have promoted tolerance through cell therapies and ex vivo manipulation of patient cells (i.e., takes
place outside the body), to our knowledge, we will be unique in our approach of using in-body induction of apoptosis to promote tolerance
to specific tissues. In addition, ADI treatment itself will not require additional hospitalization but only an injection of minute
amounts of the therapeutic drug into the skin.
Moreover,
preclinical studies have demonstrated that ADI treatment significantly and substantially prolongs graft survival, in addition to successfully
“reversing” other established immune-mediated inflammatory processes.
License Agreement
with Loma Linda University (“LLU”)
On
March 15, 2018, we entered into a License Agreement with LLU, which was subsequently amended on July 1, 2020. Pursuant to the LLU License
Agreement, we obtained the exclusive royalty-bearing worldwide license to all intellectual property, including patents, technical information,
trade secrets, proprietary rights, technology, know-how, data, formulas, drawings, and specifications, owned or controlled by LLU and/or
any of its affiliates (the “LLU Patent and Technology Rights”) and related to therapy for immune-mediated inflammatory diseases
(the ADI™ technology). In consideration for the LLU License Agreement, we issued 13 shares of common stock to LLU.
PEARSANTA, INC.
Formed
in January 2023, our subsidiary Pearsanta™, Inc. (“Pearsanta”) seeks to take personalized medicine to a whole new level
by delivering “Health by the Numbers.” Since its founding, Pearsanta has been building the platform for enabling our vision
of lab quality testing, anytime, anywhere. Our plan for Pearsanta’s platform is for it to be the transactional backbone for sample
collection, sample processing (on- and off-site), and reporting. This will require the development and convergence of multiple components
developed by Pearsanta, or through transactions with third parties, including collection devices, “lab-on-a-chip” technologies,
Lab Developed Test (LDT) assays, a data-driven analysis engine, and telemedicine. According to a comprehensive research report by Market
Research Future, the clinical and consumer diagnostic market is estimated to hit $429.3 billion by 2030.
We
believe that timely and personalized testing enables far more informed treatment decisions. Pearsanta’s platform is being developed
as a seamless digital healthcare solution. This platform will integrate at-location sample collection, Point-of-Care (“POC”)
and LDT assays, and an analytical reporting engine, with telemedicine-enabled visits with licensed physicians to review test results
and, if necessary, order a prescription. Pearsanta’s goal of extending its platform to enable consumers to monitor their health
more proactively as the goal is to provide a more complete picture about someone’s dynamic health status, factoring in genetic
makeup and their response to medication. The POC component of Pearsanta would enable diagnostic testing at-home, at work, in pharmacies,
and more to generate results quickly so that an individual can access necessary treatment faster. With certain infections, prescribing
the most effective treatment according to one’s numbers can prevent hospital emergency room admissions and potentially life-threatening
consequences.
Examples
of indication-focused tests for the Test2Treat platform will include the evaluation for advanced urinary tract infections (“UTIs”),
COVID-19/flu/respiratory syncytial virus, sexually transmitted infections, gut health, pharmacogenomics (i.e., how your genes affect
the way your body responds to certain therapeutics), and sepsis. We believe that these offerings are novel and needed as the current
standard of care using broad spectrum antibiotic treatment can be ineffective and potentially life-threatening. For example, improperly
prescribed antibiotics may approach 50% of outpatient cases. Further, according to an article published in Physician’s Weekly,
only 1% of board-certified critical care medicine physicians are trained in infectious disease.
Licensed Technologies
– AditxtScoreTM
We
intend to sublicense to Pearsanta an exclusive worldwide sub-license for commercializing the AditxtScore™ technology which provides
a personalized comprehensive profile of the immune system. AditxtScore is intended to detect individual immune responses to viruses,
bacteria, peptides, drugs, supplements, bone marrow and solid organ transplants, and cancer. It has broad applicability to many other
agents of clinical interest impacting the immune system, including those not yet identified such as emerging infectious agents.
AditxtScore
is being designed to enable individuals and their healthcare providers to understand, manage and monitor their immune profiles and to
stay informed about attacks on or by their immune system. We believe AditxtScore can also assist the medical community and individuals
by being able to anticipate the immune system’s potential response to viruses, bacteria, allergens, and foreign tissues such as
transplanted organs. This technology may be able to serve as a warning signal, thereby allowing for more time to respond appropriately.
Its advantages include the ability to provide simple, rapid, accurate, high throughput assays that can be multiplexed to determine the
immune status with respect to several factors simultaneously, in approximately 3-16 hours. In addition, it can determine and differentiate
between distinct types of cellular and humoral immune responses (e.g., T and B cells and other cell types). It also provides for simultaneous
monitoring of cell activation and levels of cytokine release (i.e., cytokine storms).
We
are actively involved in the regulatory approval process for AditxtScore assays for clinical use and securing manufacturing, marketing,
and distribution partnerships for application in the various markets. To obtain regulatory approval to use AditxtScore as a clinical
assay, we have conducted validation studies to evaluate its performance in detection of antibodies and plan to continue conducting additional
validation studies for new applications in autoimmune diseases.
Advantages
The
sophistication of the AditxtScore technology includes the following:
| ● | greater
sensitivity/specificity. |
| ● | 20-fold
higher dynamic range, greatly reducing signal to noise compared to conventional assays. |
| ● | ability
to customize assays and multiplex a large number of analytes with speed and efficiency. |
| ● | ability
to test for cellular immune responses (i.e., T and B cells and cytokines). |
| ● | proprietary
reporting algorithm. |
License Agreement
with Leland Stanford Junior University (“Stanford”)
On
February 3, 2020, we entered into an exclusive license agreement (the “February 2020 License Agreement”) with Stanford with
regard to a patent concerning a method for detection and measurement of specific cellular responses. Pursuant to the February 2020 License
Agreement, we received an exclusive worldwide license to Stanford’s patent with regard to use, import, offer, and sale of Licensed
Products (as defined in the agreement). The license to the patented technology is exclusive, including the right to sublicense, beginning
on the effective date of the agreement, and ending when the patent expires. Under the exclusivity agreement, we acknowledged that Stanford
had already granted a non-exclusive license in the Nonexclusive Field of Use, under the Licensed Patents in the Licensed Field of Use
in the Licensed Territory (as those terms are defined in the “February 2020 License Agreement”). However, Stanford agreed
not to grant further licenses under the Licensed Patents in the Licensed Field of Use in the Licensed Territory. On December 29, 2021,
we entered into an amendment to the February 2020 License Agreement which extended our exclusive right to license the technology deployed
in AditxtScoreTM and securing worldwide exclusivity in all fields of use of the licensed technology.
ADIVIR, INC.
Formed
in April of 2023, Adivir™, Inc., is Aditxt’s most recently formed wholly owned subsidiary, dedicated to the clinical and
commercial development efforts of innovative antiviral products, which have the potential to address a wide range of infectious diseases,
including those that currently lack viable treatment options.
Background
On
April 18, 2023, we entered into an Asset Purchase Agreement (the “Asset Purchase Agreement”) with Cellvera Global Holdings
LLC (“Cellvera Global”), Cellvera Holdings Ltd. (“BVI Holdco”), Cellvera, Ltd. (“Cellvera Ltd.”),
Cellvera Development LLC (“Cellvera Development” and together with Cellvera Global, BVI Holdco, Cellvera Ltd. and Cellvera
Development (the “Sellers”), AiPharma Group Ltd. (“Seller Owner” and collectively with the Sellers, “Cellvera”),
and the legal representative of Cellvera, pursuant to which, the Company will purchase Cellvera’s 50% ownership interest in G Response
Aid FZE (“GRA”), certain other intellectual property and all goodwill related thereto (the “Acquired Assets”). Unless
expressly stated otherwise herein, capitalized terms used but not defined herein have the meanings ascribed to them in the Asset Purchase
Agreement. Pursuant to the Asset Purchase Agreement, the consideration for the Acquired Assets consists of (A) $24.5 million, comprised
of: (i) the forgiveness of the Company’s $14.5 million loan to Cellvera Global, and (ii) approximately $10 million in cash, and
(B) future revenue sharing payments for a term of seven years. GRA holds an exclusive, worldwide license for the antiviral medication,
Avigan® 200mg, excluding Japan, China and Russia. The other 50% interest in GRA is held by Agility, Inc. (“Agility”).
Additionally,
upon the closing, the Share Exchange Agreement previously entered into as of December 28, 2021, between Cellvera Global Holdings, LLC
f/k/a AiPharma Global Holdings, LLC (together with other affiliates and subsidiaries) and the Company, and all other related agreements
will be terminated.
The
obligations of the Company to consummate the Closing under the Asset Purchase Agreement are subject to the satisfaction or waiver, at
or prior to the Closing of certain conditions, including but not limited to, the following:
| (i) | Satisfactory
completion of due diligence; |
| (ii) | Completion
by the Company of financing sufficient to consummate the transactions contemplated by the
Asset Purchase Agreement; |
| (iii) | Receipt
by the Company of all required Consents from Governmental Bodies for the Acquisition, including
but not limited to, any consents required to complete the transfer and assignment of Cellvera’s
membership interests in GRA; |
| (iv) | Receipt
of executed payoff letters reflecting the amount required to be fully pay all of each of
Seller’s and Seller Owner’s Debt to be paid at Closing; |
| (v) | Receipt
by the Company of a release from Agility; |
| (vi) | Execution
of an agreement acceptable to the Company with respect to the acquisition by the Company
of certain intellectual property presently held by a third party; |
| (vii) | Execution
of an amendment to an asset purchase agreement previously entered into by Cellvera with a
third party that effectively grants the Company the rights to acquire the intellectual property
from the third party under such agreement; |
| (viii) | Receipt
of a fairness opinion by the Company with respect to the transactions contemplated by the
Asset Purchase Agreement; and |
| (ix) | Receipt
by the Company from the Seller Owner of written consent, whether through its official liquidator
or the Board of Directors of Seller Owner, to the sale and purchase of the Acquired Assets
and Assumed Liabilities pursuant to the Assert Purchase Agreement. |
There
can be no assurance that the conditions to closing will be satisfied or that the proposed acquisition will be completed as proposed or
at all.
Our
commitment to building our antiviral portfolio is strategic and timely. We believe that there has never has there been a more important
time to address the growing global need to uncover new treatments or commercialize existing ones that treat life-threatening global viral
infections.
Our Team
Aditxt
has assembled an entrepreneurial team of experts from a variety of different business, engineering, and scientific fields, and commercial
backgrounds, with collective experience that ranges from founding startup innovation companies, to developing and marketing biopharmaceutical
and diagnostic products, to designing clinical trials, and management of private and public companies. We have deep experience in identifying
and accessing promising health innovations and developing them into products and services with the ability to scale. We understand the
capital markets, both public and private, as well as M&A and facilitating complex IPOs.
Going Concern
We were incorporated on September
28, 2017 and have not generated significant revenues to date. During the three months ended March 31, 2024 we had a net loss of $14,868,694
and cash of $88,671 as of March 31, 2024.
We are currently over 90
days past due on a significant number of vendor obligations. The Company will require significant additional capital to operate in the
normal course of business and fund clinical studies in the long-term. We believe our remaining funds on hand will not be sufficient to
fund our operations for the next 12 months and such creates substantial doubt about our ability to continue as a going concern beyond
one year.
Financial Results
We
have a limited operating history. Therefore, there is limited historical financial information upon which to base an evaluation of our
performance. Our prospects must be considered in light of the uncertainties, risks, expenses, and difficulties frequently encountered
by companies in their early stages of operations. Our condensed consolidated financial statements as of December 31, 2023, show a net
loss of $32,390,447. We expect to incur additional net expenses over the next several years as we continue to maintain and expand our
existing operations. The amount of future losses and when, if ever, we will achieve profitability are uncertain.
Results of Operations
Results of operations for the three months
ended March 31, 2024 and 2023
We generated revenue of $79,680
and $218,415 for the three months ended March 31, 2024 and 2023, respectively. Cost of for the three months ended March 31, 2024 and
2023 was $65,799 and $178,309, respectively.
During the three months ended
March 31, 2024, we incurred a loss from operations of $11,549,527. This is due to general and administrative expenses of $3,363,748,
which includes approximately $1,214,004 in payroll expenses, $1,145,679 in professional fees, and $20,477 in stock-based compensation.
Research and development expenses were $8,145,266, which includes $201,579 in consulting expenses and $6,712,663 in stock-based compensation.
Sales and marketing expenses were $40,513.
During the three months ended
March 31, 2023, we incurred a loss from operations of $5,822,001. This is due to general and administrative expenses of $4,368,843, which
includes $274,315 in stock-based compensation, research and development of $1,387,541, which includes $62,633 in stock-based compensation,
and sales and marketing expenses of negative $65,617, which includes $2,503 in stock-based compensation. The $1,387,541 in research and
development is mainly comprised of $415,429 in consulting expenses, $673,377 in compensation and $62,633 in stock-based compensation.
Sales and marketing expenses were $65,617 which includes $2,503 in stock-based compensation.
The decrease in expenses
during the three months ended March 31, 2024 compared to the three months ended March 31, 2023 was due to decreased research and development
spend and the termination of a sales and marketing vendor.
Results of operations
for the years ended December 31, 2023 and 2022
We generated revenue of $645,176
and $933,715 for the years ended December 31, 2023 and 2022, respectively. Cost of sales for the years ended December 31, 2023 and 2022
was $756,836 and $766,779, respectively.
During the year ended December
31, 2023, we incurred a loss from operations of $26,062,425. This is due primarily to general and administrative expenses of $18,607,142.
This includes approximately $9,641,000 in payroll expenses, $4,484,000 in professional fees, and $1,133,077 in stock-based compensation.
Research and development expenses were $7,074,339 which includes $1,815,068 in consulting expenses and $262,154 in stock-based compensation.
Sales and marketing expenses were $269,284, which includes $6,787 in stock-based compensation.
During the year ended December
31, 2022, we incurred a loss from operations of $25,480,098. This is due to general and administrative expenses of $15,985,552, which
includes $1,516,805 in stock-based compensation, research and development of $7,268,084, which includes $591,518 in stock-based compensation,
sales and marketing expenses of $1,849,460, which includes $1,023,045 in stock-based compensation and impairment on note receivable of
$534,938. The $7,268,084 in research and development is mainly comprised of $2,145,382 in consulting expenses, and $3,375,757 in compensation
offset by a one-time adjustment to research and development purchases. During the year, the Company transitioned from purchasing certain
inventory items to internally manufacturing these items.
Liquidity and Capital
Resources
We
have incurred substantial operating losses since inception and expect to continue to incur significant operating losses for the foreseeable
future and may never become profitable. As of March 31, 2024, we had an accumulated deficit of $142,470,799. We had working capital of
$(15,706,647) as of March 31, 2024. During the three months ended March 31, 2024, we purchased zero in fixed assets.
Our
condensed consolidated financial statements have been prepared assuming that we will continue as a going concern.
We
have funded our operations from proceeds from the sale of equity and debt securities. On July 2, 2020, we completed our IPO and raised
approximately $9.5 million in net proceeds. At the time of the IPO, we believed that these funds would be sufficient to fund our operations
for the foreseeable future.
On
September 10, 2020, we completed a follow-on public offering. In connection therewith, we issued 1,200 units, or Follow-On Units, excluding
the underwriters’ option to cover overallotments, at an offering price of $8,000.00 per Follow-On Unit, resulting in gross proceeds
of approximately $9.6 million.
On
January 25, 2021, the Company entered into a securities purchase agreement with an institutional accredited investor (the “Investor”)
for the sale of a $6,000,000 senior secured convertible note (the “Convertible Note”). The Convertible Note had a term of
24 months, was originally convertible at a price of $8,000.00 per share and was issued at an original issuance discount of $1,000,000.
On August 30, 2021, the Company entered into a defeasance and waiver agreement with the Investor, pursuant to which the Noteholder has
agreed in exchange for (a) a cash payment by the Company to the Investor of $1.2 million (the Cash Payment”), (b) a waiver, in
part of the conversion price adjustment provision such that the January 2021 Note shall be convertible into 2,401 shares of common stock
(without giving effect to the conversion notice received by the company form the Noteholder prior to the date hereof totaling (503 shares)
(the “Shares”), and (c) a voluntary and permanent reduction by the Company of the exercise price of the warrant to purchase
400 shares of the common stock of the Company (the “January 2021 Warrant”) to $5,060 per share. As of December 31, 2022,
the outstanding principle of the convertible note had been converted to 2,401 shares of common stock.
On
August 30, 2021, the Company completed a registered direct offering and raised approximately $10.1 million in net proceeds.
On
October 20, 2021, the Company completed a public offering for net proceeds of $3.8 million. As part of this offering, we issued 1,417
shares of the Company’s common stock
On
December 6, 2021, the Company completed a public offering for net proceeds of $16.0 million. As part of this offering, we issued 4,123
units consisting of shares of the Company’s common stock and warrant to purchase shares of the Company’s common stock and
4,164 pre-funded warrants. The warrant issued as part of the units had an exercise price of $2,300.00 and the prefunded warrants had
an exercise price of $0.001.
On
September 20, 2022, the Company completed a public offering for net proceeds of $18.1 million (the “September 2022 Offering”).
As part of the September 2022 Offering, we issued 30,608 of shares of the Company’s common stock, pre-funded warrants to purchase
52,725 shares of the Company’s common stock and warrants to purchase 83,333 shares of the Company’s common stock. The
warrants have an exercise price of $240.00 and the pre-funded warrants have an exercise price of $0.04.
On
April 20, 2023, the Company entered into a securities purchase agreement (the “Purchase Agreement”) with an institutional
investor, pursuant to which the Company agreed to sell to such investor pre-funded warrants (the “Pre-Funded Warrants”) to
purchase up to 39,634 shares of common stock of the Company (the “Common Stock”) at a purchase price of $48.76 per Pre-Funded
Warrant. Concurrently with the sale of the Pre-Funded Warrants, pursuant to the Purchase Agreement in a concurrent private placement,
for each Pre-Funded Warrant purchased by the investor, such investor received from the Company an unregistered warrant (the “Warrant”)
to purchase two shares of Common Stock. The warrants have an exercise price of $34.40 per share and are exercisable for a three-year
period. In addition, the Company issued a warrant to the placement agent to purchase up to 2,378 shares of common stock at an exercise
price of $61.00 per share.
On
August 31, 2023, the Company entered into a securities purchase agreement (the “August Purchase Agreement”) with an
institutional investor for the issuance and sale in a private placement (the “Private Placement”) of (i) pre-funded warrants
(the “Pre-Funded Warrants”) to purchase up to 1,000,000 shares of the Company’s common stock at an exercise price of
$0.001 per share, and (ii) warrants (the “Common Warrants”) to purchase up to 1,000,000 shares of the Company’s Common
Stock at an exercise price of $10.00 per share. The Private Placement closed on September 6, 2023. The net proceeds to the Company from
the Private Placement were approximately $9 million, after deducting placement agent fees and expenses and estimated offering expenses
payable by the Company. The Company utilized net proceeds received from the Private Placement for (i) payment of approximately $3.1 million
in outstanding obligations, (ii) repayment of approximately $0.4 million of outstanding debt, and (iii) continuing operating expenses
and working capital.
On December 29, 2023, the
Company entered into a securities purchase agreement (the “Purchase Agreement”) with an institutional investor (“the
“December Purchaser”) for the issuance and sale in a private placement (the “December Private Placement”) of
(i) pre-funded warrants (the “December Pre-Funded Warrants”) to purchase up to 1,237,114 shares of the Company’s Common
Stock, par value $0.001 at an exercise price of $0.001 per share, and (ii) warrants (the “December Common Warrants”) to purchase
up to 2,474,228 shares of the Company’s Common Stock, at a purchase price of $4.85 per share.
Pursuant to the Purchase
Agreement, the Company agreed to reduce the exercise price of certain outstanding warrants to purchase Common Stock of the Company (“Certain
Outstanding Warrants”) held by the Purchaser to $4.60 per share in consideration for the cash payment by the December Purchaser
of $0.125 per share of Common Stock underlying the Certain Outstanding Warrants, effective immediately.
The December Private Placement
closed on January 4, 2024. The net proceeds to the Company from the December Private Placement were approximately $5.5 million, after
deducting placement agent fees and expenses and estimated offering expenses payable by the Company.
In addition, the Company
agreed to pay H.C. Wainwright & Co., LLC (“Wainwright”) certain expenses and issued to Wainwright or its designees warrants
(the “December Placement Agent Warrants”) to purchase up to an aggregate of 74,227 shares of Common Stock at an exercise
price equal to $6.0625 per share. The December Placement Agent Warrants are exercisable immediately upon issuance and have a term of
exercise equal to three years from the date of issuance.
We
will need significant additional capital to continue to fund our operations and the clinical trials for our product candidates. We may
seek to sell common stock, preferred stock or convertible debt securities, enter into a credit facility or another form of third-party
funding or seek other debt financing. In addition, we may seek to raise cash through collaborative agreements or from government grants.
The sale of equity and convertible debt securities may result in dilution to our stockholders and certain of those securities may have
rights senior to those of our common shares. If we raise additional funds through the issuance of preferred stock, convertible debt securities,
or other debt financing, these securities or other debt could contain covenants that would restrict our operations. Any other third-party
funding arrangement could require us to relinquish valuable rights.
The
source, timing, and availability of any future financing will depend principally upon market conditions, and, more specifically, on the
progress of our clinical development program. Funding may not be available when needed, at all, or on terms acceptable to us. Lack of
necessary funds may require us to, among other things, delay, scale back or eliminate expenses including some or all our planned development,
including our clinical trials. While we may need to raise funds in the future, we believe the current cash reserves should be sufficient
to fund our operation for the foreseeable future. Because of these factors, we believe that this creates doubt about our ability to continue
as a going concern.
Contractual Obligations
The
following table shows our contractual obligations as of March 31, 2024:
| | |
Payment Due by Year | |
| | |
Total | | |
2024 | | |
2025 | | |
2026 | |
| Lease | |
$ | 1,853,227 | | |
$ | 718,751 | | |
$ | 710,546 | | |
$ | 423,930 | |
Critical Accounting
Polices and Estimates
Our
condensed consolidated financial statements are prepared in accordance with generally accepted accounting principles in the United States.
The preparation of our condensed consolidated financial statements and related disclosures requires us to make estimates, assumptions
and judgments that affect the reported amount of assets, liabilities, revenue, costs and expenses, and related disclosures. We believe
that our critical accounting policies described under the heading “Management’s Discussion and Analysis of Financial Condition
and Plan of Operations—Critical Accounting Policies” in our Prospectus, dated September 1, 2020, filed with the SEC pursuant
to Rule 424(b), are critical to fully understanding and evaluating our financial condition and results of operations. The following involve
the most judgment and complexity:
| ● | Research
and development |
| ● | Stock-based
compensation expense |
Accordingly,
we believe the policies set forth above are critical to fully understanding and evaluating our financial condition and results of operations.
If actual results or events differ materially from the estimates, judgments and assumptions used by us in applying these policies, our
reported financial condition and results of operations could be materially affected.
Off-Balance Sheet
Arrangements
We
did not have during the periods presented, and we do not currently have, any off-balance sheet arrangements, as defined in the rules
and regulations of the SEC.
JOBS Act
On
April 5, 2012, the JOBS Act was enacted. Section 107 of the JOBS Act provides that an “emerging growth company”
can take advantage of the extended transition period provided in Section 7(a)(2)(B) of the Securities Act, for complying with new
or revised accounting standards. In other words, an “emerging growth company” can delay the adoption of certain accounting
standards until those standards would otherwise apply to private companies.
When
favorable, we have chosen to take advantage of the extended transition periods available to emerging growth companies under the JOBS
Act for complying with new or revised accounting standards until those standards would otherwise apply to private companies provided
under the JOBS Act.
We
are in the process of evaluating the benefits of relying on other exemptions and reduced reporting requirements provided by the JOBS Act. Subject
to certain conditions set forth in the JOBS Act, as an “emerging growth company,” we intend to rely on certain of these exemptions,
including without limitation, (i) providing an auditor’s attestation report on our system of internal controls over financial
reporting pursuant to Section 404(b) of the Sarbanes-Oxley Act and (ii) complying with any requirement that may be adopted
by the Public Company Accounting Oversight Board (“PCAOB”) regarding mandatory audit firm rotation or a supplement to the
auditor’s report providing additional information about the audit and the financial statements, known as the auditor discussion
and analysis. We will remain an “emerging growth company” until the earliest of (i) the last day of the fiscal year
in which we have total annual gross revenues of $1.07 billion or more; (ii) the last day of our fiscal year following the fifth
anniversary of the date of the completion of our IPO (December 31, 2025); (iii) the date on which we have issued more than $1 billion
in nonconvertible debt during the previous three years; or (iv) the date on which we are deemed to be a large accelerated filer
under the rules of the SEC.
Recently Issued and
Adopted Accounting Pronouncements
See
Note 3 - Summary of Significant Accounting Policies to the accompanying condensed consolidated financial statements for a description
of other accounting policies and recently issued accounting pronouncements.
Recent Developments
See
Note 12 – Subsequent Event to the accompanying condensed consolidated financial statements for a description of material recent
developments.
BUSINESS
Our Mission
We believe the world needs—and
deserves—a new approach to health innovation that is focused on harnessing the power of large groups of stakeholders who work together
to ensure that the most promising treatments make it into the hands of people who need them most.
We were incorporated in the
State of Delaware on September 28, 2017, and our headquarters are located in Mountain View, California. The company was founded with
a mission of bringing stakeholders together, to transform promising innovations into products and services that could address some of
the most challenging needs. The socialization of innovation through engaging stakeholders in every aspect of it, is key to transforming
more innovations, more rapidly, and more efficiently.
At inception,
the first innovation we took on was an immune modulation technology titled ADI/Adimune with a focus on prolonging life and enhancing
life quality of patients that have undergone organ transplants. Since then, we expanded our portfolio of innovations, and we continue
to evaluate a variety of promising health innovations.
Our Model
Our
mission is to advance promising health innovations. Our business model begins with identifying and securing innovations through licensing
or acquisition. Once an innovation is secured and becomes “An Aditxt Company,” we seek to accelerate its growth through supporting
and scaling its innovation, operations, and commercialization.
Our
model focuses on identifying promising health innovation and surrounding it with activation resources that take a systemized approach
to bringing that idea to life. We seek to engage various stakeholders for each of our programs on every level: This includes identifying
researchers and research institution partners, such as Stanford University; leading health institutions to get critical trials underway,
such as the Mayo Clinic; manufacturing partners who enable us to take innovations from preclinical to clinical; municipalities and governments,
such as the city of Richmond and the state of Virginia and public health agencies launch our programs, such as Pearsanta’s laboratory;
and thousands of shareholders around the globe. What was once a promising innovation becomes a purposeful product that has the power
to change lives.
We are
not about a single idea or a single innovation. It is about making sure the right innovation is made possible. With any type of innovation,
there is power in numbers and power in people who believe in our mission of “Making Promising Innovations Possible, Together.”

Our Value Proposition
Far too often, promising treatment
or technology does not reach commercialization due to lack of funds and critical infrastructure. As a result, potentially life-changing
and lifesaving treatments are not available to the individuals who so desperately need them.
Our mission is to make promising
innovation a reality faster and more efficiently. Since inception, we have sought to provide the critical infrastructure consisting of
a highly experienced team and stakeholders skilled in product development, operations, commercialization, engagement and content, finance
and accounting, people, and legal. Our ecosystem has established access to industry leaders, top-rated research and medical institutions,
universities, manufacturing and distribution companies, and critical infrastructure such as CLIA-certified state-of-the art labs and
GMP manufacturing.
We bring the holistic concept
of an efficient ecosystem for advancing and accelerating innovations. Our process: We seek to discover, identify, and license or acquire
promising innovations. We then form and build out a subsidiary around each innovation and support the subsidiaries through innovation,
operations, commercialization, and corporate functions that seek to thrive and grow as a successful, monetizable business.
The Shifting Landscape of Health Innovation
Health
innovation requires significant resources. The convergence of biotech and high-tech offers new possibilities of accelerating breakthrough
innovations faster and more efficiently. This approach reflects our mission of “Making Promising Innovations Possible, Together”.
People deserve innovative solutions,
which have never been more within reach. We believe the best idea, best product and the best solution will come from creating an ecosystem
where all stakeholders, such as vendors, customers, municipalities, and shareholders contribute. When we disrupt the way we’re
innovating, through our collaborative model, we believe we can move faster and more efficiently to activate viable solutions that have
the potential to make a measurable impact.
Our Growth Strategy
The era of precision and personalized
medicine is here. We believe that people around the globe would benefit from health diagnostics and treatments that more accurately pinpoint
the problems and more precisely treat the condition.
In addition to our current programs,
Adimune and Pearsanta, we look to bring in future health innovations in the areas of software and AI, medical devices, therapeutics,
and other technologies that take a fundamentally different approach to health because they prioritize precision medicine, timely disease
root cause analysis, and targeted treatments.
Year over year, we plan to continue
building our infrastructure and adding more personalized and precision health innovations that align with our mission. These opportunities
may come in different forms such as IP, an early-stage company, or a late-stage company. We will continue to scale our systemized approach
to the innovation process, making large-scale automation and enterprise systems available to Aditxt portfolio companies at every stage
of their growth. Specifically, certain subsidiaries will need to grow through further M&A activities, operational infrastructure
implementation, and development or acquisition of critical technologies.
Our Team
Aditxt has assembled an entrepreneurial
team of experts from a variety of different business, engineering, and scientific fields, and commercial backgrounds, with collective
experience that ranges from founding startup innovation companies, to developing and marketing biopharmaceutical and diagnostic products,
to designing clinical trials, and management of private and public companies. We have deep experience in identifying and accessing promising
health innovations and developing them into products and services with the ability to scale. We understand the capital markets, both
public and private, as well as M&A and facilitating complex IPOs.
THE ADITXT PROGRAMS
ADIMUNE, INC.
Formed in January 2023, Adimune™,
Inc. (“Adimune”) is focused on leading Aditxt’s immune modulation therapeutic programs. Adimune’s proprietary
immune modulation product Apoptotic DNA Immunotherapy, or ADI-100™, which utilizes a novel approach that mimics the way our bodies
naturally induce tolerance to our own tissues. It includes two DNA molecules designed to deliver signals to induce tolerance. ADI-100
has been successfully tested in several preclinical models (skin grafting, psoriasis, type 1 diabetes, multiple sclerosis).
In May 2023, Adimune and Mayo
Clinic signed a clinical trial agreement to advance clinical studies targeting autoimmune diseases of the central nervous system (“CNS”)
with the initial focus on the rare, but debilitating, autoimmune disease Stiff Person Syndrome (“SPS”). According to the
National Organization of Rare Diseases, the exact incidence and prevalence of SPS is unknown; however, one estimate places the incidence
at approximately 1 in 1 million individuals in the general population.
Pending approval by the International
Review Board, a human trial for SPS will get underway in the second half of 2023 or the first half of 2024 with enrollment of 10-15 patients,
some of whom also have type 1 diabetes. ADI-100 will be tested for safety and efficacy. ADI-100 is designed to tolerize against an antigen
known as glutamic acid decarboxylase (“GAD”), which is implicated in type-1 diabetes, psoriasis, and in many autoimmune diseases
of the CNS.
Background
The
discovery of immunosuppressive (anti-rejection and monoclonal) drugs over 40 years ago has made possible life-saving organ transplantation
procedures and blocking of unwanted immune responses in autoimmune diseases. However, immune suppression leads to significant undesirable
side effects, such as increased susceptibility to life-threatening infections and cancers, because it indiscriminately and broadly suppresses
immune function throughout the body. While the use of these drugs has been justifiable because they prevent or delay organ rejection,
their use for treatment of autoimmune diseases and allergies may not be acceptable because of the aforementioned side effects. Furthermore,
transplanted organs often ultimately fail despite the use of immune suppression, and about 40% of transplanted organs survive no more
than 5 years.
Through
Aditxt, Adimune has the right of use to the exclusive worldwide license for commercializing ADI nucleic acid-based technology (which
is currently at the pre-clinical stage) from Loma Linda University. (See below). ADI uses a novel approach that mimics the way the body
naturally induces tolerance to our own tissues (“therapeutically induced immune tolerance”). While immune suppression requires
continuous administration to prevent rejection of a transplanted organ, induction of tolerance has the potential to retrain the immune
system to accept the organ for longer periods of time. ADI may allow patients to live with transplanted organs with significantly reduced
immune suppression. ADI is a technology platform which we believe can be engineered to address a wide variety of indications.
We are
developing ADI™ products for organ transplantation including skin allografting, autoimmune diseases, and allergies, with the initial
focus on psoriasis, type 1 diabetes and skin allografting, indications for which we have compelling preclinical data. To submit a Biologics
License Application (“BLA”) for a biopharmaceutical product, clinical safety and efficacy must be demonstrated in clinical
studies conducted with human subjects. For products in our class of drugs, the first-in-human trials will be a combination of Phase I
(safety/tolerability) and Phase II (efficacy) in affected subjects. To obtain approval to initiate the Phase I/IIa studies, an Investigational
New Drug or Clinical Trial Application will be submitted that will include a compilation of non-clinical efficacy data as well as manufacturing
and pre-clinical safety/toxicology data. To date, we have conducted non-clinical studies in a stringent model of skin transplantation
using genetically mismatched donor and recipient animals demonstrating a 3-fold increase in the survival of the skin allograft in animals
that were tolerized with ADI™ compared to animals that receive immune suppression alone. Prolongation of graft life was observed
despite discontinuation of immune suppression after the first 5 weeks. In a non-obese diabetic mouse model of type 1 diabetes, we showed
reversal of hyperglycemia with 80% of the animals showing durable glycemic control for the 40-week study period. Additionally, in an
induced non-clinical model for psoriasis, ADI™ treatment resulted in a 69% reduction in skin thickness and a 38% decrease in skin
flaking (two clinical parameters for assessment of psoriasis skin lesions). The Phase I/IIa studies in psoriasis will evaluate the safety/tolerability
of ADI™ in patients diagnosed with psoriasis. Since the drug will be administered in subjects diagnosed with psoriasis, effectiveness
of the drug to improve psoriatic lesions will also be evaluated. In the type 1 diabetes clinical studies, newly diagnosed subjects will
receive ADI™ treatment to evaluate safety and efficacy. In another Phase I/IIa study, patients requiring skin allografts will receive
weekly intra-dermal injections of ADI™ in combination with standard immune suppression to assess safety/tolerability and possibility
of reducing levels of immunosuppressive drugs as well as prolongation of graft life.
New,
focused therapeutic approaches are needed that modulate only the immune cells involved in rejection of the transplanted organ, as this
approach can be safer for patients than indiscriminate immune suppression. Such approaches are referred to as immune tolerance, and when
therapeutically induced, may be safer for patients and potentially allow long-termer survival of transplanted tissues and organs.
In the
late 1990s, academic research on these approaches was conducted at the Transplant Center in Loma Linda University (“LLU”)
in connection with a project that secured initial grant funding from the U.S. Department of Defense. The focus of that project was induction
of tolerance for skin allografting for burn victims. Twenty years of research at LLU and an affiliated incubator led to a series of discoveries
that have been translated into a large patent portfolio of therapeutic approaches that may be applied to the modulation of the immune
system to induce tolerance to self and transplanted organs.
Advantages
ADI™
is a nucleic acid-based technology (e.g., DNA-based), which we believe selectively suppresses only those immune cells involved
in attacking or rejecting self and transplanted tissues and organs. It does so by tapping into the body’s natural process of cell
turnover (apoptosis) to retrain the immune system to stop unwanted attacks on self or transplanted tissues. Apoptosis is a natural process
used by the body to clear dying cells and to allow recognition and tolerance to self-tissues. ADI™ triggers this process by enabling
the cells of the immune system to recognize the targeted tissues as “self”. Conceptually, it is designed to retrain the immune
system to accept the tissues, similar to how natural apoptosis reminds our immune system to be tolerant to our own “self”
tissues.
While
efforts have been made by various groups to promote tolerance through cell therapies and ex vivo manipulation of patient
cells (takes place outside the body), we believe we will be unique in our approach of using in-body induction of apoptosis to promote
tolerance to specific tissues. In addition, ADI treatment itself will not require additional hospitalization, only an injection
of minute amounts of the therapeutic drug into the skin.
Reduce Chronic Rejection
Moreover,
preclinical studies have demonstrated that ADI treatment significantly and substantially prolongs graft survival, in addition to successfully
“reversing” other established immune-mediated inflammatory processes.
While
immunosuppressants control acute rejection during the early time-period after receiving an organ, chronic rejection of the organ that
occurs one or more years after the transplant procedure continues to pose a major challenge for organ recipients.
Chronic
rejection has been likened to autoimmunity (a misdirected immune response that occurs when the immune system goes awry), where specific
tissues in the transplanted organ become targets of immune attack. In other words, chronic rejection may not be caused just by differences
between the donor and the recipient, but rather by an immune response by the recipient to specific tissues in the organ. Our pre-clinical
studies suggest that ADI™ has the ability to tolerize to specific tissues in a transplanted organ, and conceivably, reduce incidences
of chronic rejection.
Reduce immune suppression
Studies
in animal models have shown that conditioning/desensitizing the animals to receive the transplant, prolongs the survival of the transplanted
tissue or organ. These studies have used repeated exposure to low doses of protein components in specific organs to reduce immunologic
recognition and attack on the transplanted organ.
Based
on some of our data, we believe that with ADI™ treatment, recipients can be conditioned/desensitized, thereby retraining the immune
system to more readily accept the organ and also reduce the levels of immunosuppressive drugs needed post-transplantation.
Preformed Antibodies
Studies
have shown that presence of preformed antibodies prior to transplantation procedures increases the rate of organ rejection. Preformed
antibodies can develop in previously transplanted patients, patients who have given birth, and patients who have previously received
blood transfusions. With more than 113,000 patients on transplant waiting lists in the U.S. alone, patients with pre-existing antibodies
have much lower chances of qualifying to receive organs due to their increased risk of rejection – even with immune suppression.
Sadly,
transplanted patients have a probability of needing re-transplantation at some point due to eventual chronic rejection of their transplanted
organ, with the possible exception of some newborn recipients. With increased incidence of preformed antibodies, these patients may never
have the opportunity to receive another organ. Based on experimental data, we believe that ADI™ may have the potential to address
this issue providing these individuals better opportunities for receiving an organ.
Technology Platform
ADI™
utilizes a novel approach that mimics the way our bodies naturally induce tolerance to our own tissues. It is a technology platform,
which we believe can be engineered to address a wide variety of indications. ADI™ includes two DNA molecules which are designed
to deliver signals to induce tolerance. The first DNA molecule encodes a pro-apoptotic protein, which induces ‘programmed’
cell death (apoptosis). This is a core component of the technology because it is intended to greatly increase the recruitment of
dendritic cells, which are implicated in regulating the immune system. The second DNA molecule encodes the protein of interest (guiding
antigen), which is modified to promote a path of tolerance. The guiding antigen is intended to result in tolerance induction specific
to the tissue where the protein is found.

ADI™
has shown efficacy in several preclinical models (skin grafting, psoriasis, type 1 diabetes, alopecia areata and multiple sclerosis)
and its efficacy can be attributed to multiple factors:
| |
1. |
ADI™
does not rely on a single mechanistic approach. It has multiple components (interchangeable target antigen, apoptosis, methylated
plasmid DNA) that affect different arms of the immune system, which can be manipulated. |
| |
2. |
ADI™
activates key immune cells known to maintain tolerance in test animals and humans. |
| |
3. |
ADI™
has been successfully applied to a stringent transplantation model. |
| |
4. |
ADI™ lends itself
to repeat dosing, which may be required to achieve its full potential therapeutic effect. |
Proof
of Concept: Skin Grafting
Results
shown are 5 weeks post-transplantation
The
proof-of-concept experiment performed in transplantation was a skin allograft transplantation procedure in which the donor skin was obtained
from white BALB/c mice and transplanted to black C57BL/6 mice. The experiment was designed to address a more challenging scenario where
the donor tissue was obtained from a donor which is genetically mismatched with the recipient. This is unlike clinical scenarios where
the donor and recipient are genetically matched as much as possible. While these experiments were repeated in several separate experiments,
the results shown here were obtained from a study conducted with 14 mice in the ADI™ treatment group and 7 mice in the control
group. Prior to submission of an Investigational New Drug or Clinical Trial Application, additional non-clinical studies will be conducted
to establish the precise protocol (e.g. timing of vaccine administration, dosing, and appropriate immunosuppressive agents that will
be used in combination with ADI™) that will be used in the clinical trials. Pre-clinical safety/toxicology studies have already
been conducted by a GLP lab to ensure product safety for clinical testing. These studies have shown no signs of toxicity to ADI™
treatment in mice.


Proof
of Concept: Psoriasis

| |
● |
Psoriasis causes increased
skin thickness and scaling in an established 10-day psoriasis model |
| |
● |
ADI™ treatment resulted
in a 69% reduction in skin thickening and 38% reduction in scaling over the 10-day study period |
Proof
of Concept: Type 1 Diabetes

Typically,
90% of female NOD mice developed spontaneous autoimmune diabetes. Disease progression may be different for individual animals.

ADI™
was administered once a week for 8 weeks after each animal developed hyperglycemia. All animals responded with 80% showing durable response
for the entire 40-week study period.
| |
● |
Type 1 or autoimmune diabetes
is a condition where the body’s immune system mistakenly attacks cells in the pancreas resulting in diminished production of
insulin |
| |
● |
ADI™ incorporates
an antigen (GAD) expressed in the pancreas |
| |
● |
Administration of ADI™
using GAD as the antigen over an 8-week period in animals with T1D restores insulin production and reverses hyperglycemia |
Pre-clinical
and Clinical Plans
The
resources and efforts used for the IND-enabling work summarized below supports both the psoriasis and TID clinical programs
High-level
objectives for psoriasis clinical program:
| |
● |
Completion of IND-enabling
work. Aditxt has initiated GMP manufacturing of clinical grade material that will be used for the first-in-human studies in subjects
with psoriatic lesions. Included in the manufacturing program is stability studies; the regulatory agency requires one month of stability
data for the GMP material for submission of the clinical trial application (CTA). Stability data will continue to be gathered while
the clinical trials are ongoing and up to 24 months. Aditxt has also completed the in-life portion of the toxicology studies. Safety
data have been recorded and Aditxt is now awaiting immunotoxicology data, which are forthcoming. |
| |
|
|
| |
● |
Upon completion of GMP
manufacturing and toxicology studies, a CTA will be submitted in Q4 2022 to initiate the Phase I/II FIH clinical trials. |
The
FIH clinical studies will combine Phase I (designed to test clinical safety) and Phase IIa (designed to obtain proof of effectiveness
in human subjects), in subjects with psoriatic skin lesions. We have selected this indication for several reasons, including:
| |
1. |
Our existing preclinical
data have shown promising results in reducing scaling and skin thickness in the mouse model; |
| |
2. |
The relative ease of visualization
of healing of psoriatic lesions; and |
| |
3. |
The need for therapies
that suitable and justifiable in individuals with mild to moderate psoriasis (current biologic therapies are primarily used in moderate
to severe cases). |
We
have identified a contract research organization with capabilities to conduct a multi-center study and ability to recruit the needed
number of subjects to complete the clinical trials. Upon approval by the regulatory agency clinical trials will be initiated.
High-level
objectives for type 1 diabetes (T1D) clinical program:
| |
● |
Completion of IND-enabling
work. Aditxt has initiated GMP manufacturing of clinical grade material that will be used for the first-in-human studies in subjects
with psoriatic lesions. Included in the manufacturing program is stability studies; the regulatory agency requires one month of stability
data for the GMP material for submission of the clinical trial application (CTA). Stability data will continue to be gathered while
the clinical trials are ongoing and up to 24 months. Aditxt has also completed the in-life portion of the toxicology studies. Safety
data have been recorded and Aditxt is now awaiting immunotoxicology data, which are forthcoming. |
| |
● |
Clinical Phase I/II Study
to demonstrate safety and clinical proof-of-concept in T1D |
Our
clinical studies will combine Phase I (designed to test clinical safety) and Phase II (designed to obtain proof of effectiveness in human
subjects), in T1D patients. We have selected this indication for several reasons, including:
| |
1. |
Our existing preclinical
data have shown promising results using ADI™ to reverse hyperglycemia in the mouse model; and |
| |
|
|
| |
2. |
There is currently no treatment
for T1D and the only option for patients suffering from T1D is insulin replacement therapy. |
We
will be identifying clinical trial centers with adequate patients. Upon approval by the FDA and/or the applicable regulatory agency clinical
trials will be initiated.
High-level
objectives for skin allograft clinical program:
| |
● |
Completion of preclinical
studies to identify the appropriate protocol for dosing and combination of ADI™ with immune suppression protocols. |
| |
|
|
| |
● |
Completion of IND-enabling
work including GMP manufacturing and toxicology studies. |
| |
|
|
| |
● |
Clinical Phase I/II Study
to demonstrate safety and clinical proof-of-concept in patients requiring skin allografts. |
Our
clinical studies will combine Phase I (designed to test clinical safety) and Phase II (designed to obtain proof of effectiveness in human
subjects), in patients requiring skin allografts. We have selected this indication for several reasons, including:
| |
1. |
Our existing preclinical
data have shown promising results using ADI™ to prolong skin allograft survival in mismatched mouse model; and |
| |
|
|
| |
2. |
The relative ease of visualization
of graft quality without the need for biopsies. |
We
will be identifying clinical trial centers with adequate patients. Upon approval by the FDA and/or the applicable regulatory agency clinical
trials will be initiated.
We
are developing our immune monitoring platforms with the objective of utilizing them as clinical assays in pre-clinical and clinical studies.
The multiplex technologies could potentially allow evaluation of more analytes with less tissue samples.
Drug
Approval Process
In
the United States, FDA approval is required before any new drugs can be introduced to the market. We currently have a product candidate
for our first-in-human studies, but as of the date of report, we have not submitted an application to the regulatory agencies for approval.
We
are working with a contract manufacturer who has the know-how, product ingredients including plasmid DNA molecules, and our patent-pending
bacterial strain. Several batch runs have been successfully completed to demonstrate our ability to produce the DNA plasmids in a GMP
facility. Based on validation studies, we are reasonably confident in our ability to produce clinical grade product candidates at larger
scales. The contract manufacturer has provided a proposal for manufacturing of our clinical grade material, which will be signed and
accepted once we are ready to initiate GMP manufacturing. We are not currently party to an agreement with this contract manufacturer.
The
product candidate selected for clinical trials must be subjected to pre-clinical safety/toxicology studies by an independent GLP (Good
Laboratory Practice) laboratory to demonstrate its suitability for clinical testing in human patients. Upon completion of manufacturing
and safety/toxicology testing, an Investigational New Drug (IND) application will be prepared for submission to the regulatory agencies.
Upon
receipt of clearance to initiate clinical testing, the ADI™ product can be tested in human patients. Our product will be tested
in clinical trials, one in patients with psoriasis and one in patients who require skin allografting. Therefore, our first-in-human studies
will be combined Phase I/Phase II studies in which safety and efficacy data will be obtained. We plan to start with in skin indications
(psoriasis and skin allografting) because we believe these indications will be most efficient in providing safety and efficacy data in
clinical trials. In parallel, we will continue to develop additional product formulations for other indications.
We
are developing our immune monitoring platforms with the objective of utilizing them as clinical assays in pre-clinical and clinical studies.
The multiplex technologies could potentially allow evaluation of more analytes with less tissue samples.
Target
Market
Psoriasis
affects close to 100 million people worldwide and presents a large market estimated at over $20 billion annually. Treatments range include
topical and systemic therapeutics including vitamin D analogs, steroids, retinoids, immunosuppressants and biologics (i.e. monoclonal
antibodies). While in more recent years, several classes of biologics have entered the market, most are primarily used for patients suffering
from moderate to severe psoriasis because of their impairment of systemic immune responsiveness to infections and cancers. Aditxt believes
that products based on the ADI™ platform will not be associated with similar side effects and can be targeted for use in mild to
moderate cases.
T1D
is one of the most common chronic disorders in children and affects nearly 2 million Americans, and has an incidence and prevalence increasing
at alarming rates in industrialized countries. Current treatment consists of daily delivery of insulin as replacement therapy, but administration
of the hormone can induce life-threatening hypoglycemia and does not completely prevent morbidity and mortality associated with the disease.
Aditxt is leveraging the ADI™ technology to develop a new class of immunotherapy designed to arrest the autoimmune destruction
of the insulin producing beta cells of the pancreas. This will be the first therapy to accomplish that long sought after goal, thus increasing
life span and quality of life for up to 40,000 of US citizens and about 300,000 people around the world who develop T1D each year, with
a 3-5% increase in yearly incidence.
In
the U.S. alone, there are over 36,000 patients who receive organ transplantations each year, with more than 113,000 on transplant waiting
lists.
The
field of organ transplantation has been made possible and continues to rely on broad-acting immunosuppressive drugs, high levels of which
can result in a compromised immune system that renders organ recipients susceptible to cancer and potentially life-threatening infections
including re-activation of latent viruses.
In
addition, immunosuppressants control acute rejection during the early time-period after receiving an organ but chronic rejection of the
organ remains an unmet challenge for surgeons and transplant recipients.
While
efforts have been made by various groups to promote tolerance through cell therapies and ex vivo manipulation of patient cells,
these procedures take place outside the body and typically require hospitalization.
Moreover,
transplanted patients will need re-transplantation at some point, with the possible exception of some newborn recipients. With increased
incidence of preformed antibodies, these patients may never have the opportunity to receive another organ. Preformed antibodies can develop
in previously transplanted patients, patients who have given birth, and patients who have previously received blood transfusions. These
patients have much lower chances at qualifying to receive organs due to their increased risk of rejection - even with immune suppression.
The potential to reduce formation of preformed antibodies in these patients will provide better opportunities for them to receive another
transplanted organ.
There
are gaps between current approaches and what the market needs. We believe that ADI™ addresses these gaps. ADI™ is easy to
administer (does not require ex-vivo treatment of patient cells), it does not appear to suppress the immune system, it may allow
patients to live with transplanted organs with significantly reduced immune suppression, it may provide for long-term survival of transplanted
tissues and organs, may be more effective because it does not rely on a single immune pathway/mechanism, and potentially provides patients
with pre-existing antibodies a chance to qualify to receive organs.
While
these advantages present opportunities for unmet medical needs in the field of organ transplantation, the industry in which we operate
is highly competitive. A small company such as us will meet significant challenges including regulatory requirements for approval of
a new class of therapeutic agents, challenges in large scale manufacturing and marketing, cost of developing a novel therapeutic agent,
which may require co-development partners who may or may not be willing to work with us, and the willingness of transplant surgeons to
adopt our therapeutic vaccines in their existing immune suppression protocols. These challenges pose risks that we may not be able to
overcome.
License
Agreement with Loma Linda University -
On
March 8, 2018, we entered into an Assignment Agreement (the “Assignment Agreement”) with Sekris Biomedical, Inc. (“Sekris”).
Sekris was a party to a License Agreement with Loma Linda University (“LLU”), entered into and made effective on May 25,
2011, and amended on June 24, 2011, July 16, 2012 and December 27, 2012 (the “Original Agreement,” and together with the
Assignment Agreement, the “Sekris Agreements”). Pursuant to the Assignment Agreement, Sekris transferred and assigned all
of its rights and obligations in and to liabilities under the Original Agreement, of whatever kind or nature, to us. In exchange, on
March 8, 2018, we issued a warrant to Sekris to purchase up to 10,000 shares of our Common Stock (the “Sekris Warrant”).
The warrant was immediately exercisable and has an exercise price of $200.00 per share. The expiration date of the warrant is March 8,
2023. On March 15, 2018, as amended on July 1, 2020, we entered into a LLU License Agreement directly with Loma Linda University, which
amends and restates the Sekris Agreements.
Pursuant
to the LLU License Agreement, we obtained the exclusive royalty-bearing worldwide license in and to all intellectual property, including
patents, technical information, trade secrets, proprietary rights, technology, know-how, data, formulas, drawings, and specifications,
owned or controlled by LLU and/or any of its affiliates (the “LLU Patent and Technology Rights”) and related to therapy for
immune-mediated inflammatory diseases (the ADI™ technology). In consideration for the LLU License Agreement, we issued 500 shares
of Common Stock to LLU.
Pursuant
to the LLU License Agreement, we are required to pay an annual license fee to LLU. Also, we paid LLU $455,000 in July 2020 for outstanding
milestone payments and license fees. We are also required to pay to LLU milestone payments in connection with certain development milestones.
Specifically, we are required to make the following milestone payments: $175,000 on March 31, 2022; $100,000 on March 31, 2024; $500,000
on March 31, 2026; and $500,000 on March 31, 2027. Additionally, as consideration for prior expenses incurred by LLU to prosecute, maintain
and defend the LLU Patent and Technology Rights, we made the following payments to LLU: $70,000 due at the end of December 2018, and
a final payment of $60,000 due at the end of March 2019. We are required to defend the LLU Patent and Technology Rights during the term
of the LLU License Agreement. Additionally, we will owe royalty payments of (i) 1.5% of Net Product Sales and Net Service Sales on any
Licensed Products (defined as any finished pharmaceutical products which utilizes the LLU Patent and Technology Rights in its development,
manufacture or supply), and (ii) 0.75% of Net Product Sales and Net Service Sales for Licensed Products and Licensed Services not covered
by a valid patent claim for technology rights and know-how for a three (3) year period beyond the expiration of all valid patent claims.
We also are required to produce a written progress report to LLU, discussing our development and commercialization efforts, within 45
days following the end of each year. All intellectual property rights in and to LLU Patent and Technology Rights shall remain with LLU
(other than improvements developed by or on our behalf).
The
LLU License Agreement shall terminate on the last day that a patent granted to us by LLU is valid and enforceable or the day that the
last patent application licensed to us is abandoned. The LLU License Agreement may be terminated by mutual agreement or by us upon 90
days written notice to LLU. LLU may terminate the LLU License Agreement in the event of (i) non-payments or late payments of royalty,
milestone and license maintenance fees not cured within 90 days after delivery of written notice by LLU, (ii) a breach of any non-payment
provision (including the provision that requires us to meet certain deadlines for milestone events (each, a “Milestone Deadline”))
not cured within 90 days after delivery of written notice by LLU and (iii) LLU delivers notice to us of three or more actual breaches
of the LLU License Agreement by us in any 12-month period. Additional Milestone Deadlines include: (i) submission of an IND/clinical
trial application to initiate first-in-human clinical trials on or before March 31, 2022, (ii) the completion of first-in-human (phase
I/II) clinical trials by March 31, 2024, (iii) the completion of Phase III clinical trials by March 31, 2026 and (iv) biologic licensing
approval by the FDA by March 31, 2027.
PEARSANTA,
INC.
Formed in January 2023, our
subsidiary PearsantaTM, Inc. (“Pearsanta”) is seeking to take personalized medicine to a whole new level by delivering
“Health by the Numbers”. On November 22, 2023, Pearsanta entered into an assignment agreement with FirstVitals LLC, an entity
controlled by Pearsanta’s CEO, Ernie Lee (“FirstVitals”), pursuant to which FirstVitals assigned its rights in certain
intellectual property and website domain to Pearsanta in consideration of the issuance of 500,000 shares of Pearsanta common stock to
FirstVitals. On December 18, 2023, the board of directors of Pearsanta adopted the Pearsanta 2023 Omnibus Equity Incentive Plan (the
“Pearsanta Omnibus Incentive Plan”), pursuant to which it reserved 15 million shares of common stock of Pearsanta for future
issuance under the Pearsanta Omnibus Incentive Plan and the Pearsanta 2023 Parent Service Provider Equity Incentive Plan (the “Pearsanta
Parent Service Provider Plan”) and approved the issuance of 9.32 million shares of Pearsanta common stock under the Pearsanta Parent
Service Provider Plan.
Since
its foundation, we have been building the platform for enabling our vision of Any-test, Anytime, Anywhere. Our plan for Pearsanta’s
platform is for it to be the transactional backbone for sample collection, sample processing (on- and off-site), and reporting. This
requires the development and convergence of multiple components developed by Pearsanta or through transactions with third parties including
collection devices, Lab on Chip technologies, Lab Developed Test (LDT) assays, data-driven analysis engine, and telemedicine. According
to a comprehensive research report by Market Research Future, the clinical and consumer diagnostic market is estimated to hit $429.3
billion by 2030.
We
believe that timely and personalized screening, enables for more informed decisions about treatments. Pearsanta’s platform is being
developed to be a seamless digital healthcare solution. This platform will integrate at-location sample collection, Point-of-Care (POC)
and LDT, and analytical reporting engine, with telemedicine enabled visits with licensed physicians to review test results and, if necessary,
order a prescription. Pearsanta’s goal of extending its platform to provide a more complete picture about someone’s health
status and how their dynamic status combined with their genetic makeup may affect their response to medication. The POC component of
Pearsanta would enable diagnostic testing at-home, at work, in pharmacies, and more to generate results quickly so that an individual
can access necessary treatment faster. With certain infections, prescribing the most effective treatment according to one’s numbers
can prevent hospital emergency room admissions and potentially life-threatening consequences.
Examples
of potential indication-focused tests for the Test2Treat platform will include the evaluation for advanced urinary tract infections (“UTIs”),
COVID-19/flu/respiratory syncytial virus, sexually transmitted infections, gut health, pharmacogenomics (i.e., how your genes affect
the way your body responds to certain therapeutics), and sepsis. These offerings are novel and needed. We believe that the current standard
of care using broad spectrum antibiotic treatment can be ineffective and life-threatening. For example, according to a recent CDC report,
improperly prescribed antibiotics are used in 50% of outpatient cases. Further, according to a recent article published in Physicans
Weekly, only 1% of board-certified critical care medicine physicians are trained in infectious disease.
Licensed
Technologies - AditxtScoreTM
We
intend to sublicense to Pearsanta an exclusive worldwide sub-license for commercializing the AditxtScore™ technology which provides
a personalized comprehensive profile of the immune system. AditxtScore is intended to detect individual immune responses to viruses,
bacteria, peptides, drugs, supplements, bone marrow and solid organ transplants and cancer. It has broad applicability to many other
agents of clinical interest impacting the immune system, including those not yet identified such as emerging infectious agents.
AditxtScore
will seek to enable individuals and their healthcare providers to understand, manage and monitor their immune profiles and to stay informed
about attacks on or by their immune system. We believe AditxtScore can also assist the medical community and individuals by being able
to anticipate the immune system’s potential response to viruses, bacteria, allergens, and foreign tissues such as transplanted
organs. This technology may be able serve as warning signal thereby allowing for more time to respond appropriately. Its advantages include
the ability to provide simple, rapid, accurate, high throughput assays that can be multiplexed to determine the immune status with respect
to several factors simultaneously, in 3-16 hours. In addition, it can determine and differentiate between distinct types of cellular
and humoral immune responses (T and B cells and other cell types). It also provides for simultaneous monitoring of cell activation and
levels of cytokine release (i.e., cytokine storms).
This
capability may be possible by having the ability to determine the body’s potential response and for developing a plan to deal with
an undesirable reaction by the immune system. Its advantages include the ability to provide a simple, rapid, accurate, high throughput
assays that can be multiplexed to determine the immune status with respect to several factors simultaneously, in 3-16 hours. In addition,
it can determine and differentiate between various types of cellular and humoral immune responses (T and B cells and other cell types).
It also provides for simultaneous monitoring of cell activation and levels of cytokine release (i.e., cytokine storms).
We
are also evaluating plans to obtain regulatory approval for AditxtScore’s use as a clinical assay and seeking to secure manufacturing,
marketing and distribution partnerships for application in the various markets. To obtain regulatory approval to use AditxtScore™
as a clinical assay, we have conducted validation studies to evaluate its performance in detection of antibodies and plan to continue
conducting additional validation studies for new applications in autoimmune diseases.
We
plan to utilize AditxtScore™ in our upcoming pre-clinical and clinical studies to monitor subjects’ immune response before,
during and after ADI™ drug administration. We are also evaluating plans to obtain regulatory approval for AditxtScore™’s
use as a clinical assay and seeking to secure manufacturing, marketing and distribution partnerships for application in the various markets.
To obtain regulatory approval to use AditxtScore™ as a clinical assay, we have conducted validation studies to evaluate its performance
in detection of antibodies and plan to continue conducting additional validation studies for new applications in autoimmune diseases
and transplantation.
(1) Organ
Rejection
Typically,
by the time a transplanted or a native organ shows signs of failure, the damage is already done, and reversal of the tissue injury becomes
challenging. Access to early warning signs of damage would be invaluable to reverse or even prevent the damage. We believe that there
are currently no practical, efficient assays available to measure cellular immune responses and available tools do not provide timely
information for patients. AditxtScore™ can be used to provide a sensitive and rapid tool for pre-transplant monitoring and to determine
T and B cell response and to differentiate between various types of cellular immune responses. It can be multiplexed providing information
about the number of cells responding as well as quantifying the amounts of various cytokines released by the cells in the same assay.
Determination of cellular response has valuable applications for prediction, monitoring, early detection, and treatment of disease, including
organ failure/rejection, as well as treatment efficacy. It can also reveal dysfunction of the immune system that can potentially contribute
to more severe disease.
(2)
Autoimmunity
Our
immune system develops to differentiate self from non-self. In autoimmunity, the body’s ability to distinguish this difference
is impaired. Detection of early signs of immune misrecognition may allow earlier intervention to reduce tissue destruction and to potential
reverse the process more effectively. Better tools are needed to recognize immune responses to our own tissues earlier, and with more
sensitivity and accuracy. We believe that AditxtScore™ harnesses the promise to develop such tools that can be used for early diagnosis,
evaluation of treatment effectiveness and determination of the need for maintenance therapies when needed.
(3) Allergies
Our
immune system protects us by acting as a barrier against foreign substances and by eliminating them when they penetrate our bodies. Once
the initial exposure has occurred, memory cells develop to prepare the body against a future exposure. This process is called immunity.
In certain situations, however, instead of immunity, the immune system develops memory cells that result in a more severe reaction during
a future exposure to the same substance. This type of response is called a hypersensitivity response, commonly known as an allergic response.
AditxtScore™ can be used to develop multiplex panels each designed to test and monitor immune response to allergens. Based on the
ability of this technology to run multiple tests in a single assay, 100 or more substances can potentially be tested simultaneously.
(4) Drug/Vaccine
Response
We
believe that there are currently no effective assays to predict and easily assess responses to drugs or vaccines. To determine whether
an individual has responded to a particular vaccine, antibody titers are measured. This process may take several days or even weeks.
Furthermore, for vaccines that require a series of injections, titers are not measured between injections and may not be known for months.
AditxtScore™ can be used to determine whether a patient is a responder or non-responder (e.g. individuals with a suppressed immune
response may be non-responders). It can provide an effective and rapid tool for potentially determining beneficial responses to a vaccine
and can be used to monitor levels of immune responsiveness post vaccination. It can allow evaluation of multiple vaccines in a single
test (for memory B cell detection). We believe that this application can be useful for vaccines, cancer therapeutics anti-rejection drugs,
anti-viral drugs, among others.
(5) Disease
Susceptibility
Disease
susceptibility can vary from one individual to another, and it can be a function of various factors, including genetic variability and
differences in human leukocyte antigens (HLA) encoded by major histocompatibility complex (MHC) and responsible for regulation of the
immune system in humans. People with certain HLA types may have higher or lower susceptibility to diseases. AditxtScore™ can be
used to develop assays to evaluate differences in HLA types in individuals to help elucidate the relationship between certain HLA types
and susceptibility to various diseases.
(6) Infectious
Diseases
We
believe that infectious diseases can cause a major predicament for scientific and medical professionals, epidemiologists, and infectious
disease specialists, who need to determine how to treat patients in real-time while efficacious therapies are still being developed.
Proper decision making requires understanding why some affected individuals show minor or no symptoms, some recover, and others die.
We feel that this is fundamental to creating effective targeted therapeutics which may differ depending on the underlying profile of
the individual at risk for, or with, disease. The immune system plays a major role in how any given individual responds to the infectious
agent. This response can be inadequate or too robust or appropriately effective. Regardless, the kinetics of the response by the cellular
and humoral (antibody) immune systems to the infectious agent are often unknown. A basic critical question, then, is what do the dynamics
of the immune response look like from exposure to and through the disease period and during convalescence for those who survive and those
who don’t; and how might vaccines and therapies alter these profiles such that predictions of vaccine/drug efficacy could be inferred
prior to vaccination/treatment and/or disease severity or progression be prognosticated. We believe that AditxtScore™ can be used
to help address these questions with multiplex assays each designed to test and monitor the immune response to infectious agents.
License
Agreement with Leland Stanford Junior University (“Stanford”)
On
February 3, 2020, we entered into an exclusive license agreement (the “February 2020 License Agreement”) with Stanford with
regard to a patent concerning a method for detection and measurement of specific cellular responses. Pursuant to the February 2020 License
Agreement, other than as described below, we received an exclusive worldwide license to Stanford’s patent with regard to use, import,
offer, and sale of Licensed Products (as defined in the agreement). The license to the patented technology is exclusive, including the
right to sublicense, beginning on the effective date of the agreement and ending when the patent expires. Under the exclusivity agreement,
we acknowledged that Stanford had already granted a non-exclusive license in the Nonexclusive Field of Use, under the Licensed Patents
in the Licensed Field of Use in the Licensed Territory (as those terms are defined in the February 2020 License Agreement”). However,
Stanford agreed to not grant further licenses under the Licensed Patents in the Licensed Field of Use in the Licensed Territory. On December
29, 2021, we entered into an amendment to the February 2020 License Agreement which extended our exclusive right to license the technology
deployed in AditxtScoreTM and securing worldwide exclusivity in all fields of use of the licensed technology.
We
were obligated to pay and paid a fee of $25,000 to Stanford within 60 days of February 3, 2020. We also issued 375 shares of the Company’s
Common Stock to Stanford. An annual licensing maintenance fee is payable by us on the first anniversary of the February 2020 License
Agreement in the amount of $40,000 for 2021 through 2024 and $60,000 starting in 2025 until the license expires upon the expiration of
the patent. The Company is required to pay and has paid $25,000 for the issuances of certain patents. The Company will pay milestone
fees of $50,000 on the first commercial sales of a licensed product and $25,000 at the beginning of any clinical study for regulatory
clearance of an in vitro diagnostic product developed and a potential licensed product. We are also required to: (i) provide a listing
of the management team or a schedule for the recruitment of key management positions by March 31, 2020 (which has been completed), (ii)
provide a business plan covering projected product development, markets and sales forecasts, manufacturing and operations, and financial
forecasts until at least $10,000,000 in revenue by June 30, 2020 (which has been completed), (iii) conduct validation studies by September
30, 2020 (which has been completed), (iv) hold a pre-submission meeting with the FDA by September 30, 2020 (which has been completed),
(v) submit a 510(k) application to the FDA, Emergency Use Authorization (“EUA”), or a Laboratory Developed Test (“LDT”)
by March 31, 2021, (which has been completed), (vi) develop a prototype assay for human profiling by December 31, 2021 (which has been
completed), (vii) execute at least one partnership for use of the technology for transplant, autoimmunity, or infectious disease purposes
by March 31, 2022, and (viii) will provide further development and commercialization milestones for specific fields of use in writing
by December 31, 2022.
In
addition to the annual license maintenance fees outlined above, we will pay Stanford royalties on Net Sales (as such term is defined
in the February 2020 License Agreement) during the of the term of the agreement as follows: 4% when Net Sales are below or equal to $5
million annually or 6% when Net Sales are above $5 million annually. The February 2020 License Agreement may be terminated upon our election
on at least 30 days advance notice to Stanford, or by Stanford if we: (i) are delinquent on any report or payment; (ii) are not diligently
developing and commercializing Licensed Product; (iii) miss certain performance milestones; (iv) are in breach of any provision of the
February 2020 License Agreement; or (v) provide any false report to Stanford. Should any events in the preceding sentence occur, we have
a thirty (30) day cure period to remedy such violation.
Plan
of Operations
The
initial application of the platform was AditxtScore™ for COVID-19, which was designed to provide a more complete assessment of
an individual’s infection and immunity status with respect to the SARS-CoV-2 virus. Infection status is determined by evaluating
the presence or absence of the virus, and immunity status by measuring levels of antibodies against viral antigens and their ability
to neutralize the virus.
In
early 2021, we established our AditxtScore™ Immune Monitoring Center in Richmond, Virginia (the “Center”). The Center
operates as a Clinical Laboratory Improvement Amendments (CLIA) certified facility for the processing of our AditxtScore™ for COVID-19
Lab Developed Test (LDT) for our prospective channel partners, including labs and hospitals.
In
August 2020, we filed for an Emergency Use Authorization (EUA) with the FDA with the ultimate objective of filing a 510(K) application.
On January 14, 2022, we submitted requests to obtain two EUAs for our antibody and neutralizing tests following an announcement on November
15, 2021 by the Department of Health and Human Services that COVID-19 related tests will require FDA review and FDA’s position
that COVID-19 tests that have been in use prior to the announcement must submit applications for EUAs but can continue to operate unless
informed otherwise. In the meantime, we are providing AditxtScore™ as a service as a Laboratory Developed Test (LDT) to assess
immunity status to COVID-19.
The
public health emergency declaration for COVID-19 ended in May 2023. Thus, COVID-related assays are no longer considered as priority for
review by FDA and will not be considered for EUAs. Assays that are not developed for evaluation of infection or immunity status to SARS-CoV-2
will continue to be offered as LDTs.
Intellectual
Property (IP)
We
strive to protect and enhance the proprietary technology, inventions, and improvements that are commercially important to our business,
including seeking, maintaining and defending patent rights, whether developed internally or licensed from third parties. Our policy is
to seek to protect our proprietary position by, among other methods, filing patent applications in the United States and in jurisdictions
outside of the United States, to protect our proprietary technology, inventions, improvements and product candidates that are important
to the development and implementation of our business. We also rely on trade secrets and know-how relating to our proprietary technology
and product candidates, continuing innovation, and in-licensing opportunities to develop, strengthen and maintain our proprietary position
in the field of immuno-therapy. We also plan to rely on data exclusivity, market exclusivity, and patent term extensions when available.
Our commercial success will depend in part on our ability to obtain and maintain patent and other proprietary protection for our technology,
inventions, and improvements; to preserve the confidentiality of our trade secrets; to obtain and maintain licenses to use intellectual
property owned by third parties; to defend and enforce our proprietary rights, including any patents that we may own in the future; and
to operate without infringing on the valid and enforceable patents and other proprietary rights of third parties.
Our
innovation portfolio includes: (1) ADI™ immune modulation technologies, which are currently at the pre-clinical stage and are designed
to retrain the immune system to induce tolerance with an objective of addressing rejection of transplanted organs, autoimmune diseases,
and allergies; and (2) AditxtScore™ immune monitoring technologies designed to provide a personalized comprehensive profile of
the immune system. Both categories are protected by multiple families of patents and patent applications, including several issued U.S.
and non-U.S. patents.
The
projected expiration dates for the ADI™ patents and patents issuing from pending applications extend until 2043 for some patents.
As of the date of this report, our patent portfolio for ADI™ includes both patents and patent applications licensed from LLU or
Stanford and patent applications owned solely by Aditxt, including 120 granted patents, 2 allowed patent applications and 30 pending
patient applications in U.S. and other regions. These patents and patent applications cover three different technical aspects of ADI™,
treatment of autoimmune diseases and type 1 diabetes, treatment of organ transplantation, and development of a new class of immunotherapeutics
for various indications. The patents and patent applications cover both methods of treatment for these indications as well as compositions
of matter including plasmids that are able to induce tolerance to antigens or prevention of immune attack on antigens, depending on the
indication, along with methods of producing such plasmids.
The
AditxtScore™ technology is also protected by multiple families of patents and patent applications, including several issued U.S.
and non-U.S. patents. The projected expiration dates for these AditxtScore™ patents and patents issuing from pending applications
ranges from 2037 to 2043. As of the date of this report, our patent portfolio for AditxtScore™ includes both patents and patent
applications licensed from Stanford and patent applications owned solely by Aditxt, including granted patents and 12 applications. These
patents and patent applications encompass methods, systems and kits for detection and measurement of specific immune responses.
We
also possess and/or in-license substantial know-how and trade secrets relating to the development and commercialization of our product
candidates, including related manufacturing processes and technology. We plan to continue expanding and strengthening our IP portfolio
with additional patent applications in the future.
In
March 2021, we signed an agreement with a regulatory consultant based in Munich, Germany, which will play a central role in navigating
the first ADI™ therapeutic program through the clinical trial and regulatory process. The firm has been working with the Aditxt’s
ADI™ team to submit a clinical trial application to the regulatory agency in Germany. Psoriasis is the first indication being
targeted for clinical trial in the ADI™ therapeutics pipeline. Other candidates that are advancing toward clinical trials
include ADI™ for type 1 diabetes and skin allografting.
Advantages
The
sophistication of the AditxtScore technology includes the following:
| |
● |
Greater sensitivity/specificity |
| |
● |
20- fold higher dynamic
range, greatly reducing signal to noise compared to conventional assays |
| |
● |
Ability to customize assays
and multiplex a large number of analytes with speed and efficiency |
| |
● |
Ability to test for cellular
immune responses, i.e. B & T cell, cytokines. |
| |
● |
Proprietary reporting algorithm. |
ADIVIR,
INC.
Formed
in April of 2023, Adivir™, Inc., is Aditxt’s most recently formed wholly owned subsidiary, dedicated to the clinical and
commercial development efforts of innovative antiviral products. These products have the potential to address a wide range of infectious
diseases, including those that currently lack viable treatment options.
Background
On
April 18, 2023, we entered into an Asset Purchase Agreement (the “Asset Purchase Agreement”) with Cellvera Global Holdings
LLC (“Cellvera Global”), Cellvera Holdings Ltd. (“BVI Holdco”), Cellvera, Ltd. (“Cellvera Ltd.”),
Cellvera Development LLC (“Cellvera Development” and together with Cellvera Global, BVI Holdco, Cellvera Ltd. and Cellvera
Development (the “Sellers”), AiPharma Group Ltd. (“Seller Owner” and collectively with the Sellers, “Cellvera”),
and the legal representative of Cellvera, pursuant to which, the Company will purchase Cellvera’s 50% ownership interest in G Response
Aid FZE (“GRA”), certain other intellectual property and all goodwill related thereto (the “Acquired Assets”). Unless
expressly stated otherwise herein, capitalized terms used but not defined herein have the meanings ascribed to them in the Asset Purchase
Agreement. Pursuant to the Asset Purchase Agreement, the consideration for the Acquired Assets consists of (A) $24.5 million, comprised
of: (i) the forgiveness of the Company’s $14.5 million loan to Cellvera Global, and (ii) approximately $10 million in cash, and
(B) future revenue sharing payments for a term of seven years. GRA holds an exclusive, worldwide license for the antiviral medication,
Avigan® 200mg, excluding Japan, China and Russia. The other 50% interest in GRA is held by Agility, Inc. (“Agility”).
Additionally,
upon the closing, the Share Exchange Agreement previously entered into as of December 28, 2021, between Cellvera Global Holdings, LLC
f/k/a AiPharma Global Holdings, LLC (together with other affiliates and subsidiaries) and the Company, and all other related agreements
will be terminated.
The
obligations of the Company to consummate the Closing are subject to the satisfaction or waiver, at or prior to the Closing of certain
conditions, including but not limited to, the following:
| |
(i) |
Satisfactory completion
of due diligence; |
| |
(ii) |
Completion by the Company
of financing sufficient to consummate the transactions contemplated by the Asset Purchase Agreement; |
| |
(iii) |
Receipt by the Company
of all required Consents from Governmental Bodies for the Acquisition, including but not limited to, any consents required to complete
the transfer and assignment of Cellvera’s membership interests in GRA; |
| |
(iv) |
Receipt of executed payoff
letters reflecting the amount required to be fully pay all of each of Seller’s and Seller Owner’s Debt to be paid at
Closing; |
| |
(v) |
Receipt by the Company
of a release from Agility; |
| |
(vi) |
Execution of an agreement
acceptable to the Company with respect to the acquisition by the Company of certain intellectual property presently held by a third
party; |
| |
(vii) |
Execution of an amendment
to an asset purchase agreement previously entered into by Cellvera with a third party that effectively grants the Company the rights
to acquire the intellectual property from the third party under such agreement; |
| |
(viii) |
Receipt of a fairness opinion
by the Company with respect to the transactions contemplated by the Asset Purchase Agreement; and |
| |
(ix) |
Receipt by the Company
from the Seller Owner of written consent, whether through its official liquidator or the Board of Directors of Seller Owner, to the
sale and purchase of the Acquired Assets and Assumed Liabilities pursuant to the Assert Purchase Agreement. |
There
can be no assurance that the conditions to closing will be satisfied or that the proposed acquisition will be completed as proposed or
at all.
Our
commitment to building our antiviral portfolio is strategic and timely. We believe that there has never has there been a more important
time to address the growing global need to uncover new treatments or commercialize existing ones that treat life-threatening global viral
infections.
MDNA
On
December 17, 2023, we entered into an Asset Purchase Agreement (the “Purchase Agreement”) with
Pearsanta, Inc., our majority owned subsidiary (“Pearsanta”) and MDNA Life Sciences, Inc. (“MDNA”), pursuant
to which Pearsanta agreed to acquire certain intellectual property and other specified assets relating to MDNA’s early cancer detection
platform (the “Acquired Assets”). MDNA’s Mitomic™ technology provides a tool for identifying biomarkers associated
with various diseases that lead to mtDNA mutations. The Acquired Assets include, but are not limited to, the following:
| ● | The
Mitomic Endometriosis Test (MET™) is in development as a blood-based assay for diagnosis
of endometriosis. This test aims to provide early diagnostic insights, potentially reducing
delays in diagnosing endometriosis. |
| ● | The
Mitomic Prostate Test (MPT™) is currently under development as a blood-based assay
for diagnosis of prostate cancer. We believe that this test holds the potential to provide
more specific and clinically informative data especially in the prostate-specific antigen
(PSA) grey zone. It aims to address the challenges of over-diagnosis and mitigate risks associated
with low-grade cancers. |
Pursuant
to the Purchase Agreement, the consideration for the transaction was to consist of: (i) an upfront working capital payment of $500,000
(the “Upfront Working Capital Payment”), which is payable upon the satisfaction of certain conditions set forth in the Purchase
Agreement, (ii) a working capital payment at closing of $500,000, (iii) 50,000 shares of our Common Stock, (iv) a warrant to purchase
50,000 shares of our Common Stock exercisable for a term of 5 years at an exercise price equal to the opening price per share of our
Common Stock as of the Closing Date (as defined below), and (v) 5,000 shares of Pearsanta Series A Preferred Stock, par value $0.001
per share (the “Pearsanta Preferred Stock”), provided, however, that if the value of such Pearsanta Preferred Stock, on an
as-converted basis, at the time of the pricing of the Pearsanta common stock in connection with the sale of shares of Pearsanta common
stock to the public in a firm-commitment underwritten public offering pursuant to an effective registration statement under the Securities
Act of 1933, as amended does not equal $25,000,000, an additional amount of Pearsanta Preferred Stock (“Additional Pearsanta Preferred
Stock”) so that the sum of the value of the Pearsanta Preferred Stock plus the Additional Pearsanta Preferred Stock (if any) shall
equal $25,000,000. The Pearsanta Preferred Stock shall have such rights, powers, and preferences as set forth in the form of Certificate
of Designation of Series A Preferred Stock, the form of which is attached as Exhibit D to the Purchase Agreement.
On
January 4, 2024, we entered into a First Amendment to Asset Purchase Agreement with Pearsanta and MDNA, pursuant to which the parties
agreed to: (i) the removal of the Upfront Working Capital Payment, (ii) the removal of the Closing Working Capital Payment (as defined
in the Purchase Agreement”), and (iii) to increase the maximum amount of payments to be made by us under the Transition Services
Agreement (as defined below) from $2.2 million to $3.2 million.
On
January 4, 2024, Pearsanta and MDNA entered into a Transition Services Agreement (the “Transition Services Agreement”), pursuant
to which MDNA agreed that it would perform, or cause certain of its affiliates or third parties to perform, certain services as described
in the Transition Services Agreement for a term of three months in consideration for the payment by Pearsanta of certain fees as provided
in the Transition Services Agreement, in an amount not to exceed $3.2 million.
On
January 4, 2024, we completed its acquisition of the Acquired Assets and issued to MDNA 50,000 shares of our Common Stock, a warrant
to purchase 50,000 shares of our Common Stock, and the Pearsanta Preferred Stock.
Properties
We
lease property consisting of office and laboratory space located at 2569 Wyandotte, St., Suite 101 Mountain View, CA 94043. The lease
expires on August 31, 2024, subject to extension. We are currently three months in arrears on our Mountain View lease.
We
lease property consisting of office space located at 532 Broadhollow Road, Suite 118, Melville, NY 11747. The lease expires on December
31, 2025, subject to extension. We are currently three months in arrears on our Melville lease.
We
lease property consisting of office and laboratory space located at 737 N. 5th Street Richmond, Virginia 23219. The lease
expires on August 31, 2026, subject to extension. We are currently 3.75 months in arrears on our Richmond lease.
Legal Proceedings
From
time to time, we may become involved in various lawsuits and legal proceedings, which arise in the ordinary course of business. Litigation
is subject to inherent uncertainties, and an adverse result in these or other matters may arise from time to time that may harm our business.
We are currently not aware of any such legal proceedings or claims that will have, individually or in the aggregate, a material adverse
effect on our business, financial condition or operating results.
Changes
in and Disagreements with Accountants on Accounting and Financial Disclosure.
None.
Employees
We
have thirty - nine (39) full time employees. We consider the relations with our employees to be good.
MANAGEMENT
Executive
Officers and Directors
Set
forth below is certain information with respect to the individuals who are our directors and executive officers as of December 31, 2023:
| Name |
|
Age |
|
Positions |
| Amro Albanna |
|
54 |
|
Chief Executive
Officer, Director |
| Corinne Pankovcin |
|
57 |
|
Chief Commercialization
Officer |
| Shahrokh Shabahang,
D.D.S., MS, Ph.D. |
|
61 |
|
Chief Innovation
Officer, Director |
| Rowena Albanna |
|
58 |
|
Chief Operating
Officer |
| Thomas J. Farley |
|
51 |
|
Chief Financial
Officer |
| Charles Nelson |
|
70 |
|
Director |
| Brian Brady |
|
45 |
|
Director |
| Jeffrey W. Runge,
M.D. |
|
68 |
|
Director |
Amro
Albanna - Chief Executive Officer
Mr.
Albanna has been our Chief Executive Officer and a Director since we were formed in 2017. He also served as our President from our inception
through September 2021. In 2010, Mr. Albanna co-founded Innovation Economy Corporation (“IEC”), formed to license and commercialize
innovations and create a group of life and health subsidiaries. From 2010 until 2017, Mr. Albanna was Chief Executive Officer and a Director
of IEC and Olfactor Laboratories, Inc., a majority-owned subsidiary of IEC. From 2010 to August 2016, he was the Chief Executive Officer
and a Director of Nano Engineered Applications, Inc., another majority-owned subsidiary of IEC. In 2003, Mr. Albanna founded Qmotions,
Inc. (subsequently renamed Deal A Day Group Corp.). He served as its Chief Executive Officer and a Director until 2011. Qmotions used
3-D spatial tracking and pattern recognition technologies to develop motion-capturing video game controllers. In 2002, Mr. Albanna was
a co-founder of Digital Angel Corporation - a company formed via the merger of three private companies (one being TTC below) into a fourth
publicly traded company (American Stock Exchange) and was placed in charge of commercializing its GPS/wireless technologies. Around that
time, Mr. Albanna co-founded an incubator for startups at the University of California, Riverside Research Park which was acquired in
2007. In 1997, he founded Timely Technology Corporation (“TTC”), which designed and developed e-commerce software for education,
retail and finance. TTC was acquired in 2000 by a Nasdaq-listed company. Mr. Albanna graduated from California State University San Bernardino
in 1991 with a B.S. in Business Administration with concentration in Computer Information Systems. He completed graduate coursework in
Computer Science and Engineering at California State University, Long Beach from 1992 to 1993. In 2019, Mr. Albanna completed coursework
in Immunology and Genetics at Harvard Medical School HMX online learning platform.
Corinne
Pankovcin — Chief Commercialization Officer
Ms.
Pankovcin has been our Chief Commercialization Officer since April 12, 2023. Ms. Pankovcin served as our President from September 2021
through April 2023. Ms. Pankovcin served as our Chief Financial Officer from July 2020 through August 2021. From December 2015 to July
2019, Ms. Pankovcin was the Chief Financial Officer and Managing Director and Treasurer of Business Development Corporation of America
(“BDCA”), a business development company. Prior thereto, from January 2011 to August 2015, Ms. Pankovcin was the Chief Financial
Officer and Treasurer of Blackrock Capital Investment Corporation (NASDAQ: BKCC), and a Managing Director of Finance at BlackRock Investment
Management LLC. Prior to joining BlackRock, Ms. Pankovcin was a senior member of Finance & Accounting of Alternative Investments
and served as Chief Financial Officer for the Global Emerging Markets products group at AIG Capital Partners. Ms. Pankovcin began her
career with PricewaterhouseCoopers LLP, where she ultimately held the role of Senior Manager of Business Assurance for Consumer Products,
Manufacturing, and Middle Market industries from 1991 to 2001. Ms. Pankovcin earned her B.S. in Accounting from Dowling College and her
Master’s Degree in Business Administration from Hofstra University. She is a Certified Public Accountant.
Shahrokh
Shabahang, D.D.S., MS, Ph.D. - Chief Innovation Officer
Dr.
Shabahang has been our Chief Innovation Officer and Director since our inception. In 2009, Dr. Shabahang co-founded Sekris Biomedical
Inc. to incubate immunotherapy technologies. He served as its Chairman of the board and Chief Executive Officer since its inception.
In 2004, Dr. Shabahang joined Genelux Corporation to lead its clinical development program and to serve as board secretary. Genelux developed
an oncolytic virus technology for treatment of cancer, co-invented by Dr. Shabahang. During his tenure from 2004-2007, Genelux raised
$20M+ and obtained regulatory approval to initiate First-In-Human clinical studies in Europe with patients who had not responded to chemotherapy.
In 2001, Dr. Shabahang became the Director of the Microbiology and Molecular Biology Lab at Loma Linda University (“LLU”).
He led the research and development of an antimicrobial therapeutic agent for treatment of dental infections, which was licensed and
marketed by one of the largest dental distribution companies. Dr. Shabahang attended the University of California, Santa Barbara from
1982 to 1984 and later received his DDS from the University of Pacific in 1987. He earned his PhD in Microbiology and Molecular Genetics
at LLU in 2001. During the same year, he established his laboratory at LLU to study infectious diseases and host immune responses.
Rowena
Albanna - Chief Operating Officer
Ms.
Albanna has been our Chief Operating Officer since July 2020. From 2017 to immediately prior to her appointment as Chief Operating Officer,
Ms. Albanna was an independent operations consultant for the Company. Prior thereto, from 2013 to 2017, Ms. Albanna was the Chief Operating
Officer of Innovation Economy Corporation (“IEC”), formed to license and commercialize innovations and create a group of
life and health subsidiaries. From 2010 to 2013, Ms. Albanna was Senior Vice President of IEC. From 2004 to 2009, Ms. Albanna was the
founder and principal of Weezies, an online-based business focused on building and operating e-commerce stores and affiliate marketing
sites. From 2003 to 2004, Ms. Albanna was the head of Product Development and Engineering of Qmotions Inc. Qmotions used 3-D spatial
tracking and pattern recognition technologies to develop motion-capturing video game controllers. In 2002, Ms. Albanna was VP of Product
Development at Digital Angel Systems where she led the development of devices which combined GPS, wireless, and biosensing. Prior to
that, Ms. Albanna held multiple product development roles with increasing responsibilities for various technology companies in the areas
of financial, medical, telecommunications, integrated circuit layout design, and defense. Ms. Albanna is a co-inventor of two patents
related to systems for localizing, monitoring, and sensing objects. Ms. Albanna received a Bachelor of Science degree in Computer Science
with a minor in Mathematics from California State University, San Bernardino in 1988. Ms. Albanna is the wife of Amro Albanna, our Chief
Executive Officer.
Thomas
J. Farley, CPA - Chief Financial Officer
Mr.
Farley has been the Chief Financial Officer since September 2021. Prior to this, Mr. Farley was the Principal Accounting Officer and
Controller from October of 2020 to September 2021. From December 2015 to June 2020, Mr. Farley was the Controller of Business Development
Corporation of America (“BDCA”), a publicly listed business development company. Prior thereto, from January 2011 to August
2015, Mr. Farley was the Senior Controller of Blackrock Capital Investment Corporation (NASDAQ: BKCC). Prior to joining BlackRock Capital
Investment Corporation, Mr. Farley was a Senior Controller for PineBridge Investments Emerging Markets practice. Mr. Farley was also
an Accounting Manager for Bessemer Venture Partners prior to his tenure at PineBridge. Mr. Farley began his career with PricewaterhouseCoopers
LLP, from 1996 to 2001. Mr. Farley earned his B.S. in Accounting from Long Island University and is a Certified Public Accountant.
Brian
Brady - Director
Mr.
Brady has served as a Director since December 1, 2018. Mr. Brady has also been the Director of Investments at a large hospital system
since March 2016, where he is responsible for the management of investment activity related to the organization and personal investments
of the family that owns that company. From December 2011 to March 2016, Mr. Brady was the Vice President/Portfolio Manager at a wealth
advisory firm, where he served in an investment advisory role, including asset and portfolio management. Mr. Brady graduated in 2001
with a Bachelor’s degree in Finance from the University of Illinois at Chicago and in 2014 with a Master of Business Administration
degree from the University of Chicago. We believe that Mr. Brady’s extensive experience with financial markets and management of
investment activities qualifies him to serve as a director of our Company.
Charles
Nelson - Director
Mr.
Nelson has served as a director since November 2023. Prior to his appointment as a member of the Board, Mr. Nelson was a consultant to
the Company from September 2020 through September 2023. He began his financial career as a market representative with American International
Group and in 1979 joined Dean Witter Reynolds as a Financial Advisor, working with high net worth and institutional clients. In 1980,
he joined Drexel Burnham and Lambert, and subsequently, at Ladenberg Thalmann and then at Auerbach Pollack and Richardson originating
equity and investment banking transactions. Over the last 20 years, Mr. Nelson has been involved with financing companies in the fintech,
healthcare and bio-pharma spaces through private equity and public financing including listings on the Nasdaq and the NYSE. We believe
that Mr. Nelson’s extensive experience in capital markets qualifies him to serve as a director of our Company.
Jeffrey
W. Runge, M.D - Director
Dr.
Runge has served as a director since July 2020. From 2008 to the present, Dr. Runge has been the President and founder of Biologue, Inc.,
which provides consulting in biodefense, medical preparedness and injury control. From 2001 through August of 2008, Dr. Runge served
in the Bush administration, first as the head of the National Highway Traffic Safety Administration, and, beginning in September 2005,
as the Department of Homeland Security’s (DHS) first Chief Medical Officer. Dr. Runge founded the DHS Office of Health Affairs
and was confirmed by the United States Senate as DHS’ first Assistant Secretary for Health Affairs in December of 2007. Dr. Runge
also served as Acting DHS Undersecretary for Science and Technology from February through August 2006. In his role at DHS, Dr. Runge
oversaw the operations of the department’s biodefense activities, medical preparedness and workforce health protection, as well
as fulfilling DHS’ responsibilities in medical countermeasure development. Prior to his government service, Dr. Runge was Assistant
Chairman and Director of Clinical Research in the Department of Emergency Medicine at Carolinas Medical Center in Charlotte, NC, from
1984 through 2001. Additionally, Dr. Runge is a Senior Advisor at The Chertoff Group, a firm providing advisory services in business
risk management, security and homeland defense. Since 2010, Dr. Runge has served on the boards of two public companies, including their
Audit and Compensation committees, both of which underwent strategic acquisitions. He has also served as President and CEO of a SEC-regulated
startup company in the health sector. Dr. Runge earned his medical degree from the Medical University of South Carolina and his undergraduate
degree from the University of the South. We believe that Dr. Runge’s experience in medicine, medical research, public service,
business and his prior service on public corporate boards qualifies him to serve as a director of our Company.
Board
Leadership Structure and Risk Oversight
The
Board oversees our business and considers the risks associated with our business strategy and decisions. The Board currently implements
its risk oversight function as a whole. Each of the Board committees, when established, will also provide risk oversight in respect of
its areas of concentration and reports material risks to the Board for further consideration.
Term
of Office
Officers
hold office until his or her successor is elected and qualified. Directors are appointed to serve for one year until the meeting of the
Board following the annual meeting of stockholders and until their successors have been elected and qualified.
Director
Independence
We
use the definition of “independence” of The Nasdaq Stock Exchange LLC (“Nasdaq”) listing rules to make this determination.
Nasdaq listing rules provide that an “independent director” is one who the board “affirmatively determines” has
no “material relationship” with the company “either directly or as a partner, shareholder or officer of an organization
that has a relationship with the Company. Nasdaq listing rules provide that a director cannot be considered independent if:
| ● | the
director is, or has been within the last three (3) years, an employee of the Company or an
immediate family member of director is, or has been within the last three (3) years, an executive
officer of the Company; |
| ● | the
director has received, or has an immediate family member who is an executive officer of the
Company and has received, during any twelve-month period within the last three (3) years,
more than $120,000 compensation directly from the Company (not including compensation received
for director service, pension plan payments or deferred compensation for prior service not
contingent on continued service); |
| ● | the
director or an immediate family member is a current partner of the Company’s internal
or external auditor; the director is a current employee of the auditor; an immediate family
member is a current employee of the auditor and personally works on the Company’s audit;
or the director or an immediate family member was within the last three (3) years a partner
or employee of the auditor and personally worked on the Company’s audit within that
time; |
| ● | the
director or an immediate family member is, or has been within the last three (3) years, employed
as an executive officer of another company where any of the Company’s present executive
officers at the same time serves or served on that company’s compensation committee;
or |
| ● | the
director is a current employee, or an immediate family member is a current executive officer,
of an organization that has made to or received from the Company payments for property or
services in an amount which, in any of the last three fiscal (3) years, exceeds greater of
2% of such other company’s consolidated gross revenues or $1 million. Charitable contributions
not considered “payments” for purposes of this prohibition but contributions
meeting these thresholds must be disclosed on the Company’s website or in its annual
proxy statement or its Annual Report on Form 10-K. |
Under
such definitions, we consider Mr. Nelson, Mr. Brady, and Dr. Runge to be “independent.” Nasdaq listing rules permits a phase-in
period of up to one year for an issuer registering securities in an initial public offering to comply with its requirement that a majority
of the board of directors be made up of independent directors. However, our common stock is not currently quoted or listed on any national
exchange or interdealer quotation system with a requirement that a majority of our Board be independent and, therefore, the Company is
not subject to any director independence requirements. We are subject to Nasdaq’s director independence requirements and are required
to structure our board of directors accordingly.
Committees
of the Board
Our
board of directors has established three standing committees: Audit, Compensation, and Nominating and Corporate Governance. Each of these
standing committees operate pursuant to its respective charter. The committee charters are reviewed annually by the Nominating and Corporate
Governance Committee. If appropriate, and in consultation with the chairs of the other committees, the Nominating and Corporate Governance
Committee may propose revisions to the charters. The responsibilities of each committee are described in more detail below.
Nasdaq
listing rules permits a phase-in period for an issuer registering securities in an initial public offering to meet the Audit Committee,
Compensation Committee and Nominating and Corporate Governance Committee independence requirements. Under the initial public offering
phase-in period, only one member of each committee is required to satisfy the heightened independence requirements at the time our registration
statement becomes effective, a majority of the members of each committee must satisfy the heightened independence requirements within
90 days following the effectiveness of our registration statement, and all members of each committee must satisfy the heightened independence
requirements within one year from the effectiveness of our registration statement.
The
composition and functions of each committee are described below.
| Name | |
Independent | | |
Audit | | |
Nominating
and Corporate Governance | | |
Compensation | |
| Amro Albanna | |
| | | |
| | | |
| | | |
| | |
| Shahrokh Shabahang, D.D.S., MS,
Ph.D. | |
| | | |
| | | |
| | | |
| | |
| Brian Brady | |
| X | | |
| X | * | |
| X | | |
| X | |
| Charles Nelson | |
| X | | |
| X | | |
| X | | |
| X | * |
| Jeffrey Runge, M.D. | |
| X | | |
| X | | |
| X | * | |
| X | |
| * | Chairman
of the committee |
Audit
Committee
The
Audit Committee, among other things, is responsible for:
| ● | appointing;
approving the compensation of; overseeing the work of; and assessing the independence, qualifications,
and performance of the independent auditor; |
| ● | reviewing
the internal audit function, including its independence, plans, and budget; |
| ● | approving,
in advance, audit and any permissible non-audit services performed by our independent auditor; |
| ● | reviewing
our internal controls with the independent auditor, the internal auditor, and management; |
| ● | reviewing
the adequacy of our accounting and financial controls as reported by the independent auditor,
the internal auditor, and management; |
| ● | overseeing
our financial compliance system; and |
| ● | overseeing
our major risk exposures regarding the Company’s accounting and financial reporting
policies, the activities of our internal audit function, and information technology. |
The
Board has affirmatively determined that each member of the Audit Committee meets the additional independence criteria applicable to audit
committee members under SEC rules and Nasdaq listing rules. The Board has adopted a written charter setting forth the authority and responsibilities
of the Audit Committee. The Board has affirmatively determined that each member of the Audit Committee is financially literate, and that
Mr. Brady meets the qualifications of an Audit Committee financial expert.
The
Audit Committee consists of Mr. Brady, Mr. Nelson, and Dr. Runge. Mr. Brady chairs the Audit Committee.
Compensation
Committee
The
Compensation Committee is responsible for:
| ● | reviewing
and making recommendations to the Board with respect to the compensation of our officers
and directors, including the CEO; |
| ● | overseeing
and administering the Company’s executive compensation plans, including equity-based
awards; |
| ● | negotiating
and overseeing employment agreements with officers and directors; and |
| ● | overseeing
how the Company’s compensation policies and practices may affect the Company’s
risk management practices and/or risk-taking incentives. |
The
Board has adopted a written charter setting forth the authority and responsibilities of the Compensation Committee.
The
Compensation Committee consists of Mr. Brady, Mr. Nelson, and Dr. Runge. Mr. Nelson serves as chairman of the Compensation Committee.
The Board has affirmatively determined that each member of the Compensation Committee meets the independence criteria applicable to compensation
committee members under SEC rules and Nasdaq listing rules.
Nominating
and Corporate Governance Committee
The
Nominating and Corporate Governance Committee, among other things, is responsible for:
| ● | reviewing
and assessing the development of the executive officers and considering and making recommendations
to the Board regarding promotion and succession issues; |
| ● | evaluating
and reporting to the Board on the performance and effectiveness of the directors, committees
and the Board as a whole; |
| ● | working
with the Board to determine the appropriate and desirable mix of characteristics, skills,
expertise and experience, including diversity considerations, for the full Board and each
committee; |
| ● | annually
presenting to the Board a list of individuals recommended to be nominated for election to
the Board; |
| ● | reviewing,
evaluating, and recommending changes to the Company’s Corporate Governance Principles
and Committee Charters; |
| ● | recommending
to the Board individuals to be elected to fill vacancies and newly created directorships; |
| ● | overseeing
the Company’s compliance program, including the Code of Conduct; and |
| ● | overseeing
and evaluating how the Company’s corporate governance and legal and regulatory compliance
policies and practices, including leadership, structure, and succession planning, may affect
the Company’s major risk exposures. |
The
Board of Directors has adopted a written charter setting forth the authority and responsibilities of the Nominating and Corporate Governance
Committee.
The
Nominating and Corporate Governance Committee consists of Dr. Runge, Mr. Brady, and Mr. Kiaie. Dr. Runge serves as chairman of the Nominating
and Corporate Governance Committee. The Company’s Board of Directors has determined that each member of the Nominating and Corporate
Governance Committee is independent within the meaning of the independent director guidelines of Nasdaq listing rules.
Compensation
Committee Interlocks and Insider Participation
None of the Company’s
executive officers serves, or in the past has served, as a member of the board of directors or compensation committee, or other committee
serving an equivalent function, of any entity that has one or more executive officers who serve as members of the Company’s board
of directors or its compensation committee. None of the members of the Company’s compensation committee is, or has ever been, an
officer or employee of the Company. There are no interlocking relationships as defined in the applicable SEC rules.
Code
of Business Conduct and Ethics
The
Company’s board of directors adopted a code of business conduct and ethics applicable to its employees, directors and officers,
in accordance with applicable U.S. federal securities laws and the corporate governance rules of the Nasdaq Capital Market. The code
of business conduct and ethics is publicly available on the Company’s website. Any substantive amendments or waivers of the code
of business conduct and ethics or code of ethics for senior financial officers may be made only by the Company’s board of directors
and will be promptly disclosed as required by applicable U.S. federal securities laws and the corporate governance rules of the Nasdaq
Capital Market.
Corporate
Governance Guidelines
The
Company’s board of directors has adopted corporate governance guidelines in accordance with the corporate governance rules of the
Nasdaq Capital Market.
Involvement
in Certain Legal Proceedings
To
our knowledge, none of our current directors or executive officers has, during the past ten years:
| ● | been
convicted in a criminal proceeding or been subject to a pending criminal proceeding (excluding
traffic violations and other minor offenses); |
| ● | had
any bankruptcy petition filed by or against the business or property of the person, or of
any partnership, corporation or business association of which he or she was a general partner
or executive officer, either at the time of the bankruptcy filing or within two years prior
to that time; |
| ● | been
subject to any order, judgment, or decree, not subsequently reversed, suspended or vacated,
of any court of competent jurisdiction or federal or state authority, permanently or temporarily
enjoining, barring, suspending or otherwise limiting, his involvement in any type of business,
securities, futures, commodities, investment, banking, savings and loan, or insurance activities,
or to be associated with persons engaged in any such activity; |
| ● | been
found by a court of competent jurisdiction in a civil action or by the SEC or the Commodity
Futures Trading Commission to have violated a federal or state securities or commodities
law, and the judgment has not been reversed, suspended, or vacated; |
| ● | been
the subject of, or a party to, any federal or state judicial or administrative order, judgment,
decree, or finding, not subsequently reversed, suspended or vacated (not including any settlement
of a civil proceeding among private litigants), relating to an alleged violation of any federal
or state securities or commodities law or regulation, any law or regulation respecting financial
institutions or insurance companies including, but not limited to, a temporary or permanent
injunction, order of disgorgement or restitution, civil money penalty or temporary or permanent
cease-and-desist order, or removal or prohibition order, or any law or regulation prohibiting
mail or wire fraud or fraud in connection with any business entity; or |
| ● | been
the subject of, or a party to, any sanction or order, not subsequently reversed, suspended
or vacated, of any self-regulatory organization (as defined in Section 3(a)(26) of the Securities
Exchange Act of 1934, as amended (the Exchange Act)), any registered entity (as defined in
Section 1(a)(29) of the Commodity Exchange Act), or any equivalent exchange, association,
entity or organization that has disciplinary authority over its members or persons associated
with a member. |
Except
as set forth above and in our discussion below in “Certain Relationships and Related Transactions,” none of our directors
or executive officers has been involved in any transactions with us or any of our directors, executive officers, affiliates or associates
which are required to be disclosed pursuant to the rules and regulations of the SEC.
Other
than as set forth below, we are not currently a party to any legal proceedings, the adverse outcome of which, individually or in the
aggregate, we believe will have a material adverse effect on our business, financial condition or operating results.
The
Company, Amro Albanna, our Chief Executive Officer, and Dr. Shahrokh Shabahang, our Chief Innovation Officer, have been named as cross-defendants
in a counterclaim filed by Christopher Sechrist in an action entitled Shahrokh Shabahang v. Christopher Sechrist, San Bernardino County
Superior Court Case No. CIVDS1831323. In a cross-complaint, Mr. Sechrist contends that he was a partner in a dental practice with Dr.
Shabahang, and that disputes arose as between those partners. Neither the Company nor Mr. Albanna were partners in, or otherwise have
an interest in, the dental practice. Notwithstanding, and seemingly based solely on the fact that Dr. Shabahang became the Chief Innovation
Officer for the Company, Mr. Sechrist has brought claims against the Company and Mr. Albanna. Both the Company and Mr. Albanna believe
that the Counterclaims filed by Mr. Sechrist have no factual or legal merit, and they intend to vigorously defend themselves in the action
and to seek a dismissal of the case as against them as soon as possible. On May 26, 2020, Mr. Sechrist filed a request for dismissal
as to the Company and Mr. Albanna with the Superior Court of California, County of San Bernardino, San Bernardino District. The clerk
of the court entered the dismissal with prejudice on May 26, 2020.
Our
Chief Executive Officer, Amro Albanna, is a party to litigation matters unrelated to the Company or any of its properties. Such litigations
relate to Innovation Economy Corporation (IEC), a company in which Mr. Albanna served as the CEO and a Director from 2010 until 2017,
and its wholly-owned subsidiaries (Innovation Economy Corporation d/b/a ieCrowd). The first litigation (ieCrowd v. Kim, et. al, Superior
Court, Riverside County) was originally commenced by IEC and its subsidiary after Mr. Albanna was no longer affiliated with IEC, against
certain third-party defendants based upon claims related to their misconduct and mismanagement. Such defendants subsequently brought
a countersuit against IEC and its subsidiary, in which they named Mr. Albanna and others as defendants, alleging that they were misled
to invest in IEC and its subsidiary based upon misrepresentations by, among others, Mr. Albanna. The cases have now been consolidated.
Mr. Albanna believes that the counteraction commenced by the third parties against him is without merit and intends to defend himself.
The second matter (Calabria v. ieCrowd) was commenced by Calabria Ventures (the “Calabria Action”) more than 2 years after
Mr. Albanna was no longer affiliated with IEC, related to uncollected rent. Mr. Albanna believes that the action commenced against him
is without merit and intends to defend himself. IEC (either directly or through its Director and officer insurance policy) has covered
all related legal costs to date. On August 5, 2020, the plaintiff in the Calabria Action filed a request for dismissal as to Mr. Albanna
with the Superior Court of California, County of Riverside. The clerk of the court entered the dismissal without prejudice on August
5, 2020.
EXECUTIVE
AND DIRECTOR COMPENSATION
The
following table represents information regarding the total compensation for the named executive officers of the Company as of December
31, 2023 and 2022:
Name
and Principal Position | |
Year | | |
Salary
($) | | |
Bonus
($) | | |
Stock
Awards
($) | | |
Option
Awards
($) | | |
Restricted
Stock
Units
($) | | |
All
Other
Compensation
($)(4) | | |
Total
($) | |
| Amro
Albanna | |
| 2023 | | |
| 432,119 | | |
| - | | |
| - | | |
| 47,114 | | |
| - | | |
| 40,000 | | |
| 519,233 | |
| Chief
Executive Officer and Director | |
| 2022 | | |
| 500,000 | | |
| - | | |
| | | |
| - | | |
| - | | |
| - | | |
| 500,000 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Shahrokh
Shabahang, D.D.S., MS, Ph.D. | |
| 2023 | | |
| 293,502 | | |
| - | | |
| - | | |
| 35,336 | | |
| - | | |
| 30,000 | | |
| 358,837 | |
| Chief
Innovation Officer | |
| 2022 | | |
| 325,000 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 325,000 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Corinne
Pankovcin | |
| 2023 | | |
| 346,774 | | |
| - | | |
| - | | |
| 23,557 | | |
| - | | |
| 20,000 | | |
| 390,331 | |
Chief
Commercialization Officer,
Former President(1), Former Chief Financial Officer(2) | |
| 2022 | | |
| 385,000 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 385,000 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Thomas
J. Farley | |
| 2023 | | |
| 337,894 | | |
| - | | |
| - | | |
| 23,557 | | |
| - | | |
| 20,000 | | |
| 381,451 | |
| Chief
Financial Officer(4) | |
| 2022 | | |
| 360,833 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| | | |
| 360,833 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Matthew
Shatzkes | |
| 2023 | | |
| 198,670 | | |
| 890,893 | | |
| - | | |
| | | |
| - | | |
| 34,076 | | |
| 1,123,639 | |
| Chief
Legal Officer & General Counsel(5) | |
| 2022 | | |
| 368,958 | | |
| 246,697 | | |
| - | | |
| - | | |
| 218,064 | | |
| - | | |
| 833,719 | |
Option
awards represent granted options at the fair market value as of the date of grant. Restricted stock units represent granted restricted
stock units at the fair market value as of the date of grant.
| (1) | In
February 2023, the Company formed a subsidiary, Pearsanta, Inc. in order to accelerate the
growth of the Company’s AditxtScore program through future strategic revenue and growth
oriented transactions. In connection with the formation of Pearsanta and Corinne Pankovcin’s
anticipated role in driving such strategic revenue and growth oriented transactions, Ms.
Pankovcin’ s title was changed from President to Chief Commercialization Officer, effective
April 12, 2023. |
| (2) | Ms.
Pankovcin served as the Company’s Chief Financial Officer from July 2020 through September
25, 2021. She was appointed as our President on September 25, 2021. Ms. Pankovcin’s
title was changed from President to Chief Commercialization Officer effective April 12, 2023. |
| (3) | Mr.
Shatzkes joined Aditxt in January of 2022. Mr. Shatzkes departed Aditxt in July of 2023. |
| | |
| (4) | All
other compensation is inclusive of Pearsanta, Inc. option grants to Mr. Albanna, Dr. Shabahang,
Ms. Pankovcin, and Mr. Farley. Mr. Shatzkes received consideration in connection with the
Separation and General Release agreement. |
Employment
Agreements
Amro
Albanna, Chief Executive Officer
On
November 14, 2021, the Company entered into an Amended and Restated Employment Agreement with Mr. Amro Albanna, the Chief Executive Officer
of the Company (the “Amro Employment Agreement”). Pursuant to the Amro Employment Agreement, Mr. Albanna will receive (i)
a base salary at the annual rate of $280,000 for the remainder of calendar year 2021, and effective January 1, 2022, $500,000 (prorated
for any partial year) payable in bimonthly installments (ii) the opportunity to earn an annual bonus of 2% of the Company’s earnings
before interest, taxes, depreciation, and amortization (EBITDA) with respect to an applicable year for which the bonus is payable, provided
that such bonus will not exceed two (2) times Mr. Albanna’s base salary, and (iii) eligible to earn an annual discretionary bonus
as determined by the Board or its Compensation Committee in their sole discretion. In addition, for calendar year 2021, Mr. Albanna will
be eligible to earn an additional discretionary bonus as determined by the Company.
The
term of Mr. Albanna’s engagement under the Amro Employment Agreement commences as of the Effective Date (as defined in the Amro
Employment Agreement) and continues until November 14, 2023, unless earlier terminated in accordance with the terms of the Amro Employment
Agreement. The term of Mr. Albanna’s Employment Agreement is automatically renewed for successive one (1) year periods until terminated
by Mr. Albanna or the Company.
Under
the Amro Employment Agreement, termination of Mr. Albanna by the Company for “Cause,” “Death,” or “Disability,”
(as such terms are defined in the Amro Employment Agreement), or resignation by Mr. Albanna without “Good Reason” (as defined
in the Amro Employment Agreement), will not require the Company to pay severance to Mr. Albanna. Upon any such termination, Mr. Albanna
will be entitled to receive any Accrued Compensation (as defined in the Amro Employment Agreement), which in the case of termination
by the Company for Cause or resignation by Mr. Albanna for Good Reason will not include payment of pro rata bonus; provided,
however, if termination of Mr. Albanna by the Company without “Cause” or resignation by Mr. Albanna for “Good
Reason,” then under the Amro Employment Agreement will require the Company to pay severance to Mr. Albanna. Upon any such termination,
Mr. Albanna will be entitled to receive any Accrued Compensation and, subject to Mr. Albanna’s execution of an irrevocable release,
receive (i) on the sixtieth day (60th) day following termination, a lump sum amount equal to twelve (12) months base salary then in effect
as of the date of termination, less applicable taxes and withholdings; (ii) provide reimbursement to Mr. Albanna’s medical insurance
premiums for a period of twelve (12) months following the date of termination; and (iii) cause any equity awards granted prior to the
Effective Date (as defined in the Amro Employment Agreement), that are then outstanding and unvested to immediately vest and, with respect
to all options and stock appreciation rights, to become fully exercisable.
Notwithstanding
the foregoing, under the Amro Employment Agreement, termination of Mr. Albanna by the Company without Cause or resignation by Mr. Albanna
for Good Reason and a Change of Control (as defined in the Amro Employment Agreement) of the Company occurs within six (6) months after
such termination, or within twenty-four (24) months prior to such termination, the Company will pay severance to Mr. Albanna in connection
to such termination. Upon such termination, Mr. Albanna will be entitled to receive any Accrued Compensation, and subject to Mr. Albanna’s
execution of an irrevocable release, receive (i) on the sixtieth (60th) day of termination, a lump sum cash-payment equal to the product
of three times Mr. Albanna’s salary then in effect as of the date of termination, less applicable taxes and withholdings; (ii)
provide reimbursement to Mr. Albanna’s medical insurance premiums for a period of twenty-four (24) months following the date of
termination; and (iii) notwithstanding any provision of any stock incentive plan, stock option agreement, realization bonus, restricted
stock agreement or other agreement relating to capital stock of the Company, cause any equity awards granted prior to the that are then
outstanding and unvested to immediately vest and, with respect to all options and stock appreciation rights, to become fully exercisable
for twenty-four (24) months (but not later than when the award would otherwise expire).
The
Amro Employment Agreement also contains customary non-solicitation and non-competition covenants, which covenants remain in effect for
twelve (12) months following any cessation of employment with respect to Mr. Albanna. To the extent any of the payments or benefits provided
for under the Amro Employment Agreement or any other agreement or arrangement between Mr. Albanna and the Company (collectively, the
“Payments”), (a) constitute an “excess parachute payment” within the meaning of Section 280G (“Section
280G”) of the Internal Revenue Code of 1986, as amended and restated (the “Code”), and (b) would otherwise be subject
to the excise tax imposed by Section 4999 of the Code (“Section 4999”), then the Company will pay or provide the greater
(whichever gives Mr. Albanna the highest net after-tax amount) of (i) all of the Payments or (ii) the portion of Payments not in excess
of the greatest amount of Payments that can be paid that would not result in the imposition of the excise tax under Section 4999.
Corinne
Pankovcin, Chief Commercialization Officer
On
November 14, 2021, Aditxt, Inc. (the “Company”) entered into a new employment agreement (the “Pankovcin Employment
Agreement”) with the Company’s President, Corinne Pankovcin, pursuant to which Ms. Pankovcin will continue to serve as the
Company’s President and Secretary until the date upon which Ms. Pankovcin’s employment may be terminated in accordance with
the terms of the Pankovcin Employment Agreement.
The
term of Ms. Pankovcin’s engagement under the Pankovcin Employment Agreement commences as of the Effective Date (as defined in the
Pankovcin Employment Agreement) and continues until November 14, 2023, unless earlier terminated in accordance with the terms of the
Pankovcin Employment Agreement. The term of Ms. Pankovcin’s Employment Agreement is automatically renewed for successive one (1)
year periods until terminated by Ms. Pankovcin or the Company.
Pursuant
to the Pankovcin Employment Agreement, Ms. Pankovcin will receive: (i) a base salary at the annual rate of $250,000 for the remainder
of calendar year 2021, and effective January 1, 2022, $385,000 (prorated for any partial year) payable in bimonthly installments and
(ii) eligible to earn an annual discretionary bonus with a target amount of 45% of Base Compensation, which is based on the achievement
of performance objectives, which will be determined by the Board and Compensation Committee. In addition, for calendar year 2021, Ms.
Pankovcin shall be eligible to earn an additional discretionary bonus as determined by the Company.
Under
the Pankovcin Employment Agreement, termination of Ms. Pankovcin by the Company for “Cause,” “Death,” or “Disability,”
(as such terms are defined in the Pankovcin Employment Agreement), or resignation by Ms. Pankovcin for “Good Reason” (as
defined in the Pankovcin Employment Agreement), will not require the Company to pay severance to Ms. Pankovcin. Upon any such termination,
Ms. Pankovcin will be entitled to receive any Accrued Compensation (as defined in the Pankovcin Employment Agreement), which in the case
of termination by the Company for Cause or resignation by Ms. Pankovcin for Good Reason will not include payment of pro rata bonus; provided,
however, if termination of Ms. Pankovcin by the Company without “Cause” or resignation by Ms. Pankovcin for
“Good Reason,” then under the Pankovcin Employment Agreement will require the Company to pay severance to Ms. Pankovcin.
Upon any such termination, Ms. Pankovcin will be entitled to receive any Accrued Compensation and, subject to Ms. Pankovcin’s execution
of an irrevocable release, receive: (i) on the sixtieth day (60th) day following termination, a lump sum amount equal to twelve (12)
months base salary then in effect as of the date of termination, less applicable taxes and withholdings; (ii) provide reimbursement to
Ms. Pankovcin’s medical insurance premiums for a period of twelve (12) months following the date of termination; and (iii) cause
any equity awards granted prior to the Effective Date (as defined in the Pankovcin Employment Agreement), that are then outstanding and
unvested to immediately vest and, with respect to all options and stock appreciation rights, to become fully exercisable.
Notwithstanding
the foregoing, under the Pankovcin Employment Agreement, termination of Ms. Pankovcin by the Company without Cause or resignation by
Ms. Pankovcin for Good Reason and a Change of Control (as defined in the Pankovcin Employment Agreement) of the Company occurs within
six (6) months after such termination, or within twenty-four (24) months prior to such termination, the Company will pay severance to
Ms. Pankovcin in connection to such termination. Upon such termination, Ms. Pankovcin will be entitled to receive any Accrued Compensation,
and subject to Ms. Pankovcin’s execution of an irrevocable release, receive (i) on the sixtieth (60th) day of termination, a lump
sum cash-payment equal to the sum of (A) the product of two times Ms. Pankovcin’s salary then in effect as of the date of termination,
less applicable taxes and withholdings, and (B) the product of two times Ms. Pankovcin’s Target Bonus; (ii) provide reimbursement
to Ms. Pankovcin’s medical insurance premiums for a period of twenty-four (24) months following the date of termination; and (iii)
notwithstanding any provision of any stock incentive plan, stock option agreement, realization bonus, restricted stock agreement or other
agreement relating to capital stock of the Company, cause any equity awards granted prior to the that are then outstanding and unvested
to immediately vest and, with respect to all options and stock appreciation rights, to become fully exercisable for twenty-four (24)
months (but not later than when the award would otherwise expire).
The
Pankovcin Employment Agreement also contains customary non-solicitation and non-competition covenants, which covenants remain in effect
for twelve (12) months following any cessation of employment with respect to Ms. Pankovcin. To the extent any of the payments or benefits
provided for under the Pankovcin Employment Agreement or any other agreement or arrangement between Ms. Pankovcin and the Company (collectively,
the “Payments”), (a) constitute an “excess parachute payment” within the meaning of Section 280G (“Section
280G”) of the Internal Revenue Code of 1986, as amended and restated (the “Code”), and (b) would otherwise be subject
to the excise tax imposed by Section 4999 of the Code (“Section 4999”), then the Company will pay or provide the greater
(whichever gives Ms. Pankovcin the highest net after-tax amount) of (i) all of the Payments or (ii) the portion of Payments not in excess
of the greatest amount of Payments that can be paid that would not result in the imposition of the excise tax under Section 4999.
Thomas
J. Farley, Chief Financial Officer
On
November 14, 2021, Aditxt, Inc. (the “Company”) entered into a new employment agreement (the “Farley Employment Agreement”)
with the Company’s Chief Financial Officer, Thomas Farley, pursuant to which Mr. Farley will continue to serve as the Company’s
Chief Financial Officer until the date upon which Mr. Farley’s employment may be terminated in accordance with the terms of the
Farley Employment Agreement.
The
term of Mr. Farley’s engagement under the Farley Employment Agreement commences as of the Effective Date (as defined in the Farley
Employment Agreement) and continues until November 14, 2023, unless earlier terminated in accordance with the terms of the Farley Employment
Agreement. The term of Mr. Farley’s Employment Agreement is automatically renewed for successive one (1) year periods until terminated
by Mr. Farley or the Company.
Pursuant
to the Farley Employment Agreement, Mr. Farley will receive: (i) a base salary at the annual rate of $225,000 for the remainder of calendar
year 2021, and effective January 1, 2022, $355,000 (prorated for any partial year) payable in bimonthly installments and, (ii) eligible
to earn an annual discretionary bonus with a target amount of 40% of Base Compensation, which is based on the achievement of performance
objectives, which will be determined by the Board and Compensation Committee. In addition, for calendar year 2021, Mr. Farley will be
eligible to earn an additional discretionary bonus as determined by the Company.
Under
the Farley Employment Agreement, termination of Mr. Farley by the Company for “Cause,” “Death,” or “Disability,”
(as such terms are defined in the Farley Employment Agreement), or resignation by Mr. Farley without “Good Reason” (as defined
in the Farley Employment Agreement), will not require the Company to pay severance to Mr. Farley. Upon any such termination, Mr. Farley
will be entitled to receive any Accrued Compensation (as defined in the Farley Employment Agreement which in the case of termination
by the Company for Cause or resignation by Mr. Farley for Good Reason will not include payment of pro rata bonus; provided,
however, if termination of Mr. Farley by the Company without “Cause” or resignation by Mr. Farley for “Good
Reason,” then under the Farley Employment Agreement will require the Company to pay severance to Mr. Farley. Upon any such termination,
Mr. Farley will be entitled to receive any Accrued Compensation and, subject to Mr. Farley’s execution of an irrevocable release,
receive (i) on the sixtieth day (60th) day following termination, a lump sum cash-payment equal to the sum of (A) the product of two
times Mr. Farley’s salary then in effect as of the date of termination, less applicable taxes and withholdings, and (B) the product
of two times Mr. Farley’s Target Bonus (as defined in the Farley Employment Agreement); (ii) provide reimbursement to Mr. Farley’s
medical insurance premiums for a period of twelve (12) months following the date of termination; and (iii) cause any equity awards granted
prior to the Effective Date (as defined in the Farley Employment Agreement), that are then outstanding and unvested to immediately vest
and, with respect to all options and stock appreciation rights, to become fully exercisable.
Notwithstanding
the foregoing, under the Farley Employment Agreement, termination of Mr. Farley by the Company without Cause or resignation by Mr. Farley
for Good Reason and a Change of Control (as defined in the Farley Employment Agreement) of the Company occurs within six (6) months after
such termination, or within twenty-four (24) months prior to such termination, the Company will pay severance to Mr. Farley in connection
to such termination. Upon such termination, Mr. Farley will be entitled to receive any Accrued Compensation, and subject to Mr. Farley’s
execution of an irrevocable release, receive (i) on the sixtieth (60th) day of termination, a lump sum cash-payment equal to the product
of two times Mr. Farley’s salary then in effect as of the date of termination, less applicable taxes and withholdings; (ii) provide
reimbursement to Mr. Farley’s medical insurance premiums for a period of twelve (12) months following the date of termination;
and (iii) notwithstanding any provision of any stock incentive plan, stock option agreement, realization bonus, restricted stock agreement
or other agreement relating to capital stock of the Company, cause any equity awards granted prior to the that are then outstanding and
unvested to immediately vest and, with respect to all options and stock appreciation rights, to become fully exercisable (but not later
than when the award would otherwise expire).
The
Farley Employment Agreement also contains customary non-solicitation and non-competition covenants, which covenants remain in effect
for twelve (12) months following any cessation of employment with respect to Mr. Farley. To the extent any of the payments or benefits
provided for under the Farley Employment Agreement or any other agreement or arrangement between Mr. Farley and the Company (collectively,
the “Payments”), (a) constitute an “excess parachute payment” within the meaning of Section 280G (“Section
280G”) of the Internal Revenue Code of 1986, as amended and restated (the “Code”), and (b) would otherwise be subject
to the excise tax imposed by Section 4999 of the Code (“Section 4999”), then the Company will pay or provide the greater
(whichever gives Mr. Farley the highest net after-tax amount) of (i) all of the Payments or (ii) the portion of Payments not in excess
of the greatest amount of Payments that can be paid that would not result in the imposition of the excise tax under Section 4999.
Shahrokh
Shabahang, Chief Innovation Officer
On
November 14, 2021, Aditxt, Inc. (the “Company”) entered into a new employment agreement (the “Shabahang Employment
Agreement”) with the Company’s Chief Innovation Officer, Shahrokh Shabahang, pursuant to which Mr. Shabahang will continue
to serve as the Company’s Chief Innovation Officer until the date upon which Mr. Shabahang’s employment may be terminated
in accordance with the terms of the Shabahang Employment Agreement.
The
term of Mr. Shabahang’s engagement under the Shabahang Employment Agreement commences as of the Effective Date (as defined in the
Shabahang Employment Agreement) and continues until November 14, 2023, unless earlier terminated in accordance with the terms of the
Shabahang Employment Agreement. The term of Mr. Shabahang’s Employment Agreement is automatically renewed for successive one (1)
year periods until terminated by Mr. Shabahang or the Company.
Pursuant
to the Shabahang Employment Agreement, Mr. Shabahang will receive: (i) a base salary at the annual rate of $210,000 for the remainder
of calendar year 2021, and effective January 1, 2022, $325,000 (prorated for any partial year) payable in bimonthly installments, and
(ii) eligible to earn an annual discretionary bonus with a target amount of 40% of Base Compensation, which is based on the achievement
of performance objectives, which will be determined by the Board and Compensation Committee. In addition, for calendar year 2021, Mr.
Shabahang will be eligible to earn an additional discretionary bonus as determined by the Company.
Under
the Shabahang Employment Agreement, termination of Mr. Shabahang by the Company for “Cause,” “Death,” or “Disability,”
(as such terms are defined in the Shabahang Employment Agreement), or resignation by Mr. Shabahang without “Good Reason”
(as defined in the Shabahang Employment Agreement), will not require the Company to pay severance to Mr. Shabahang. Upon any such termination,
Mr. Shabahang will be entitled to receive any Accrued Compensation (as defined in the Shabahang Employment Agreement), which in the case
of termination by the Company for Cause or resignation by Mr. Shabahang for Good Reason will not include payment of pro rata bonus; provided,
however, if termination of Mr. Shabahang by the Company without “Cause” or resignation by Mr. Shabahang for
“Good Reason,” then under the Shabahang Employment Agreement will require the Company to pay severance to Mr. Shabahang.
Upon any such termination, Mr. Shabahang will be entitled to receive any Accrued Compensation and, subject to Mr. Shabahang’s execution
of an irrevocable release, receive: (i) on the sixtieth day (60th) day following termination, a lump sum cash-payment equal to the sum
of (A) the product of two times Mr. Shabahangs’s salary then in effect as of the date of termination, less applicable taxes and
withholdings, and (B) the product of two times Mr. Shabahang’s Target Bonus (as defined in the Shabahang Employment Agreement);
(ii) provide reimbursement to Mr. Shabahang’s medical insurance premiums for a period of twelve (12) months following the date
of termination; and (iii) cause any equity awards granted prior to the Effective Date (as defined in the Shabahang Employment Agreement),
that are then outstanding and unvested to immediately vest and, with respect to all options and stock appreciation rights, to become
fully exercisable.
Notwithstanding
the foregoing, under the Shabahang Employment Agreement, termination of Mr. Shabahang by the Company for without Cause or resignation
by Mr. Shabahang for Good Reason and a Change of Control (as defined in the Shabahang Employment Agreement) of the Company occurs within
six (6) months after such termination, or within twenty-four (24) months prior to such termination, the Company will pay severance to
Mr. Shabahang in connection to such termination. Upon such termination, Mr. Shabahang will be entitled to receive any Accrued Compensation,
and subject to Mr. Shabahang’s execution of an irrevocable release, receive: (i) on the sixtieth (60th) day of termination, a lump
sum cash-payment equal to the product of two times Mr. Shabahang’s salary then in effect as of the date of termination, less applicable
taxes and withholdings; (ii) provide reimbursement to Mr. Shabahang’s medical insurance premiums for a period of twenty-four (24)
months following the date of termination; and (iii) notwithstanding any provision of any stock incentive plan, stock option agreement,
realization bonus, restricted stock agreement or other agreement relating to capital stock of the Company, cause any equity awards granted
prior to the that are then outstanding and unvested to immediately vest and, with respect to all options and stock appreciation rights,
to become fully exercisable for twenty-four (24) months (but not later than when the award would otherwise expire).
The
Shabahang Employment Agreement also contains customary non-solicitation and non-competition covenants, which covenants remain in effect
for twelve (12) months following any cessation of employment with respect to Mr. Shabahang. To the extent any of the payments or benefits
provided for under the Shabahang Employment Agreement or any other agreement or arrangement between Mr. Shabahang and the Company (collectively,
the “Payments”), (a) constitute an “excess parachute payment” within the meaning of Section 280G (“Section
280G”) of the Internal Revenue Code of 1986, as amended and restated (the “Code”), and (b) would otherwise be subject
to the excise tax imposed by Section 4999 of the Code (“Section 4999”), then the Company will pay or provide the greater
(whichever gives Mr. Shabahang the highest net after-tax amount) of (i) all of the Payments or (ii) the portion of Payments not in excess
of the greatest amount of Payments that can be paid that would not result in the imposition of the excise tax under Section 4999.
Rowena
Albanna, Chief Operating Officer
On
November 14, 2021, Aditxt, Inc. (the “Company”) entered into a new employment agreement (the “Rowena Employment Agreement”)
with the Company’s Chief Operating Officer, Rowena Albanna, pursuant to which Ms. Albanna will continue to serve as the Company’s
Chief Operating Officer until the date upon which Ms. Albanna’s employment may be terminated in accordance with the terms of the
Rowena Employment Agreement.
The
term of Ms. Albanna’s engagement under the Rowena Employment Agreement commences as of the Effective Date (as defined in the Rowena
Employment Agreement) and continues until November 14, 2023, unless earlier terminated in accordance with the terms of the Rowena Employment
Agreement. The term of Ms. Albanna’s Employment Agreement is automatically renewed for successive one (1) year periods until terminated
by Ms. Albanna or the Company.
Pursuant
to the Rowena Employment Agreement, Ms. Albanna will receive: (i) a base salary at the annual rate of $210,000 for the remainder of calendar
year 2021 and effective January 1, 2022, $325,000 (prorated for any partial year) payable in bimonthly installments, and (ii) eligible
to earn an annual discretionary bonus with a target amount of 40% of Base Compensation, which is based on the achievement of performance
objectives, which will be determined by the Board and Compensation Committee. In addition, for calendar year 2021, Ms. Albanna will be
eligible to earn an additional discretionary bonus as determined by the Company.
Under
the Rowena Employment Agreement, termination of Ms. Albanna by the Company for “Cause,” “Death,” or “Disability,”
(as such terms are defined in the Rowena Employment Agreement), or resignation by Ms. Albanna for “Good Reason” (as defined
in the Rowena Employment Agreement), will not require the Company to pay severance to Ms. Albanna. Upon any such termination, Ms. Albanna
will be entitled to receive any Accrued Compensation (as defined in the Rowena Employment Agreement), which in the case of termination
by the Company for Cause or resignation by Ms. Albanna for Good Reason will not include payment of pro rata bonus; provided,
however, if termination of Ms. Albanna by the Company without “Cause” or resignation by Ms. Albanna for “Good
Reason” (as such terms are defined in the Rowena Employment Agreement), then under the Rowena Employment Agreement will require
the Company to pay severance to Ms. Albanna. Upon any such termination, Ms. Albanna will be entitled to receive any Accrued Compensation
and, subject to Ms. Albanna’s execution of an irrevocable release, receive: (i) on the sixtieth day (60th) day following termination,
a lump sum amount equal to twelve (12) months base salary then in effect as of the date of termination, less applicable taxes and withholdings;
(ii) provide reimbursement to Ms. Albanna’s medical insurance premiums for a period of twelve (12) months following the date of
termination; and (iii) cause any equity awards granted prior to the Effective Date (as defined in the Rowena Employment Agreement), that
are then outstanding and unvested to immediately vest and, with respect to all options and stock appreciation rights, to become fully
exercisable.
Notwithstanding
the foregoing, under the Rowena Employment Agreement, termination of Ms. Albanna by the Company without Cause or resignation by Ms. Albanna
for Good Reason and a Change of Control (as defined in the Rowena Employment Agreement) of the Company occurs within six (6) months after
such termination, or within twenty-four (24) months prior to such termination, the Company will pay severance to Ms. Albanna in connection
to such termination. Upon such termination, Ms. Albanna will be entitled to receive any Accrued Compensation, and subject to Ms. Albanna’s
execution of an irrevocable release, receive: (i) on the sixtieth (60th) day of termination, a lump sum cash-payment equal to the sum
of (A) the product of two times Ms. Albanna’s salary then in effect as of the date of termination, less applicable taxes and withholdings,
and (B) the product of two times Ms. Albanna’s Target Bonus; (ii) provide reimbursement to Ms. Albanna’s medical insurance
premiums for a period of twenty-four (24) months following the date of termination; and (iii) notwithstanding any provision of any stock
incentive plan, stock option agreement, realization bonus, restricted stock agreement or other agreement relating to capital stock of
the Company, cause any equity awards granted prior to the that are then outstanding and unvested to immediately vest and, with respect
to all options and stock appreciation rights, to become fully exercisable for twenty-four (24) months (but not later than when the award
would otherwise expire).
The
Rowena Employment Agreement also contains customary non-solicitation and non-competition covenants, which covenants remain in effect
for twelve (12) months following any cessation of employment with respect to Ms. Albanna. To the extent any of the payments or benefits
provided for under the Rowena Employment Agreement or any other agreement or arrangement between Ms. Albanna and the Company (collectively,
the “Payments”), (a) constitute an “excess parachute payment” within the meaning of Section 280G (“Section
280G”) of the Internal Revenue Code of 1986, as amended and restated (the “Code”), and (b) would otherwise be subject
to the excise tax imposed by Section 4999 of the Code (“Section 4999”), then the Company will pay or provide the greater
(whichever gives Ms. Albanna the highest net after-tax amount) of (i) all of the Payments or (ii) the portion of Payments not in excess
of the greatest amount of Payments that can be paid that would not result in the imposition of the excise tax under Section 4999.
Matthew
Shatzkes, Former Chief Legal Officer and General Counsel
On
January 28, 2022, Aditxt, Inc. (the “Company”) entered into an employment agreement (the “Employment Agreement”)
with Matthew Shatzkes, the Chief Legal Officer and General Counsel of the Company. Pursuant to the Employment Agreement, Mr. Shatzkes
will (i) receive a base salary at the annual rate of $385,000 (the “Base Compensation”) payable in bimonthly installments,
(ii) receive a one-time sign-on bonus (the “Sign-on Bonus”), (iii) a minimum 2022 quarterly bonus (the “Minimum 2022
Bonus”), and (iv) will be entitled to earn an annual discretionary bonus beginning in fiscal year 2022.
Following
the first anniversary of the Employment Agreement (the “Anniversary Date”), in addition to Mr. Shatzkes’ Base Compensation,
Mr. Shatzkes will be entitled to a minimum quarterly bonus (the “Subsequent Year Minimum Bonus”). Following the Anniversary
Date, in addition to Mr. Shatzkes’ Base Compensation and Subsequent Year Minimum Bonus, Mr. Shatzkes will also be eligible to earn
an annual discretionary bonus.
Under
the Employment Agreement, Mr. Shatzkes will also receive (i) a restricted stock unit award that will entitle Mr. Shatzkes to receive
150,000 shares of the Company’s common stock which shall vest immediately, and (ii) a restricted stock unit award of an additional
330,000 shares of the Company’s common stock, which shall vest ratably over eight successive equal quarterly installments over
a two-year period commencing on March 1, 2022 and ending on December 1, 2023.
The
term of Mr. Shatzkes engagement under the Employment Agreement commences on the Effective Date (as defined in the Employment Agreement)
and continues until January 16, 2024, unless earlier terminated in accordance with the terms of the Employment Agreement. The term of
Mr. Shatzkes’ Employment Agreement is automatically renewed for successive one-year periods until terminated by Mr. Shatzkes or
the Company.
Under
the Employment Agreement, termination of Mr. Shatzkes by the Company for “Cause,” “Death,” or “Disability,”
(as such terms are defined in the Employment Agreement), or resignation by Mr. Shatzkes without “Good Reason” (as defined
in the Employment Agreement), will not require the Company to pay severance to Mr. Shatzkes. Upon any such termination, Mr. Shatzkes
will be entitled to receive any Accrued Compensation (as defined in the Employment Agreement), which in the case of termination by the
Company for Cause or resignation by Mr. Shatzkes for Good Reason will not include payment of pro rata bonus. If, however, termination
of Mr. Shatzkes by the Company without “Cause”, resignation by Mr. Shatzkes for “Good Reason” or and a Change
of Control (as defined in the Employment Agreement) event occurs, then the Employment Agreement will require the Company to pay severance
to Mr. Shatzkes. Upon any such termination, Mr. Shatzkes will be entitled to receive any Accrued Compensation and, subject to Mr. Shatzkes’
execution of an irrevocable release, (i) on the sixtieth day following termination, a lump sum amount equal (a) twelve months of his
Base Compensation, Sign-on Bonus and Minimum 2022 Bonus if his Employment Agreement is terminated prior to December 31, 2022, or (b)
his Base Compensation and Subsequent Year Minimum Bonus if his Employment Agreement is terminated after December 31, 2022; (ii) provide
reimbursement to Mr. Shatzkes’ medical insurance premiums for a period of twelve months following the date of termination; and
(iii) notwithstanding any provision of any stock incentive plan, stock option agreement, realization bonus, restricted stock agreement
or other agreement relating to capital stock of the Company, cause any equity awards granted prior to that termination that are then
outstanding and unvested to immediately vest and, with respect to all options and stock appreciation rights, to become fully exercisable.
To
the extent any of the payments or benefits provided for under the Employment Agreement or any other agreement or arrangement between
Mr. Shatzkes and the Company (collectively, the “Payments”), (a) constitute an “excess parachute payment” within
the meaning of Section 280G (“Section 280G”) of the Internal Revenue Code of 1986, as amended and restated (the “Code”),
and (b) would otherwise be subject to the excise tax imposed by Section 4999 of the Code (“Section 4999”), then the Company
will pay or provide the greater (whichever gives Mr. Shatzkes the highest net after-tax amount) of (i) all of the Payments or (ii) the
portion of Payments not in excess of the greatest amount of Payments that can be paid that would not result in the imposition of the
excise tax under Section 4999.
On
July 21, 2023, Matthew Shatzkes tendered his resignation as Chief Legal Officer, General Counsel and Corporate Secretary of the Company.
In connection with his resignation, the Company entered into a Separation Agreement and General Release (the “Separation Agreement”).
Pursuant to the Separation Agreement, Mr. Shatzkes employment with the Company terminated on August 4, 2023 (the “Termination Date”).
In addition, the Company agreed to pay Mr. Shatzkes within seven days after the Termination Date: (i) $122,292.32, representing all accrued
salary and wages (inclusive of Base Compensation and earned Subsequent Quarterly Bonus amounts, as those terms are defined in Mr. Shatzkes
employment agreement), and (ii) $32,575.84, representing Mr. Shatzkes accrued, but unused paid time off. The Company also agreed to pay
Mr. Shatzkes: (i) $385,000, representing 12 months of Mr. Shatzkes Base Compensation (as that term is defined in Mr. Shatzkes employment
agreement), and (ii) $290,000, representing Mr. Shatzkes Subsequent Year Minimum Bonus (as such term is defined in Mr. Shatzkes employment
agreement), on the 60th day following the Termination Date. In addition, the Company shall reimburse Mr. Shatzkes COBRA premium for a
period of 12 months and shall cause any restricted stock units granted to Mr. Shatzkes to immediately vest as of the Termination Date.
On
August 15, 2023, the Company entered into an Amendment to Separation Agreement and General Release with Mr. Shatzkes (the “Separation
Agreement Amendment”). Pursuant to the Separation Agreement Amendment, the Company was required to pay Mr. Shatzkes, upon the earlier
of (i) September 1, 2023 or (ii) two business days following the closing of a capital raise by the Company, an amount equal to $91,060.16,
which amount represents the balance of Mr. Shatzkes’ Accrued Salary and Wages and Accrued PTO plus an additional $1,000 to serve
as consideration for entering into the Separation Agreement Amendment. In addition, under the Separation Agreement Amendment, the Company
was required to pay Mr. Shatzkes the Severance Base Compensation and the Severance Bonus upon the earlier of (i) the 60th day
following the Termination Date or (ii) two business days following the closing of a capital raise by the Company.
CERTAIN
RELATIONSHIPS AND RELATED TRANSACTIONS AND DIRECTOR INDEPENDENCE
Except
as described below and except for employment arrangements which are described under “executive compensation,” since January 1,
2018, there has not been, nor is there currently proposed, any transaction in which we are or were a participant, the amount involved
exceeds the lesser of $120,000 or 1% of the average of the total assets at December 31, 2022 and 2021, and any of our directors,
executive officers, holders of more than 5% of our Common Stock or any immediate family member of any of the foregoing had or will have
a direct or indirect material interest.
On
February 7, 2024, Amro Albanna, the Chief Executive Officer of the Company loaned $30,000 to the Company. The loan was evidenced by an
unsecured promissory note (the “February Note”). Pursuant to the terms of the February Note, it will accrue interest at the
Prime rate of eight and one-half percent (8.5%) per annum and is due on the earlier of August 7, 2024 or an event of default, as defined
therein.
On
December 6, 2023, Amro Albanna, the Chief Executive Officer of the Company loaned $200,000 to the Company. The loan was evidenced by
an unsecured promissory note (the “December Note”). Pursuant to the terms of the December Note, it will accrue interest at
the Prime rate of eight and one-half percent (8.5%) per annum and is due on the earlier of June 6, 2024 or an event of default, as defined
therein.
On
November 30, 2023, Amro Albanna, the Chief Executive Officer of the Company loaned $10,000 to the Company. The loan was evidenced by
an unsecured promissory note (the “November Note”). Pursuant to the terms of the November Note, it will accrue interest at
the Prime rate of eight and one-half percent (8.5%) per annum and is due on the earlier of May 30, 2024 or an event of default, as defined
therein.
On
June 12, 2023, Amro Albanna, the Chief Executive Officer of the Company and Shahrokh Shabahang, the Chief Innovation Officer of the Company,
loaned $200,000 and $100,000, respectively, to the Company. The loans were evidenced by an unsecured promissory note (the “June
Notes”). Pursuant to the terms of the June Notes, each of the June Notes will accrue interest at the Prime rate of eight and one-quarter
percent (8.25%) per annum and is due on the earlier of December 12, 2023 or an event of default, as defined therein.
On
April 21, 2023, Amro Albanna, the Chief Executive Officer of the Company, and Shahrokh Shabahang, the Chief Innovation Officer of the
Company, loaned $87,523 and $100,000, respectively, to the Company. The loans were each evidenced by an unsecured promissory note
(the “April Note”). Pursuant to the terms each April Note, it will accrue interest at the Prime rate of eight percent (8.00%)
per annum and is due on the earlier of October 21, 2023, or an event of default, as defined therein. As of September 30, 2023, the note
was fully paid off.
On
May 25, 2023, Amro Albanna, the Chief Executive Officer of the Company, loaned $200,000 to the Company. The loan was evidenced by
an unsecured promissory note (the “May Note”). Pursuant to the terms of the May Note, it will accrue interest at a rate of
eight and one-quarter percent (8.25%) per annum, the Prime rate on the date of signing, and is due on the earlier of November 25, 2023
or an event of default, as defined therein. As of September 30, 2023, the note was fully paid off.
On
June 12, 2023, Amro Albanna, the Chief Executive Officer of the Company, and Shahrokh Shabahang, the Chief Innovation Officer of the
Company, loaned $200,000 and $100,000, respectively, to the Company. The loans were evidenced by an unsecured promissory note (the
“June Note”). Pursuant to the terms of the June Note, it will accrue interest at the Prime rate of eight and one-quarter
percent (8.25%) per annum and is due on the earlier of December 12, 2023, or an event of default, as defined therein. As of September
30, 2023, the June Note was fully paid off.
On
July 11, 2023, we entered into a Subscription and Investment Representation Agreement (the “Subscription Agreement”) with
Amro Albanna, its Chief Executive Officer, who is an accredited investor (the “Purchaser”), pursuant to which the Company
agreed to issue and sell one (1) share of the Company’s Series C Preferred Stock, par value $0.001 per share (the “Preferred
Stock”), to the Purchaser for $1,000.00 in cash. The sale closed on July 11, 2023.
On
July 19, 2022, we entered into a Subscription and Investment Representation Agreement (the “Subscription Agreement”) with
Amro Albanna, its Chief Executive Officer, who is an accredited investor (the “Purchaser”), pursuant to which the Company
agreed to issue and sell one (1) share of the Company’s Series B Preferred Stock, par value $0.001 per share (the “Preferred
Stock”), to the Purchaser for $20,000.00 in cash. The sale closed on July 19, 2022. The one share of Series B Preferred Stock was
redeemed by the Company on Pctober 7, 2022 for $20,000 following the approval of the 2022 reverse stock split.
During
the years ended December 31, 2019 and 2018, Rowena Albanna, the wife of Amro Albanna, our Chief Executive Officer, provided
the Company with operations consulting services. In July 2020, Ms. Albanna joined the Company as its Chief Operating Officer. As
of December 31, 2018, $112,000 was accrued as compensation. An additional $180,000 was expensed as compensation during the year
ended December 31, 2019, and $17,000 was paid on the accrued balance. As of December 31, 2019, $275,000 remained accrued and
outstanding.
On
January 22, 2018, the Company issued an unsecured promissory note to Sekris for $40,000 that accrued interest of 4% annually. The
note was due on the earlier of July 22, 2018 or in the event of default, as defined in the agreement. This note has been repaid
as of December 31, 2019.
On
February 12, 2018, the Company issued an unsecured promissory note to Sekris for $50,000 that accrued interest of 4% annually. The
note was due on the earlier of August 12, 2018 or in the event of default, as defined in the agreement. This note has been repaid
as of December 31, 2019.
On
March 2, 2018, the Company issued an unsecured promissory note to Sekris for $10,000 that accrued interest of 4% annually. The note
was due on the earlier of September 2, 2018 or in the event of default, as defined in the agreement. This note has been repaid as
of December 31, 2019.
On
March 8, 2018, we entered into an Assignment Agreement (the “Assignment Agreement”) with Sekris. See “Summary — Overview — License
Agreement with Loma Linda University.” Dr. Shabahang, our Chief Innovative Officer, was the Chief Executive Officer of Sekris.
Sekris was subsequently dissolved in 2019.
On
March 8, 2018, we issued a warrant to purchase up to 10,000 shares of our Common Stock to Sekris. On March 2, 2018, we
issued a 4% unsecured promissory note to Sekris in the principal amount of $10,000. Principal and interest was due on September 2,
2018 or immediately upon an event of default. On February 12, 2018, we issued a 4% unsecured promissory note to Sekris in the principal
amount of $50,000. Principal and interest was due on August 12, 2018 or immediately upon an event of default. On January 22,
2018, we issued a 4% unsecured promissory note to Sekris in the principal amount of $40,000. Principal and interest was due on July 22,
2018 or immediately upon an event of default.
On
June 18, 2018, the Company issued an unsecured promissory note to Sekris for $17,502 that accrued interest of 4% annually. The note
was due on the earlier of December 18, 2018 or in the event of default, as defined in the agreement. This note has been repaid as
of December 31, 2019.
On
January 1, 2019, we entered into a consulting agreement with Rowena Albanna, the wife of Amro Albanna, our Chief Executive Officer,
to perform operations consulting services. As part of this agreement, we pay Ms. Albanna $15,000 per month for her services. This agreement
terminated on June 30, 2020. In July 2020, Ms. Albanna joined the Company as its Chief Operating Officer.
On
March 21, 2019, we issued a promissory note to Dr. Shabahang, our Chief Innovative Officer. The note has a principal amount
of $10,000, was due on September 21, 2019, and bears an interest rate of 4% per year. This note remains outstanding.
During
the year ended December 31, 2019, we assumed an aggregate of $189,625 of liabilities from Sekris in exchange for the return of 94,813 shares
of our Common Stock.
On
January 20, 2020, we issued a promissory note to Brian Brady, a member of our board of directors. The note has a principal amount
of $50,000, was due on the earlier of April 19, 2020 or within 10 days of the closing of our initial public offering. This
note carried an original issue discount of $25,000. The note was amended on April 23, 2020 to extend the maturity date to the earlier
of June 30, 2020 or within 10 days of the closing of our initial public offering. This note was repaid in July 2020.
Review,
Approval and Ratification of Related Party Transactions
Given
our small size and limited financial resources, we have not adopted formal policies and procedures for the review, approval or ratification
of transactions, such as those described above, with our executive officer(s), Director(s) and significant stockholders. We intend to
establish formal policies and procedures in the future, once we have sufficient resources and have appointed additional Directors, so
that such transactions will be subject to the review, approval or ratification of our Board of Directors, or an appropriate committee
thereof. On a moving forward basis, our Directors will continue to approve any related party transaction.
SECURITY
OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND
MANAGEMENT AND RELATED STOCKHOLDER MATTERS
The following table sets
forth certain information regarding beneficial ownership of shares of our common stock as of June 4, 2024 based on 1,993,682 shares issued
and outstanding by (i) each person known to beneficially own more than 5% of our outstanding common stock, (ii) each of our directors,
(iii) our executive officers and (iv) all directors and executive officers as a group. Shares are beneficially owned when an individual
has voting and/or investment power over the shares or could obtain voting and/or investment power over the shares within 60 days of June
4, 2024. Except as otherwise indicated, the persons named in the table have sole voting and investment power with respect to all shares
beneficially owned, subject to community property laws, where applicable. Unless otherwise indicated, the address of each beneficial
owner listed below is c/o Aditxt, Inc., 2569 Wyandotte St., Suite 101, Mountain View, CA 94043.
| | |
Number
of
shares of
Common
Stock
Beneficially
Owned | | |
Percentage | |
| Directors and Officers: | |
| | |
| |
| Amro Albanna (1) | |
| 10,103 | | |
| * | % |
| Shahrokh Shabahang, D.D.S., MS, Ph.D. (2) | |
| 7,780 | | |
| * | % |
| Corinne Pankovcin (3) | |
| 4,966 | | |
| * | % |
| Rowena Albanna (4) | |
| 4,956 | | |
| * | % |
| Brian Brady (5) | |
| 488 | | |
| * | % |
| Jeffrey Runge, M.D. (6) | |
| 483 | | |
| * | % |
| Thomas J. Farley (7) | |
| 4,812 | | |
| * | % |
| Charles Nelson (8) | |
| 731 | | |
| * | % |
| All directors and executive
officers as a group (9 persons) | |
| 34,319 | | |
| 1.7 | % |
| Sabby Volatility Warrant Master Fund, Ltd. (9) | |
| 168,734 | | |
| 8.5 | % |
| (1) | Includes
(i) 9,704 shares issuable pursuant to options that are fully vested; (ii) 228 shares beneficially
owned by the Albanna Family Trust, of which Mr. Albanna is the Trustee; (iii) 151 shares
directly owned by Mr. Albanna; and (iv) 20 Series A Warrants issued as part of the conversion
of outstanding accrued compensation through March 31, 2020. Mr. Albanna may be deemed to
beneficially own the securities held by his wife Rowena Albanna, the Company’s Chief
Operating Officer. |
| (2) | Includes
(i) 7,108 beneficially owned by Shabahang-Hatami Family Trust, of which Shahrokh Shabahang,
D.D.S., MS, Ph.D. is the Trustee; (ii) warrants to purchase 111 shares, including 24 Series
A Warrants issued as part of the conversion of outstanding accrued compensation through March
31, 2020, and 87 warrants beneficially owned by the Shabahang-Hatami Family Trust; (iii)
561 shares directly owned by Mr. Shabahang. |
| (3) | Includes
(i) 86 shares held directly by Ms. Pankovcin; and (ii) 4,880 shares issuable pursuant to
options that are fully vested. |
| (4) | Includes
(i) 86 shares held directly by Ms. Albanna; (ii) 4,852 shares issuable pursuant to options
that are fully vested; and (iii) 18 Series A Warrants issued as part of the conversion of
outstanding accrued compensation through March 31, 2020. Ms. Albanna may be deemed to beneficially
own the securities held by her husband Amro Albanna, the Company’s Chief Executive
Officer. |
| (5) | Includes
(i) 13 shares held directly by Mr. Brady; and (ii) 475 shares issuable pursuant to options
that are fully vested. |
| (6) | Includes
(i) 2 shares held by Biologue, Inc., over which Dr. Runge has voting and dispositive control;
(ii) 6 shares held directly by Dr. Runge; and (iii) 475 shares issuable pursuant to options
that are fully vested. |
| (7) | Includes
(i) 80 shares held directly by Mr. Farley and (ii) 4,732 shares issuable pursuant to options
that are fully vested. |
| (8) | Includes
(i) 261 shares held by Siu Kim Athle International, LLC., over which Mr. Nelson has voting
and dispositive control and (ii) 470 shares issuable pursuant to options that are fully vested. |
| (9) |
Sabby Management, LLC is the investment manager of Sabby Volatility Warrant Master Fund, Ltd. (“Sabby”) and shares voting and investment power with respect to these shares in this capacity. As manager of Sabby Management, LLC, Hal Mintz also shares voting and investment power on behalf of Sabby. Each of Sabby Management, LLC and Mr. Mintz disclaims beneficial ownership over the securities listed except to the extent of their pecuniary interest therein. |
DESCRIPTION
OF CAPITAL STOCK
General
The
following description of the Company’s capital stock and provisions of its Amended and Restated Certificate of Incorporation and
Amended and Restated Bylaws are summaries and are qualified by reference to the full text of the Company’s Amended and Restated
Certificate of Incorporation and Amended and Restated Bylaws.
Description
of Common Stock
The
following description of our Common Stock is a summary and does not purport to be complete. It is subject to and qualified
in its entirety by reference to our Amended and Restated Certificate of Incorporation, as amended (the “Certificate of Incorporation”)
and our Amended and Restated Bylaws (the “Bylaws”), each of which are incorporated by reference as an exhibit to our Annual
Report on Form 10-K. We encourage you to read our Certificate of Incorporation, Bylaws, and the applicable provisions of
the Delaware General Corporation Law for additional information.
Authorized
Capital Shares
Our authorized capital shares
consist of 100,000,000 shares of Common Stock, $0.001 par value per share, and 3,000,000 shares of preferred stock, $0.001 par value
per share. As of December 31, 2023, there were 1,665,214 shares of Common Stock issued and 1,665,214 shares of Common Stock outstanding.
As of December 31, 2023, there were 24,905 shares of preferred stock issued and outstanding, of which (i) 22,280 were designated as Series
A-1 Convertible Preferred Stock, and (ii) 2,625 were designated as Series B-2 Convertible Preferred Stock.
Voting
Rights
Holders
of Common Stock are entitled to one vote per share on all matters voted on by the stockholders, including the election of directors.
Our Certificate of Incorporation and Bylaws do not provide for cumulative voting in the election of directors.
Dividend
Rights
Holders
of the Company’s Common Stock are entitled to receive dividends, if any, as may be declared from time to time by the board of directors
in its discretion out of funds legally available for the payment of dividends.
Liquidation
Rights
In
the event of our liquidation, the holders of our Common Stock will be entitled to share ratably in any distribution of our assets after
payment of all debts and other liabilities and the preferences payable to holders of shares of Preferred Stock then outstanding, if any.
Applicable
Anti-Takeover Law
Set
forth below is a summary of the provisions of the Certificate of Incorporation and the Bylaws that could have the effect of delaying
or preventing a change in control of the Company. The following description is only a summary and it is qualified by refence to the Certificate
of Incorporation, the Bylaws and relevant provisions of the Delaware General Corporation Law.
Blank
Check Preferred Stock
The
Certificate of Incorporation authorizes 3,000,000 undesignated shares of Preferred Stock and permits our board of directors to issue
Preferred Stock with rights or preferences that could impede the success of any attempt to change control of the Company. For example,
our board of directors, without stockholder approval, may create or issue Preferred Stock with conversion rights that could adversely
affect the voting power of the holders of our Common Stock as well as rights to such Preferred Stock, in connection with implementing
a stockholder rights plan. This provision may be deemed to have a potential anti-takeover effect, because the issuance of such Preferred
Stock may delay or prevent a change of control of the Company. Furthermore, shares of Preferred Stock, if any are issued, may have other
rights, including economic rights, senior to Common Stock, and as a result, the issuance thereof could depress the market price of our
Common Stock.
Series
A-1 Convertible Preferred Stock
On
December 22, 2023, we filed a Certificate of Designation for its Series A-1 Preferred Stock with the Secretary of State of Delaware (the
“Series A-1 Certificate of Designations”). The following is only a summary of the Series A-1 Certificate of Designations,
and is qualified in its entirety by reference to the full text of the Series A-1 Certificate of Designations, a copy of which is filed
as Exhibit 3.1 to our Current Report on Form 8-K filed on December 26, 2023 and is incorporated by reference herein.
Designation,
Amount, and Par Value. The number of Series A-1 Preferred Stock designated is 22,280 shares. The shares of Series A-1 Preferred
Stock have a par value of $0.001 per share and a stated value of $1,000 per share.
Conversion
Price. The Series A-1 Preferred Stock will be convertible into shares of Common Stock at an initial conversion price of $4.44
(subject to adjustment pursuant to the Series A-1 Certificate of Designations) (the “Conversion Price”). The
Certificate of Designations also provides that in the event of certain Triggering Events (as defined below) any holder may, at any time,
convert any or all of such holder’s Series A-1 Preferred Stock at an alternate conversion rate equal to the product of (i) the
Alternate Conversion Price (as defined below) and (ii) the quotient of (x) the 25% redemption premium multiplied by (y) the amount of
Series A-1 Preferred Stock subject to such conversion. “Triggering Events” include, among others, (i) a suspension of trading
or the failure to be traded or listed on an eligible market for five consecutive days or more, (ii) the failure to remove restrictive
legends when required, (iii) the Company’s default in payment of indebtedness in an aggregate amount of $500,000 or more, (iv)
proceedings for a bankruptcy, insolvency, reorganization or liquidation, which are not dismissed with 30 days, (v) commencement of a
voluntary bankruptcy proceeding, and (viii) final judgments against the Company for the payment of money in excess of $100,000. “Alternate
Conversion Price” means the lowest of (i) the applicable conversion price the in effect, (ii) the greater of (x) $0.888 (the “Floor
Price”) and (y) 80% of the volume weighted average price (“VWAP”) of the Common Stock on the trading day immediately
preceding the delivery of the applicable conversion notice. Further, the Series A-1 Certificate of Designations provides that if on any
of the 90th and 180th day after each of the occurrence of any Stock Combination Event (as defined in the Series A-1 Certificate
of Designations) and the Applicable Date (as defined in the Series A-1 Certificate of Designations), the conversion price then in effect
is greater than the market price then in effect (the “Adjustment Price”), on such date then the conversion price shall automatically
lower to the Adjustment Price.
Dividends.
Holders of the Series A-1 Preferred Stock shall be entitled to receive dividends when and as declared by the Board, from time
to time, in its sole discretion, which Dividends shall be paid by the Company out of funds legally available therefor, payable, subject
to the conditions and other terms hereof, in cash, in securities of the Company or any other entity, or using assets as determined by
the Board on the Stated Value of such Preferred Share.
Liquidation. In
the event of a Liquidation Event (as defined in the Series A-1 Certificate of Designation), the holders the Series A-1 Preferred Stock
shall be entitled to receive in cash out of the assets of the Company, before any amount shall be paid to the holders of any other shares
of capital stock of the Company, equal to the greater of (A) 125% of the Conversion Amount (as defined in the Series A-1 Certificate
of Designation) on the date of such payment and (B) the amount per share such holder of Series A-1 Preferred Stock would receive if they
converted such share of Series A-1 Preferred Stock into Common Stock immediately prior to the date of such payment
Company
Redemption. The Company may redeem all, or any portion, of the Series A-1 Preferred Stock for cash, at a price per share
of Series A-1 Preferred Stock equal to 115% of the greater of (i) the Conversion Amount (as defined in the Series A-1 Certificate of
Designation)being redeemed as of the Company Optional Redemption Date (as defined in the Series A-1 Certificate of Designation) and (ii)
the product of (1) the Conversion Rate (as defined in the Series A-1 Certificate of Designation) with respect to the Conversion Amount
being redeemed as of the Company Optional Redemption Date multiplied by (2) the greatest Closing Sale Price (as defined in the Certificate
of Designation) of the Common Stock on any Trading Day during the period commencing on the date immediately preceding such Company Optional
Redemption Notice Date (as defined in the Certificate of Designation) and ending on the Trading Day immediately prior to the date the
Company makes the entire payment required to be made under the Certification of Designation.
Maximum
Percentage. Holders of Series A-1 Preferred Stock are prohibited from converting
shares of Series A-1 Preferred Stock into shares of Common Stock if, as a result of such
conversion, such holder, together with its affiliates, would beneficially own in excess of
4.99% (the “Maximum Percentage”) of the total number of shares of Common
Stock issued and outstanding immediately after giving effect to such conversion.
Voting
Rights. The holders of the Series A-1 Preferred Stock shall have no voting power and no right to vote on any matter at any
time, either as a separate series or class or together with any other series or class of share of capital stock, and shall not be entitled
to call a meeting of such holders for any purpose nor shall they be entitled to participate in any meeting of the holders of Common Stock,
except as expressly provided in the Certificate of Designations and where required by the DGCL.
Series B-1 Convertible Preferred Stock
On
January 24, 2024, we filed a Certificate of Designations for its Series B-1 Preferred Stock with the Secretary of State of Delaware (the
“Series B-1 Certificate of Designations”). The following is only a summary of
the Series B-1 Certificate of Designations, and is qualified in its entirety by reference to the full text of the Series B-1 Certificate
of Designations, a copy of which is filed as Exhibit 3.1 to this Current Report on Form 8-K and is incorporated by reference herein.
Designation,
Amount, and Par Value. The number of Series B-1 Preferred Stock designated is 6,000 shares. The shares of Series B-1 Preferred
Stock have a par value of $0.001 per share and a stated value of $1,000 per share.
Conversion
Price. The Series B-1 Preferred Stock will be convertible into shares of Common Stock at an initial conversion price of $4.06
(subject to adjustment pursuant to the Series B-1 Certificate of Designations) (the “Conversion Price”). The
Series B-1 Certificate of Designations also provides that in the event of certain Triggering Events (as defined below) any holder may,
at any time, convert any or all of such holder’s Series B-1 Preferred Stock
at an alternate conversion rate equal to the product of (i) the Alternate Conversion Price (as defined below) and (ii) the quotient of
(x) the 125% redemption premium multiplied by (y) the amount of Series B-1 Preferred
Stock subject to such conversion. “Triggering Events” include, among others, (i) a suspension of trading or the failure to
be traded or listed on an eligible market for five consecutive days or more, (ii) the failure to remove restrictive legends when required,
(iii) the Company’s default in payment of indebtedness in an aggregate amount of $500,000 or more, (iv) proceedings for a bankruptcy,
insolvency, reorganization or liquidation, which are not dismissed with 30 days, (v) commencement of a voluntary bankruptcy proceeding,
and (viii) final judgments against the Company for the payment of money in excess of $500,000. “Alternate
Conversion Price” means the lowest of (i) the applicable conversion price the in effect, (ii) the greater of (x) $0.9420
(the “Floor Price”) and (y) 80% of the lowest volume weighted average price (“VWAP”)
of the Common Stock during the five consecutive trading day period ending and including the trading day immediately preceding the delivery
of the applicable conversion notice. Further, the Series B-1 Certificate of Designations
provides that if on any of the 90th and 180th day after each of the occurrence of any Stock Combination
Event (as defined in the Series B-1 Certificate of Designations) and the Applicable
Date (as defined in the Series B-1 Certificate of Designations), the conversion
price then in effect is greater than the market price then in effect (the “Adjustment Price”), on such date then the
conversion price shall automatically lower to the Adjustment Price.
Dividends.
Holders of the Series B-1 Preferred Stock shall be entitled to receive dividends when and as declared by the Board, from time to time,
in its sole discretion, which Dividends shall be paid by the Company out of funds legally available therefor, payable, subject to the
conditions and other terms hereof, in cash, in securities of the Company or any other entity, or using assets as determined by the Board
on the Stated Value of such Preferred Share.
Liquidation. In
the event of a Liquidation Event (as defined in the Series B-1 Certificate of Designations), the holders the Series B-1 Preferred Stock
shall be entitled to receive in cash out of the assets of the Company, before any amount shall be paid to the holders of any other shares
of capital stock of the Company, equal to the greater of (A) 125% of the Conversion Amount (as defined in the Series B-1 Certificate
of Designation) on the date of such payment and (B) the amount per share such holder of Series B-1 Preferred Stock would receive if they
converted such share of Series B-1 Preferred Stock into Common Stock immediately prior to the date of such payment.
Company
Redemption. The Company may redeem all, or any portion, of the Series B-1 Preferred Stock for cash, at a price per share
of Series B-1 Preferred Stock equal to 115% of the greater of (i) the Conversion Amount (as defined in the Series B-1 Certificate of
Designations) being redeemed as of the Company Optional Redemption Date (as defined in the Series B-1 Certificate of Designations) and
(ii) the product of (1) the Conversion Rate (as defined in the Series B-1 Certificate of Designations) with respect to the Conversion
Amount being redeemed as of the Company Optional Redemption Date multiplied by (2) the greatest Closing Sale Price (as defined in the
Series B-1 Certificate of Designations) of the Common Stock on any Trading Day during the period commencing on the date immediately preceding
such Company Optional Redemption Notice Date (as defined in the Series B-1 Certificate of Designations) and ending on the Trading Day
immediately prior to the date the Company makes the entire payment required to be made under the Certification of Designation.
Maximum
Percentage. Holders of Series B-1 Preferred Stock are prohibited from converting shares of Series B-1 Preferred Stock into shares
of Common Stock if, as a result of such conversion, such holder, together with its affiliates, would beneficially own in excess of 4.99%
(the “Maximum Percentage”) of the total number of shares of Common Stock issued
and outstanding immediately after giving effect to such conversion.
Voting
Rights. The holders of the Series B-1 Preferred Stock shall have no voting power and no right to vote on any matter at any
time, either as a separate series or class or together with any other series or class of share of capital stock, and shall not be entitled
to call a meeting of such holders for any purpose nor shall they be entitled to participate in any meeting of the holders of Common Stock,
except as expressly provided in the Series B-1 Certificate of Designations and where required by the DGCL.
Series
B-2 Convertible Preferred Stock
On
December 28, 2023, we filed a Certificate of Designations for its Series B-2 Preferred Stock with the Secretary of State of Delaware
(the “Series B-2 Certificate of Designations”). The following is only a summary of the Series B-2 Certificate of Designations,
and is qualified in its entirety by reference to the full text of the Series B-2 Certificate of Designations, a copy of which is filed
as an exhibit to our Current Report on Form 8-K filed with the SEC on January 2, 2024.
Designation,
Amount, and Par Value. The number of Series B-2 Preferred Stock designated is 2,625 shares. The shares of Series B-2 Preferred
Stock have a par value of $0.001 per share and a stated value of $1,000 per share.
Conversion
Price. The Series B-2 Preferred Stock will be convertible into shares of Common Stock at an initial conversion price of $4.71
(subject to adjustment pursuant to the Series B-2 Certificate of Designations) (the “Conversion Price”). The
Series B-2 Certificate of Designations also provides that in the event of certain Triggering Events (as defined below) any holder may,
at any time, convert any or all of such holder’s Series B-2 Preferred Stock
at an alternate conversion rate equal to the product of (i) the Alternate Conversion Price (as defined below) and (ii) the quotient of
(x) the 125% redemption premium multiplied by (y) the amount of Series B-2 Preferred
Stock subject to such conversion. “Triggering Events” include, among others, (i) a suspension of trading or the failure to
be traded or listed on an eligible market for five consecutive days or more, (ii) the failure to remove restrictive legends when required,
(iii) the Company’s default in payment of indebtedness in an aggregate amount of $500,000 or more, (iv) proceedings for a bankruptcy,
insolvency, reorganization or liquidation, which are not dismissed with 30 days, (v) commencement of a voluntary bankruptcy proceeding,
and (viii) final judgments against the Company for the payment of money in excess of $500,000. “Alternate Conversion Price”
means the lowest of (i) the applicable conversion price the in effect, (ii) the greater of (x) $0.9420 (the “Floor Price”)
and (y) 80% of the lowest volume weighted average price (“VWAP”) of the Common Stock during the five consecutive trading
day period ending and including the the trading day immediately preceding the delivery of the applicable conversion notice. Further,
the Series B-2 Certificate of Designations provides that if on any of the 90th
and 180th day after each of the occurrence of any Stock Combination Event (as defined in the Series B-2 Certificate
of Designations) and the Applicable Date (as defined in the Series B-2 Certificate
of Designations), the conversion price then in effect is greater than the market price then in effect (the “Adjustment Price”),
on such date then the conversion price shall automatically lower to the Adjustment Price.
Dividends.
Holders of the Series B-2 Preferred Stock shall be entitled to receive dividends when and as declared by the Board, from time
to time, in its sole discretion, which Dividends shall be paid by the Company out of funds legally available therefor, payable, subject
to the conditions and other terms hereof, in cash, in securities of the Company or any other entity, or using assets as determined by
the Board on the Stated Value of such Preferred Share.
Liquidation. In
the event of a Liquidation Event (as defined in the Series B-2 Certificate of Designations), the holders the Series B-2 Preferred Stock
shall be entitled to receive in cash out of the assets of the Company, before any amount shall be paid to the holders of any other shares
of capital stock of the Company, equal to the greater of (A) 125% of the Conversion Amount (as defined in the Series B-2 Certificate
of Designation) on the date of such payment and (B) the amount per share such holder of Series B-2 Preferred Stock would receive if they
converted such share of Series B-2 Preferred Stock into Common Stock immediately prior to the date of such payment.
Company
Redemption. The Company may redeem all, or any portion, of the Series B-2 Preferred
Stock for cash, at a price per share of Series B-2 Preferred Stock equal to 115% of the greater
of (i) the Conversion Amount (as defined in the Series B-2 Certificate of Designations) being
redeemed as of the Company Optional Redemption Date (as defined in the Series B-2 Certificate
of Designations) and (ii) the product of (1) the Conversion Rate (as defined in the Series
B-2 Certificate of Designations) with respect to the Conversion Amount being redeemed as
of the Company Optional Redemption Date multiplied by (2) the greatest Closing Sale Price
(as defined in the Series B-2 Certificate of Designations) of the Common Stock on any Trading
Day during the period commencing on the date immediately preceding such Company Optional
Redemption Notice Date (as defined in the Series B-2 Certificate of Designations) and ending
on the Trading Day immediately prior to the date the Company makes the entire payment required
to be made under the Certification of Designation.
Maximum
Percentage. Holders of Series B-2 Preferred Stock are prohibited from converting shares of Series B-2 Preferred Stock into shares
of Common Stock if, as a result of such conversion, such holder, together with its affiliates, would beneficially own in excess of 4.99%
(the “Maximum Percentage”) of the total number of shares of Common Stock issued and outstanding immediately after giving
effect to such conversion.
Voting
Rights. The holders of the Series B-2 Preferred Stock shall have no voting power and no right to vote on any matter at any
time, either as a separate series or class or together with any other series or class of share of capital stock, and shall not be entitled
to call a meeting of such holders for any purpose nor shall they be entitled to participate in any meeting of the holders of Common Stock,
except as expressly provided in the Series B-2 Certificate of Designations and where required by the DGCL.
No
Cumulative Voting
The
Certificate of Incorporation and the Bylaws do not provide holders of our Common Stock cumulative voting rights in the election of directors.
The absence of cumulative voting could have the effect of preventing stockholders holding a minority of our shares of Common Stock from
obtaining representation on our board of directors. The absence of cumulative voting might also, under certain circumstances, render
more difficult or discourage a merger, tender offer or proxy contest favored by a majority of our stockholders, the assumption of control
by a holder of a large block of our stock or the removal of incumbent management.
Advance
Notice Requirements for Stockholder Proposals and Director Nominees
The
Bylaws require stockholders seeking to make nominations of candidates for election as directors or to bring other business before a meeting
of our stockholders to provide timely notice of their intent in writing. To be timely, a stockholder’s notice must be delivered
to the Secretary at our principal executive offices not less than 90 days nor more than 120 days prior to the first anniversary of the
immediately preceding annual meeting of the stockholders; provided, however, that in the event that the date of the annual meeting is
more than 30 days before or after such anniversary date, notice by the stockholder to be timely must be so received not later than the
close of business on the tenth day following the earlier of the date on which we first give notice or publicly announce the date of the
meeting. A stockholder’s notice must include certain information about the stockholder and the nominee or proposal as specified
in the Bylaws. These advance notice provisions may restrict the ability of the stockholders to make nominations for directors at or bring
business before a meeting of the Company’s stockholders.
Series C-1 Convertible Preferred Stock
On May 2, 2024, we filed
a Certificate of Designation for its Series C-1 Preferred Stock with the Secretary of State of Delaware (the “Series
C-1 Certificate of Designations”). The following is only a summary of the Series C-1 Certificate of Designations, and is
qualified in its entirety by reference to the full text of the Series C-1 Certificate of Designations, a copy of which is filed as Exhibit
3.1 to our Current Report on Form 8-K filed on May 8, 2024.
Designation,
Amount, and Par Value. The number of Series C-1 Preferred Stock designated is 10,853 shares. The shares of Series C-1 Preferred
Stock have a par value of $0.001 per share and a stated value of $1,000 per share.
Conversion
Price. The Series C-1 Preferred Stock will be convertible into shares of Common Stock at an initial conversion price of $2.595
(subject to adjustment pursuant to the Series C-1 Certificate of Designations) (the “Conversion
Price”). The Certificate of Designations also provides that in the event of certain
Triggering Events (as defined below) any holder may, at any time, convert any or all of such holder’s Series C-1 Preferred Stock
at an alternate conversion rate equal to the product of (i) the Alternate Conversion Price (as defined below) and (ii) the quotient of
(x) the 25% redemption premium multiplied by (y) the amount of Series C-1 Preferred Stock subject to such conversion. “Triggering
Events” include, among others, (i) a suspension of trading or the failure to be traded or listed on an eligible market for five
consecutive days or more, (ii) the failure to remove restrictive legends when required, (iii) the Company’s default in payment
of indebtedness in an aggregate amount of $500,000 or more, (iv) proceedings for a bankruptcy, insolvency, reorganization or liquidation,
which are not dismissed with 30 days, (v) commencement of a voluntary bankruptcy proceeding, and (viii) final judgments against the Company
for the payment of money in excess of $500,000. “Alternate Conversion Price” means the lowest of (i) the applicable conversion
price the in effect, (ii) the greater of (x) $0.519 (the “Floor Price”) and (y)
80% of the volume weighted average price (“VWAP”) of the Common Stock on the trading day immediately preceding the delivery
of the applicable conversion notice. Further, the Series C-1 Certificate of Designations provides that if on any of the 90th and 180th day
after each of the occurrence of any Stock Combination Event (as defined in the Series C-1 Certificate of Designations) and the Applicable
Date (as defined in the Series C-1 Certificate of Designations), the conversion price then in effect is greater than the market price
then in effect (the “Adjustment Price”), on such date then the conversion price
shall automatically lower to the Adjustment Price.
Dividends.
Holders of the Series C-1 Preferred Stock shall be entitled to receive dividends when and as declared by the Board, from time to time,
in its sole discretion, which Dividends shall be paid by the Company out of funds legally available therefor, payable, subject to the
conditions and other terms hereof, in cash, in securities of the Company or any other entity, or using assets as determined by the Board
on the Stated Value of such Preferred Share.
Liquidation. In
the event of a Liquidation Event (as defined in the Series C-1 Certificate of Designation), the holders the Series C-1 Preferred Stock
shall be entitled to receive in cash out of the assets of the Company, before any amount shall be paid to the holders of any other shares
of capital stock of the Company, equal to the greater of (A) 125% of the Conversion Amount (as defined in the Series C-1 Certificate
of Designation) on the date of such payment and (B) the amount per share such holder of Series C-1 Preferred Stock would receive if they
converted such share of Series C-1 Preferred Stock into Common Stock immediately prior to the date of such payment
Company
Redemption. The Company may redeem all, or any portion, of the Series C-1 Preferred Stock for cash, at a price per share
of Series C-1 Preferred Stock equal to 115% of the greater of (i) the Conversion Amount (as defined in the Series C-1 Certificate of
Designations) being redeemed as of the Company Optional Redemption Date (as defined in the Series C-1 Certificate of Designation) and
(ii) the product of (1) the Conversion Rate (as defined in the Series C-1 Certificate of Designation) with respect to the Conversion
Amount being redeemed as of the Company Optional Redemption Date multiplied by (2) the greatest Closing Sale Price (as defined in the
Certificate of Designation) of the Common Stock on any Trading Day during the period commencing on the date immediately preceding such
Company Optional Redemption Notice Date (as defined in the Certificate of Designation) and ending on the Trading Day immediately prior
to the date the Company makes the entire payment required to be made under the Certification of Designation.
Maximum
Percentage. Holders of Series C-1 Preferred Stock are prohibited from converting shares of Series C-1 Preferred Stock into shares
of Common Stock if, as a result of such conversion, such holder, together with its affiliates, would beneficially own in excess of 4.99%
(the “Maximum Percentage”) of the total number of shares of Common Stock issued and outstanding immediately after
giving effect to such conversion.
Voting
Rights. The holders of the Series C-1 Preferred Stock shall have no voting power and no right to vote on any matter at any
time, either as a separate series or class or together with any other series or class of share of capital stock, and shall not be entitled
to call a meeting of such holders for any purpose nor shall they be entitled to participate in any meeting of the holders of Common Stock,
except as expressly provided in the Certificate of Designations and where required by the General Corporation Law of the State of Delaware
(the “DGCL”).
Series D-1 Preferred Stock
On May 2, 2024, we filed
a Certificate of Designation for its Series D-1 Preferred Stock with the Secretary of State of Delaware (the “Series
D-1 Certificate of Designations”). The following is only a summary of the Series D-1 Certificate of Designations, and is
qualified in its entirety by reference to the full text of the Series D-1 Certificate of Designations, a copy of which is filed as Exhibit
3.1 to this Current Report on Form 8-K and is incorporated by reference herein.
The Series D-1 Certificate
of Designations provides that the share of Preferred Stock will have 418,600,000 votes and will vote together with the outstanding shares
of the Company’s Common Stock as a single class exclusively with respect to any proposal to amend the Company’s Amended and
Restated Certificate of Incorporation to increase the number of shares of Common Stock that the Company is authorized to issue. The Series
D-1Preferred Stock will be voted, without action by the holder, on any such proposal in the same proportion as shares of the Company’s
Common Stock are voted. The Series D-1 Preferred Stock otherwise has no voting rights except as otherwise required by the DGCL.
The Series D-1 Preferred
Stock is not convertible into, or exchangeable for, shares of any other class or series of stock or other securities of the Company.
The Series D-1 Preferred Stock has no rights with respect to any distribution of assets of the Company, including upon a liquidation,
bankruptcy, reorganization, merger, acquisition, sale, dissolution or winding up of the Company, whether voluntarily or involuntarily.
The holders of Series D-1 Preferred Stock will not be entitled to receive dividends of any kind.
The
outstanding share of Series D-1 Preferred Stock shall be redeemed in whole, but not in part, at any time (i) if such redemption is ordered
by the Board of Directors in its sole discretion or (ii) automatically upon the effectiveness of the amendment to increase the number
of shares of Common Stock that the Company is authorized to issue. Upon such redemption, the holder of the Preferred Stock will receive
consideration of $0.01 per share in cash.
Listing
Our Common Stock is traded
on Nasdaq Capital Market under the trading symbol “ADTX”.
Transfer Agent
The Company’s transfer
agent is VStock Transfer, LLC.
Pre-Funded Warrants
General
The
term “pre-funded” refers to the fact that the purchase price of the pre-funded warrants in this offering includes
almost the entire exercise price that will be paid under the pre-funded warrants, except for a nominal remaining exercise price
of $0.001. The purpose of the pre-funded warrants is to enable investors that may have restrictions on their ability to beneficially
own more than 4.99% (or, at the election of such purchaser, 9.99%) of our outstanding Common Stock following the consummation of this
offering the opportunity to invest capital into the Company without triggering their ownership restrictions, by receiving pre-funded warrants
in lieu of shares of our Common Stock which would result in such ownership of more than 4.99% or 9.99%, as applicable, and receiving
the ability to exercise their option to purchase the shares underlying the pre-funded warrants at a nominal price at a later
date.
The
following is a brief summary of certain terms and conditions of the pre-funded warrants being offered by us. The
following description is subject in all respects to the provisions contained in the form of pre-funded warrant, the form
of which will be filed as an exhibit to the registration statement of which this prospectus forms a part.
Exercise price
Pre-funded warrants will
have an exercise price of $0.001 per share. The exercise price is subject to appropriate adjustment in the event of certain stock dividends
and distributions, stock splits, stock combinations, reclassifications or similar events affecting our Common Stock and also upon any
distributions of assets, including cash, stock or other property to our stockholders.
Exercisability
The
pre-funded warrants are exercisable at any time after their original issuance and until exercised in full. The pre-funded warrants will
be exercisable, at the option of each holder, in whole or in part by delivering to us a duly executed exercise notice and by payment
in full of the exercise price in immediately available funds for the number of shares of Common Stock purchased upon such exercise. As
an alternative to payment in immediately available funds, the holder may elect to exercise the pre-funded warrant through a cashless
exercise, in which the holder would receive upon such exercise the net number of shares of Common Stock determined according to the formula
set forth in the pre-funded warrant. No fractional shares of Common Stock will be issued in connection with the exercise of a pre-funded
warrant.
Exercise limitations
The
pre-funded warrants may not be exercised by the holder to the extent that the holder, together with its affiliates, would beneficially
own, after such exercise more than 4.99% of the shares of our Common Stock then outstanding (including for such purpose the shares of
our Common Stock issuable upon such exercise). However, any holder may increase or decrease such beneficial ownership limitation upon
notice to us, provided that such limitation cannot exceed 9.99%, and provided that any increase in the beneficial ownership limitation
shall not be effective until 61 days after such notice is delivered. Purchasers of pre-funded warrants in this offering may also elect
prior to the issuance of the pre-funded warrants to have the initial exercise limitation set at 9.99% of our outstanding shares of Common
Stock.
Transferability
Subject
to applicable laws, the pre-funded warrants may be offered for sale, sold, transferred or assigned without our consent.
Exchange listing
There
is no established trading market for the pre-funded warrants and we do not expect a market to develop. In addition, we do not intend
to apply for the listing of the pre-funded warrants on any national securities exchange or other trading market. Without an active trading
market, the liquidity of the pre-funded warrants will be limited.
Fundamental transactions
In
the event of a fundamental transaction, as described in the pre-funded warrants and generally including any reorganization, recapitalization
or reclassification of our Common Stock, the sale, transfer or other disposition of all or substantially all of our properties or assets,
our consolidation or merger with or into another person, the acquisition of more than 50% of our outstanding Common Stock, or any person
or group becoming the beneficial owner of 50% of the voting power represented by our outstanding Common Stock, upon consummation of such
a fundamental transaction, the holders of the pre-funded warrants will be entitled to receive upon exercise of the pre-funded warrants
the kind and amount of securities, cash or other property that the holders would have received had they exercised the pre-funded warrants
immediately prior to such fundamental transaction without regard to any limitations on exercise contained in the pre-funded warrants.
No rights as a
stockholder
Except
as otherwise provided in the pre-funded warrant or by virtue of such holder’s ownership of shares of our Common Stock, the holder
of a pre-funded warrant does not have the rights or privileges of a holder of our Common Stock, including any voting rights, until the
holder exercises the pre-funded warrant. The pre-funded warrants will provide that holders have the right to participate in distributions
or dividends paid on our Common Stock.
SELLING
STOCKHOLDERS
The Common Stock being offered
by the Selling Stockholders are those issuable to the Selling Stockholders, upon exercise of the Warrants. For additional information
regarding the issuances of those securities, see “Prospectus Summary—Recent Developments—December 2023 PIPE”
above. We are registering the shares of Common Stock in order to permit the Selling Stockholders to offer the shares for resale from
time to time. Except for the ownership of the Warrants and as noted below, the Selling Stockholders have not had any material relationship
with us within the past three years.
The table below lists the
Selling Stockholders and other information regarding the beneficial ownership of the shares of Common Stock by each of the Selling Stockholders.
The second column lists the number of shares of Common Stock beneficially owned by each Selling Stockholder, based on its ownership of
the shares of Common Stock and Warrants, as of June 4, 2024, assuming exercise of the Warrants held by the Selling Stockholders on that
date, without regard to any limitations on exercises.
The third column lists the
shares of Common Stock being offered by this prospectus by the Selling Stockholders.
In accordance with the terms
of a registration rights agreement with the Selling Stockholders, this prospectus generally covers the resale of the maximum number of
shares of Common Stock issuable upon exercise of the Warrants, determined as if the outstanding Warrants were exercised in full as of
the trading day immediately preceding the date this registration statement was initially filed with the SEC, each as of the trading day
immediately preceding the applicable date of determination and all subject to adjustment as provided in the registration rights agreement,
without regard to any limitations on the exercise of the Warrants. The fourth column assumes the sale of all of the shares offered by
the Selling Stockholders pursuant to this prospectus.
Under the terms of the Warrants,
a Selling Stockholder may not exercise the Warrants to the extent such exercise would cause such Selling Stockholder, together with its
affiliates and attribution parties, to beneficially own a number of shares of Common Stock which would exceed 4.99% of our then outstanding
Common Stock following such exercise, excluding for purposes of such determination shares of Common Stock issuable upon exercise of such
Warrants which have not been exercised. The number of shares in the second and fourth columns do not reflect this limitation. The Selling
Stockholders may sell all, some or none of their shares in this offering. See “Plan of Distribution.”
| Name of
Selling Stockholder |
|
Number
of
Shares
of Common
Stock
Beneficially
Owned
Prior to
Offering(1) |
|
|
Maximum
Number of
Shares
of Common
Stock
to be Sold
in this
Offering |
|
|
Number
of
Shares
of Common
Stock
Beneficially
Owned After
Offering |
|
|
Percentage
of Shares
Beneficially
Owned after
Offering(1) |
|
| Sabby Volatility Warrant Master
Fund, Ltd.(2) |
|
|
168,734 |
|
|
|
3,711,342 |
|
|
|
168,734 |
|
|
|
8.5 |
% |
| Michael Vasinkevich(3) |
|
|
86,073 |
|
|
|
47,598 |
|
|
|
38,475 |
|
|
|
2.8 |
% |
| Michael Mirsky(3) |
|
|
25,504 |
|
|
|
14,104 |
|
|
|
11,400 |
|
|
|
* |
|
| Noam Rubinstein(3) |
|
|
16,778 |
|
|
|
9,278 |
|
|
|
7,500 |
|
|
|
* |
|
| Craig Schwabe(3) |
|
|
4,530 |
|
|
|
2,505 |
|
|
|
2,025 |
|
|
|
* |
|
| Charles Worthman(3) |
|
|
1,342 |
|
|
|
742 |
|
|
|
600 |
|
|
|
* |
|
(1) |
The ability to exercise
the Warrants held by the Selling Stockholders is subject to a beneficial ownership limitation that, at the time of initial issuance
of the Warrants was capped at 4.99% beneficial ownership of the Company’s issued and outstanding Common Stock (post-exercise).
These beneficial ownership limitations may be adjusted up or down, subject to providing advanced notice to the Company. Beneficial
ownership as reflected in the selling stockholder table reflects the total number of shares potentially issuable underlying the Warrants,
and does not give effect to these beneficial ownership limitations. Accordingly, actual beneficial ownership, as calculated in accordance
with Section 13(d) and Rule 13d-3 thereunder may be lower than as reflected in the table. |
| (2) |
Sabby
Management, LLC is the investment manager of Sabby Volatility Warrant Master Fund, Ltd. (“Sabby”) and shares voting and
investment power with respect to these shares in this capacity. As manager of Sabby Management, LLC, Hal Mintz also shares voting
and investment power on behalf of Sabby. Each of Sabby Management, LLC and Mr. Mintz disclaims beneficial ownership over the securities
listed except to the extent of their pecuniary interest therein. |
| (3) |
The selling stockholder
is affiliated with H.C. Wainwright & Co., LLC, a registered broker dealer with a registered address of H.C. Wainwright &
Co., LLC, 430 Park Ave, 3rd Floor, New York, NY 10022, and has sole voting and dispositive power over the securities held. The number
of shares beneficially owned prior to this offering consist of (a) shares of Common Stock issuable upon exercise of placement
agent warrants, which were received as compensation in connection with the concurrent Registered Direct Offering and the Private
Placements consummated by us in June 2023, and (b) shares of Common Stock issuable upon exercise of placement agent warrants,
which were received as compensation in connection with the December 2023 PIPE. The selling stockholder acquired the placement agent
warrants in the ordinary course of business and, at the time the placement agent warrants were acquired, the selling stockholder
had no agreement or understanding, directly or indirectly, with any person to distribute such securities. |
Plan
of Distribution
Each Selling Stockholder
of the securities and any of their pledgees, assignees and successors-in-interest may, from time to time, sell any or all of their securities
covered hereby on the principal Trading Market or any other stock exchange, market or trading facility on which the securities are traded
or in private transactions. These sales may be at fixed or negotiated prices. A Selling Stockholder may use any one or more of the following
methods when selling securities:
| ● | ordinary
brokerage transactions and transactions in which the broker-dealer solicits purchasers; |
| ● | block
trades in which the broker-dealer will attempt to sell the securities as agent but may position
and resell a portion of the block as principal to facilitate the transaction; |
| ● | purchases
by a broker-dealer as principal and resale by the broker-dealer for its account; |
| ● | an
exchange distribution in accordance with the rules of the applicable exchange; |
| ● | privately
negotiated transactions; |
| ● | settlement
of short sales; |
| ● | in
transactions through broker-dealers that agree with the Selling Stockholders to sell a specified
number of such securities at a stipulated price per security; |
| ● | through
the writing or settlement of options or other hedging transactions, whether through an options
exchange or otherwise; |
| ● | a
combination of any such methods of sale; or |
| ● | any
other method permitted pursuant to applicable law. |
The Selling Stockholders
may also sell securities under Rule 144 or any other exemption from registration under the Securities Act of 1933, as amended (the “Securities
Act”), if available, rather than under this prospectus. Broker-dealers engaged by the Selling Stockholders may arrange for other
brokers-dealers to participate in sales. Broker-dealers may receive commissions or discounts from the Selling Stockholders (or, if any
broker-dealer acts as agent for the purchaser of securities, from the purchaser) in amounts to be negotiated, but, except as set forth
in a supplement to this prospectus, in the case of an agency transaction not in excess of a customary brokerage commission in compliance
with FINRA Rule 2121; and in the case of a principal transaction a markup or markdown in compliance with FINRA Rule 2121.
In connection with the sale
of the securities or interests therein, the Selling Stockholders may enter into hedging transactions with broker-dealers or other financial
institutions, which may in turn engage in short sales of the securities in the course of hedging the positions they assume. The Selling
Stockholders may also sell securities short and deliver these securities to close out their short positions, or loan or pledge the securities
to broker-dealers that in turn may sell these securities. The Selling Stockholders may also enter into option or other transactions with
broker-dealers or other financial institutions or create one or more derivative securities which require the delivery to such broker-dealer
or other financial institution of securities offered by this prospectus, which securities such broker-dealer or other financial institution
may resell pursuant to this prospectus (as supplemented or amended to reflect such transaction).
The Selling Stockholders
and any broker-dealers or agents that are involved in selling the securities may be deemed to be “underwriters” within the
meaning of the Securities Act in connection with such sales. In such event, any commissions received by such broker-dealers or agents
and any profit on the resale of the securities purchased by them may be deemed to be underwriting commissions or discounts under the
Securities Act. Each Selling Stockholder has informed us that it does not have any written or oral agreement or understanding, directly
or indirectly, with any person to distribute the securities.
We are required to pay certain
fees and expenses incurred by us incident to the registration of the securities. We have agreed to indemnify the Selling Stockholders
against certain losses, claims, damages and liabilities, including liabilities under the Securities Act.
We agreed to keep this prospectus
effective until the earlier of (i) the date on which the securities may be resold by the Selling Stockholders without registration and
without regard to any volume or manner-of-sale limitations by reason of Rule 144, without the requirement for us to be in compliance
with the current public information under Rule 144 under the Securities Act or any other rule of similar effect or (ii) all of the securities
have been sold pursuant to this prospectus or Rule 144 under the Securities Act or any other rule of similar effect. The resale securities
will be sold only through registered or licensed brokers or dealers if required under applicable state securities laws. In addition,
in certain states, the resale securities covered hereby may not be sold unless they have been registered or qualified for sale in the
applicable state or an exemption from the registration or qualification requirement is available and is complied with.
Under
applicable rules and regulations under the Exchange Act, any person engaged in the distribution of the resale securities may not simultaneously
engage in market making activities with respect to the Common Stock for the applicable restricted period, as defined in Regulation M,
prior to the commencement of the distribution. In addition, the Selling Stockholders will be subject to applicable provisions of the
Exchange Act and the rules and regulations thereunder, including Regulation M, which may limit the timing of purchases and sales of the
Common Stock by the Selling Stockholders or any other person. We will make copies of this prospectus available to the Selling Stockholders
and have informed them of the need to deliver a copy of this prospectus to each purchaser at or prior to the time of the sale (including
by compliance with Rule 172 under the Securities Act).
LEGAL
MATTERS
Sheppard, Mullin, Richter
& Hampton LLP, New York, New York, will pass upon the validity of the shares of our Common Stock offered hereby.
EXPERTS
dbbmckennon, an independent
registered public accounting firm, has audited our financial statements included in our Annual Report on Form 10-K for the year ended
December 31, 2023, as set forth in their report, which includes an explanatory paragraph as to our ability to continue as a going concern,
dated April 16, 2024, which is included in this prospectus and elsewhere in the registration statement, given on the authority of such
firm as experts in accounting and auditing.
WHERE
YOU CAN FIND MORE INFORMATION
We have filed with the SEC
a registration statement on Form S-1 under the Securities Act with respect to the securities offered by this prospectus. This prospectus,
which constitutes a part of the registration statement, does not contain all of the information set forth in the registration statement,
some of which is contained in exhibits to the registration statement as permitted by the rules and regulations of the SEC. For further
information with respect to us and our securities, we refer you to the registration statement, including the exhibits filed as a part
of the registration statement. Statements contained in this prospectus concerning the contents of any contract or any other document
is not necessarily complete. If a contract or document has been filed as an exhibit to the registration statement, please see the copy
of the contract or document that has been filed. Each statement is this prospectus relating to a contract or document filed as an exhibit
is qualified in all respects by the filed exhibit. We are subject to the informational requirements of the Exchange Act and in accordance
therewith file annual, quarterly and current reports, proxy statements and other information with the SEC. The SEC maintains an Internet
website that contains reports, proxy statements and other information about issuers, like us, that file electronically with the SEC.
The address of that website is www.sec.gov. The registration statement and the documents referred to below under “Incorporation
of Documents By Reference” are also available on our website, www.astrotechcorp.com. We have not incorporated by reference
into this prospectus the information on our website, and you should not consider it to be a part of this prospectus.
INDEX TO AUDITED CONSOLIDATED
FINANCIAL STATEMENTS
INDEX TO UNAUDITED INTERIM
CONSOLIDATED FINANCIAL STATEMENTS
REPORT OF INDEPENDENT REGISTERED
PUBLIC ACCOUNTING FIRM
To the Board of Directors and Stockholders of
Aditxt, Inc.
Opinion on the Financial Statements
We have audited the accompanying consolidated
balance sheets of Aditxt, Inc. and its subsidiaries (the “Company”) as of December 31, 2023 and 2022, the related consolidated
statements of operations, stockholders’ equity, and cash flows, for the years ended December 31, 2023 and 2022, and the related
notes (collectively referred to as the “financial statements”). In our opinion, the financial statements present fairly,
in all material respects, the financial position of the Company as of December 31, 2023 and 2022, and the results of its operations and
its cash flows for the years then ended, in conformity with accounting principles generally accepted in the United States of America.
Going Concern
The accompanying financial statements have been
prepared assuming the Company will continue as a going concern. As discussed in Note 2 to the financial statements, the Company’s
net losses and negative cash flow from operations, raise substantial doubt about its ability to continue as a going concern. Management’s
plans in regard to these matters are also described in Note 2. The financial statements do not include any adjustments that might result
from the outcome of this uncertainty.
Basis for Opinion
These financial statements are the responsibility
of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our
audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (“PCAOB”)
and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable
rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the
standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial
statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged
to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding
of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s
internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to
assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that
respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial
statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well
as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/ dbbmckennon
We have served as the Company’s auditor
since 2018.
San Diego, California
April 16, 2024
ADITXT, INC.
CONSOLIDATED BALANCE SHEETS
| | |
December 31, | | |
December 31, | |
| | |
2023 | | |
2022 | |
| ASSETS | |
| | |
| |
| CURRENT ASSETS: | |
| | |
| |
| Cash | |
$ | 97,102 | | |
$ | 2,768,640 | |
| Accounts receivable, net | |
| 408,326 | | |
| 527,961 | |
| Inventory | |
| 745,502 | | |
| 950,093 | |
| Prepaid expenses | |
| 217,390 | | |
| 496,869 | |
| Subscription receivable | |
| 5,444,628 | | |
| - | |
| TOTAL CURRENT ASSETS | |
| 6,912,948 | | |
| 4,743,563 | |
| | |
| | | |
| | |
| Fixed assets, net | |
| 1,898,243 | | |
| 2,318,863 | |
| Intangible assets, net | |
| 9,444 | | |
| 107,000 | |
| Deposits | |
| 106,410 | | |
| 355,366 | |
| Right of use asset - long term | |
| 2,200,299 | | |
| 3,160,457 | |
| Deferred issuance costs | |
| - | | |
| 50,000 | |
| Investment in Evofem | |
| 22,277,211 | | |
| - | |
| Deposit on acquisition | |
| 11,173,772 | | |
| - | |
| TOTAL ASSETS | |
$ | 44,578,327 | | |
$ | 10,735,249 | |
| | |
| | | |
| | |
| LIABILITIES AND STOCKHOLDERS’
EQUITY | |
| | | |
| | |
| CURRENT LIABILITIES: | |
| | | |
| | |
| Accounts payable and accrued expenses | |
$ | 8,554,959 | | |
$ | 1,958,502 | |
| Notes payable - related party | |
| 375,000 | | |
| - | |
| Notes payable, net of discount | |
| 15,653,477 | | |
| - | |
| Financing on fixed assets | |
| 147,823 | | |
| 409,983 | |
| Deferred rent | |
| 158,612 | | |
| 188,581 | |
| Lease liability - current | |
| 999,943 | | |
| 1,086,658 | |
| TOTAL CURRENT LIABILITIES | |
| 25,889,814 | | |
| 3,643,724 | |
| | |
| | | |
| | |
| Settlement liability | |
| 1,600,000 | | |
| - | |
| Lease liability - long term | |
| 1,041,744 | | |
| 1,885,218 | |
| | |
| | | |
| | |
| TOTAL LIABILITIES | |
| 28,531,558 | | |
| 5,528,942 | |
| | |
| | | |
| | |
| STOCKHOLDERS’ EQUITY | |
| | | |
| | |
| Preferred stock, $0.001 par value, 3,000,000 shares
authorized, zero shares issued and outstanding, respectively | |
| - | | |
| - | |
| Series A-1 Convertible Preferred stock, $0.001 par value,
22,280 shares authorized, 22,280 and zero shares issued and outstanding, respectively | |
| 22 | | |
| - | |
| Series B Preferred stock, $0.001 par value, 1 share authorized,
zero and zero shares issued and outstanding, respectively | |
| - | | |
| - | |
| Series B-2 Convertible Preferred stock, $0.001 par value,
2,625 shares authorized, 2,625 and zero shares issued and outstanding, respectively | |
| 3 | | |
| - | |
| Series C Preferred stock, $0.001 par value, 1 share authorized,
zero and zero shares issued and outstanding, respectively | |
| - | | |
| - | |
| Common stock, $0.001 par value, 100,000,000 shares
authorized, 1,318,969 and 107,698 shares issued and 1,318,918 and 107,647 shares outstanding,
respectively | |
| 1,319 | | |
| 108 | |
| Treasury stock, 51 and 51 shares, respectively | |
| (201,605 | ) | |
| (201,605 | ) |
| Additional paid-in capital | |
| 143,997,710 | | |
| 100,448,166 | |
| Accumulated deficit | |
| (127,741,072 | ) | |
| (95,040,362 | ) |
| TOTAL ADITXT,
INC. STOCKHOLDERS’ EQUITY | |
| 16,056,377 | | |
| 5,206,307 | |
| | |
| | | |
| | |
| NON-CONTROLLING
INTEREST | |
| (9,608 | ) | |
| - | |
| | |
| | | |
| | |
| TOTAL STOCKHOLDERS’
EQUITY | |
| 16,046,769 | | |
| 5,206,307 | |
| | |
| | | |
| | |
| TOTAL LIABILITIES
AND STOCKHOLDERS’ EQUITY | |
$ | 44,578,327 | | |
$ | 10,735,249 | |
See accompanying notes to the consolidated financial
statements.
ADITXT, INC.
CONSOLIDATED STATEMENTS OF OPERATIONS
| | |
Year Ended | | |
Year Ended | |
| | |
December 31,
2023 | | |
December 31,
2022 | |
| REVENUE | |
| | |
| |
| Sales | |
$ | 645,176 | | |
$ | 933,715 | |
| Cost of goods sold | |
| 756,836 | | |
| 766,779 | |
| Gross profit (loss) | |
| (111,660 | ) | |
| 166,936 | |
| | |
| | | |
| | |
| OPERATING EXPENSES | |
| | | |
| | |
| General and administrative expenses $1,133,077, and $1,516,805
in stock-based compensation, respectively | |
| 18,607,142 | | |
| 15,985,552 | |
| Research and development, includes $262,154, and $591,518
in stock-based compensation, respectively | |
| 7,074,339 | | |
| 7,268,084 | |
| Sales and marketing $6,787, and $1,023,045 in stock-based
compensation, respectively | |
| 269,284 | | |
| 1,849,460 | |
| Impairment on notes receivable | |
| - | | |
| 543,938 | |
| Total operating expenses | |
| 25,950,765 | | |
| 25,647,034 | |
| | |
| | | |
| | |
| NET LOSS FROM OPERATIONS | |
| (26,062,425 | ) | |
| (25,480,098 | ) |
| | |
| | | |
| | |
| OTHER EXPENSE | |
| | | |
| | |
| Interest expense | |
| (4,195,127 | ) | |
| (753,038 | ) |
| Interest income | |
| 10,166 | | |
| 57,348 | |
| Other income | |
| - | | |
| 58,960 | |
| Amortization of debt discount | |
| (2,194,773 | ) | |
| (1,533,048 | ) |
| Gain on note exchange agreement | |
| 51,712 | | |
| - | |
| Total other expense | |
| (6,328,022 | ) | |
| (2,169,778 | ) |
| | |
| | | |
| | |
| Net loss before income taxes | |
| (32,390,447 | ) | |
| (27,649,876 | ) |
| Income tax provision | |
| - | | |
| - | |
| | |
| | | |
| | |
| NET LOSS | |
$ | (32,390,447 | ) | |
$ | (27,649,876 | ) |
| | |
| | | |
| | |
| NET LOSS ATTRIBUTABLE
TO NON-CONTROLLING INTEREST | |
| (9,608 | ) | |
| - | |
| | |
| | | |
| | |
| NET LOSS ATTRIBUTABLE
TO ADITXT, INC. & SUBSIDIARIES | |
$ | (32,380,839 | ) | |
$ | (27,649,876 | ) |
| | |
| | | |
| | |
| Deemed Dividend | |
| (319,871 | ) | |
| (37,667 | ) |
| | |
| | | |
| | |
| NET LOSS ATTRIBUTABLE
TO COMMON STOCKHOLDERS | |
$ | (32,700,710 | ) | |
$ | (27,687,553 | ) |
| | |
| | | |
| | |
| Net loss per share - basic and diluted | |
$ | (108.15 | ) | |
$ | (597.12 | ) |
| | |
| | | |
| | |
| Weighted average number of shares
outstanding during the period - basic and diluted | |
| 302,356 | | |
| 46,369 | |
See accompanying notes to the consolidated financial
statements.
ADITXT, INC.
CONSOLIDATED STATEMENTS OF STOCKHOLDERS’
EQUITY
YEARS ENDED DECEMBER 31, 2023 AND 2022
| | |
Preferred
Shares Outstanding | | |
Preferred
Shares Par | | |
Preferred
A-1 Shares Outstanding | | |
Preferred
A-1 Shares Par | | |
Preferred B
Shares Outstanding | | |
Preferred B
Shares Par | | |
Preferred
B-2 Shares Outstanding | | |
Preferred
B-2 Shares Par | | |
Preferred C
Shares Outstanding | | |
Preferred C
Shares Par | | |
Common
Shares Outstanding | | |
Common
Shares Par | | |
Treasury
Stock | | |
Additional
Paid-in Capital | | |
Accumulated
Deficit | | |
Non-
Controlling Interest | | |
Total
Stockholders’
Equity | |
| Balance
December 31, 2022 | |
| - | | |
| - | | |
$ | - | | |
| - | | |
| - | | |
$ | - | | |
| - | | |
$ | - | | |
| - | | |
$ | - | | |
| 107,647 | | |
$ | 108 | | |
$ | (201,605 | ) | |
$ | 100,448,166 | | |
$ | (95,040,362 | ) | |
$ | - | | |
$ | 5,206,307 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Stock
option compensation | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 589,014 | | |
| - | | |
| - | | |
| 609,014 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Restricted
stock unit compensation | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 308,479 | | |
| - | | |
| - | | |
| 308,479 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Issuance
of restricted stock units for compensation | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 157 | | |
| 2 | | |
| - | | |
| (2 | ) | |
| - | | |
| - | | |
| - | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Sale
of common stock | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 8,463 | | |
| 9 | | |
| - | | |
| 507,007 | | |
| - | | |
| - | | |
| 507,016 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Issuance
of shares for services | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 74,675 | | |
| 75 | | |
| - | | |
| 484,450 | | |
| - | | |
| - | | |
| 484,525 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Issuance
of shares of Pearsanta Common Stock for IP | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 10,000 | | |
| - | | |
| - | | |
| 10,000 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Warrants
issued for cash, net of issuance costs | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 1,581,467 | | |
| - | | |
| - | | |
| 1,581,467 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Exercise
of warrants | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 1,055,374 | | |
| 1,057 | | |
| - | | |
| (57 | ) | |
| - | | |
| - | | |
| 1,000 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Sale
of Series C Preferred shares to related party | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 1 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 1,000 | | |
| - | | |
| - | | |
| 1,000 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Issuance
of shares for debt issuance costs | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 31,251 | | |
| 32 | | |
| - | | |
| 354,806 | | |
| - | | |
| - | | |
| 354,838 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Issuance
of warrants for offering, net of issuance costs | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 14,411,028 | | |
| - | | |
| - | | |
| 14,411,028 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Modification
of warrants | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 319,871 | | |
| (319,871 | ) | |
| - | | |
| - | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Redemption
of Series C Preferred shares to related party | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| (1 | ) | |
| - | | |
| - | | |
| - | | |
| - | | |
| (1,000 | ) | |
| - | | |
| - | | |
| (1,000 | ) |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Series
A-1 Preferred shares issued for exchange agreement | |
| - | | |
| - | | |
| 22,280 | | |
| 22 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 22,277,211 | | |
| - | | |
| - | | |
| 22,277,233 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Note
exchange agreement | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 2,625 | | |
| 3 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 2,686,306 | | |
| - | | |
| - | | |
| 2,686,309 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Rounding
from reverse stock split | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 41,351 | | |
| 36 | | |
| - | | |
| (36 | ) | |
| - | | |
| - | | |
| - | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Net
loss | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| (32,380,839 | ) | |
| (9,608 | ) | |
| (32,390,447 | ) |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Balance
December 31, 2023 | |
| - | | |
| - | | |
$ | 22,280 | | |
| 22 | | |
| - | | |
$ | - | | |
| 2,625 | | |
$ | 3 | | |
| - | | |
$ | - | | |
| 1,318,918 | | |
$ | 1,319 | | |
$ | (201,605 | ) | |
$ | 143,997,710 | | |
$ | (127,741,072 | ) | |
$ | (9,608 | ) | |
$ | 16,046,769 | |
See accompanying notes to the consolidated
financial statements.
ADITXT, INC.
CONSOLIDATED STATEMENTS OF STOCKHOLDERS’
EQUITY
YEARS ENDED DECEMBER 31, 2023 AND 2022
| | |
Preferred
Shares Outstanding | | |
Preferred
Shares Par | | |
Preferred
A-1 Shares Outstanding | | |
Preferred
A-1 Shares Par | | |
Preferred B
Shares Outstanding | | |
Preferred B
Shares Par | | |
Preferred
B-2 Shares Outstanding | | |
Preferred
B-2 Shares Par | | |
Preferred C
Shares Outstanding | | |
Preferred C
Shares Par | | |
Common
Shares Outstanding | | |
Common
Shares Par | | |
Treasury
Stock | | |
Additional
Paid-in Capital | | |
Accumulated
Deficit | | |
Non-Controlling
Interest | | |
Total
Stockholders’ Equity | |
| Balance December 31, 2021 | |
| - | | |
$ | - | | |
| - | | |
$ | - | | |
| - | | |
$ | - | | |
| - | | |
$ | - | | |
| - | | |
$ | - | | |
| 22,220 | | |
$ | 22 | | |
$ | (201,605 | ) | |
$ | 77,735,165 | | |
$ | (67,352,809 | ) | |
$ | - | | |
$ | 10,180,773 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Stock option and warrant
compensation | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 1,413,904 | | |
| - | | |
| - | | |
| 1,413,904 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Issuance of shares for vested
restricted stock units | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 463 | | |
| 4 | | |
| - | | |
| 1,209,902 | | |
| - | | |
| - | | |
| 1,209,906 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Issuance of shares for services | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 3,707 | | |
| 5 | | |
| - | | |
| 507,553 | | |
| - | | |
| - | | |
| 507,558 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Exercise of warrants, modification
of warrants, and issuance of warrants | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 4,486 | | |
| 5 | | |
| - | | |
| 1,203,764 | | |
| - | | |
| - | | |
| 1,203,769 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Sale of Series B Preferred
shares to related party | |
| - | | |
| - | | |
| - | | |
| - | | |
| 1 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 20,000 | | |
| - | | |
| - | | |
| 20,000 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Redemption of Series B Preferred
shares to related party | |
| - | | |
| - | | |
| - | | |
| - | | |
| (1 | ) | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| (20,000 | ) | |
| - | | |
| - | | |
| (20,000 | ) |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Shares issued as inducement
on loans, net of issuance costs | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 1,195 | | |
| 2 | | |
| - | | |
| 146,520 | | |
| - | | |
| - | | |
| 146,522 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Warrants issued with loans | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 878,622 | | |
| - | | |
| - | | |
| 878,622 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Reset provision on warrants
and modification of warrants | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 37,677 | | |
| (37,677 | ) | |
| - | | |
| - | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Issuance of shares for debt
issuance costs | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 262 | | |
| 1 | | |
| - | | |
| 96,029 | | |
| - | | |
| - | | |
| 96,030 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Exercise of warrants | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 44,173 | | |
| 45 | | |
| - | | |
| (45 | ) | |
| - | | |
| - | | |
| - | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Issuance of shares and warrants
for offering, net of issuance costs | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 30,609 | | |
| 31 | | |
| - | | |
| 17,232,276 | | |
| - | | |
| - | | |
| 17,232,307 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Issuance costs related to
exercise of warrants, modification of warrants, and issuance of warrants | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| (94,195 | ) | |
| - | | |
| - | | |
| (94,195 | ) |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Issuance of shares for settlement of AP | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 231 | | |
| 1 | | |
| - | | |
| 79,999 | | |
| - | | |
| - | | |
| 80,000 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Rounding from reverse stock
split | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 301 | | |
| (8 | ) | |
| - | | |
| (5 | ) | |
| - | | |
| - | | |
| (13 | ) |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Net
loss | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| (27,649,876 | ) | |
| - | | |
| (27,649,876 | ) |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Balance December 31, 2022 | |
| - | | |
$ | - | | |
| - | | |
$ | - | | |
| - | | |
$ | - | | |
| - | | |
$ | - | | |
| - | | |
$ | - | | |
| 107,647 | | |
$ | 108 | | |
$ | (201,605 | ) | |
$ | 100,448,166 | | |
$ | (95,040,362 | ) | |
$ | - | | |
$ | 5,206,307 | |
See accompanying notes to the consolidated financial
statements.
ADITXT, INC.
CONSOLIDATED STATEMENTS OF CASH FLOWS
| | |
Year Ended | | |
Year Ended | |
| | |
December 31,
2023 | | |
December 31,
2022 | |
| CASH FLOWS FROM OPERATING ACTIVITIES: | |
| | |
| |
| Net loss | |
$ | (32,390,447 | ) | |
$ | (27,649,876 | ) |
| Adjustments to reconcile net loss to net cash used in operating
activities | |
| | | |
| | |
| Stock-based compensation | |
| 1,402,018 | | |
| 3,131,368 | |
| Depreciation expense | |
| 435,027 | | |
| 428,977 | |
| Amortization of intangible assets | |
| 107,556 | | |
| 107,000 | |
| Amortization of debt discount | |
| 2,821,629 | | |
| 1,533,048 | |
| Impairment on notes receivable | |
| - | | |
| 543,938 | |
| Disposal of fixed assets | |
| - | | |
| 6,976 | |
| Gain on note exchange agreement | |
| (51,712 | ) | |
| - | |
| Changes in operating assets and liabilities: | |
| | | |
| | |
| Accounts receivable | |
| 119,635 | | |
| (438,117 | ) |
| Prepaid expenses | |
| 279,479 | | |
| (36,767 | ) |
| Deposits | |
| 248,956 | | |
| 23,884 | |
| Inventory | |
| 204,591 | | |
| (455,396 | ) |
| Accounts payable and accrued expenses | |
| 6,646,457 | | |
| 412,959 | |
| Settlement liability | |
| 1,600,000 | | |
| - | |
| Net cash used in operating activities | |
| (18,576,811 | ) | |
| (22,392,006 | ) |
| | |
| | | |
| | |
| CASH FLOWS FROM INVESTING ACTIVITIES: | |
| | | |
| | |
| Purchase of fixed assets | |
| (14,407 | ) | |
| (367,079 | ) |
| Tenant improvement allowance receivable | |
| - | | |
| 125,161 | |
| Net cash used in investing activities | |
| (14,407 | ) | |
| (241,918 | ) |
| | |
| | | |
| | |
| CASH FLOWS FROM FINANCING ACTIVITIES: | |
| | | |
| | |
| Proceeds from notes - related party | |
| 1,062,523 | | |
| 80,000 | |
| Proceeds from notes and convertible notes payable, net of offering costs | |
| 7,903,445 | | |
| 2,795,000 | |
| Repayments of note payable - related party | |
| (687,523 | ) | |
| (80,000 | ) |
| Repayments of note payable | |
| (3,152,488 | ) | |
| (3,206,887 | ) |
| Sale of Series B Preferred shares to related party | |
| - | | |
| 20,000 | |
| Redemption of Series B Preferred shares to related party | |
| - | | |
| (20,000 | ) |
| Common stock and warrants issued for cash, net of issuance costs | |
| 11,054,883 | | |
| 17,233,307 | |
| Sale of Series C Preferred shares to related party | |
| 1,000 | | |
| - | |
| Redemption of Series C Preferred shares to related party | |
| (1,000 | ) | |
| - | |
| Exercise of warrants, modification of warrants, and issuance of warrants | |
| 1,000 | | |
| 1,109,574 | |
| Payments on financing on fixed asset | |
| (262,160 | ) | |
| (400,491 | ) |
| Net cash provided by financing activities | |
| 15,919,680 | | |
| 17,530,503 | |
| | |
| | | |
| | |
| NET INCREASE (DECREASE) IN CASH | |
| (2,671,538 | ) | |
| (5,103,421 | ) |
| | |
| | | |
| | |
| CASH AT BEGINNING OF YEAR | |
| 2,768,640 | | |
| 7,872,061 | |
| | |
| | | |
| | |
| CASH AT END OF YEAR | |
$ | 97,102 | | |
$ | 2,768,640 | |
| | |
| | | |
| | |
| Supplemental cash flow information: | |
| | | |
| | |
| Cash paid for income taxes | |
$ | - | | |
$ | - | |
| Cash paid for interest expense | |
$ | 2,726,020 | | |
$ | 753,038 | |
| | |
| | | |
| | |
| Issuance of shares for the settlement of accounts payable | |
$ | - | | |
$ | 80,000 | |
| Debt discount from warrants issued with convertible note
payable | |
$ | - | | |
$ | 878,622 | |
| Debt discount from shares issued as inducement for note
payable | |
$ | - | | |
$ | 146,522 | |
| Shares issued for debt offering costs | |
$ | 354,838 | | |
$ | 96,030 | |
| Warrant modification | |
$ | 319,871 | | |
$ | 37,677 | |
| Deferred issuance costs | |
$ | - | | |
$ | 50,000 | |
| Issuance of shares of Pearsanta Common Stock for IP | |
$ | 10,000 | | |
$ | - | |
| Assumption of notes payable from Evofem merger agreement | |
$ | 11,173,750 | | |
$ | - | |
| Series A-1 Preferred shares issued for exchange agreement | |
$ | 22,277,233 | | |
$ | - | |
| Accrued intertest rolled into notes payable | |
$ | 701,315 | | |
$ | - | |
| Series B-2 Preferred shares issued in note exchange agreement | |
$ | 2,686,306 | | |
$ | - | |
See accompanying notes to the consolidated financial
statements.
ADITXT, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
NOTE 1 – ORGANIZATION AND NATURE OF BUSINESS
Company Background
Overview
We are a biotech innovation company with a mission
of prolonging life and enhancing its quality by improving the health of the immune system. We are an innovation company developing and
commercializing technologies with a focus on monitoring and modulating the immune system. Our immune reprogramming technologies are currently
at the pre-clinical stage and are designed to retrain the immune system to induce tolerance with an objective of addressing rejection
of transplanted organs, autoimmune diseases, and allergies. Our immune monitoring technologies are designed to provide a personalized
comprehensive profile of the immune system and we plan to utilize them in our upcoming reprogramming clinical trials to monitor subjects’
immune response before, during and after drug administration.
On January 1, 2023, the Company formed Adimune,
Inc., a Delaware wholly owned subsidiary.
On January 1, 2023, the Company formed Pearsanta,
Inc., a Delaware majority owned subsidiary.
On April 13, 2023, the Company formed Adivir,
Inc., a Delaware wholly owned subsidiary.
On August 24, 2023, the Company formed Adivue,
Inc., a Delaware wholly owned subsidiary.
On October 16, 2023, the Company formed Adicure,
Inc., a Delaware wholly owned subsidiary.
Reverse Stock Split
On September 13, 2022, the Company effectuated
a 1 for 50 reverse stock split (the “2022 Reverse Split”). The Company’s stock began trading on a split-adjusted
basis effective on the Nasdaq Stock Market on September 14, 2022. There was no change to the number of authorized shares of the Company’s
common stock.
On August 17, 2023, the Company effectuated a
1 for 40 reverse stock split (the “2023 Reverse Split”). The Company’s stock began trading on a split-adjusted
basis effective on the Nasdaq Stock Market on August 18, 2023. There was no change to the number of authorized shares of the Company’s
common stock. All share amounts referenced in this report are adjusted to reflect the 2023 Reverse Split.
Offerings
On August 31, 2021, the Company completed a registered
direct offering (“August 2021 Offering”). In connection therewith, the Company issued 2,292 shares of common stock,
at a purchase price of $4,800.00 per share, resulting in gross proceeds of approximately $11.0 million. In a concurrent private
placement, the Company issued warrants to purchase up to 2,292 shares. The warrants have an exercise price of $5,060.00 per
share and are exercisable for a five-year period commencing months from the date of issuance. The warrants exercise price
was subsequently repriced to $3,000.00. In addition, the Company issued a warrant to the placement agent to purchase up to 115 shares
of common stock at an exercise price of $6,000.00 per share.
On October 18, 2021, the Company entered into
an underwriting agreement with Revere Securities LLC, relating to the public offering (the “October 2021 Offering”) of 1,417 shares
of the Company’s common stock (the “Shares”) by the Company. The Shares were offered, issued, and sold at a price to
the public of $3,000.00 per share under a prospectus supplement and accompanying prospectus filed with the SEC pursuant to an effective
shelf registration statement filed with the SEC on Form S-3 (File No. 333-257645), which was declared effective by the SEC on July 13,
2021. The October 2021 Offering closed on October 20, 2021 for gross proceeds of $4.25 million. The Company utilized a portion of
the proceeds, net of underwriting discounts of approximately $3.91 million from the October 2021 Offering to fund certain obligations
of the Company.
On December 6, 2021, the Company completed a
public offering for net proceeds of $16.0 million (the “December 2021 Offering”). As part of the December 2021 Offering,
we issued 4,123 units consisting of shares of the Company’s common stock and warrant to purchase shares of the Company’s
common stock and 4,164 prefunded warrants. The warrant issued as part of the units had an exercise price of $2,300.00 and
the prefunded warrants had an exercise price of $0.04. On June 15, 2022, the Company entered an agreement with a holder of certain warrants
in the December 2021 Offering. (See Note 10)
On September 20, 2022, the Company completed
a public offering for net proceeds of $17.2 million (the “September 2022 Offering”). As part of the September 2022 Offering,
we issued 30,608 of shares of the Company’s common stock, pre-funded warrants to purchase 52,725 shares of
common stock, and warrants to purchase 83,333 shares of the Company’s common stock. The warrants had an exercise price
of $240.00 and the pre-funded warrants had an exercise price of $0.04.
On April 20, 2023, the Company entered into a
securities purchase agreement (the “April Purchase Agreement”) with an institutional investor, pursuant to which the Company
agreed to sell to such investor pre-funded warrants (the “April Pre-Funded Warrants”) to purchase up to 39,634 shares
of common stock of the Company (the “Common Stock”) at a purchase price of $48.76 per April Pre-Funded Warrant. The
April Pre-Funded Warrants (and shares of common stock underlying the April Pre-Funded Warrants) were offered by the Company pursuant
to its shelf registration statement on Form S-3 (File No. 333-257645), which was declared effective by the Securities and Exchange Commission
on July 13, 2021. Concurrently with the sale of the April Pre-Funded Warrants, pursuant to the Purchase Agreement in a concurrent
private placement, for each April Pre-Funded Warrant purchased by the investor, such investor received from the Company an unregistered
warrant (the “Warrant”) to purchase two shares of Common Stock. The warrants have an exercise price of $34.40 per
share, and are exercisable for a three year period. In addition, the Company issued a warrant to the placement agent to purchase
up to 2,378 shares of common stock at an exercise price of $61.00 per share. The closing of the sales of these securities under the April
Purchase Agreement took place on April 24, 2023. The gross proceeds from the offering were approximately $1.9 million, prior to
deducting placement agent’s fees and other offering expenses payable by the Company.
On August 31, 2023, the “Company entered
into a securities purchase agreement (the “August Purchase Agreement”) with an institutional investor for the issuance
and sale in a private placement (the “Private Placement”) of (i) pre-funded warrants (the “August Pre-Funded Warrants”)
to purchase up to 1,000,000 shares of the Company’s common stock at an exercise price of $0.001 per share, and (ii) warrants (the
“Common Warrants”) to purchase up to 1,000,000 shares of the Company’s Common Stock at an exercise price of $10.00
per share. The Private Placement closed on September 6, 2023. The net proceeds to the Company from the Private Placement were approximately
$9 million, after deducting placement agent fees and expenses and estimated offering expenses payable by the Company. The Company used
the net proceeds received from the Private Placement for (i) the payment of approximately $3.1 million in outstanding obligations, (ii)
the repayment of approximately $0.4 million of outstanding debt, and (iii) the balance for continuing operating expenses and working
capital.
On December 29, 2023, the Company entered into
a securities purchase agreement (the “Purchase Agreement”) with an institutional investor (“the “Purchaser”)
for the issuance and sale in a private placement (the “Private Placement”) of (i) pre-funded warrants (the “Pre-Funded
Warrants”) to purchase up to 1,237,114 shares of the Company’s common stock, par value $0.001 (the “Common Stock”)
at an exercise price of $0.001 per share, and (ii) warrants (the “Common Warrants”) to purchase up to 2,474,228 shares of
the Company’s Common Stock, at a purchase price of $4.85 per share. The Private Placement closed and the funds were received on
January 4, 2024. The net proceeds to the Company from the Private Placement were approximately $5.4 million, after deducting placement
agent fees and expenses and estimated offering expenses payable by the Company. The Company intends to use the net proceeds received
from the Private Placement for continuing operating expenses and working capital.
Risks and Uncertainties
The Company has a limited operating history and
is in the very early stages of generating revenue from intended operations. The Company’s business and operations are sensitive
to general business and economic conditions in the U.S. and worldwide along with local, state, and federal governmental policy decisions.
A host of factors beyond the Company’s control could cause fluctuations in these conditions. Adverse conditions may include: changes
in the biotechnology regulatory environment, technological advances that render our technologies obsolete, availability of resources
for clinical trials, acceptance of technologies into the medical community, and competition from larger, more well-funded companies.
These adverse conditions could affect the Company’s financial condition and the results of its operations.
NOTE 2 – GOING CONCERN ANALYSIS
Management Plans
The Company was incorporated on September 28,
2017 and has not generated significant revenues to date. During the year ended December 31, 2023, the Company had a net loss of $32,390,447 and
negative cash flow from operating activities of $18,576,811. As of December 31, 2023, the Company’s cash balance was $97,102.
As of December 31, 2023, the Company had approximately
$1.8 million of availability to sell under its shelf registration statement on Form S-3. Upon the filing of the Company’s annual
report on Form 10-K on April 17, 2023, the Company’s aggregate market value of the voting and non-voting equity held by non-affiliates
was below $75.0 million. As a result, the maximum amount that the Company can sell under its shelf registration statement on Form
S-3 during any 12 month period is equal to one-third of the aggregate market value of the voting and non-voting equity held by non-affiliates
of the Company.
On November 21, 2023, the Company received written
notice from Nasdaq that we had regained compliance with the Public Float Rule. On December 29, 2023, the Company received written notice
from Nasdaq that we had regained compliance with the Stockholders’ Equity Rule but will be subject to a Mandatory Panel Monitor
for a period of one year.
If we are delisted from Nasdaq, but obtain a
substitute listing for our common stock, it will likely be on a market with less liquidity, and therefore experience potentially more
price volatility than experienced on Nasdaq. Stockholders may not be able to sell their shares of common stock on any such substitute
market in the quantities, at the times, or at the prices that could potentially be available on a more liquid trading market. As a result
of these factors, if our common stock is delisted from Nasdaq, the value and liquidity of our common stock, warrants and pre-funded warrants
would likely be significantly adversely affected. A delisting of our common stock from Nasdaq could also adversely affect our ability
to obtain financing for our operations and/or result in a loss of confidence by investors, employees and/or business partners.
The Company continues to actively pursue numerous
capital raising transactions with the objective of obtaining sufficient bridge funding to meet the Company’s existing capital needs
as well as more substantial capital raises to meet the Company’s longer-term needs.
In addition, factors such as stock price, volatility,
trading volume, market conditions, demand and regulatory requirements may adversely affect the Company’s ability to raise capital
in an efficient manner. Because of these factors, the Company believes that this creates substantial doubt with the Company’s ability
to continue as a going concern.
In addition to the shelf registration, the Company
has the ability to raise capital from equity or debt through private placements or public offerings pursuant to a registration statement
on Form S-1. We may also secure loans from related parties.
The financial statements included in this report
do not include any adjustments to reflect the possible future effects on the recoverability and classification of assets or the amounts
and classification of liabilities that may result from the matters discussed herein. The Company’s ability to continue as a going
concern is dependent upon the ability to complete clinical studies and implement the business plan, generate sufficient revenues and
to control operating expenses. In addition, the Company is consistently focused on raising capital, strategic acquisitions and alliances,
and other initiatives to strengthen the Company.
NOTE 3 – SUMMARY OF SIGNIFICANT ACCOUNTING
POLICIES
Basis of Presentation
The Company’s financial statements have
been prepared in accordance with accounting principles generally accepted in the United States of America (“U.S. GAAP”) and
the rules and regulations of the Securities and Exchange Commission (“SEC”).
Principles of Consolidation
The consolidated financial statements include
the accounts of Aditxt, Inc., its wholly owned subsidiaries and, one majority owned subsidiary. All significant intercompany balances
and transactions have been eliminated in the consolidated financial statements.
Use of Estimates
The preparation of financial statements in conformity
with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure
of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenue and expense during the
reporting period. Actual results could differ from those estimates. Significant estimates underlying the financial statements include
the collectability of notes receivable, the reserve on insurance billing, value of preferred shares issued, our investments in preferred
shares, estimation of discounts on non-interest bearing borrowing, and the fair value of stock options and warrants.
Fair Value Measurements and Fair Value
of Financial Instruments
The Company adopted Financial Accounting Standards
Board (“FASB”) Accounting Standards Codification (“ASC”) Topic 820, Fair Value Measurements. ASC Topic 820 clarifies
the definition of fair value, prescribes methods for measuring fair value, and establishes a fair value hierarchy to classify the inputs
used in measuring fair value as follows:
| |
Level 1 |
- |
Inputs are unadjusted quoted
prices in active markets for identical assets or liabilities available at the measurement date. |
| |
Level 2 |
- |
Inputs are unadjusted quoted
prices for similar assets and liabilities in active markets, quoted prices for identical or similar assets and liabilities in markets
that are not active, inputs other than quoted prices that are observable, and inputs derived from or corroborated by observable market
data. |
| |
Level 3 |
- |
Inputs are unobservable
inputs which reflect the reporting entity’s own assumptions on what assumptions the market participants would use in pricing
the asset or liability based on the best available information. |
The Company did not identify any assets or liabilities
that are required to be presented on the balance sheets at fair value in accordance with ASC Topic 820.
Due to the short-term nature of all financial
assets and liabilities, their carrying value approximates their fair value as of the balance sheet dates. (See Note 9)
Concentrations of Credit Risk
Financial instruments that potentially subject
the Company to concentrations of credit risk consist primarily of cash and cash equivalents and accounts receivable.
The Company maintains its cash accounts at financial
institutions which are insured by the Federal Deposit Insurance Corporation. At times, the Company may have deposits in excess of federally
insured limits.
Substantially all the Company’s accounts
receivable are with companies in the healthcare industry, individuals, and the U.S. government. However, concentration of credit risk
is mitigated due to the Company’s number of customers. In addition, for receivables due from U.S. government agencies, the Company
does not believe the receivables represent a credit risk as these are related to healthcare programs funded by the U.S. government and
payment is primarily dependent upon submitting the appropriate documentation.
Cash and Cash Equivalents
Cash and cash equivalents include short-term,
liquid investments.
Inventory
Inventory consists of laboratory materials and
supplies used in laboratory analysis. We capitalize inventory when purchased. Inventory is valued at the lower of cost or net realizable
value on a first-in, first-out basis. We periodically perform obsolescence assessments and write off any inventory that is no longer
usable.
Fixed Assets
Fixed assets are stated at cost less accumulated
depreciation. Cost includes expenditures for furniture, office equipment, laboratory equipment, and other assets. Maintenance and repairs
are charged to expense as incurred. When assets are sold, retired, or otherwise disposed of, the cost and accumulated depreciation are
removed from the accounts and any resulting gain or loss is reflected in operations. The costs of fixed assets are depreciated using
the straight-line method over the estimated useful lives or lease life of the related assets.
Useful lives assigned to fixed assets are as
follows:
| Computers |
|
Three years to five years |
| Lab Equipment |
|
Seven to ten years |
| Office Furniture |
|
Five to ten years |
| Other fixed assets |
|
Five to ten years |
| Leasehold Improvements |
|
Shorter of estimated useful life or remaining lease
term |
Intangible Assets
Intangible assets are stated at cost less accumulated
amortization. For intangible assets that have finite lives, the assets are amortized using the straight-line method over the estimated
useful lives of the related assets. For intangible assets with indefinite lives, the assets are tested periodically for impairment.
Investments
The following table sets forth a summary of the
changes in equity investments. This investment has been recorded at cost in accordance with ASC 321.
| | |
For the year
ended
December 31,
2023 | |
| | |
| |
| As of December 31, 2022 | |
| - | |
| Purchase of equity investments | |
| 22,711,211 | |
| Unrealized gains | |
| - | |
| As of December 31, 2023 | |
$ | 22,711,221 | |
This investment is included in its own line item
on the Company’s consolidated balance sheet.
Non-marketable equity investments (for which
we do not have significant influence or control) are investments without readily determinable fair values that are recorded based on
initial cost minus impairment, if any, plus or minus adjustments resulting from observable price changes in orderly transactions for
identical or similar securities, if any. All gains and losses on investments in non-marketable equity securities, realized and unrealized,
are recognized in investment and other income (expense), net.
We monitor equity method and non-marketable equity
investments for events or circumstances that could indicate the investments are impaired, such as a deterioration in the investee’s
financial condition and business forecasts and lower valuations in recently completed or anticipated financings, and recognize a charge
to investment and other income (expense), net for the difference between the estimated fair value and the carrying value. For equity
method investments, we record impairment losses in earnings only when impairments are considered other-than-temporary.
Accounts Receivable and Allowance for Doubtful
Accounts
Accounts receivable are stated at the amount
management expects to collect from outstanding balances. The Company generally does not require collateral to support customer receivables.
The Company determines if receivables are past due based on days outstanding, and amounts are written off when determined to be uncollectible
by management. As of December 31, 2023 and 2022, there was an allowance for doubtful accounts of zero and $18,634, respectively.
Accounts receivable is made up on billed and unbilled of $236,605 and $171,721 as of December 31, 2023 and $527,961 and zero as of December
31, 2022, respectively.
Income Taxes
Deferred
tax assets and liabilities are recognized for the future tax consequences attributable to differences between the financial statement
carrying amounts of existing assets and liabilities and their respective tax bases and operating loss and tax credit carry forwards.
Deferred tax assets and liabilities are measured using enacted tax rates expected to apply to taxable income in the years in which those
temporary differences are expected to be recovered or settled. The effect on deferred tax assets and liabilities of a change in tax rates
is recognized in income in the period that includes the enactment date. At December 31, 2023 and December 31, 2022, the Company had a
full valuation allowance against its deferred tax assets.
Offering Costs
Offering costs incurred in connection with equity
are recorded as a reduction of equity and offering costs incurred in connection with debt are recorded as a reduction of debt as a debt
discount. Equity instruments issued as offering costs have zero net effect on the Company’s equity.
Revenue Recognition
In accordance with ASC 606 (Revenue From Contracts
with Customers), revenue is recognized when a customer obtains control of promised services. The amount of revenue recognized reflects
the consideration to which the Company expects to be entitled to receive in exchange for these services. To achieve this core principle,
the Company applies the following five steps:
| |
1) |
Identify the contract
with a customer |
| |
2) |
Identify the performance
obligations in the contract |
| |
3) |
Determine the transaction
price |
| |
4) |
Allocate the transaction
price to performance obligations in the contract |
| |
5) |
Recognize revenue when
or as the Company satisfies a performance obligation |
Revenues reported from services relating to the
AditxtScore™ are recognized when the AditxtScoreTM report is delivered to the customer. The services performed
include the analysis of specimens received in the Company’s CLIA laboratory and the generation of results which are then delivered
upon completion.
The Company recognizes revenue in the following
manner for the following types of customers:
Client Payers:
Client payers include physicians or other entities
for which services are billed based on negotiated fee schedules. The Company principally estimates the allowance for credit losses for
client payers based on historical collection experience and the period of time the receivable has been outstanding.
Cash Pay:
Customers are billed based on established patient
fee schedules or fees negotiated with physicians on behalf of their patients. Collection of billings is subject to credit risk and the
ability of the patients to pay.
Insurance:
Reimbursements from healthcare insurers are based
on fee for service schedules. Net revenues recognized consist of amounts billed net of contractual allowances for differences between
amounts billed and the estimated consideration the Company expects to receive from such payers, collection experience, and the terms
of the Company’s contractual arrangements.
Leases
Under Topic 842 (Leases), operating lease expense
is generally recognized evenly over the term of the lease. The Company has operating leases consisting of office space, laboratory space,
and lab equipment.
Leases with an initial term of twelve months
or less are not recorded on the balance sheet. We combine the lease and non-lease components in determining the lease liabilities and
right of use (“ROU”) assets.
Stock-Based Compensation
The Company accounts for stock-based compensation
costs under the provisions of ASC 718, Compensation—Stock Compensation, which requires the measurement and recognition of compensation
expense related to the fair value of stock-based compensation awards that are ultimately expected to vest. Stock-based compensation expense
recognized includes the compensation cost for all stock-based payments granted to employees, officers, and directors based on the grant
date fair value estimated in accordance with the provisions of ASC 718. ASC 718 is also applied to awards modified, repurchased, or cancelled
during the periods reported. Stock-based compensation is recognized as expense over the employee’s requisite vesting period and
over the nonemployee’s period of providing goods or services.
Patents
The Company incurs fees from patent licenses,
which are reflected in research and development expenses, and are expensed as incurred. During the years ended December 31, 2023 and
2022, the Company incurred patent licensing fees of $123,541 and $263,273, respectively.
Research and Development
We incur research and development costs during
the process of researching and developing our technologies and future offerings. We expense these costs as incurred unless such costs
qualify for capitalization under applicable guidance. During the years ended December 31, 2023 and 2022, the Company incurred research
and development costs of $7,074,339 and $7,268,084, respectively.
Non-controlling
Interest in Subsidiary
Non-controlling
interests represent the Company’s subsidiary’s cumulative results of operations and changes in deficit attributable to non-controlling
shareholders. During the years ended December 31, 2023 and 2022, the Company recognized $9,608 and $0 in net loss attributable to non-controlling
interest in Pearsanta. The Company owns approximately 97.5% of Pearsanta, Inc., as of December 31, 2023.
Basic and Diluted Net Loss per Common Share
Basic loss per common share is computed by dividing
the net loss by the weighted average number of shares of common stock outstanding for each period. Diluted loss per share is computed
by dividing the net loss attributable of common stockholders by the weighted average number of shares of common stock outstanding plus
the dilutive effect of shares issuable through the common stock equivalents. The weighted-average number of common shares outstanding
excludes common stock equivalents because their inclusion would be anti-dilutive. As of December 31, 2023, 45,572 stock options, 0 unvested restricted
stock units, 5,047,451 warrants, 22,280 shares of preferred series A-1 stock, and 2,625 shares of preferred series B-2 stock were excluded
from dilutive earnings per share as their effects were anti-dilutive. As of December 31, 2022, 1,105 stock options, 180 unvested
restricted stock units, and 127,251 warrants were excluded from dilutive earnings per share as their effects were anti-dilutive.
Recent Accounting Pronouncements
The FASB issues ASUs to amend the authoritative
literature in ASC. There have been several ASUs to date, including those above, that amend the original text of ASC. Management believes
that those issued to date either (i) provide supplemental guidance, (ii) are technical corrections, (iii) are not applicable to us or
(iv) are not expected to have a significant impact on our financial statements.
NOTE 4 – FIXED ASSETS
The Company’s fixed assets include the
following on December 31, 2023:
| | |
Cost Basis | | |
Accumulated
Depreciation | | |
Net | |
| Computers | |
$ | 378,480 | | |
$ | (320,473 | ) | |
$ | 58,007 | |
| Lab Equipment | |
| 2,585,077 | | |
| (859,612 | ) | |
| 1,725,465 | |
| Office Furniture | |
| 56,656 | | |
| (13,866 | ) | |
| 42,790 | |
| Other Fixed Assets | |
| 8,605 | | |
| (2,084 | ) | |
| 6,521 | |
| Leasehold Improvements | |
| 120,440 | | |
| (54,980 | ) | |
| 65,460 | |
| Total Fixed Assets | |
$ | 3,149,258 | | |
$ | (1,251,015 | ) | |
$ | 1,898,243 | |
The Company’s fixed assets include the
following on December 31, 2022:
| | |
Cost Basis | | |
Accumulated
Depreciation | | |
Net | |
| Computers | |
$ | 376,429 | | |
$ | (197,907 | ) | |
$ | 178,522 | |
| Lab Equipment | |
| 2,572,720 | | |
| (579,015 | ) | |
| 1,993,705 | |
| Office Furniture | |
| 56,656 | | |
| (8,200 | ) | |
| 48,456 | |
| Other Fixed Assets | |
| 8,605 | | |
| (1,224 | ) | |
| 7,381 | |
| Leasehold Improvements | |
| 120,440 | | |
| (29,641 | ) | |
| 90,799 | |
| Total Fixed Assets | |
$ | 3,134,850 | | |
$ | (815,987 | ) | |
$ | 2,318,863 | |
Depreciation expense was $435,027 and $428,977
for the years ended December 31, 2023 and 2022, respectively. As of December 31, 2023 and 2022, the fixed assets that serve as collateral
subject to the financed asset liability have a carrying value of $1,316,830 and $1,359,091, respectively.
Financed Assets:
In October 2020, the Company purchased two pieces
of lab equipment and financed them for a period of twenty-four months with a monthly payment of $19,487, with an interest rate of 8%.
As of December 31, 2023, the Company has one payment in arrears.
In January of 2021, the Company purchased one
piece of lab equipment and financed it for a period of twenty-four months with a monthly payment of $9,733, with an interest rate of 8%.
As of December 31, 2023, the Company has one payment in arrears.
In March of 2021, the Company purchased five
pieces of lab equipment and financed them for a period of twenty-four months with a monthly payment of $37,171, with an interest rate
of 8%. As of December 31, 2023, the Company has four payments in arrears.
As of December 31, 2023, all lab equipment financing
agreements have matured and are in default status.
NOTE 5 – INTANGIBLE ASSETS
The Company’s intangible assets include
the following on December 31, 2023:
| | |
Cost Basis | | |
Accumulated
Amortization | | |
Net | |
| Proprietary Technology | |
$ | 321,000 | | |
$ | (321,000 | ) | |
$ | - | |
| Intellectual property | |
| 10,000 | | |
| 556 | | |
| 9,444 | |
| Total Intangible Assets | |
$ | 321,000 | | |
$ | (321,556 | ) | |
$ | 9,444 | |
The Company’s intangible assets include
the following on December 31, 2022:
| | |
Cost Basis | | |
Accumulated
Amortization | | |
Net | |
| Proprietary Technology | |
$ | 321,000 | | |
$ | (214,000 | ) | |
$ | 107,000 | |
| Total Intangible Assets | |
$ | 321,000 | | |
$ | (214,000 | ) | |
$ | 107,000 | |
Amortization expense was $107,556 and $107,000 for
the years ended December 31, 2023 and 2022, respectively. The Company’s proprietary technology is being amortized over its estimated
useful life of three years.
NOTE 6 – RELATED PARTY TRANSACTIONS
On January 28, 2022, the Company granted 9,600 restricted
stock units to an officer of the Company pursuant to the Company’s 2021 Equity Incentive Plan. The Company recognized $146,613 in
stock-based compensation for the issuance of these vested and unvested restricted stock units during the year ended December 31, 2022.
(Note 11)
On July 19, 2022, the Company entered into a
Subscription and Investment Representation Agreement with its Chief Executive Officer (the “Purchaser”), pursuant to which
the Company agreed to issue and sell one (1) share of the Company’s Series B Preferred Stock (the “Series B Preferred Stock”),
par value $0.001 per share, to the Purchaser for $20,000 in cash.
On July 19, 2022, the Company filed a certificate
of designation (the “Certificate of Designation”) with the Secretary of State of Delaware, effective as of the time of filing,
designating the rights, preferences, privileges and restrictions of the share of Series B Preferred Stock. The Certificate of Designation
provides that the share of Series B Preferred Stock will have 250,000,000 votes and will vote together with the outstanding
shares of the Company’s common stock as a single class exclusively with respect to any proposal to amend the Company’s Restated
Certificate of Incorporation to effect a reverse stock split of the Company’s common stock. The Series B Preferred Stock will be
voted, without action by the holder, on any such proposal in the same proportion as shares of common stock are voted. The Series B Preferred
Stock otherwise has no voting rights except as otherwise required by the General Corporation Law of the State of Delaware.
The Series B Preferred Stock is not convertible
into, or exchangeable for, shares of any other class or series of stock or other securities of the Company. The Series B Preferred Stock
has no rights with respect to any distribution of assets of the Company, including upon a liquidation, bankruptcy, reorganization, merger,
acquisition, sale, dissolution or winding up of the Company, whether voluntarily or involuntarily. The holder of the Series B Preferred
Stock will not be entitled to receive dividends of any kind.
The outstanding share of Series B Preferred Stock
shall be redeemed in whole, but not in part, at any time (i) if such redemption is ordered by the Board of Directors in its sole discretion
or (ii) automatically upon the effectiveness of the amendment to the Certificate of Incorporation implementing a reverse stock split.
Upon such redemption, the holder of the Series B Preferred Stock will receive consideration of $20,000 in cash. On September 13,
2022, the share was redeemed.
On July 19, 2022, the Company filed a certificate
of designation (the “Certificate of Designation”) with the Secretary of State of Delaware, effective as of the time of filing,
designating the rights, preferences, privileges and restrictions of the share of Series B Preferred Stock. The Certificate of Designation
provides that the share of Preferred Stock will have 250,000,000 votes and will vote together with the outstanding shares of
the Company’s common stock as a single class exclusively with respect to any proposal to amend the Company’s Restated Certificate
of Incorporation to effect a reverse stock split of the Company’s common stock. The Series B Preferred Stock will be voted, without
action by the holder, on any such proposal in the same proportion as shares of common stock are voted. The Series B Preferred Stock otherwise
has no voting rights except as otherwise required by the General Corporation Law of the State of Delaware.
On July 21, 2022, the Chief Executive Officer
loaned $80,000 to the Company. The loan was evidenced by an unsecured promissory note (the “July 2022 Promissory Note”).
Pursuant to the terms of the July 2022 Promissory Note, it will accrue interest at a rate of four and three-quarters percent (4.75%)
per annum, the Prime rate on the date of signing, and is due on the earlier of January 22, 2023, or an event of default. On October 7,
2022, the Company fully repaid the $80,000 July 2022 Promissory Note and $812 of accrued interest to its Chief Executive Officer. The
Chief Executive Officer and the Company entered the July 2022 Promissory Note on July 21, 2022.
On April 21, 2023, Amro Albanna, the Chief Executive
Officer of the Company, and Shahrokh Shabahang, the Chief Innovation Officer of the Company, loaned $87,523 and $100,000, respectively,
to the Company. The loans were each evidenced by an unsecured promissory note (the “April Note”). Pursuant to the terms each
April Note, it will accrue interest at the Prime rate of eight percent (8.00%) per annum and is due on the earlier of October 21, 2023,
or an event of default, as defined therein. As of December 31, 2023, the note was fully paid off.
On May 25, 2023, Amro Albanna, the Chief Executive
Officer of the Company, loaned $200,000 to the Company. The loan was evidenced by an unsecured promissory note (the “May Note”).
Pursuant to the terms of the May Note, it will accrue interest at a rate of eight and one-quarter percent (8.25%) per annum, the Prime
rate on the date of signing, and is due on the earlier of November 25, 2023 or an event of default, as defined therein. As of December
31, 2023, the note was fully paid off.
On June 12, 2023, Amro Albanna, the Chief Executive
Officer of the Company, and Shahrokh Shabahang, the Chief Innovation Officer of the Company, loaned $200,000 and $100,000, respectively,
to the Company. The loans were evidenced by an unsecured promissory note (the “June Note”). Pursuant to the terms of the
June Note, it will accrue interest at the Prime rate of eight and one-quarter percent (8.25%) per annum and is due on the earlier of
December 12, 2023, or an event of default, as defined therein. As of December 31, 2023, the June Note was fully paid off.
On July 11, 2023, the Company entered into a
Subscription and Investment Representation Agreement with the Purchaser, pursuant to which the Company agreed to issue and sell one (1)
share of the Company’s Series C Preferred Stock (the “Series C Preferred Stock”), par value $0.001 per share,
to the Purchaser for $1,000 in cash.
On July 11, 2023, the Company filed a certificate
of designation (the “Certificate of Designation”) with the Secretary of State of Delaware, effective as of the time of filing,
designating the rights, preferences, privileges and restrictions of the share of Series C Preferred Stock. The Certificate of Designation
provides that the share of Series C Preferred Stock will have 250,000,000 votes and will vote together with the outstanding
shares of the Company’s common stock as a single class exclusively with respect to any proposal to amend the Company’s Restated
Certificate of Incorporation to effect a reverse stock split of the Company’s common stock. The Series C Preferred Stock will be
voted, without action by the holder, on any such proposal in the same proportion as shares of common stock are voted. The Series C Preferred
Stock otherwise has no voting rights except as otherwise required by the General Corporation Law of the State of Delaware.
The Series C Preferred Stock is not convertible
into, or exchangeable for, shares of any other class or series of stock or other securities of the Company. The Series C Preferred Stock
has no rights with respect to any distribution of assets of the Company, including upon a liquidation, bankruptcy, reorganization, merger,
acquisition, sale, dissolution or winding up of the Company, whether voluntarily or involuntarily. The holder of the Series C Preferred
Stock will not be entitled to receive dividends of any kind.
The outstanding share of Series C Preferred Stock
shall be redeemed in whole, but not in part, at any time (i) if such redemption is ordered by the Board of Directors in its sole discretion
or (ii) automatically upon the effectiveness of the amendment to the Certificate of Incorporation implementing a reverse stock split.
Upon such redemption, the holder of the Series C Preferred Stock will receive consideration of $1,000 in cash. On August 17, 2023,
the share was redeemed.
On November 30, 2023, Amro Albanna, the Chief
Executive Officer of the Company, loaned $10,000 to the Company. The loan was evidenced by an unsecured promissory note (the “November
Note”). Pursuant to the terms of the November Note, it will accrue interest at a rate of eight and a half percent (8.50%) per annum,
the Prime rate on the date of signing, and is due on the earlier of May 30, 2024 or an event of default, as defined therein. As of December
31, 2023, there was a remaining principal balance of $10,000 on the November Loan and accrued interest of $72.
On December 6, 2023, Amro Albanna, the Chief
Executive Officer of the Company, loaned $200,000 to the Company. The loan was evidenced by an unsecured promissory note (the “First
December Note”). Pursuant to the terms of the First December Note, it will accrue interest at a rate of eight and a half percent
(8.50%) per annum, the Prime rate on the date of signing, and is due on the earlier of June 6, 2024 or an event of default, as defined
therein. As of December 31, 2023, there was a remaining principal balance of $200,000 on the First December Loan and accrued interest
of $1,164.
On December 20, 2023, Amro Albanna, the Chief
Executive Officer of the Company, loaned $165,000 to the Company. The loan was evidenced by an unsecured promissory note (the “Second
December Note”). Pursuant to the terms of the Second December Note, it will accrue interest at a rate of eight and a half percent
(8.50%) per annum, the Prime rate on the date of signing, and is due on the earlier of June 20, 2024 or an event of default, as defined
therein. As of December 31, 2023, there was a remaining principal balance of $165,000 on the Second December Loan and accrued interest
of $423.
See Note 12 for additional loans incurred or
paid subsequent to December 31, 2023.
NOTE 7 – NOTES PAYABLE
On February 21, 2023, the Company entered into
an agreement for the purchase and sale of future receipts (the “Future Receipts Agreement”) with a commercial funding source
pursuant to which the Company agreed to sell to the funder certain future trade receipts in the aggregate amount of $2,160,000 (the
“Future Receipts Purchased Amount” for gross proceeds to the Company of $1,500,000, less origination fees of $75,000. Pursuant
to the Future Receipts Agreement, the Company granted the funder a security interest in all of the Company’s present and future
accounts receivable in an amount not to exceed the Future Receipts Purchased Amount. The Future Receipts Purchased Amount shall be repaid
by the Company in 28 weekly installments of approximately $77,000 with the final payment due on September 5, 2023. On May 30, 2023,
the Company entered into the May Loan (as defined below) for gross proceeds to the Company of $2,000,000, less origination fees of $100,000
and less the full outstanding balance under the Future Receipts Agreement of $1,157,143, resulting in net proceeds to the Company of
$742,857.
On April 4, 2023, the Company entered into a
Business Loan and Security Agreement (the “April Loan Agreement”) with a commercial funding source (the “April Lender”),
pursuant to which the Company obtained a loan from the April Lender in the principal amount of $1,060,000, which includes origination
fees of $60,000 (the “April Loan”). Pursuant to the April Loan Agreement, the Company granted the April Lender a continuing
secondary security interest in; (i) any and all amounts owed to the Company now or in the future from any merchant processor processing
charges made by customers of the Company via credit card or debit card transactions, and (ii) all other tangible and intangible property.
The total amount of interest and fees payable by the Company to the April Lender under the April Loan (the “April Repayment Amount”)
will be (i) $1,000,000 if paid prior to April 6, 2023, (ii) $1,219,000 if paid prior to April 10, 2023, or (iii) $1,590,000 if
paid after April 10, 2023, and will be repaid in 20 weekly installments of $79,500 commencing on April 10, 2023 and ending on August
21, 2023. On April 24, 2023, the Company entered into the Loan Agreement (as defined below) for gross proceeds of $1,000,000, less the
full outstanding balance under the April Loan Agreement of $139,500, resulting net proceeds to the Company of $860,500.
On April 24, 2023, the Company entered into a
Business Loan and Security Agreement (the “Loan Agreement”) with a commercial funding source (the “Lender”),
pursuant to which the Company obtained a loan from the Lender in the principal amount of $1,060,000, which includes origination fees
of $60,000 (the “Loan”). Pursuant to the Loan Agreement, the Company granted the Lender a continuing secondary security
interest in; (i) any and all amounts owed to the Company now or in the future from any merchant processor processing charges made by
customers of the Company via credit card or debit card transactions, and (ii) all other tangible and intangible property. The total amount
of interest and fees payable by the Company to the Lender under the Loan (the “April Repayment Amount”) will be $1,590,000 and
will be repaid in 20 weekly installments of $79,500. On August 23, 2023, the April Repayment Amount was restructured in connection
with the August Loan Agreement, as defined below.
On May 30, 2023, the Company entered into a Business
Loan and Security Agreement (the “May Loan Agreement”) with a commercial funding source (the “May Lender”), pursuant
to which the Company obtained a loan from the Lender in the principal amount of $2,000,000, which includes origination fees of $100,000 (the
“May Loan”). Pursuant to the May Loan Agreement, the Company granted the May Lender a continuing secondary security interest
in; (i) any and all amounts owed to the Company now or in the future from any merchant processor processing charges made by customers
of the Company via credit card or debit card transactions, and (ii) all other tangible and intangible property. The total amount of interest
and fees payable by the Company to the Lender under the Loan will be $2,880,000 (the “May Repayment Amount) and will be repaid
in 28 weekly installments of $102,857. On October 5, 2023 the May Repayment Amount was restructured in connection with the
October MCA Agreement (as defined below).
On July 3, 2023, the Company entered into a Business
Loan and Security Agreement (the “July Loan Agreement”) with a commercial funding source (the “July Lender’’),
pursuant to which the Company obtained a loan from the Lender in the principal amount of $215,000, which includes origination fees of
$10,750 (the “July Loan”). Pursuant to the July Loan Agreement, the Company granted the July Lender a continuing secondary
security interest in certain collateral (as defined in the July Loan Agreement). The total amount of interest and fees payable by the
Company to the Lender under the Loan (the “July Repayment Amount”) will be (i) $322,285 and will be repaid in 13 weekly installments
of $24,500 with a final payment of $3,785 in the fourteenth week. As of December 31, 2023, the note was fully paid off. On August 23,
2023, the July Repayment Amount was restructured in connection with the August Loan Agreement, as defined below.
On August 23, 2023, the Company entered into
a Business Loan and Security Agreement (the “August Loan Agreement”) with a commercial funding source (the “August
Lender’’), pursuant to which the Company obtained a loan from the Lender in the principal amount of $1,400,000, which includes
origination fees of $70,000 (the “August Loan”). Pursuant to the August Loan Agreement, the Company granted the August Lender
a continuing secondary security interest in certain collateral (as defined in the August Loan Agreement). The total amount of interest
and fees payable by the Company to the Lender under the Loan (the “Repayment Amount”) will be (i) $2,079,000 (the “August
Repayment Amount”) and will be repaid in 21 weekly installments of $99,000 On November 7, 2023 the August Repayment Amount was
restructured in connection with the November Loan Agreement (as defined below).
On October 5, 2023, the Company entered into
an Agreement for the Purchase and Sale of Future Receipts (the “October MCA Agreement”) pursuant to which the existing funder
(the “Funder”) increased the existing outstanding amount to $4,470,000 (the “October MCA Purchased Amount”) for
gross proceeds to the Company of $3,000,000, less origination fees of $240,000 and the outstanding balance under the existing agreement
of $1,234,461, resulting in net proceeds to the Company of $1,525,539. Pursuant to the October MCA Agreement, the Company granted the
Funder a security interest in all of the Company’s present and future accounts receivable in an amount not to exceed the October
MCA Purchased Amount. The October MCA Purchased Amount shall be repaid by the Company in 30 weekly installments of $149,000. The October
Purchased Amount may be prepaid by the Company via a payment of $3,870,000 if repaid within 30 days, $4,110,000 if repaid within 60 days
and $4,230,000 if repaid within 90 days. As of December 31, 2023 the October MCA Agreement has an outstanding principal balance of $2,498,245.
The October MCA Agreement is currently in default status.
On November 7, 2023, the Company entered into
a Business Loan and Security Agreement (the “November Loan Agreement”) with the lender (the “Lender”), pursuant
to which the Company obtained a loan from the Lender in the principal amount of $2,100,000, which satisfied the outstanding balance on
the August Loan of $1,089,000 and includes origination fees of $140,000 (the “November Loan”). Pursuant to the November Loan
Agreement, the Company granted the Lender a continuing secondary security interest in certain collateral (as defined in the November
Loan Agreement). The total amount of interest and fees payable by us to the Lender under the November Loan will be $3,129,000, which
will be repaid in 34 weekly installments ranging from $69,000 - $99,000. As of December 31, 2023 the November Loan has an outstanding
principal balance of $1,990,699. The November Loan Agreement is currently in default status.
On November 24, 2023, the Company entered into
a loan with a principal of $53,099. The loan was evidenced by an unsecured promissory note (the “Second November Note”).
Pursuant to the terms of the Second November Note, it will accrue interest at a rate of eight and a half percent (8.50%) per annum, the
Prime rate on the date of signing, and is due on the earlier of May 24, 2024 or an event of default, as defined therein. As of December
31, 2023, there was a remaining principal balance of $53,099 on the Second December Loan and accrued interest of $458.
Securities Purchase Agreement
On July 3, 2023, the Company entered into a Securities
Purchase Agreement (the “First Tranche Securities Purchase Agreement”) with an accredited investor pursuant to which the
Company issued and sold a secured promissory note in the principal amount of $375,000 (the “First Tranche Note”) resulting
in gross proceeds to the Company of $250,000. In connection with the issuance of the First Tranche Note, the Company issued 3,907 shares
of its common stock (the “First Tranche Commitment Shares”) as a commitment fee to the investor. Pursuant to the First Tranche
Securities Purchase Agreement, the Company was obligated to and obtained approval of its shareholders (“First Tranche Shareholder
Approval”) with respect to the issuance of any securities in connection with the First Tranche Securities Purchase Agreement and
the First Tranche Note in excess of 19.99% of the Company’s issued and outstanding shares on the closing date, which was equal
to 33,792 shares of the Company’s common stock. The Company recognized a total debt discount of $164,775 on the Note from the issuance
of stock and original issuance discount. The First Tranche Note has a maturity date of December 31, 2023, and is convertible following
First Tranche Shareholder Approval and the occurrence of an Event of Default (as defined in the July Note) at a conversion price of $18.00
per share.
In connection with the First Tranche Securities
Purchase Agreement and the issuance of the First Tranche Note, the Company and certain of its subsidiaries also entered into a Security
Agreement with the investor (the “First Tranche Security Agreement”) pursuant to which it granted the investor a security
interest in certain Collateral (as defined in the First Tranche Security Agreement) to secure its obligations under the First Tranche
Note. In addition, the Company entered into a registration rights agreement with the investor pursuant to which the Company agreed to
prepare and file with the U.S. Securities and Exchange Commission a registration statement covering the resale of the First Tranche Commitment
Shares and any shares of the Company’s common stock issuable upon conversion of the First Tranche Note within 120 days of the closing
date and to have such registration statement declared effective within 150 days of the closing date. As of December 31, 2023, the First
Tranche Note was fully paid off.
On July 24, 2023, the Company entered into a
Securities Purchase Agreement (the “Second Tranche Securities Purchase Agreement”) with an accredited investor pursuant to
which the Company issued and sold a secured promissory note in the principal amount of $2,625,000 (the “Second Tranche Note”)
resulting in gross proceeds to the Company of $1,750,000. In connection with the issuance of the Second Tranche Note, the Company agreed
to issue a total of 27,344 shares of its common stock (the “Second Tranche Commitment Shares”) as a commitment fee to the
investor. At the request of the investor, the Company issued 17,278 Second Tranche Commitment Shares and will issue the remaining 10,066
Second Tranche Commitment Shares within 120 days, subject to the investor’s discretion. Pursuant to the Second Tranche Securities
Purchase Agreement, the Company was obligated to and obtained approval of its shareholders (“Second Tranche Shareholder Approval”)
with respect to the issuance of any securities in connection with the Second Tranche Securities Purchase Agreement and the Second Tranche
Note in excess of 19.99% of the Company’s issued and outstanding shares on the closing date, which was equal to 38,026 shares of
the Company’s common stock. The company recognized a total debt discount of $1.0 million on the Second Tranche Note from the issuance
of stock and original issuance discount. The Note has a maturity date of December 31, 2023 and is convertible following Second Tranche
Shareholder Approval and the occurrence of an Event of Default (as defined in the Second Tranche Note) at a conversion price of $15.60
per share.
In connection with the Second Tranche Securities
Purchase Agreement and the issuance of the Second Tranche Note, the Company and certain of its subsidiaries also entered into a Security
Agreement with the investor (the “Second Tranche Security Agreement”) pursuant to which it granted the investor a security
interest in certain Collateral (as defined in the Second Tranche Security Agreement) to secure its obligations under the Second Tranche
Note. In addition, the Company entered into a registration rights agreement with the investor pursuant to which the Company agreed to
prepare and file with the U.S. Securities and Exchange Commission a registration statement covering the resale of the Second Tranche
Commitment Shares and any shares of the Company’s common stock issuable upon conversion of the Second Tranche Note within 90 days
of the closing date and to have such registration statement declared effective within 120 days of the closing date. As of December 31,
2023, $2,625,000 in outstanding principal on the Second Tranche Note and accrued interest of $113,021 was converted into 2,625 shares
of the Company’s Series B-2 Preferred Stock (See Note 10).
Evofem Merger
In connection with the Agreement and Plan of
Merger (the “Merger Agreement”) with Adicure, Inc., a Delaware corporation and wholly owned subsidiary of the Company (“Merger
Sub”) and Evofem Biosciences, Inc., a Delaware corporation (“Evofem”), the Company, Evofem and the holders (the “Holders”)
of certain senior indebtedness (the “Notes”) entered into an Assignment Agreement dated December 11, 2023 (the “Assignment
Agreement”), pursuant to which the Holders assigned the Notes to the Company in consideration for the issuance by the Company of
(i) an aggregate principal amount of $5 million in secured notes of the Company due on January 2, 2024 (the “January 2024 Secured
Notes”), (ii) an aggregate principal amount of $8 million in secured notes of the Company due on September 30, 2024 (the “September
2024 Secured Notes”), (iii) an aggregate principal amount of $5 million in ten-year unsecured notes (the “Unsecured Notes”),
and (iv) payment of $154,480 in respect of net sales of Phexxi in respect of the calendar quarter ended September 30, 2023, which amount
is due and payable on December 14, 2023. The January 2024 Secured Notes are secured by certain intellectual property assets of the Company
and its subsidiaries pursuant to an Intellectual Property Security Agreement (the “IP Security Agreement”) entered into in
connection with the Assignment Agreement. The September 2024 Secured Notes are secured by the Notes and certain associated security documents
pursuant to a Security Agreement (the “Security Agreement”) entered into in connection with the Assignment Agreement. As
of December 31, 2023, there was a remaining principal balance of $13,000,000 on the Notes.
Subject to the terms and conditions set forth
in the Merger Agreement, at the effective time of the Merger (the “Effective Time”), (i) all issued and outstanding shares
of common stock, par value $0.0001 per share of Evofem (“Evofem Common Stock”), other than any shares of Evofem Common Stock
held by the Company or Merger Sub immediately prior to the Effective Time, will be converted into the right to receive an aggregate of
610,000 shares of the Company’s common stock, par value $0.001 per share (“Company Common Stock”); and (ii) all issued
and outstanding shares of Series E-1 Preferred Stock, par value $0.0001 of Evofem (the “Evofem Unconverted Preferred Stock”),
other than any shares of Evofem Unconverted Preferred Stock held by the Company or Merger Sub immediately prior to the Effective Time,
will be converted into the right to receive an aggregate of 2,327 shares of Series A-1 Preferred Stock, par value $0.001 of the Company
(the “Company Preferred Stock”), having such rights, powers, and preferences set forth in the form of Certificate of Designation
of Series A-1 Preferred Stock, the form of which is attached as Exhibit C to the Merger Agreement.
The respective obligations of each of the Company,
Merger Sub and Evofem to consummate the closing of the Merger (the “Closing”) are subject to the satisfaction or waiver,
at or prior to the closing of certain conditions, including but not limited to, the following:
| |
(i) |
approval by the Company’s
shareholders and Evofem shareholders; |
| |
(ii) |
the registration statement
on Form S-4 pursuant to which the shares of the Company Common Stock issuable in the Merger being declared effective by the U.S.
Securities and Exchange Commission; |
| |
(iii) |
the entry into a voting
agreement by the Company and certain members of Evofem management; |
| |
(iv) |
all preferred stock of
Evofem other than the Evofem Unconverted Preferred Stock shall have been converted to Evofem Common Stock; |
| |
(v) |
Evofem shall have received
agreements (the “Evofem Warrant Holder Agreements”) from all holders of Evofem warrants which provide: |
a. waivers with respect to any fundamental
transaction, change in control or other similar rights that such warrant holder may have under any such Evofem warrants, and (b) an agreement
to such Evofem warrants to exchange such warrants for not more than an aggregate (for all holders of Evofem warrants) of 551 shares of
Company Preferred Stock;
| |
(vi) |
Evofem shall have cashed
out any other holder of Evofem warrants who has not provided an Evofem Warrant Holder Agreement; and |
| |
(vii) |
Evofem shall have obtained
waivers from the holders of the convertible notes of Evofem (the “Evofem Convertible Notes”) with respect to any fundamental
transaction rights that such holder may have under the Evofem Convertible Notes, including any right to vote, consent, or otherwise
approve or veto any of the transactions contemplated under the Merger Agreement. |
The obligations of the Company and Merger Sub
to consummate the Closing are subject to the satisfaction or waiver, at or prior to the Closing of certain conditions, including but
not limited to, the following:
| |
(i) |
the Company shall have
obtained agreements from the holders of Evofem Convertible Notes and purchase rights they hold to exchange such Convertible Notes
and purchase rights for not more than an aggregate (for all holders of Evofem Convertible Notes) of 86,153 shares of Company Preferred
Stock; |
| |
(ii) |
the Company shall have
received waivers form the holders of certain of the Company’s securities which contain prohibitions on variable rate transactions;
and |
| |
(iii) |
the Company, Merger Sub
and Evofem shall work together between the Execution Date and the Effective Time to determine the tax treatment of the Merger and
the other transactions contemplated by the Merger Agreement. |
The obligations of the Company to consummate
the Closing are subject to the satisfaction or waiver, at or prior to the Closing of certain conditions, including but not limited to,
the following:
| |
(i) |
the Company shall have
regained compliance with the stockholders’ equity requirement in Nasdaq Listing Rule 5550(b)(1) and shall meet all other applicable
criteria for continued listing, subject to any panel monitor imposed by Nasdaq. |
As the January 2024 Secured Notes and September
2024 Secured Notes did not contain a stated interest rate, the Company calculated an imputed interest rate of 26.7% based on the Company’s
weighted average cost of capital for the period in which the January 2024 Secured Notes and September 2024 Secured Notes were outstanding.
This amounted to approximately $1.8 million which was recorded as a discount to be amortized over the life of the January 2024 Secured
Notes and September 2024 Secured Notes.
See Note 12 for amendments entered into subsequent
to year end.
NOTE 8 – LEASES
Our lease agreements generally do not provide
an implicit borrowing rate; therefore, an internal incremental borrowing rate is determined based on information available at lease commencement
date for purposes of determining the present value of lease payments. We used the incremental borrowing rate on December 31, 2023 and
2022 for all leases that commenced prior to that date. In determining this rate, which is used to determine the present value of future
lease payments, we estimate the rate of interest we would pay on a collateralized basis, with similar payment terms as the lease and
in a similar economic environment.
Our corporate headquarters is located in Richmond,
Virginia, where we lease approximately 25,000 square feet. The lease expires in August 31, 2026, subject to extension.
As of December 31, 2023 the Company is 1.75 months in arrears on this lease.
We also lease approximately 5,810 square
feet of laboratory and office space in Mountain View, California. The lease expires in August 31, 2024, subject to extension. As
of December 31, 2023 the Company is 1 month in arrears on this lease.
Additionally, we lease approximately 3,150 square
feet of office space in Melville, New York. The lease expires in December 31, 2025, subject to extension. As of December 31, 2023
the Company is 1 month in arrears on this lease.
Lease Costs
| | |
Year Ended
December 31,
2023 | | |
Year Ended
December 31,
2022 | |
| Components of total lease costs: | |
| | |
| |
| Operating lease expense | |
$ | 1,140,949 | | |
$ | 1,396,875 | |
| Total lease costs | |
$ | 1,140,949 | | |
$ | 1,396,875 | |
Lease Positions as of December 31, 2023 and
2022
ROU lease assets and lease liabilities for our
operating leases are recorded on the balance sheet as follows:
| | |
December 31,
2023 | | |
December 31,
2022 | |
| Assets | |
| | |
| |
| Right of use asset –
long term | |
$ | 2,200,299 | | |
$ | 3,160,457 | |
| Total right of use asset | |
$ | 2,200,299 | | |
$ | 3,160,457 | |
| | |
| | | |
| | |
| Liabilities | |
| | | |
| | |
| Operating lease liabilities – short term | |
$ | 999,943 | | |
$ | 1,086,658 | |
| Operating lease liabilities –
long term | |
| 1,041,744 | | |
| 1,885,218 | |
| Total lease liability | |
$ | 2,041,687 | | |
$ | 2,971,876 | |
Lease Terms and Discount Rate as of December
31, 2023
| Weighted average remaining lease term (in years) – operating leases | |
| 1.92 | |
| Weighted average discount rate – operating leases | |
| 8.00 | % |
Maturities of leases are as follows:
Year Ended December 31, 2023
| 2024 | |
$ | 1,004,982 | |
| 2025 | |
| 710,546 | |
| 2026 | |
| 423,930 | |
| Total lease payments | |
$ | 2,139,458 | |
| Less imputed interest | |
| (97,771 | ) |
| Less current portion | |
| (999,943 | ) |
| Total maturities, due beyond one year | |
$ | 1,041,744 | |
See Note 12 for additional disclosure regarding
the Company’s leases.
NOTE 9 – COMMITMENTS & CONTINGENCIES
License Agreement with Loma Linda University
On March 15, 2018, as amended on July 1, 2020,
we entered into a LLU License Agreement directly with Loma Linda University.
Pursuant to the LLU License Agreement, we obtained
the exclusive royalty-bearing worldwide license in and to all intellectual property, including patents, technical information, trade
secrets, proprietary rights, technology, know-how, data, formulas, drawings, and specifications, owned or controlled by LLU and/or any
of its affiliates (the “LLU Patent and Technology Rights”) and related to therapy for immune-mediated inflammatory diseases
(the ADI™ technology). In consideration for the LLU License Agreement, we issued 13 shares of common stock to LLU.
Pursuant to the LLU License Agreement, we are
required to pay an annual license fee to LLU. Also, we paid LLU $455,000 in July 2020 for outstanding milestone payments and license
fees. We are also required to pay to LLU milestone payments in connection with certain development milestones. Specifically, we are required
to make the following milestone payments to LLU: $175,000 on March 31, 2022; $100,000 on March 31, 2024; $500,000 on March 31, 2026;
and $500,000 on March 31, 2027. In lieu of the $175,000 milestone payment due on March 31, 2023, the Company paid LLU an extension fee
of $100,000. Upon payment of this extension fee, an additional year will be added for the March 31, 2023 milestone. Additionally, as
consideration for prior expenses incurred by LLU to prosecute, maintain and defend the LLU Patent and Technology Rights, we made the
following payments to LLU: $70,000 at the end of December 2018, and a final payment of $60,000 at the end of March 2019. We are required
to defend the LLU Patent and Technology Rights during the term of the LLU License Agreement. Additionally, we will owe royalty payments
of (i) 1.5% of Net Product Sales (as such terms are defined under the LLU License Agreement) and Net Service Sales on any Licensed Products
(defined as any finished pharmaceutical products which utilizes the LLU Patent and Technology Rights in its development, manufacture
or supply), and (ii) 0.75% of Net Product Sales and Net Service Sales for Licensed Products and Licensed Services (as such terms are
defined under the LLU License Agreement) not covered by a valid patent claim for technology rights and know-how for a three (3) year
period beyond the expiration of all valid patent claims. We also are required to produce a written progress report to LLU, discussing
our development and commercialization efforts, within 45 days following the end of each year. All intellectual property rights in and
to LLU Patent and Technology Rights shall remain with LLU (other than improvements developed by or on our behalf).
The LLU License Agreement shall terminate on
the last day that a patent granted to us by LLU is valid and enforceable or the day that the last patent application licensed to us is
abandoned. The LLU License Agreement may be terminated by mutual agreement or by us upon 90 days written notice to LLU. LLU may terminate
the LLU License Agreement in the event of (i) non-payments or late payments of royalty, milestone and license maintenance fees not cured
within 90 days after delivery of written notice by LLU, (ii) a breach of any non-payment provision (including the provision that requires
us to meet certain deadlines for milestone events (each, a “Milestone Deadline”)) not cured within 90 days after delivery
of written notice by LLU and (iii) LLU delivers notice to us of three or more actual breaches of the LLU License Agreement by us in any
12-month period. Additional Milestone Deadlines include: (i) the requirement to have regulatory approval of an IND application to initiate
first-in-human clinical trials on or before March 31, 2023, which will be extended to March 31, 2024 with a payment of a $100,000 extension
fee, (ii) the completion of first-in-human (phase I/II) clinical trials by March 31, 2024, (iii) the completion of Phase III clinical
trials by March 31, 2026 and (iv) biologic licensing approval by the FDA by March 31, 2027.
License Agreement with Leland Stanford Junior University
On February 3, 2020, we entered into an exclusive
license agreement (the “February 2020 License Agreement”) with Stanford regarding a patent concerning a method for detection
and measurement of specific cellular responses. Pursuant to the February 2020 License Agreement, we received an exclusive worldwide license
to Stanford’s patent regarding use, import, offer, and sale of Licensed Products (as defined in the agreement). The license to
the patented technology is exclusive, including the right to sublicense, beginning on the effective date of the agreement, and ending
when the patent expires. Under the exclusivity agreement, we acknowledged that Stanford had already granted a non-exclusive license in
the Nonexclusive Field of Use, under the Licensed Patents in the Licensed Field of Use in the Licensed Territory (as those terms are
defined in the February 2020 License Agreement”). However, Stanford agreed to not grant further licenses under the Licensed Patents
in the Licensed Field of Use in the Licensed Territory. On December 29, 2021, we entered into an amendment to the February 2020 License
Agreement which extended our exclusive right to license the technology deployed in AditxtScoreTM and securing worldwide
exclusivity in all fields of use of the licensed technology.
We were obligated to pay and paid a fee of $25,000
to Stanford within 60 days of February 3, 2020. We also issued 10 shares of the Company’s common stock to Stanford.
An annual licensing maintenance fee is payable by us on the first anniversary of the February 2020 License Agreement in the amount of
$40,000 for 2021 through 2024 and $60,000 starting in 2025 until the license expires upon the expiration of the patent. The Company is
required to pay and has paid $25,000 for the issuances of certain patents. The Company will pay milestone fees of $50,000 on the first
commercial sales of a licensed product and $25,000 at the beginning of any clinical study for regulatory clearance of an in vitro diagnostic
product developed and a potential licensed product. The Company paid a milestone fee for a clinical study for regulatory clearance of
an in vitro diagnostic product developed and a potential licensed product of $25,000 in March of 2022. We are also required to: (i) provide
a listing of the management team or a schedule for the recruitment of key management positions by March 31, 2020 (which has been completed),
(ii) provide a business plan covering projected product development, markets and sales forecasts, manufacturing and operations, and financial
forecasts until at least $10,000,000 in revenue by June 30, 2020 (which has been completed), (iii) conduct validation studies by September
30, 2020 (which has been completed), (iv) hold a pre-submission meeting with the FDA by September 30, 2020 (which has been completed),
(iv) submit a 510(k) application to the FDA, Emergency Use Authorization (“EUA”), or a Laboratory Developed Test (“LDT”)
by March 31, 2021 (which has been completed), (vi) develop a prototype assay for human profiling by December 31, 2021 (which has been
completed), (vii) execute at least one partnership for use of the technology for transplant, autoimmunity, or infectious disease purposes
by March 31, 2022 (which has been completed) and (viii) provided further development and commercialization milestones for specific fields
of use in writing prior to December 31, 2022.
In addition to the annual license maintenance
fees outlined above, we will pay Stanford royalties on Net Sales (as such term is defined in the February 2020 License Agreement) during
the of the term of the agreement as follows: 4% when Net Sales are below or equal to $5 million annually or 6% when Net Sales are above
$5 million annually. The February 2020 License Agreement may be terminated upon our election on at least 30 days advance notice to Stanford,
or by Stanford if we: (i) are delinquent on any report or payment; (ii) are not diligently developing and commercializing Licensed Product;
(iii) miss certain performance milestones; (iv) are in breach of any provision of the February 2020 License Agreement; or (v) provide
any false report to Stanford. Should any events in the preceding sentence occur, we have a thirty (30) day cure period to remedy such
violation.
Asset Purchase Agreement
On April 18, 2023, the Company entered into an
Asset Purchase Agreement (the “Asset Purchase Agreement”) with Cellvera Global Holdings LLC (“Cellvera Global”),
Cellvera Holdings Ltd. (“BVI Holdco”), Cellvera, Ltd. (“Cellvera Ltd.”), Cellvera Development LLC (“Cellvera
Development” and together with Cellvera Global, BVI Holdco, Cellvera Ltd. and Cellvera Development (the “Sellers”),
AiPharma Group Ltd. (“Seller Owner” and collectively with the Sellers, “Cellvera”), and the legal representative
of Cellvera, pursuant to which, the Company will purchase Cellvera’s 50% ownership interest in G Response Aid FZE (“GRA”),
certain other intellectual property and all goodwill related thereto (the “Acquired Assets”). Unless expressly stated
otherwise herein, capitalized terms used but not defined herein have the meanings ascribed to them in the Asset Purchase Agreement. Pursuant
to the Asset Purchase Agreement, the consideration for the Acquired Assets consists of (A) $24.5 million, comprised of: (i)
the forgiveness of the Company’s $14.5 million loan to Cellvera Global, and (ii) approximately $10 million in cash, and
(B) future revenue sharing payments for a term of seven years. GRA holds an exclusive, worldwide license for the antiviral medication,
Avigan® 200mg, excluding Japan, China and Russia. The other 50% interest in GRA is held by Agility, Inc. (“Agility”).
Additionally, upon the closing, the Share Exchange Agreement previously entered into as of December 28, 2021, between Cellvera Global
Holdings, LLC f/k/a AiPharma Global Holdings, LLC (together with other affiliates and subsidiaries) and the Company, and all other related
agreements will be terminated.
The obligations of the
Company to consummate the closing are subject to the satisfaction or waiver, at or prior to the Closing of certain conditions, including
but not limited to, the following:
| |
(i) |
Satisfactory completion
of due diligence; |
| |
|
|
| |
(ii) |
Completion by the Company
of financing sufficient to consummate the transactions contemplated by the Asset Purchase Agreement; |
| |
|
|
| |
(iii) |
Receipt by the Company
of all required Consents from Governmental Bodies for the Acquisition, including but not limited to, any consents required to complete
the transfer and assignment of Cellvera’s membership interests in GRA; |
| |
(iv) |
Receipt of executed payoff
letters reflecting the amount required to be fully pay all of each of Seller’s and Seller Owner’s Debt to be paid at
Closing; |
| |
|
|
| |
(v) |
Receipt by the Company
of a release from Agility; |
| |
|
|
| |
(iv) |
Execution of an agreement
acceptable to the Company with respect to the acquisition by the Company of certain intellectual property presently held by a third
party; |
| |
|
|
| |
(v) |
Execution of an amendment
to an asset purchase agreement previously entered into by Cellvera with a third party that effectively grants the Company the rights
to acquire the intellectual property from the third party under such agreement; |
| |
|
|
| |
(vi) |
Receipt of a fairness opinion
by the Company with respect to the transactions contemplated by the Asset Purchase Agreement; and |
| |
|
|
| |
(vii) |
Receipt by the Company
from the Seller Owner of written consent, whether through its official liquidator or the Board of Directors of Seller Owner, to the
sale and purchase of the Acquired Assets and Assumed Liabilities pursuant to the Assert Purchase Agreement. |
Departure of Officer
On July 21, 2023, Matthew Shatzkes tendered his
resignation as Chief Legal Officer, General Counsel and Corporate Secretary of the Company. In connection with his resignation, the Company
entered into a Separation Agreement and General Release (the “Separation Agreement”) with Mr. Shatzkes. Pursuant to the Separation
Agreement, Mr. Shatzkes’ employment with the Company terminated on August 4, 2023 (the “Termination Date”). In addition,
the Company agreed to pay Mr. Shatzkes’ within seven days after the Termination Date: (i) $122,292, representing all accrued salary
and wages (inclusive of Base Compensation and earned Subsequent Quarterly Bonus amounts, as those terms are defined in Mr. Shatzkes’
employment agreement), and (ii) $32,576, representing Mr. Shatzkes accrued, but unused paid time off (collectively, the “Initial
Payment”). The Company also agreed to pay Mr. Shatzkes: (i) $385,000, representing 12 months of Mr. Shatzkes’ Base Compensation
(as that term is defined in Mr. Shatzkes employment agreement), and (ii) $290,000, representing Mr. Shatzkes Subsequent Year Minimum
Bonus (as such term is defined in Mr. Shatzkes employment agreement), on the 60th day following the Termination Date. In addition,
the Company shall reimburse Mr. Shatzkes COBRA premium for a period of 12 months and shall cause any restricted stock units granted to
Mr. Shatzkes to immediately vest as of the Termination Date. As of December 31, 2023, the Company has completed all obligations under
the Separation Agreement.
Contingent Liability
On September 7, 2023, the Company received a
demand letter from the holder of certain warrants issued by the Company in April 2023. The demand letter alleged that the investor suffered
more than $2 million in damages as a result of the Company failing to register the shares of the Company’s common stock underlying
the warrants as required under the securities purchase agreement. The Company denies the amount of the liability claimed by the investor
and intends to defend itself vigorously against any such claims. The Company is engaged in ongoing discussions with the investor and,
as a result, has accrued a loss of $1.6 million relating to the potential liability. This liability was settled subsequent to December
31, 2023. (See Note 12)
Letter of Intent Termination
On August 1, 2023, the Company and Natural State
Genomics and Natural State Laboratories mutually agreed to terminate the Amended and Restated Non-Binding Letter of Intent dated June
12, 2023.
EvoFem Merger Agreement
On December 11, 2023 (the “Execution Date”),
Aditxt, Inc., a Delaware corporation (the “Company”) entered into an Agreement and Plan of Merger (the “Merger Agreement”)
with Adicure, Inc., a Delaware corporation and wholly owned subsidiary of the Company (“Merger Sub”) and Evofem Biosciences,
Inc., a Delaware corporation (“Evofem”), pursuant to which, Merger Sub will be merged into and with Evofem (the “Merger”),
with Evofem surviving the Merger as a wholly owned subsidiary of the Company.
In connection with the Merger Agreement the Company
assumed $13.0 million in notes payable held by Evofem (see Note 7) and assumed a payable for $154,480 (see Note 7). These items were
capitalized on the Company’s balance sheet to deposit on acquisition as of December 31, 2023. The Company recognized a debt discount
of $1,826,250. As of December 31, 2023, there was an unamortized discount of $1,633,389.
Subject to the terms and conditions set forth
in the Merger Agreement, at the effective time of the Merger (the “Effective Time”), (i) all issued and outstanding shares
of common stock, par value $0.0001 per share of Evofem (“Evofem Common Stock”), other than any shares of Evofem Common Stock
held by the Company or Merger Sub immediately prior to the Effective Time, will be converted into the right to receive an aggregate of
610,000 shares of the Company’s common stock, par value $0.001 per share (“Company Common Stock”); and (ii) all issued
and outstanding shares of Series E-1 Preferred Stock, par value $0.0001 of Evofem (the “Evofem Unconverted Preferred Stock”),
other than any shares of Evofem Unconverted Preferred Stock held by the Company or Merger Sub immediately prior to the Effective Time,
will be converted into the right to receive an aggregate of 2,327 shares of Series A-1 Preferred Stock, par value $0.001 of the Company
(the “Company Preferred Stock”), having such rights, powers, and preferences set forth in the form of Certificate of Designation
of Series A-1 Preferred Stock. (See Note 10)
Evofem Exchange Agreement
On December 22, 2023, the Company entered into
an Exchange Agreement (the “Exchange Agreement”) with the holders of an aggregate of 22,280 shares of Series F-1 Convertible
Preferred Stock of Evofem (the “Evofem Series F-1 Preferred Stock”) agreed to exchange their respective shares of Evofem
Series F-1 Preferred Stock for an aggregate of 22,280 shares of a new series of convertible preferred stock of the Company designated
as Series A-1 Convertible Preferred Stock, $0.001 par value, (the “Series A-1 Preferred Stock”), having a total value of
$22,277,233. (see Note 10) This investment has been recorded at cost in accordance with ASC 321.
NOTE 10 – STOCKHOLDERS’ EQUITY
Common Stock
On May 24, 2021, the Company increased the number
of authorized shares of the Company’s common stock, par value $0.001 per share, from 27,000,000 to 100,000,000 (the
“Authorized Shares Increase”) by filing a Certificate of Amendment (the “Certificate of Amendment”) to its Amended
and Restated Certificate of Incorporation with the Secretary of State of the State of Delaware. In accordance with the General Corporation
Law of the State of Delaware, the Authorized Shares Increase and the Certificate of Amendment were approved by the stockholders of the
Company at the Company’s Annual Meeting of Stockholders on May 19, 2021. On September 13, 2022, the Company effectuated a
1 for 50 reverse stock split (the “2022 Reverse Split”). The Company’s stock began trading at the 2022 Reverse
Split price effective on the Nasdaq Stock Market on September 14, 2022. There was no change to the number of authorized shares of the
Company’s common stock. On August 17, 2023, the Company effectuated a 1 for 40 reverse stock split (the “2023 Reverse Split”). The
Company’s stock began trading at the 2023 Reverse Split price effective on the Nasdaq Stock Market on August 17, 2023. There was
no change to the number of authorized shares of the Company’s common stock.
Formed in January 2023, our majority owned subsidiary
Pearsanta™, Inc. (“Pearsanta”) seeks to take personalized medicine to a new level by delivering “Health by the
Numbers.” On November 22, 2023, Pearsanta entered into an assignment agreement with FirstVitals LLC, an entity controlled by Pearsanta’s
CEO, Ernie Lee (“FirstVitals”), pursuant to which FirstVitals assigned its rights in certain intellectual property and website
domain to Pearsanta in consideration of the issuance of 500,000 shares of Pearsanta common stock to FirstVitals. On December 18, 2023,
the board of directors of Pearsanta adopted the Pearsanta 2023 Omnibus Equity Incentive Plan (the “Pearsanta Omnibus Incentive
Plan”), pursuant to which it reserved 15 million shares of common stock of Pearsanta for future issuance under the Pearsanta Omnibus
Incentive Plan and the Pearsanta 2023 Parent Service Provider Equity Incentive Plan (the “Pearsanta Parent Service Provider Plan”)
and approved the issuance of 9.32 million options, exercisable into shares of Pearsanta common stock under the Pearsanta Parent Service
Provider Plan and the issuance of 4.0 million options, exercisable into shares of Pearsanta common stock, subject to vesting, and 1.0
million restricted common stock shares under the Pearsanta Omnibus Incentive Plan.
During the year ended December 31, 2023, the
Company issued 74,675 shares of common stock and recognized expense of $484,525 in stock-based compensation for consulting
services. The stock-based compensation for consulting services is calculated by the number of shares multiplied by the closing price
on the effective date of the contract. The Company recognized expense of $308,479 in stock-based compensation related to the RSUs
for the year ended December 31, 2023. The stock-based compensation for shares issued or RSUs granted during the period were valued based
on the fair market value on the date of grant. During the year ended December 31, 2023, the Company issued 1,055,374 shares of common
stock for the exercise of warrants.
During the year ended December 31, 2022, the
Company issued 3,707 shares of common stock and recognized expense of $507,558 in stock-based compensation for consulting services. The
Company also granted 292 RSUs, 463 vested and resulted in the issuance of shares. As a result, the Company recognized expense of $1,209,906
in stock-based compensation. The stock-based compensation for shares issued or RSU’s granted during the period were valued based
on the fair market value on the date of grant. During the year ended December 31, 2022, the Company issued 48,659 shares of common stock
for the exercise of warrants.
On December 20, 2022, the Company entered into
an At The Market Offering Agreement (the “ATM”) with H.C. Wainwright & Co., LLC as agent (the “Agent”), pursuant
to which the Company may offer and sell, from time to time through the Agent, shares of the Company’s common stock having an aggregate
offering price of up to $50,000,000 (the “Shares”).
The offer and sale of the Shares was made pursuant
to a shelf registration statement on Form S-3 and the related prospectus (File No. 333-257645) filed by the Company with the SEC on July
2, 2021, amended on July 6, 2021 and declared effective by the SEC on July 13, 2021, under the Securities Act of 1933, as amended.
For the year ended December 31, 2023, the Company
sold 8,463 Shares at an average price of $62.05 per share under the ATM. The sale of Shares generated net proceeds of
$507,016 after paying commissions and related fees.
On April 20, 2023, the Company entered into an
amendment to the ATM, pursuant to which the Company and the Agent agreed to reduce the aggregate gross sales price of the Shares under
the ATM from $50,000,000 to zero.
Preferred Stock
The Company is authorized to issue 3,000,000 shares
of preferred stock, par value $0.001 per share. There were 24,905 and zero shares of preferred stock outstanding as of
December 31, 2023 and 2022, respectively.
Issuance of Series A-1 Preferred Stock:
On December 11, 2023 (the “Execution Date”),
the Company entered into an Agreement and Plan of Merger (the “Merger Agreement”) with Adicure, Inc., a Delaware corporation
and wholly owned subsidiary of the Company (“Merger Sub”) and Evofem Biosciences, Inc., a Delaware corporation (“Evofem”),
pursuant to which, Merger Sub will be merged into and with Evofem (the “Merger”), with Evofem surviving the Merger as a wholly
owned subsidiary of the Company.
Subject to the terms and conditions set forth
in the Merger Agreement, at the effective time of the Merger (the “Effective Time”), (i) all issued and outstanding shares
of common stock, par value $0.0001 per share of Evofem (“Evofem Common Stock”), other than any shares of Evofem Common Stock
held by the Company or Merger Sub immediately prior to the Effective Time, will be converted into the right to receive an aggregate of
610,000 shares of the Company’s common stock, par value $0.001 per share (“Company Common Stock”); and (ii) all issued
and outstanding shares of Series E-1 Preferred Stock, par value $0.0001 of Evofem (the “Evofem Unconverted Preferred Stock”),
other than any shares of Evofem Unconverted Preferred Stock held by the Company or Merger Sub immediately prior to the Effective Time,
will be converted into the right to receive an aggregate of 2,327 shares of Series A-1 Preferred Stock, par value $0.001 of the Company
(the “Company Preferred Stock”), having such rights, powers, and preferences set forth in the form of Certificate of Designation
of Series A-1 Preferred Stock. See Series A-1 Preferred Stock certificate of designation incorporated by reference to this document.
On December 22, 2023, the Company entered into
an Exchange Agreement (the “Exchange Agreement”) with the holders (the “Holders”) of an aggregate of 22,280 shares
of Series F-1 Convertible Preferred Stock of Evofem (the “Evofem Series F-1 Preferred Stock”) agreed to exchange their respective
shares of Evofem Series F-1 Preferred Stock for an aggregate of 22,280 shares of a new series of convertible preferred stock of the Company
designated as Series A-1 Convertible Preferred Stock, $0.001 par value, (the “Series A-1 Preferred Stock”).
The following is only a summary of the Series
A-1 Certificate of Designations, and is qualified in its entirety by reference to the full text of the Series A-1 Certificate of Designations,
a copy of which is filed as Exhibit 3.1 to our Current Report on Form 8-K filed on December 26, 2023 and is incorporated by reference
herein.
Designation, Amount, and Par Value: The number
of Series A-1 Preferred Stock designated is 22,280 shares. The shares of Series A-1 Preferred Stock have a par value of $0.001 per share
and a stated value of $1,000 per share.
Conversion Price: The Series A-1 Preferred Stock
will be convertible into shares of Common Stock at an initial conversion price of $4.44 (subject to adjustment pursuant to the Series
A-1 Certificate of Designations) (the “Conversion Price”). The Certificate of Designations also provides that in the event
of certain Triggering Events (as defined below) any holder may, at any time, convert any or all of such holder’s Series A-1 Preferred
Stock at an alternate conversion rate equal to the product of (i) the Alternate Conversion Price (as defined below) and (ii) the quotient
of (x) the 25% redemption premium multiplied by (y) the amount of Series A-1 Preferred Stock subject to such conversion. “Triggering
Events” include, among others, (i) a suspension of trading or the failure to be traded or listed on an eligible market for five
consecutive days or more, (ii) the failure to remove restrictive legends when required, (iii) the Company’s default in payment
of indebtedness in an aggregate amount of $500,000 or more (the Company is currently in default for payments greater than $500,000),
(iv) proceedings for a bankruptcy, insolvency, reorganization or liquidation, which are not dismissed with 30 days, (v) commencement
of a voluntary bankruptcy proceeding, and (viii) final judgments against the Company for the payment of money in excess of $100,000.
“Alternate Conversion Price” means the lowest of (i) the applicable conversion price the in effect, (ii) the greater of (x)
$0.888 (the “Floor Price”) and (y) 80% of the volume weighted average price (“VWAP”) of the Common Stock on the
trading day immediately preceding the delivery of the applicable conversion notice. Further, the Series A-1 Certificate of Designations
provides that if on any of the 90th and 180th day after each of the occurrence of any Stock Combination Event (as defined in the Series
A-1 Certificate of Designations) and the Applicable Date (as defined in the Series A-1 Certificate of Designations), the conversion price
then in effect is greater than the market price then in effect (the “Adjustment Price”), on such date then the conversion
price shall automatically lower to the Adjustment Price.
Dividends: Holders of the Series A-1 Preferred
Stock shall be entitled to receive dividends when and as declared by the Board, from time to time, in its sole discretion, which Dividends
shall be paid by the Company out of funds legally available therefor, payable, subject to the conditions and other terms hereof, in cash,
in securities of the Company or any other entity, or using assets as determined by the Board on the Stated Value of such Preferred Share.
Liquidation: In the event of a Liquidation Event
(as defined in the Series A-1 Certificate of Designation), the holders the Series A-1 Preferred Stock shall be entitled to receive in
cash out of the assets of the Company, before any amount shall be paid to the holders of any other shares of capital stock of the Company,
equal to the greater of (A) 125% of the Conversion Amount (as defined in the Series A-1 Certificate of Designation) on the date of such
payment and (B) the amount per share such holder of Series A-1 Preferred Stock would receive if they converted such share of Series A-1
Preferred Stock into Common Stock immediately prior to the date of such payment
Company Redemption: The Company may redeem all,
or any portion, of the Series A-1 Preferred Stock for cash, at a price per share of Series A-1 Preferred Stock equal to 115% of the greater
of (i) the Conversion Amount (as defined in the Series A-1 Certificate of Designation)being redeemed as of the Company Optional Redemption
Date (as defined in the Series A-1 Certificate of Designation) and (ii) the product of (1) the Conversion Rate (as defined in the Series
A-1 Certificate of Designation) with respect to the Conversion Amount being redeemed as of the Company Optional Redemption Date multiplied
by (2) the greatest Closing Sale Price (as defined in the Certificate of Designation) of the Common Stock on any Trading Day during the
period commencing on the date immediately preceding such Company Optional Redemption Notice Date (as defined in the Certificate of Designation)
and ending on the Trading Day immediately prior to the date the Company makes the entire payment required to be made under the Certification
of Designation.
Maximum Percentage: Holders of Series A-1 Preferred
Stock are prohibited from converting shares of Series A-1 Preferred Stock into shares of Common Stock if, as a result of such conversion,
such holder, together with its affiliates, would beneficially own in excess of 4.99% (the “Maximum Percentage”) of the total
number of shares of Common Stock issued and outstanding immediately after giving effect to such conversion.
Voting Rights: The holders of the Series A-1
Preferred Stock shall have no voting power and no right to vote on any matter at any time, either as a separate series or class or together
with any other series or class of share of capital stock, and shall not be entitled to call a meeting of such holders for any purpose
nor shall they be entitled to participate in any meeting of the holders of Common Stock, except as expressly provided in the Certificate
of Designations and where required by the DGCL.
Issuance of Series B Preferred Stock:
On July 19, 2022, the Company entered into a
Subscription and Investment Representation Agreement with its Chief Executive Officer (the “Purchaser”), pursuant to which
the Company agreed to issue and sell one (1) share of the Company’s Series B Preferred Stock (the “Preferred Stock”),
par value $0.001 per share, to the Purchaser for $20,000 in cash.
On July 19, 2022, the Company filed a certificate
of designation (the “Certificate of Designation”) with the Secretary of State of Delaware, effective as of the time of filing,
designating the rights, preferences, privileges and restrictions of the share of Preferred Stock. The Certificate of Designation provides
that the share of Preferred Stock will have 250,000,000 votes and will vote together with the outstanding shares of the Company’s
common stock as a single class exclusively with respect to any proposal to amend the Company’s Restated Certificate of Incorporation
to effect a reverse stock split of the Company’s common stock. The Preferred Stock will be voted, without action by the holder,
on any such proposal in the same proportion as shares of common stock are voted. The Preferred Stock otherwise has no voting rights except
as otherwise required by the General Corporation Law of the State of Delaware.
The Preferred Stock is not convertible into,
or exchangeable for, shares of any other class or series of stock or other securities of the Company. The Preferred Stock has no rights
with respect to any distribution of assets of the Company, including upon a liquidation, bankruptcy, reorganization, merger, acquisition,
sale, dissolution or winding up of the Company, whether voluntarily or involuntarily. The holder of the Preferred Stock will not be entitled
to receive dividends of any kind. See Series B Preferred Stock certificate of designation incorporated by reference to this document.
The outstanding share of Preferred Stock shall
be redeemed in whole, but not in part, at any time (i) if such redemption is ordered by the Board of Directors in its sole discretion
or (ii) automatically upon the effectiveness of the amendment to the Certificate of Incorporation implementing a reverse stock split.
Upon such redemption, the holder of the Preferred Stock will receive consideration of $20,000 in cash.
Redemption of Series B Preferred Stock
On October 7, 2022, the Company paid $20,000 in
consideration for the one share of Preferred Stock which was redeemed on September 13, 2022.
Issuance of Series B-2 Preferred Stock:
On December 29, 2023, the Company entered into
an Exchange Agreement (the “Note Exchange Agreement”) with the Noteholder, pursuant to which the Noteholder agreed, subject
to the terms and conditions set forth therein, to exchange the Note, including all accrued but unpaid interest thereon, for an aggregate
of 2,625 shares of a new series of convertible preferred stock of the Company, designated as Series B-2 Convertible Preferred Stock,
$0.001 par value (the “Series B-2 Preferred Stock”). See Series B-2 Preferred Stock certificate of designation incorporated
by reference to this document.
The following is only a summary of the Series
B-2 Certificate of Designations, and is qualified in its entirety by reference to the full text of the Series B-2 Certificate of Designations,
a copy of which is filed as an exhibit to our Current Report on Form 8-K filed with the SEC on January 2, 2024.
Designation, Amount, and Par Value: The number
of Series B-2 Preferred Stock designated is 2,625 shares. The shares of Series B-2 Preferred Stock have a par value of $0.001 per share
and a stated value of $1,000 per share.
Conversion Price: The Series B-2 Preferred Stock
will be convertible into shares of Common Stock at an initial conversion price of $4.71 (subject to adjustment pursuant to the Series
B-2 Certificate of Designations) (the “Conversion Price”). The Series B-2 Certificate of Designations also provides that
in the event of certain Triggering Events (as defined below) any holder may, at any time, convert any or all of such holder’s Series
B-2 Preferred Stock at an alternate conversion rate equal to the product of (i) the Alternate Conversion Price (as defined below) and
(ii) the quotient of (x) the 125% redemption premium multiplied by (y) the amount of Series B-2 Preferred Stock subject to such conversion.
“Triggering Events” include, among others, (i) a suspension of trading or the failure to be traded or listed on an eligible
market for five consecutive days or more, (ii) the failure to remove restrictive legends when required, (iii) the Company’s default
in payment of indebtedness in an aggregate amount of $500,000 or more(the Company is currently in default for payments greater than $500,000),
(iv) proceedings for a bankruptcy, insolvency, reorganization or liquidation, which are not dismissed with 30 days, (v) commencement
of a voluntary bankruptcy proceeding, and (viii) final judgments against the Company for the payment of money in excess of $500,000.
“Alternate Conversion Price” means the lowest of (i) the applicable conversion price the in effect, (ii) the greater of (x)
$0.9420 (the “Floor Price”) and (y) 80% of the lowest volume weighted average price (“VWAP”) of the Common Stock
during the five consecutive trading day period ending and including the trading day immediately preceding the delivery of the applicable
conversion notice. Further, the Series B-2 Certificate of Designations provides that if on any of the 90th and 180th day after each of
the occurrence of any Stock Combination Event (as defined in the Series B-2 Certificate of Designations) and the Applicable Date (as
defined in the Series B-2 Certificate of Designations), the conversion price then in effect is greater than the market price then in
effect (the “Adjustment Price”), on such date then the conversion price shall automatically lower to the Adjustment Price.
Dividends: Holders of the Series B-2 Preferred
Stock shall be entitled to receive dividends when and as declared by the Board, from time to time, in its sole discretion, which Dividends
shall be paid by the Company out of funds legally available therefor, payable, subject to the conditions and other terms hereof, in cash,
in securities of the Company or any other entity, or using assets as determined by the Board on the Stated Value of such Preferred Share.
Liquidation: In the event of a Liquidation Event
(as defined in the Series B-2 Certificate of Designations), the holders the Series B-2 Preferred Stock shall be entitled to receive in
cash out of the assets of the Company, before any amount shall be paid to the holders of any other shares of capital stock of the Company,
equal to the greater of (A) 125% of the Conversion Amount (as defined in the Series B-2 Certificate of Designation) on the date of such
payment and (B) the amount per share such holder of Series B-2 Preferred Stock would receive if they converted such share of Series B-2
Preferred Stock into Common Stock immediately prior to the date of such payment.
Company Redemption: The Company may redeem all,
or any portion, of the Series B-2 Preferred Stock for cash, at a price per share of Series B-2 Preferred Stock equal to 115% of the greater
of (i) the Conversion Amount (as defined in the Series B-2 Certificate of Designations) being redeemed as of the Company Optional Redemption
Date (as defined in the Series B-2 Certificate of Designations) and (ii) the product of (1) the Conversion Rate (as defined in the Series
B-2 Certificate of Designations) with respect to the Conversion Amount being redeemed as of the Company Optional Redemption Date multiplied
by (2) the greatest Closing Sale Price (as defined in the Series B-2 Certificate of Designations) of the Common Stock on any Trading
Day during the period commencing on the date immediately preceding such Company Optional Redemption Notice Date (as defined in the Series
B-2 Certificate of Designations) and ending on the Trading Day immediately prior to the date the Company makes the entire payment required
to be made under the Certification of Designation.
Maximum Percentage: Holders of Series B-2 Preferred
Stock are prohibited from converting shares of Series B-2 Preferred Stock into shares of Common Stock if, as a result of such conversion,
such holder, together with its affiliates, would beneficially own in excess of 4.99% (the “Maximum Percentage”) of the total
number of shares of Common Stock issued and outstanding immediately after giving effect to such conversion.
Voting Rights: The holders of the Series B-2
Preferred Stock shall have no voting power and no right to vote on any matter at any time, either as a separate series or class or together
with any other series or class of share of capital stock, and shall not be entitled to call a meeting of such holders for any purpose
nor shall they be entitled to participate in any meeting of the holders of Common Stock, except as expressly provided in the Series B-2
Certificate of Designations and where required by the DGCL.
Series C Preferred Stock
On July 11, 2023, the Company filed a certificate
of designation (the “Certificate of Designation”) with the Secretary of State of Delaware, effective as of the time of filing,
designating the rights, preferences, privileges and restrictions of the share of Preferred Stock. The Certificate of Designation provides
that the share of Preferred Stock will have 250,000,000 votes and will vote together with the outstanding shares of the Company’s
common stock as a single class exclusively with respect to any proposal to amend the Company’s Amended and Restated Certificate
of Incorporation to effect a reverse stock split of the Company’s common stock. The Preferred Stock will be voted, without action
by the holder, on any such proposal in the same proportion as shares of common stock are voted. The Preferred Stock otherwise has no
voting rights except as otherwise required by the General Corporation Law of the State of Delaware.
The Preferred Stock is not convertible into,
or exchangeable for, shares of any other class or series of stock or other securities of the Company. The Preferred Stock has no rights
with respect to any distribution of assets of the Company, including upon a liquidation, bankruptcy, reorganization, merger, acquisition,
sale, dissolution or winding up of the Company, whether voluntarily or involuntarily. The holder of the Preferred Stock will not be entitled
to receive dividends of any kind.
The outstanding share of Preferred Stock shall
be redeemed in whole, but not in part, at any time (i) if such redemption is ordered by the Board of Directors in its sole discretion
or (ii) automatically upon the effectiveness of the amendment to the Certificate of Incorporation implementing a reverse stock split.
Upon such redemption, the holder of the Preferred Stock will receive consideration of $1,000 in cash. As of December 31, 2023, the share
has been redeemed and the consideration has been paid.
On July 11, 2023, the Company entered into a
Subscription and Investment Representation Agreement (the “Subscription Agreement”) with Amro Albanna, its Chief Executive
Officer, who is an accredited investor (the “Purchaser”), pursuant to which the Company agreed to issue and sell one (1)
share of the Company’s Series C Preferred Stock, par value $0.001 per share (the “Preferred Stock”), to the Purchaser
for $1,000 in cash. The sale closed on July 11, 2023. The Subscription Agreement contains customary representations and warranties and
certain indemnification rights and obligations of the parties. See Series C Preferred Stock certificate of designation incorporated by
reference to this document. On August 17, 2023, the share was redeemed.
Stock-Based Compensation
In October 2017, our Board of Directors adopted
the Aditx Therapeutics, Inc. 2017 Equity Incentive Plan (the “2017 Plan”). The 2017 Plan provides for the grant of equity
awards to directors, employees, and consultants. The Company is authorized to issue up to 2,500,000 shares of our common
stock pursuant to awards granted under the 2017 Plan. The 2017 Plan is administered by our Board of Directors, and expires ten years
after adoption, unless terminated earlier by the Board of Directors. All shares of our common stock pursuant to awards under the
2017 Plan have been awarded.
On February 24, 2021, our Board of Directors
adopted the Aditx Therapeutics, Inc. 2021 Omnibus Equity Incentive Plan (the “2021 Plan”). The 2021 Plan provides for grants
of nonqualified stock options, incentive stock options, stock appreciation rights, restricted stock and restricted stock units, and other
stock-based awards (collectively, the “Awards”). Eligible recipients of Awards include employees, directors or independent
contractors of the Company or any affiliate of the Company. The Compensation Committee of the Board of Directors (the “Committee”)
administers the 2021 Plan. A total of 60,000 shares of common stock, par value $0.001 per share, of the Company may be
issued pursuant to Awards granted under the 2021 Plan. The exercise price per share for the shares to be issued pursuant to an exercise
of a stock option will be no less than one hundred percent (100%) of the Fair Market Value (as defined in the 2021 Plan) of a share of
Common Stock on the date of grant. The 2021 Plan was submitted and approved by the Company’s stockholders at the 2021 annual meeting
of stockholders, held on May 19, 2021.
During the years ended December 31, 2023 and
2022, the Company granted 44,445 and 0 new options. respectively.
For the
year ended December 31, 2023, the fair value of each option granted was estimated using
the assumption and/or factors in the Black-Scholes Model as follows:
| | |
| |
| Exercise price | |
$ | 5.01 | |
| Expected dividend yield | |
| 0 | % |
| Risk free interest rate | |
| 4.49 | % |
| Expected life in years | |
| 10 | |
| Expected volatility | |
| 164 | % |
The risk-free interest rate assumption for options
granted is based upon observed interest rates on the United States Government Bond Equivalent Yield appropriate for the expected term
of option.
The Company determined the expected volatility
assumption for options granted using the historical volatility of comparable public companies’ common stock. The Company will continue
to monitor peer companies and other relevant factors used to measure expected volatility for future option
grants, until such time that the Company’s common stock has enough market history to use historical volatility.
The dividend yield assumption for option
granted is based on the Company’s history and expectation of dividend payouts. The Company has never declared nor paid any
cash dividends on its common stock, and the Company does not anticipate paying any cash dividends in the foreseeable future.
The Company recognizes option
forfeitures as they occur, as there is insufficient historical data to accurately determine future forfeitures rates.
The following is an analysis of the stock option
grant activity under the Plan:
| Vested and Nonvested Stock Options | |
Number | | |
Weighted
Average
Exercise
Price | | |
Weighted
Average
Remaining
Life | |
| Outstanding December 31, 2022 | |
| 1,127 | | |
$ | 6,802.93 | | |
| 5.74 | |
| Granted | |
| 44,445 | | |
| 5.01 | | |
| 9.86 | |
| Exercised | |
| - | | |
| - | | |
| - | |
| Expired or forfeited | |
| - | | |
| - | | |
| - | |
| Outstanding December 31, 2023 | |
| 45,572 | | |
$ | 173.12 | | |
| 9.74 | |
| Nonvested Stock Options | |
Number | | |
Weighted-
Average
Exercise
Price | |
| Nonvested on December 31, 2022 | |
| 55 | | |
$ | 3,840 | |
| Granted | |
| 44,445 | | |
| 5.01 | |
| Vested | |
| (44,500 | ) | |
| 9.75 | |
| Forfeited | |
| - | | |
| - | |
| Nonvested on December 31, 2023 | |
| - | | |
$ | - | |
As of December 31, 2023 there were 45,572 exercisable
options; these options had a weighted average exercise price $173.12. These options had a grant date fair value of $221,005.
On December 18, 2023, our Board of Directors
adopted the Pearsanta, Inc. 2023 Omnibus Equity Incentive Plan (the “Pearsanta 2023 Plan”) and the 2023 Parent Service Provider
Equity Incentive Plan (the “Pearsanta Parent 2023 Plan”), collectively (the “Pearsanta Plans”). The Pearsanta
Plans provides for grants of nonqualified stock options, incentive stock options, stock appreciation rights, restricted stock and restricted
stock units, and other stock-based awards (collectively, the “Pearsanta Awards”). Eligible recipients of Pearsanta Awards
include employees, directors or independent contractors of the Company or any affiliate of the Company. The Board of Directors administers
the Pearsanta Plans. The Pearsanta 2023 Plan consists of a total of 15,000,000 shares of Pearsanta common stock, par value
$0.001 per share, which may be issued pursuant to Pearsanta Awards granted under the Pearsanta 2023 Plan. The Pearsanta Parent 2023
Plan consists of a total of 9,320,000 shares of Pearsanta common stock, par value $0.001 per share, which may be issued
pursuant to Pearsanta Awards granted under the Pearsanta Parent 2023 Plan. The exercise price per share for the shares to be issued pursuant
to an exercise of a stock option will be no less than one hundred percent (100%) of the Fair Market Value (as defined in the Pearsanta
Plans) of a share of Common Stock on the date of grant.
During the years ended December 31, 2023 and
2022, Pearsanta granted 4,000,000 and 0 new options under the Pearsanta 2023 Plan, respectively.
During the years ended December 31, 2023 and
2022, Pearsanta granted 9,320,000 and 0 new options under the Pearsanta Parent 2023 Plan, respectively.
For the
year ended December 31, 2023, the fair value of each option granted was estimated using
the assumption and/or factors in the Black-Scholes Model as follows:
| |
|
|
|
|
| Exercise price |
|
$ |
0.02 |
|
| Expected dividend yield |
|
|
0 |
% |
| Risk free interest rate |
|
|
3.95 |
% |
| Expected life in years |
|
|
10 |
|
| Expected volatility |
|
|
194 |
% |
The risk-free interest rate assumption for warrants
granted is based upon observed interest rates on the United States Government Bond Equivalent Yield appropriate for the expected term
of option.
The Company determined the expected volatility
assumption for options granted using the historical volatility of comparable public companies’ common stock. The Company will continue
to monitor peer companies and other relevant factors used to measure expected volatility for future option
grants, until such time that the Company’s common stock has enough market history to use historical volatility.
The dividend yield assumption for option
granted is based on the Company’s history and expectation of dividend payouts. The Company has never declared nor paid any
cash dividends on its common stock, and the Company does not anticipate paying any cash dividends in the foreseeable future.
The following is an analysis of the stock option
grant activity under the Pearsanta Plans:
| Vested and Nonvested Stock Options | |
Number | | |
Weighted
Average
Exercise
Price | | |
Weighted
Average
Remaining
Life | |
| Outstanding December 31, 2022 | |
| - | | |
$ | - | | |
| - | |
| Granted | |
| 13,320,000 | | |
| 0.02 | | |
| 9.97 | |
| Exercised | |
| - | | |
| - | | |
| - | |
| Expired or forfeited | |
| - | | |
| - | | |
| - | |
| Outstanding December 31, 2023 | |
| 13,320,000 | | |
$ | 0.02 | | |
| 9.97 | |
| Nonvested
Stock Options |
|
Number |
|
|
Weighted-
Average
Exercise
Price |
|
| Nonvested on December 31, 2022 |
|
|
- |
|
|
$ |
- |
|
| Granted |
|
|
13,320,000 |
|
|
|
0.02 |
|
| Vested |
|
|
(9,320,000 |
) |
|
|
0.02 |
|
| Forfeited |
|
|
- |
|
|
|
- |
|
| Nonvested on December 31, 2023 |
|
|
4,000,000 |
|
|
$ |
0.02 |
|
As of December 31, 2023 there were 9,320,000 exercisable
options; these options had a weighted average exercise price $0.02. These options had a grant date fair value of $265,929.
The Company recognized stock-based compensation
expense related to all options granted and vesting expense of $589,014 during the year ended December 31, 2023, of which $385,640 is
included in general and administrative expenses and $203,374 is included in research and development expenses in the accompanying
statements of operations. The remaining value to be expensed is $77,812 as of December 31, 2023. The weighted average vesting
term is 2.17 years as of December 31, 2023. The Company recognized stock-based compensation expense related to all options
granted and vesting expense of $791,187 during the year ended December 31, 2022, of which $555,772 is included in general and administrative
expenses and $235,415 is included in research and development expenses in the accompanying statements of operations.
Warrants
For the
year ended December 31, 2023, the fair value of each warrant granted was estimated using
the assumption and/or factors in the Black-Scholes Model as follows:
| |
|
|
|
|
| Exercise price |
|
$ |
300-2,300 |
|
| Expected dividend yield |
|
|
0 |
% |
| Risk free interest rate |
|
|
1.13%-3.47 |
% |
| Expected life in years |
|
|
5-5.50 |
|
| Expected volatility |
|
|
147-165 |
% |
For the
year ended December 31, 2022, the fair value of each warrant granted was estimated using the assumption and/or factors in the Black-Scholes
Model as follows:
| |
|
|
|
|
| Exercise price |
|
$ |
7.50-20.00 |
|
| Expected dividend yield |
|
|
0 |
% |
| Risk free interest rate |
|
|
2.55%-3.47 |
% |
| Expected life in years |
|
|
5.00-5.50 |
|
| Expected volatility |
|
|
147%-165 |
% |
The risk-free interest rate assumption for warrants
granted is based upon observed interest rates on the United States Government Bond Equivalent Yield appropriate for the expected term
of warrants.
The Company determined the expected volatility
assumption for warrants granted using the historical volatility of comparable public companies’ common stock. The Company will
continue to monitor peer companies and other relevant factors used to measure expected volatility for future warrant grants, until such
time that the Company’s common stock has enough market history to use historical volatility.
The dividend yield assumption for warrants granted
is based on the Company’s history and expectation of dividend payouts. The Company has never declared nor paid any cash dividends
on its common stock, and the Company does not anticipate paying any cash dividends in the foreseeable future.
The Company recognizes warrant forfeitures as
they occur, as there is insufficient historical data to accurately determine future forfeitures rates.
A summary of warrant issuances are as follows:
| Vested and Nonvested Warrants | |
Number | | |
Weighted
Average
Exercise
Price | | |
Weighted
Average
Remaining
Life | |
| Outstanding December 31, 2022 | |
| 127,281 | | |
$ | 514.97 | | |
| 4.54 | |
| Granted | |
| 5,975,936 | | |
| 3.92 | | |
| 2.72 | |
| Exercised | |
| (1,055,374 | ) | |
| 0.24 | | |
| - | |
| Expired or forfeited | |
| (393 | ) | |
| 8,249.36 | | |
| - | |
| Outstanding December 31, 2023 | |
| 5,047,450 | | |
$ | 14.11 | | |
| 2.73 | |
On September 1, 2023, the Company recognized
a deemed dividend resulting in the issuance of 9,086 warrants, 6,128 of which were immediately exercised.
| Nonvested Warrants | |
Number | | |
Weighted-
Average
Exercise
Price | |
| Nonvested on December 31, 2022 | |
| 2,500 | | |
$ | 300.00 | |
| Granted | |
| 5,975,936 | | |
| 3.92 | |
| Vested | |
| (5,978,436 | ) | |
| 4.04 | |
| Forfeited | |
| - | | |
| - | |
| Nonvested on December 31, 2023 | |
| - | | |
$ | - | |
The Company recognized stock-based compensation
expense related to warrants granted and vesting expense of zero and $609,748 during the years ended December 31, 2023
and 2022, respectively, of which $105,049 is included in general and administrative and $504,699 is included in sales and marketing in
the accompanying Statements of Operations. The remaining value to be expensed is zero as of December 31, 2023. The weighted
average vesting term is zero years as of December 31, 2023.
On April 20, 2023, the Company entered into a
securities purchase agreement (the “Purchase Agreement”) with an institutional investor, pursuant to which the Company agreed
to sell to such investor pre-funded warrants (the “Pre-Funded Warrants”) to purchase up to 39,634 shares of common stock
of the Company (the “Common Stock”) at a purchase price of $48.76 per Pre-Funded Warrant, resulting in proceeds of approximately
$1.6 million after deducting approximately $291,000 in commissions and closing fees. Concurrently with the sale of the Pre-Funded Warrants,
pursuant to the Purchase Agreement in a concurrent private placement, for each Pre-Funded Warrant purchased by the investor, such investor
received from the Company an unregistered warrant (the “Warrant”) to purchase two shares of Common Stock. The warrants have
an exercise price of $34.40 per share and are exercisable for a three year period. In addition, the Company issued a warrant to the placement
agent to purchase up to 2,379 shares of common stock at an exercise price of $61.00 per share and were valued at $56,742 using a Black
Scholes valuation model. As these warrants were considered offering costs, they had a zero net effect on the Company’s equity.
On August 31, 2023, the Company entered into
a securities purchase agreement (the “August Purchase Agreement”) with an institutional investor for the issuance and
sale in a private placement (the “Private Placement”) of (i) pre-funded warrants (the “Pre-Funded Warrants”)
to purchase up to 1,000,000 shares of the Company’s common stock at an exercise price of $0.001 per share, and (ii) warrants (the
“Common Warrants”) to purchase up to 1,000,000 shares of the Company’s Common Stock at an exercise price of $10.00
per share. 60,000 warrants were also issued to the placement agent. These warrants had an exercise price of $12.50 and a term of 5.5
years. The Common Warrants were valued at $32.3 million and the 60,000 warrants issued to the placement agents were valued at $1.9 million
using a Black Scholes valuation model. As these warrants were considered offering costs, they had a zero net effect on the Company’s
equity. The Private Placement closed on September 6, 2023. The net proceeds to the Company from the Private Placement were approximately
$9 million, after deducting placement agent fees and expenses and estimated offering expenses payable by the Company. The Company used
the net proceeds received from the Private Placement for (i) the payment of approximately $3.1 million in outstanding obligations, (ii)
the repayment of approximately $0.4 million of outstanding debt, and (iii) the balance for continuing operating expenses and working
capital.
On December 29, 2023, the Company entered into
a securities purchase agreement (the “Purchase Agreement”) with an institutional investor (“the “Purchaser”)
for the issuance and sale in a private placement (the “Private Placement”) of (i) pre-funded warrants (the “Pre-Funded
Warrants”) to purchase up to 1,237,114 shares of the Company’s common stock, par value $0.001 (the “Common Stock”)
at an exercise price of $0.001 per share, and (ii) warrants (the “Common Warrants”) to purchase up to 2,474,228 shares of
the Company’s Common Stock, at a purchase price of $4.85 per share. As of December 31, 2023, the Company had not received the funds
from the Purchase Agreement resulting in a $5,444,628 receivable. These funds were received on January 4, 2024.
The Common Warrants are exercisable immediately
upon issuance at an exercise price of $4.60 per share and have a term of exercise equal to three years from the date of issuance. The
Pre-Funded Warrants are exercisable immediately and may be exercised at any time until the Pre-Funded Warrants are exercised in full.
A holder of Pre-Funded Warrants or Warrants (together with its affiliates) may not exercise any portion of a warrant to the extent that
the holder would own more than 4.99% (or, at the election of the holder 9.99%) of the Company’s outstanding common stock immediately
after exercise.
Pursuant to the Purchase Agreement, the Company
agreed to reduce the exercise price of certain outstanding warrants to purchase Common Stock of the Company (“Outstanding Warrants”)
held by the Purchaser to $4.60 per share in consideration for the cash payment by the Purchaser of $0.125 per share of Common Stock underlying
the Outstanding Warrants, effective immediately. The Company issued a warrant to the placement agent to purchase up to 74,227 shares
of common stock at an exercise price of $6.06 per share and were valued at $470,772 using a Black Scholes valuation model. As these warrants
were considered offering costs, they had a zero net effect on the Company’s equity.
Restricted Stock Units
A summary of Restricted Stock Units (“RSUs”)
issuances are as follows:
| Nonvested RSUs | |
Number | | |
Weighted
Average
Price | |
| Nonvested December 31, 2022 | |
| 187 | | |
$ | 1,856,21 | |
| Granted | |
| - | | |
| - | |
| Vested | |
| (170 | ) | |
| 2,714.15 | |
| Forfeited | |
| (35 | ) | |
| 1,345.77 | |
| Rounding for Reverse Split | |
| 18 | | |
| - | |
| Nonvested December 31, 2023 | |
| - | | |
$ | - | |
The Company recognized stock-based compensation
expense related to RSUs granted and vesting expense of $308,479 and $1,843,902 during the years ended December 31, 2023 and
2022, respectively. Of the $308,479, $242,915 is included in general and administrative, $58,777 is included in research and
development, and $6,787 is included in sales and marketing in the accompanying Statements of Operations. Of the $1,843,902, $1,237,182 is
included in general and administrative and $606,720 is included in research and development in the accompanying Statements of Operations.
The remaining value to be expensed is $0 with a weighted average vesting term of 0 years as of December 31, 2023.
During the year ended December 31, 2023, the
Company granted a total of zero RSUs. During the year ended December 31, 2023, 170 RSUs vested and the Company issued 157 shares
of common stock for the 170 vested RSUs.
Pearsanta Restricted Stock Award
During the year ended December 31, 2023, Pearsanta
granted a total of 1,000,000 immediately vested restricted stock awards under the Pearsanta 2023 Plan. The Company recognized stock-based
compensation expense related to the Pearsanta restricted stock awards of $20,000.
NOTE 11 – INCOME TAXES
For the years ended December 31, 2023 and
2022, the Company did not record a current or deferred income tax expense or benefit due to current and historical losses incurred by
the Company. The Company’s losses before income taxes consist solely of losses from domestic operations.
A reconciliation of income tax expense (benefit)
computed at the statutory federal income tax rate to income taxes as reflected in the financial statements is as follows:
| | |
2023 | | |
2022 | |
| Income taxes at U.S. statutory rate | |
| 21 | % | |
| 21 | % |
| State income taxes | |
| 0.8 | | |
| 1.6 | |
| Tax Credits | |
| 0.5 | | |
| 1.0 | |
| Permanent Differences/Others | |
| (1.9 | ) | |
| (10.5 | ) |
| Change in valuation allowance | |
| (20.5 | ) | |
| (13.1 | ) |
| Total provision for income taxes | |
| 0 | % | |
| 0 | % |
Deferred taxes are recognized for temporary differences
between the basis of assets and liabilities for financial statement and income tax purposes. The significant components of the Company’s
deferred tax assets and liabilities as of December 31, 2023 and 2022 are comprised of the following:
| | |
Years Ended December 31, | |
| | |
2023 | | |
2022 | |
| Deferred tax assets | |
| | |
| |
| Net operating loss carryforwards | |
$ | 18,555,428 | | |
$ | 13,499,811 | |
| Tax credits carryforwards | |
| 796,320 | | |
| 430,468 | |
| Stock-based compensation | |
| 1,580,038 | | |
| 1,511,849 | |
| Lease liability | |
| 486,473 | | |
| 722,126 | |
| Section 174 Capitalization | |
| 2,207,611 | | |
| 1,547,343 | |
| Loss on impairment of debt | |
| 3,326,129 | | |
| 3,288,363 | |
| Other | |
| 92,704 | | |
| 114,973 | |
| Total deferred tax assets | |
| 27,044,703 | | |
| 21,114,933 | |
| Valuation allowance | |
| (26,414,533 | ) | |
| (20,217,400 | ) |
| Net deferred tax assets | |
| 630,170 | | |
| 897,533 | |
| Deferred tax liabilities | |
| | | |
| | |
| Right of use assets | |
| (486,473 | ) | |
| (722,127 | ) |
| Fixed assets | |
| (143,697 | ) | |
| (175,406 | ) |
| Total deferred tax liabilities | |
| (630,170 | ) | |
| (897,533 | ) |
| Net deferred taxes | |
$ | — | | |
$ | — | |
The Company has evaluated the positive and negative
evidence bearing upon its ability to realize its deferred tax assets, which are comprised primarily of net operating loss carryforwards
and tax credits. Management has considered the Company’s history of cumulative net losses in the United States, estimated future
taxable income and prudent and feasible tax planning strategies and has concluded that it is more likely than not that the Company will
not realize the benefits of its U.S. federal and state deferred tax assets. Accordingly, a full valuation allowance has been established
against these net deferred tax assets as of December 31, 2023 and 2022, respectively. The Company reevaluates the positive and negative
evidence at each reporting period. The Company’s valuation allowance increased during 2023 by approximately $6.2 million primarily
due to the generation of net operating loss and tax credit carryforwards and the capitalization of research and experimental expenditures.
The Company’s valuation allowance increased during 2022 by approximately $3.5 million primarily due to the generation of net
operating loss and tax credit carryforwards and the capitalization of research and experimental expenditures.
As of December 31, 2023 and 2022, the Company
had U.S. federal net operating loss carryforwards of $75.2 million and $56.6 million, respectively, which may be available
to offset future income tax liabilities. The 2017 Tax Cuts and Jobs Act (“ TCJA”) will generally allow losses incurred after
2017 to be carried over indefinitely, but will generally limit the net operating loss deduction to the lesser of the net operating loss
carryover or 80% of a corporation’s taxable income (subject to Section 382 of the Internal Revenue Code of 1986, as amended).
Also, there will be no carryback for losses incurred after 2017. Losses incurred prior to 2018 will generally be deductible to the extent
of the lesser of a corporation’s net operating loss carryover or 100% of a corporation’s taxable income and be available
for twenty years from the period the loss was generated. The Company has federal net operating losses generated following 2017
of $75.1 million, which do not expire. The federal net operating losses generated prior to 2018 of $0.1 million will expire
at various dates through 2037. The CARES Act temporarily allows the Company to carryback net operating losses arising in 2018, 2019
and 2020 to the five prior tax years. In addition, net operating losses generated in these years could fully offset prior year taxable
income without the 80% of the taxable income limitation under the TCJA which was enacted on December 22, 2017. The Company has been
generating losses since its inception, as such the net operating loss carryback provision under the CARES Act is not applicable to the
Company.
As of December 31, 2023 and 2022, the Company
also had U.S. state net operating loss carryforwards (post-apportioned) of $28.2 million and $26.2 million, respectively, which
may be available to offset future income tax liabilities and expire at various dates through 2042.
As of December 31, 2023, the Company had
$0.1 million federal tax credit carryforwards available to reduce future tax liabilities which expire at various dates through 2042.
As of December 31, 2022, the Company had $0.1 federal tax credit carryforwards. As of December 31, 2023 and 2022, the Company
had state research and development tax credit carryforwards of approximately $0.4 million and $0.2 million, respectively, which
may be available to reduce future tax liabilities and can be carried over indefinitely.
Utilization of the U.S. federal and state net
operating loss and research and development credit carryforwards may be subject to a substantial annual limitation under Section 382
and Section 383 of the Internal Revenue Code of 1986, as amended, and corresponding provisions of state law, due to ownership changes
that have occurred previously or that could occur in the future. These ownership changes may limit the amount of net operating loss and
research and development credit carryforwards that can be utilized annually to offset future taxable income and tax liabilities, respectively.
The Company has not completed a study to assess whether a change of ownership has occurred, or whether there have been multiple ownership
changes since its formation. Any limitation may result in expiration of a portion of the net operating loss carryforwards or research
and development tax credit carryforwards before utilization.
The Company has not, as of yet, conducted a study
of research and development tax credit carryforwards. Such a study, once undertaken by the Company, may result in an adjustment to the
research and development tax credit carryforwards; however, a full valuation allowance has been provided against the Company’s
research and development tax credits and, if an adjustment is required, this adjustment would be offset by an adjustment to the valuation
allowance. Thus, there would be no impact to the balance sheet or statement of operations if an adjustment is required.
The Company files tax returns in the United States,
California, Virginia, and New York. The Company is subject to U.S. federal and state tax examinations by tax authorities for the tax
years ended December 31, 2019 through present. As of December 31, 2023 and 2022, the Company has recorded no liability for unrecognized
tax benefits, interest, or penalties related to federal and state income tax matters and there currently no pending tax examinations.
The Company will recognize interest and penalties related to uncertain tax positions in income tax expense.
NOTE 12 – SUBSEQUENT EVENTS
Closing of Private Placement
On December 29, 2023, the Company entered into
a securities purchase agreement (the “Purchase Agreement”) with an institutional investor (“the “December Purchaser”)
for the issuance and sale in a private placement (the “December Private Placement”) of (i) pre-funded warrants (the “December
Pre-Funded Warrants”) to purchase up to 1,237,114 shares of the Company’s Common Stock, par value $0.001 at an exercise price
of $0.001 per share, and (ii) warrants (the “December Common Warrants”) to purchase up to 2,474,228 shares of the Company’s
Common Stock, at a purchase price of $4.85 per share.
Pursuant to the Purchase Agreement, the Company
agreed to reduce the exercise price of certain outstanding warrants to purchase Common Stock of the Company (“Certain Outstanding
Warrants”) held by the Purchaser to $4.60 per share in consideration for the cash payment by the December Purchaser of $0.125 per
share of Common Stock underlying the Certain Outstanding Warrants, effective immediately.
The December Private Placement closed on January
4, 2024. The net proceeds to the Company from the December Private Placement were approximately $5.5 million, after deducting placement
agent fees and expenses and estimated offering expenses payable by the Company.
In addition, the Company agreed to pay H.C. Wainwright
& Co., LLC (“Wainwright”) certain expenses and issued to Wainwright or its designees warrants (the “December Placement
Agent Warrants”) to purchase up to an aggregate of 74,227 shares of Common Stock at an exercise price equal to $6.0625 per share.
The December Placement Agent Warrants are exercisable immediately upon issuance and have a term of exercise equal to three years from
the date of issuance.
Secured Notes Amendments and Assignment
On January 2, 2024, the Company and certain holders
of the secured notes (the “Holders”) entered into amendments to the January 2024 Secured Notes (“Amendment No. 1 to
January 2024 Secured Notes”), pursuant to which the maturity date of the January 2024 Notes was extended to January 5, 2024.
On January 5, 2024, the Company and the Holders
entered into amendments to the January 2024 Secured Notes (“Amendment No. 2 to January 2024 Secured Notes”) and amendments
to the September 2024 Secured Notes (“Amendment No. 1 to September 2024 Secured Notes”), pursuant to which the Company and
the Holders agreed that in consideration of a principal payment in the aggregate amount of $1 million on the January 2024 Secured Notes
and in increase in the aggregate principal balance of $250,000 on the September 2024 Secured Notes, that the maturity date of the January
2024 Secured Notes would be further extended to January 31, 2024.
On January 31, 2024, the Company and the Holders
entered into amendments to the January 2024 Secured Notes (“Amendment No. 3 to January 2024 Secured Notes”), pursuant to
which the maturity date of the January 2024 Notes was extended to February 29, 2024. In addition, on January 31, 2024, the Company and
the Holders entered into amendments to the September 2024 Secured Notes (“Amendment No. 2 to September 2024 Secured Notes”),
pursuant to which the Company and the Holders agreed that in consideration of a principal payment in the aggregate amount of $1.25 million
on the January 2024 Secured Notes and in increase in the aggregate principal balance of $300,000 on the September 2024 Secured Notes.
Pursuant to Amendment No. 3 to the January 2024
Secured Notes, the Company was required to make the Additional Consideration payment no later than February 9, 2024. As a result of the
Company’s failure to make the Additional Consideration payment by February 9, 2023, the January 2024 Secured Notes and the September
2024 Secured Notes were in default and the entire principal balance of the January 2024 Secured Notes and the September 2024 Secured
Notes, without demand or notice, were due and payable.
As a result of the defaults on the January 2024
Secured Notes and the September 2024 Secured Notes, the Company was in default on the Business Loan and Security Agreement dated January
24, 2024 (the January Business Loan”), which has a current balance of approximately $5.2 million, and the Business Loan and Security
Agreement dated November 7, 2023 (the “November Business Loan”) which had a current balance of approximately $2.7 million.
On February 26, 2024, the Company and the Holders
entered into an Assignment Agreement (the “February Assignment Agreement”), pursuant to which the Company assigned all remaining
amounts due under the January 2024 Secured Notes, the September 2024 Secured Notes and the Unsecured Notes (collectively, the “Notes”)
back to the Holders. In connection with the February Assignment Agreement, the Company and the Holders entered into a payoff letter (the
“Payoff Letter”) and amendments to the January 2024 Secured Notes (“Amendment No. 4 to January 2024 Secured Notes”),
pursuant to which the maturity date of the January 2024 Secured Notes was extended to March 31, 2024 and the outstanding balance under
the Notes, after giving effect to the transactions contemplated by the February Assignment Agreement as applied pursuant to the Payoff
Letter, was adjusted to $250,000. On April 15, 2024, the Company has repaid the $250,000.
Settlement Agreement
On January 3, 2024, the Company entered into
a settlement agreement and general release with an investor (the “Settlement Agreement”), pursuant to which the Company and
the investor agreed to settle an action filed in the United States District Court in the Southern District of New York by an investor
against the Company (the “Action”) in consideration of the issuance by the Company of shares of the Company’s Common
Stock (the “Settlement Shares”). The number of Settlement Shares to be issued will be equal to $1.6 million divided by the
closing price of the Company’s Common Stock on the day prior to court approval of the joint motion. Following the issuance of the
Settlement Shares, the Investor will file a dismissal stipulation in the Action.
On January 17, 2024, the Company issued 296,296
Settlement Shares to the investor. The Settlement Shares were issued pursuant to an exemption from registration pursuant to Section 3(a)(10)
under the Securities Act of 1933, as amended.
Closing of MDNA Transaction
On January 4, 2024 (the “Closing Date”),
the Company completed its acquisition of certain assets and issued to MDNA Lifesciences, Inc. (“MDNA”): the Company’s
Common Stock, the Company’s Warrants, and the Pearsanta Preferred Stock. The Company expects to account for this transaction as
an asset acquisition.
On January 4, 2024, the Company, Pearsanta and
MDNA entered into a First Amendment to Asset Purchase Agreement (the “First Amendment to Asset Purchase Agreement”), pursuant
to which the parties agreed to: (i) the removal of an upfront working capital payment, (ii) the removal of a Closing Working Capital
Payment (as defined in the Purchase Agreement”), and (iii) to increase the maximum amount of payments to be made by Aditxt under
the Transition Services Agreement (as defined below) from $2.2 million to $3.2 million.
On January 4, 2024, Pearsanta and MDNA entered
into a Transition Services Agreement (the “Transition Services Agreement”), pursuant to which MDNA agreed that it would perform,
or cause certain of its affiliates or third parties to perform, certain services as described in the Transition Services Agreement for
a term of three months in consideration for the payment by Pearsanta of certain fees as provided in the Transition Services Agreement,
in an amount not to exceed $3.2 million.
Evofem Merger Agreement and Amendments
As previously reported in a Current Report on
Form 8-K filed by the Company, on December 11, 2023 the Company entered into an Agreement and Plan of Merger (the “Merger Agreement”)
with Adicure, Inc., a Delaware corporation and wholly owned subsidiary of the Company (“Merger Sub”) and Evofem Biosciences,
Inc., a Delaware corporation (“Evofem”), pursuant to which, Merger Sub will be merged into and with Evofem (the “Merger”),
with Evofem surviving the Merger as a wholly owned subsidiary of the Company.
On January 8, 2024, the Company, Adicure, Inc.,
a Delaware corporation and wholly owned subsidiary of the Company (“Merger Sub”), and Evofem Biosciences, Inc., a Delaware
corporation (“Evofem”) entered into the First Amendment (the “First Amendment to Merger Agreement”), to the Agreement
and Plan of Merger (the “Merger Agreement”) pursuant to which the parties agreed to extend the date by which the joint proxy
statement would be filed with the SEC until February 14, 2024.
On January 30, 2024, the Company, Adicure and
Evofem entered into the Second Amendment to the Merger Agreement (the “Second Amendment to Merger Agreement”) to amend (i)
the date of the Parent Loan (as defined in the Merger Agreement) to Evofem to be February 29, 2024, (ii) to change the date by which
Evofem may terminate the Merger Agreement for failure to receive the Parent Loan to be February 29, 2024, and (iii) to change the filing
date for the Joint Proxy Statement (as defined in the Merger Agreement) to April 1, 2024.
On February 29, 2024, the Company, Adicure and
Evofem entered into the Third Amendment to the Merger Agreement (the “Third Amendment to Merger Agreement”) in order to (i)
make certain conforming changes to the Merger Agreement regarding the Notes, (ii) extend the date by which the Company and Evofem will
file the joint proxy statement until April 30, 2024, and (iii) remove the requirement that the Company make the Parent Loan (as defined
in the Merger Agreement) by February 29, 2024 and replace it with the requirement that the Company make an equity investment into Evofem
consisting of (a) a purchase of 2,000 shares of Evofem Series F-1 Preferred Stock for an aggregate purchase price of $2.0 million on
or prior to April 1, 2024, and (b) a purchase of 1,500 shares of Evofem Series F-1 Preferred Stock for an aggregate purchase price of
$1.5 million on or prior to April 30, 2024. As of the date of this filing the Company has not purchased the 2,000 shares of EvoFem Series
F-1 Preferred Stock.
Business Loan Agreement
On January 24, 2024, the Company entered into
a Business Loan and Security Agreement (the “January Loan Agreement”) with a commercial funding source (the “Lender”),
pursuant to which the Company obtained a loan from the Lender in the principal amount of $3,600,000, which includes origination fees
of $252,000 (the “January Loan”). Pursuant to the January Loan Agreement, the Company granted the Lender a continuing secondary
security interest in certain collateral (as defined in the January Loan Agreement). The total amount of interest and fees payable by
the Company to the Lender under the January Loan will be $5,364,000, which will be repayable by the Company in 30 weekly installments
of $178,800. The Company received net proceeds from the January Loan of $814,900 following repayment of the outstanding balance on the
October Purchased Amount of $2,533,100.
Brain Scientific Assignment Agreement
On January 24, 2024, the Company entered into
an Assignment and Assumption Agreement (the “Brain Assignment Agreement”) with the agent (the “Agent”) of certain
secured creditors (the “Brain Creditors”) of Brain Scientific, Inc., a Nevada corporation (“Brain Scientific”)
and Philip J. von Kahle (the “Brain Seller”), as assignee of Brain Scientific and certain affiliated entities (collectively,
the “Brain Companies”) under an assignment for the benefit of creditors pursuant to Chapter 727 of the Florida Statutes.
Pursuant to the Brain Assignment Agreement, the Agent assigned its rights under that certain Asset Purchase and Settlement Agreement
dated October 31, 2023 between the Seller and the Agent (the “Brain Asset Purchase Agreement”) to the Company in consideration
for the issuance by the Company of an aggregate of 6,000 shares of a new series of convertible preferred stock of the Company, designated
as Series B-1 Convertible Preferred Stock, $0.001 par value (the “Series B-1 Preferred Stock”). The shares of Series B-1
Preferred Stock were issued pursuant to a Securities Purchase Agreement entered into by and between the Company and each of the purchasers
signatory thereto (the “Brain Purchase Agreement”).
In connection with the Brain Assignment Agreement,
on January 24, 2024, the Company entered into a Patent Assignment with the Brain Seller (the “Brain Patent Assignment”),
pursuant to which the Seller assigned all of its rights, titles and interests in certain patents and patent applications that were previously
held by the Brain Companies to the Company.
Series B-1 Preferred Stock Certificate of
Designation
On January 24, 2024, the Company filed a Certificate
of Designations for its Series B-1 Preferred Stock with the Secretary of State of Delaware. See Series B-1 Preferred Stock certificate
of designation incorporated by reference to this document.
Officer Promissory Notes
On January 8, 2024, the Company fully repaid
the November Note, First December Note, and Second December Note to Amro Albanna, the Company’s Chief Executive Officer.
On February 7, 2024, Amro Albanna, the Chief
Executive Officer of the Company loaned $30,000 to the Company. The loan was evidenced by an unsecured promissory note (the “February
7th Note”). Pursuant to the terms of the February 7th Note, it will accrue interest at the Prime rate of eight and one-half percent
(8.5%) per annum and is due on the earlier of August 7, 2024 or an event of default, as defined therein.
On February 15, 2024, Amro Albanna, the Chief
Executive Officer of the Company loaned $205,000 to the Company. The loan was evidenced by an unsecured promissory note (the “February
15th Note”). Pursuant to the terms of the February 15th Note, it will accrue interest at the Prime rate of eight and one-half percent
(8.5%) per annum and is due on the earlier of August 15, 2024 or an event of default, as defined therein.
On February 29, 2024, Amro Albanna, the Chief
Executive Officer of the Company, and Shahrokh Shabahang, the Chief Innovation Officer of the Company, loaned $117,000 and $115,000,
respectively, to the Company. The loans were evidenced by an unsecured promissory note (the “February 29th Notes”). Pursuant
to the terms of the February 29th Notes, it will accrue interest at the Prime rate of eight and one-half percent (8.5%) per annum and
is due on the earlier of August 29, 2024 or an event of default, as defined therein.
Engagement Letter with Dawson James Securities,
Inc.
On February 16, 2024, the “Company”
entered into an engagement letter (the “Dawson Engagement Letter”) with Dawson James Securities, Inc.(“Dawson”),
pursuant to which the Company engaged Dawson to serve as financial advisor with respect to one or more potential business combinations
involving the Company for a term of twelve months. Pursuant to the Dawson Engagement Letter, the Company agreed to pay Dawson an initial
fee of $1.85 million (the “Dawson Initial Fee”), which amount is payable on the later of (i) the closing of an offering resulting
in gross proceeds to the Company of greater than $4.9 million, or (ii) five days after the execution of the Dawson Engagement Letter.
At the Company’s option, the Dawson Initial Fee may be paid in securities of the Company. In addition, with respect to any business
combination (i) that either is introduced to the Company by Dawson following the date of the Dawson Engagement Letter or (ii) that with
respect to which the Company hereafter requests Dawson to provide M&A advisory services, the Company shall compensate Dawson in an
amount equal to 5% of the Total Transaction Value (as defined in the Engagement Letter) with respect to the first $20.0 million in Total
Transaction Value plus 10.0% of the Total Transaction Value that is in excess of $20.0 million (the “Transaction Fee”). The
Transaction Fee is payable upon the closing of a business combination transaction.
Lease Default
On March 6, 2024, the Company received correspondence
from 532 Realty Associates, LLC (the “Landlord”) that the Company is in default under that certain Agreement of Lease dated
November 3, 2021 by and between the Landlord and the Company (the “New York Lease”) for failure to pay Basic Rent and Additional
Rent (as each term is defined in the New York Lease) in the aggregate amount of $40,707 (the “Past Due Rent”).
Promissory Note
On March 7, 2024, Sixth Borough Capital Fund,
LP loaned $300,000 to the Company. The loan was evidenced by an unsecured promissory note (the “Sixth Borough Note”). Pursuant
to the terms of the Sixth Borough Note, it will accrue interest at the Prime rate of eight and one-half percent (8.5%) per annum and
is due on the earlier of March 31, 2024 or an event of default, as defined therein.
Appili Arrangement Agreement
On April 1, 2024 (the “Execution Date”),
the Company, entered into an Arrangement Agreement (the “Arrangement Agreement”) with Adivir, Inc., a Delaware corporation
and wholly owned subsidiary of the Company (“Adivir” or the “Buyer”), and Appili Therapeutics, Inc., a Canadian
corporation (“Appili”), pursuant to which, Adivir will acquire all of the issued and outstanding Class A common shares of
Appili (the “Appili Shares”) on the terms and subject to the conditions set forth therein. The acquisition of the Appili
Shares (the “Arrangement”) will be completed by way of a statutory plan of arrangement under the Canada Business Corporation
Act.
At the effective time of the Arrangement (the
“Effective Time”), each Appili Share outstanding immediately prior to the Effective Time (other than Appili Shares held by
a registered holder of Appili Shares who has validly exercised such holder’s dissent rights) will be deemed to be assigned and
transferred by the holder thereof to the Buyer in exchange for (i) $0.0467 in cash consideration per share for an aggregate cash payment
of $5,668,222 (the “Cash Consideration”) and (ii) 0.002745004 of a share of common stock of Aditxt or an aggregate of 332,876
shares (the “Consideration Shares” and together with the Cash Consideration, the “Transaction Consideration”).
In connection with the transaction, each outstanding option and warrant of Appili will be cashed-out based on the implied in-the-money
value of the Transaction Consideration, which is expected to result in an additional aggregate cash payment of approximately $341,000
(based on the number of issued and outstanding options and warrants and exchange rates as of the date of the Arrangement Agreement).
Promissory Note
On April
10, 2024, Sixth Borough Capital Fund, LP loaned $230,000 to the Company. The loan was evidenced by an unsecured promissory note (the
“April Sixth Borough Note”). Pursuant to the terms of the April Sixth Borough Note, it will accrue interest at the Prime
rate of eight and one-half percent (8.5%) per annum and is due on the earlier of April 19, 2024 or an event of default, as defined therein.
On May 9, 2024, at which point the balance of the loan was $35,256, Sixth Borough loaned an additional $20,000 to the Company bringing
the balance of the loan to $55,256.03.
Subsequent Events – Unaudited
Evofem Termination,
Reinstatement, and Fourth Amendement
On April 26, 2024, the
Company received notice from Evofem (the “Termination Notice”) that Evofem was exercising its right to terminate the Merger
Agreement as a result of the Company’s failure to provide the Initial Parent Equity Investment (as defined in the Merger Agreement,
as amended).
On May 2, 2024, the
Company, Adifem, Inc. f/k/a Adicure, Inc. and Evofem entered into the Reinstatement and Fourth Amendment to the Merger Agreement (the
“Fourth Amendment”) in order to waive and amend, among other things, the several provisions listed below.
Amendments to Article
VI: Covenants and Agreement
Article VI of the Merger
Agreement is amended to:
| ● | reinstate
the Merger Agreement, as amended by the Fourth Amendment, as if never terminated; |
| | | |
| ● | reflect
the Company’s payment to Evofem, in the amount of $1,000,000 (the “Initial Payment”),
via wire initiated by May 2, 2024; |
| | | |
| ● | delete
Section 6.3, which effectively eliminates the “no shop” provision, and the several
defined terms used therein; |
| | | |
| ● | add a new
defined term “Company Change of Recommendation;” and |
| | | |
| ● | revise
section 6.10 of the Merger Agreement such that, after the Initial Payment, and upon the closing
of each subsequent capital raise by the Company (each a “Parent Subsequent Capital
Raise”), the Company shall purchase that number of shares of Evofem’s Series
F-1 Preferred Stock, par value $0.0001 per share (the “Series F-1 Preferred Stock”),
equal to forty percent (40%) of the gross proceeds of such Parent Subsequent Capital Raise
divided by 1,000, up to a maximum aggregate amount of $2,500,000 or 2,500 shares of Series
F-1 Preferred Stock. A maximum of$1,500,000 shall be raised prior to June 17, 2024 and $1,000,000
prior to July 1, 2024 (the “Parent Capital Raise”). |
Amendments to Article
VIII: Termination
Article VIII of the
Merger Agreement is amended to:
| ● | extend
the date after which either party may terminate from May 8, 2024 to July 15, 2024; |
| | | |
| ● | revise
Section 8.1(d) in its entirety to allow Company to terminate at any time after there has
been a Company Change of Recommendation, provided that Aditxt must receive ten day written
notice and have the opportunity to negotiate a competing offer in good faith; and |
| | | |
| ● | amend and
restate Section 8.1(f) in its entirety, granting the Company the right to terminate the agreement
if (a) the full $1,000,000 Initial Payment required by the Fourth Amendment has not been
paid in full by May 3, 2024 (b) $1,500,000 of the Parent Capital Raise Amount has not been
paid to the Company by June 17, 2024, (c) $1,000,000 of the Parent Capital Raise Amount has
not been paid to the Company by July 1, 2024, or (d) Aditxt does not pay any portion of the
Parent Equity Investment within five calendar days after each closing of a Parent Subsequent
Capital Raise. |
Equity Line of
Credit
On May 2, 2024, Aditxt
entered into a Common Stock Purchase Agreement (the “ELOC Purchase Agreement”) with an equity line investor (the “ELOC
Investor”), pursuant to which the ELOC Investor has agreed to purchase from the Company, at the Company’s direction from
time to time, in its sole discretion, from and after the date effective date of the Registration Statement (as defined below) and until
the termination of the ELOC Purchase Agreement in accordance with the terms thereof, shares of the Company’s common stock having
a total maximum aggregate purchase price of $150,000,000 (the “ELOC Purchase Shares”), upon the terms and subject to the
conditions and limitations set forth in the ELOC Purchase Agreement.
In connection with the
ELOC Purchase Agreement, the Company also entered into a Registration Rights Agreement with the Investor (the “Registration Rights
Agreement”), pursuant to which the Company agreed to file a registration statement with the Securities and Exchange Commission
covering the resale of the shares of common stock issued to the ELOC Investor pursuant to the ELOC Purchase Agreement (the “ELOC
Registration Statement”) by the later of (i) the 30th calendar day following the closing date, and (ii) the second business day
following Stockholder Approval (defined below).
The Company may, from
time to time and at its sole discretion, direct the ELOC Investor to purchase shares of its common stock upon the satisfaction of certain
conditions set forth in the ELOC Purchase Agreement at a purchase price per share based on the market price of the Company’s common
stock at the time of sale as computed under the ELOC Purchase Agreement. There is no upper limit on the price per share that the ELOC
Investor could be obligated to pay for common stock under the ELOC Purchase Agreement. The Company will control the timing and amount
of any sales of its common stock to the Investor, and the Investor has no right to require us to sell any shares to it under the ELOC
Purchase Agreement. Actual sales of shares of common stock to the Investor under the ELOC Purchase Agreement will depend on a variety
of factors to be determined by the Company from time to time, including (among others) market conditions, the trading price of its common
stock and determinations by the Company as to available and appropriate sources of funding for the Company and its operations. The Investor
may not assign or transfer its rights and obligations under the ELOC Purchase Agreement.
Under the applicable
Nasdaq rules, in no event may the Company issue to the ELOC Investor under the ELOC Purchase Agreement more than 332,876 shares of common
stock, which number of shares is equal to 19.99% of the shares of the common stock outstanding immediately prior to the execution of
the ELOC Purchase Agreement (the “Exchange Cap”), unless (i) the Company obtains stockholder approval to issue shares of
common stock in excess of the Exchange Cap in accordance with applicable Nasdaq rules (“Stockholder Approval”), or (ii) the
average price per share paid by the ELOC Investor for all of the shares of common stock that the Company directs the ELOC Investor to
purchase from the Company pursuant to the ELOC Purchase Agreement, if any, equals or exceeds the official closing sale price on the Nasdaq
Capital Market immediately preceding the delivery of the applicable purchase notice to the Investor and (B) the average of the closing
sale prices of the Company’s common stock on the Nasdaq Capital market for the five business days immediately preceding the delivery
of such purchase notice.
In all cases, the Company
may not issue or sell any shares of common stock to the ELOC Investor under the ELOC Purchase Agreement which, when aggregated with all
other shares of the Company’s common stock then beneficially owned by the Investor and its affiliates, would result in the Investor
beneficially owning more than 4.99% of the outstanding shares of the Company’s common stock.
As consideration for
the ELOC Investor’s commitment to purchase shares of common stock at the Company’s direction upon the terms and subject to
the conditions set forth in the ELOC Purchase Agreement, the Company shall pay the ELOC Investor a commitment fee as outlined in the
ELOC Purchase Agreement, which is payable on the later of (i) January 2, 2025 and (ii) the trading day following the date on which Stockholder
Approval is obtained.
May 2024 PIPE
On May 2, 2024, Aditxt
entered into a Securities Purchase Agreement (the “May 2024 PIPE Purchase Agreement”) with certain accredited investors,
pursuant to which the Company agreed to issue and sell to such investors in a private placement (the “May 2024 Private Placement”)
(i) an aggregate of 4,186 shares of the Company’s Series C-1 Convertible Preferred Stock (the “Series C-1 Preferred Stock”),
(ii) an aggregate of 4,186 shares of the Company’s Series D-1 Preferred Stock (the “Series D-1 Preferred Stock”), and
(iii) warrants (the “May 2024 PIPE Warrants”) to purchase up to an aggregate of 1,613,092 shares of the Company’s common
stock.
The May 2024 PIPE Warrants
are exercisable commencing six months following the initial issuance date at an initial exercise price of $2.47 per share and expire
five years from the date of issuance.
Lease Default
On May 10, 2024, the
Company received written notice (the “Lease Default Notice”) from LS Biotech Eight, LLC (the “Landlord”)
that the Company was in violation of its obligation to (i) pay Base Rent (as defined in the Lease) and Additional Rent (as defined in
the Lease) in the amount of $431,182.32 in the aggregate, together with administrative charges and interest, as well as (ii) replenish
the Security Deposit (as defined in the Lease) in the amount of $159,375.00, all as required under that certain Lease Agreement dated
as of May 4, 2021 by and between the Landlord and the Company (the “Lease”). Pursuant to the Lease Default Notice,
the Landlord has demanded that a payment of $590,557.31 plus administrative charges and interest, which shall accrue at the Default Rate
(as defined in the Lease) be made no later than May 17, 2024.
The
Company is working with the Landlord to come to an amicable resolution. However, no assurance can be given that the parties will reach
an amicable resolution on a timely basis, on favorable terms, or at all.
ADITXT, INC.
CONSOLIDATED BALANCE SHEETS
(unaudited)
| | |
March 31, | | |
December 31, | |
| | |
2024 | | |
2023 | |
| ASSETS | |
| | |
| |
| CURRENT ASSETS: | |
| | |
| |
| Cash | |
$ | 88,671 | | |
$ | 97,102 | |
| Accounts receivable, net | |
| 427,044 | | |
| 408,326 | |
| Inventory | |
| 622,768 | | |
| 745,502 | |
| Prepaid expenses | |
| 456,884 | | |
| 217,390 | |
| Subscription receivable | |
| - | | |
| 5,444,628 | |
| TOTAL CURRENT ASSETS | |
| 1,595,367 | | |
| 6,912,948 | |
| | |
| | | |
| | |
| Fixed assets, net | |
| 2,021,760 | | |
| 1,898,243 | |
| Intangible assets, net | |
| 8,611 | | |
| 9,444 | |
| Deposits | |
| 132,496 | | |
| 106,410 | |
| Right of use asset - long term | |
| 1,940,076 | | |
| 2,200,299 | |
| Investment in Evofem | |
| 22,277,211 | | |
| 22,277,211 | |
| Deposit on acquisition | |
| - | | |
| 11,173,772 | |
| TOTAL ASSETS | |
$ | 27,975,521 | | |
$ | 44,578,327 | |
| | |
| | | |
| | |
| LIABILITIES AND STOCKHOLDERS’ EQUITY | |
| | | |
| | |
| CURRENT LIABILITIES: | |
| | | |
| | |
| Accounts payable and accrued expenses | |
$ | 9,360,680 | | |
$ | 8,554,959 | |
| Notes payable - related party | |
| 467,000 | | |
| 375,000 | |
| Notes payable, net of discount | |
| 5,678,182 | | |
| 15,653,477 | |
| Financing on fixed assets | |
| 147,823 | | |
| 147,823 | |
| Deferred rent | |
| 147,350 | | |
| 158,612 | |
| Lease liability - current | |
| 900,979 | | |
| 999,943 | |
| Advance on private placement | |
| 600,000 | | |
| - | |
| TOTAL CURRENT LIABILITIES | |
| 17,302,014 | | |
| 25,889,814 | |
| | |
| | | |
| | |
| Settlement liability | |
| - | | |
| 1,600,000 | |
| Lease liability - long term | |
| 891,747 | | |
| 1,041,744 | |
| | |
| | | |
| | |
| TOTAL LIABILITIES | |
| 18,193,761 | | |
| 28,531,558 | |
| | |
| | | |
| | |
| COMMITMENTS AND CONTINGENCIES | |
| | | |
| | |
| | |
| | | |
| | |
| STOCKHOLDERS’ EQUITY | |
| | | |
| | |
| Preferred stock, $0.001 par value, 3,000,000 shares
authorized, zero shares issued and outstanding, respectively | |
| - | | |
| - | |
| Series A-1 Convertible Preferred stock, $0.001 par value, 22,280
shares authorized, 22,280 and 22,280 shares issued and outstanding, respectively | |
| 22 | | |
| 22 | |
| Series B Preferred stock, $0.001 par value, 1 share authorized,
zero and zero shares issued and outstanding, respectively | |
| - | | |
| - | |
| Series B-1 Convertible Preferred stock, $0.001 par value, 6,000
shares authorized, 6,000 and zero shares issued and outstanding, respectively | |
| 6 | | |
| - | |
| Series B-2 Convertible Preferred stock, $0.001 par value, 2,625
shares authorized, 2,625 and 2,625 shares issued and outstanding, respectively | |
| 3 | | |
| 3 | |
| Series C Preferred stock, $0.001 par value, 1 share authorized,
zero and zero shares issued and outstanding, respectively | |
| - | | |
| - | |
| Common stock, $0.001 par value, 100,000,000 shares
authorized, 1,665,265 and 1,318,968 shares issued and 1,665,214 and 1,318,918 shares outstanding,
respectively | |
| 1,665 | | |
| 1,319 | |
| Treasury stock, 51 and 51 shares, respectively | |
| (201,605 | ) | |
| (201,605 | ) |
| Additional paid-in capital | |
| 152,601,043 | | |
| 143,997,710 | |
| Accumulated deficit | |
| (142,470,799 | ) | |
| (127,741,072 | ) |
| TOTAL ADITXT, INC. STOCKHOLDERS’
EQUITY | |
| 9,930,335 | | |
| 16,056,377 | |
| | |
| | | |
| | |
| NON-CONTROLLING INTEREST | |
| (148,575 | ) | |
| (9,608 | ) |
| | |
| | | |
| | |
| TOTAL STOCKHOLDERS’
EQUITY | |
| 9,781,760 | | |
| 16,046,769 | |
| | |
| | | |
| | |
| TOTAL LIABILITIES AND
STOCKHOLDERS’ EQUITY | |
$ | 27,975,521 | | |
$ | 44,578,327 | |
See accompanying notes to the consolidated financial
statements.
ADITXT, INC.
CONSOLIDATED STATEMENTS
OF OPERATIONS
(unaudited)
| | |
Three Months
Ended | | |
Three Months
Ended | |
| | |
March 31,
2024 | | |
March 31,
2023 | |
| REVENUE | |
| | |
| |
| Sales | |
$ | 79,680 | | |
$ | 218,415 | |
| Cost of goods sold | |
| 65,799 | | |
| 178,309 | |
| Gross profit (loss) | |
| 13,881 | | |
| 40,106 | |
| | |
| | | |
| | |
| OPERATING EXPENSES | |
| | | |
| | |
| General and administrative expenses, includes $24,573, and $274,315
in stock-based compensation, respectively | |
| 3,363,748 | | |
| 4,368,843 | |
| Research and development, includes $6,712,663, and $62,633 in
stock-based compensation, respectively | |
| 8,145,266 | | |
| 1,387,541 | |
| Sales and marketing $0, and $2,503 in stock-based
compensation, respectively | |
| 40,513 | | |
| 65,617 | |
| Total operating expenses | |
| 11,549,527 | | |
| 5,822,001 | |
| | |
| | | |
| | |
| NET LOSS FROM OPERATIONS | |
| (11,535,646 | ) | |
| (5,781,895 | ) |
| | |
| | | |
| | |
| OTHER EXPENSE | |
| | | |
| | |
| Interest expense | |
| (2,489,045 | ) | |
| (198,492 | ) |
| Interest income | |
| 377 | | |
| 9,074 | |
| Amortization of debt discount | |
| (635,710 | ) | |
| (13,393 | ) |
| Loss on note exchange agreement | |
| (208,670 | ) | |
| - | |
| Total other expense | |
| (3,333,048 | ) | |
| (202,811 | ) |
| | |
| | | |
| | |
| Net loss before income taxes | |
| (14,868,694 | ) | |
| (5,984,706 | ) |
| Income tax provision | |
| - | | |
| - | |
| | |
| | | |
| | |
| NET LOSS | |
$ | (14,868,694 | ) | |
$ | (5,984,706 | ) |
| | |
| | | |
| | |
| NET LOSS ATTRIBUTABLE
TO NON-CONTROLLING INTEREST | |
| (138,967 | ) | |
| - | |
| | |
| | | |
| | |
| NET LOSS ATTRIBUTABLE
TO ADITXT, INC. & SUBSIDIARIES | |
$ | (14,729,727 | ) | |
$ | (5,984,706 | ) |
| | |
| | | |
| | |
| Net loss per share - basic and diluted | |
$ | (9.14 | ) | |
$ | (52.46 | ) |
| | |
| | | |
| | |
| Weighted average number of shares outstanding
during the period - basic and diluted | |
| 1,610,872 | | |
| 114,072 | |
See accompanying notes to the consolidated financial
statements.
ADITXT, INC.
CONSOLIDATED STATEMENTS
OF STOCKHOLDERS’ EQUITY
THREE MONTHS ENDED MARCH 31, 2024 AND 2023
(unaudited)
| | |
Preferred
A-1
Shares
Outstanding | | |
Preferred
A-1
Shares
Par | | |
Preferred
B-1
Shares
Outstanding | | |
Preferred
B-1
Shares
Par | | |
Preferred
B-2
Shares
Outstanding | | |
Preferred
B-2
Shares
Par | | |
Common
Shares
Outstanding | | |
Common
Shares
Par | | |
Treasury
Stock | | |
Additional
Paid-in
Capital | | |
Accumulated
Deficit | | |
Non-
Controlling Interest | | |
Total
Stockholders’
Equity | |
| Balance December 31, 2023 | |
| 22,280 | | |
$ | 22 | | |
| - | | |
$ | - | | |
| 2,625 | | |
$ | 3 | | |
| 1,318,918 | | |
$ | 1,319 | | |
$ | (201,605 | ) | |
$ | 143,997,710 | | |
$ | (127,741,072 | ) | |
$ | (9,608 | ) | |
$ | 16,046,769 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Stock option compensation | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 24,573 | | |
| - | | |
| - | | |
| 24,573 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| MDNA asset purchase | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 50,000 | | |
| 50 | | |
| - | | |
| 1,008,619 | | |
| - | | |
| - | | |
| 1,008,669 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Brain asset purchase | |
| - | | |
| - | | |
| 6,000 | | |
| 6 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 5,970,437 | | |
| - | | |
| - | | |
| 5,970,443 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Issuance of shares for settlement | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 296,296 | | |
| 296 | | |
| - | | |
| 1,599,704 | | |
| - | | |
| - | | |
| 1,600,000 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Net loss | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| (14,729,727 | ) | |
| (138,967 | ) | |
| (14,868,694 | ) |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Balance March 31, 2024 | |
| 22,280 | | |
$ | 22 | | |
| 6,000 | | |
$ | 6 | | |
| 2,625 | | |
$ | 3 | | |
| 1,665,214 | | |
$ | 1,665 | | |
$ | (201,605 | ) | |
$ | 152,601,043 | | |
$ | (142,470,799 | ) | |
$ | (148,575 | ) | |
$ | 9,781,760 | |
See accompanying notes to the consolidated
financial statements.
ADITXT, INC.
CONSOLIDATED STATEMENTS OF STOCKHOLDERS’
EQUITY
THREE MONTHS ENDED MARCH 31, 2024 AND 2023
(unaudited)
| | |
Preferred
A-1
Shares
Outstanding | | |
Preferred
A-1
Shares
Par | | |
Preferred
B-1
Shares
Outstanding | | |
Preferred
B-1
Shares
Par | | |
Preferred
B-2
Shares
Outstanding | | |
Preferred
B-2
Shares
Par | | |
Common
Shares
Outstanding | | |
Common
Shares
Par | | |
Treasury
Stock | | |
Additional
Paid-in
Capital | | |
Accumulated
Deficit | | |
Non-
Controlling Interest | | |
Total
Stockholders’
Equity | |
| Balance December 31, 2022 | |
| - | | |
$ | - | | |
| - | | |
$ | - | | |
| - | | |
$ | - | | |
| 107,647 | | |
$ | 108 | | |
$ | (201,605 | ) | |
$ | 100,448,166 | | |
$ | (95,040,362 | ) | |
$ | - | | |
$ | 5,206,307 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Stock option compensation | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 59,964 | | |
| - | | |
| - | | |
| 59,964 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Restricted stock unit compensation | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 111,187 | | |
| - | | |
| - | | |
| 111,187 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Issuance of restricted stock units for compensation | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 44 | | |
| 1 | | |
| - | | |
| (1 | ) | |
| - | | |
| - | | |
| - | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Sale of common stock | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 8,463 | | |
| 9 | | |
| - | | |
| 507,007 | | |
| - | | |
| - | | |
| 507,016 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Issuance of shares for services | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| 4,675 | | |
| 5 | | |
| | | |
| 168,295 | | |
| | | |
| | | |
| 168,300 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Net loss | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| (5,984,706 | ) | |
| - | | |
| (5,984,706 | ) |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Balance March 31, 2023 | |
| - | | |
$ | - | | |
| - | | |
$ | - | | |
| - | | |
$ | - | | |
| 120,829 | | |
$ | 123 | | |
$ | (201,605 | ) | |
$ | 101,294,618 | | |
$ | (101,025,068 | ) | |
$ | - | | |
$ | 68,068 | |
See accompanying notes to the consolidated financial
statements.
ADITXT, INC.
CONSOLIDATED STATEMENTS
OF CASH FLOWS
(unaudited)
| | |
Three Months Ended | | |
Three Months Ended | |
| | |
March 31,
2024 | | |
March 31,
2023 | |
| CASH FLOWS FROM OPERATING ACTIVITIES: | |
| | |
| |
| Net loss | |
$ | (14,868,694 | ) | |
$ | (5,984,706 | ) |
| Adjustments to reconcile net loss to net
cash used in operating activities | |
| | | |
| | |
| Stock-based compensation | |
| 24,573 | | |
| 339,451 | |
| Stock-based compensation from asset purchase | |
| 6,712,663 | | |
| - | |
| Depreciation expense | |
| 142,932 | | |
| 109,896 | |
| Amortization of intangible assets | |
| 833 | | |
| 26,750 | |
| Amortization of debt discount | |
| 635,710 | | |
| - | |
| Loss on note exchange agreement | |
| 208,670 | | |
| - | |
| Changes in operating assets and liabilities: | |
| | | |
| | |
| Accounts receivable | |
| (18,718 | ) | |
| 198,940 | |
| Prepaid expenses | |
| (239,494 | ) | |
| (51,533 | ) |
| Deposits | |
| (26,086 | ) | |
| (54,940 | ) |
| Inventory | |
| 122,734 | | |
| 186,563 | |
| Accounts payable and accrued expenses | |
| 1,343,946 | | |
| 1,277,788 | |
| Net cash used in operating activities | |
| (5,960,931 | ) | |
| (3,951,791 | ) |
| | |
| | | |
| | |
| CASH FLOWS FROM INVESTING ACTIVITIES: | |
| | | |
| | |
| Purchase of fixed assets | |
| - | | |
| (5,049 | ) |
| Net cash used in investing activities | |
| - | | |
| (5,049 | ) |
| | |
| | | |
| | |
| CASH FLOWS FROM FINANCING ACTIVITIES: | |
| | | |
| | |
| Proceeds from notes - related party | |
| 467,000 | | |
| - | |
| Proceeds from notes and convertible notes payable, net of offering
costs | |
| 1,269,950 | | |
| 1,245,853 | |
| Repayments of note payable - related party | |
| (375,000 | ) | |
| - | |
| Repayments of note payable | |
| (1,906,052 | ) | |
| - | |
| New principal from extension of notes, net of debt discount | |
| 451,974 | | |
| - | |
| Advance on private placement | |
| 600,000 | | |
| - | |
| Common stock and warrants issued for cash, net of issuance
costs | |
| - | | |
| 507,016 | |
| Cash from subscription receivable | |
| 5,444,628 | | |
| - | |
| Payments on financing on fixed asset | |
| - | | |
| (262,160 | ) |
| Net cash provided by financing activities | |
| 5,952,500 | | |
| 1,490,709 | |
| | |
| | | |
| | |
| NET INCREASE IN CASH | |
| (8,431 | ) | |
| (2,466,131 | ) |
| | |
| | | |
| | |
| CASH AT BEGINNING OF PERIOD | |
| 97,102 | | |
| 2,768,640 | |
| | |
| | | |
| | |
| CASH AT END OF PERIOD | |
$ | 88,671 | | |
$ | 302,509 | |
| | |
| | | |
| | |
| Supplemental cash flow information: | |
| | | |
| | |
| Cash paid for income taxes | |
$ | - | | |
$ | - | |
| Cash paid for interest expense | |
$ | 622,762 | | |
$ | 193,175 | |
| | |
| | | |
| | |
| Issuance of shares in asset purchase | |
$ | 266,448 | | |
$ | - | |
| Shares issued for settlement | |
$ | 1,600,000 | | |
$ | - | |
| Return of notes payable from Evofem merger
agreement | |
$ | 11,174,426 | | |
$ | - | |
| Accrued interest rolled into notes payable | |
$ | 538,223 | | |
$ | - | |
See accompanying notes to the consolidated financial
statements.
ADITXT, INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
NOTE 1 – ORGANIZATION AND NATURE OF BUSINESS
Company Background
Overview
We are a biotech innovation company with a mission
of prolonging life and enhancing its quality by improving the health of the immune system. We are an innovation company developing and
commercializing technologies with a focus on monitoring and modulating the immune system. Our immune reprogramming technologies are currently
at the pre-clinical stage and are designed to retrain the immune system to induce tolerance with an objective of addressing rejection
of transplanted organs, autoimmune diseases, and allergies. Our immune monitoring technologies are designed to provide a personalized
comprehensive profile of the immune system and we plan to utilize them in our upcoming reprogramming clinical trials to monitor subjects’
immune response before, during and after drug administration.
On January 1, 2023, the Company formed Adimune,
Inc., a Delaware wholly owned subsidiary.
On January 1, 2023, the Company formed Pearsanta,
Inc., a Delaware majority owned subsidiary.
On April 13, 2023, the Company formed Adivir,
Inc., a Delaware wholly owned subsidiary.
On August 24, 2023, the Company formed Adivue,
Inc., a Delaware wholly owned subsidiary.
On October 16, 2023, the Company formed Adicure,
Inc., which was renamed Adifem, Inc., a Delaware wholly owned subsidiary.
Reverse Stock Split
On August 17, 2023, the Company effectuated a
1 for 40 reverse stock split (the “2023 Reverse Split”). The Company’s stock began trading on a split-adjusted
basis effective on the Nasdaq Stock Market on August 18, 2023. There was no change to the number of authorized shares of the Company’s
common stock. All share amounts referenced in this report are adjusted to reflect the 2023 Reverse Split.
Offerings
On August 31, 2021, the Company completed a registered
direct offering (“August 2021 Offering”). In connection therewith, the Company issued 2,292 shares of common stock,
at a purchase price of $4,800.00 per share, resulting in gross proceeds of approximately $11.0 million. In a concurrent private
placement, the Company issued warrants to purchase up to 2,292 shares. The warrants have an exercise price of $5,060.00 per
share and are exercisable for a five-year period commencing months from the date of issuance. The warrants exercise price
was subsequently repriced to $3,000.00. In addition, the Company issued a warrant to the placement agent to purchase up to 115 shares
of common stock at an exercise price of $6,000.00 per share.
On October 18, 2021, the Company entered into
an underwriting agreement with Revere Securities LLC, relating to the public offering (the “October 2021 Offering”) of 1,417 shares
of the Company’s common stock (the “Shares”) by the Company. The Shares were offered, issued, and sold at a price to
the public of $3,000.00 per share under a prospectus supplement and accompanying prospectus filed with the SEC pursuant to an effective
shelf registration statement filed with the SEC on Form S-3 (File No. 333-257645), which was declared effective by the SEC on July 13,
2021. The October 2021 Offering closed on October 20, 2021 for gross proceeds of $4.25 million. The Company utilized a portion of
the proceeds, net of underwriting discounts of approximately $3.91 million from the October 2021 Offering to fund certain obligations
of the Company.
On December 6, 2021, the Company completed a
public offering for net proceeds of $16.0 million (the “December 2021 Offering”). As part of the December 2021 Offering,
we issued 4,123 units consisting of shares of the Company’s common stock and warrant to purchase shares of the Company’s
common stock and 4,164 prefunded warrants. The warrant issued as part of the units had an exercise price of $2,300.00 and
the prefunded warrants had an exercise price of $0.04. On June 15, 2022, the Company entered an agreement with a holder of certain warrants
in the December 2021 Offering. (See Note 10)
On September 20, 2022, the Company completed
a public offering for net proceeds of $17.2 million (the “September 2022 Offering”). As part of the September 2022 Offering,
we issued 30,608 of shares of the Company’s common stock, pre-funded warrants to purchase 52,725 shares of
common stock, and warrants to purchase 83,333 shares of the Company’s common stock. The warrants had an exercise price
of $240.00 and the pre-funded warrants had an exercise price of $0.04.
On April 20, 2023, the Company entered into a
securities purchase agreement (the “April Purchase Agreement”) with an institutional investor, pursuant to which the Company
agreed to sell to such investor pre-funded warrants (the “April Pre-Funded Warrants”) to purchase up to 39,634 shares
of common stock of the Company (the “Common Stock”) at a purchase price of $48.76 per April Pre-Funded Warrant. The
April Pre-Funded Warrants (and shares of common stock underlying the April Pre-Funded Warrants) were offered by the Company pursuant
to its shelf registration statement on Form S-3 (File No. 333-257645), which was declared effective by the Securities and Exchange Commission
on July 13, 2021. Concurrently with the sale of the April Pre-Funded Warrants, pursuant to the Purchase Agreement in a concurrent
private placement, for each April Pre-Funded Warrant purchased by the investor, such investor received from the Company an unregistered
warrant (the “Warrant”) to purchase two shares of Common Stock. The warrants have an exercise price of $34.40 per
share, and are exercisable for a three year period. In addition, the Company issued a warrant to the placement agent to purchase
up to 2,378 shares of common stock at an exercise price of $61.00 per share. The closing of the sales of these securities under the April
Purchase Agreement took place on April 24, 2023. The gross proceeds from the offering were approximately $1.9 million, prior to
deducting placement agent’s fees and other offering expenses payable by the Company.
On August 31, 2023, the “Company entered
into a securities purchase agreement (the “August Purchase Agreement”) with an institutional investor for the issuance
and sale in a private placement (the “Private Placement”) of (i) pre-funded warrants (the “August Pre-Funded Warrants”)
to purchase up to 1,000,000 shares of the Company’s common stock at an exercise price of $0.001 per share, and (ii) warrants (the
“Common Warrants”) to purchase up to 1,000,000 shares of the Company’s Common Stock at an exercise price of $10.00
per share. The Private Placement closed on September 6, 2023. The net proceeds to the Company from the Private Placement were approximately
$9 million, after deducting placement agent fees and expenses and estimated offering expenses payable by the Company. The Company used
the net proceeds received from the Private Placement for (i) the payment of approximately $3.1 million in outstanding obligations, (ii)
the repayment of approximately $0.4 million of outstanding debt, and (iii) the balance for continuing operating expenses and working
capital.
On December 29, 2023, the Company entered into
a securities purchase agreement (the “Purchase Agreement”) with an institutional investor (“the “Purchaser”)
for the issuance and sale in a private placement (the “Private Placement”) of (i) pre-funded warrants (the “Pre-Funded
Warrants”) to purchase up to 1,237,114 shares of the Company’s common stock, par value $0.001 (the “Common Stock”)
at an exercise price of $0.001 per share, and (ii) warrants (the “Common Warrants”) to purchase up to 2,474,228 shares of
the Company’s Common Stock, at a purchase price of $4.85 per share. The Private Placement closed and the funds were received on
January 4, 2024. The net proceeds to the Company from the Private Placement were approximately $5.4 million, after deducting placement
agent fees and expenses and estimated offering expenses payable by the Company. The Company intends to use the net proceeds received
from the Private Placement for continuing operating expenses and working capital.
Risks and Uncertainties
The Company has a limited operating history and
is in the very early stages of generating revenue from intended operations. The Company’s business and operations are sensitive
to general business and economic conditions in the U.S. and worldwide along with local, state, and federal governmental policy decisions.
A host of factors beyond the Company’s control could cause fluctuations in these conditions. Adverse conditions may include: changes
in the biotechnology regulatory environment, technological advances that render our technologies obsolete, availability of resources
for clinical trials, acceptance of technologies into the medical community, and competition from larger, more well-funded companies.
These adverse conditions could affect the Company’s financial condition and the results of its operations.
NOTE 2 – GOING CONCERN ANALYSIS
Management Plans
The Company was incorporated on September 28,
2017 and has not generated significant revenues to date. During the three months ended March 31, 2024, the Company had a net loss of
$14,868,694 and negative cash flow from operating activities of $5,960,931. As of March 31, 2024, the Company’s cash balance was
$88,671.
As of March 31, 2024, the Company had approximately
$1.8 million of availability to sell under its shelf registration statement on Form S-3. Upon the filing of the Company’s annual
report on Form 10-K on April 16, 2024, the Company’s aggregate market value of the voting and non-voting equity held by non-affiliates
was below $3.0 million. As a result, the maximum amount that the Company can sell under its shelf registration statement on Form
S-3 during any 12 month period is equal to one-third of the aggregate market value of the voting and non-voting equity held by non-affiliates
of the Company.
On November 21, 2023, the Company received written
notice from Nasdaq that we had regained compliance with the Public Float Rule. On December 29, 2023, the Company received written notice
from Nasdaq that we had regained compliance with the Stockholders’ Equity Rule but will be subject to a Mandatory Panel Monitor
for a period of one year.
If we are delisted from Nasdaq, but obtain a
substitute listing for our common stock, it will likely be on a market with less liquidity, and therefore experience potentially more
price volatility than experienced on Nasdaq. Stockholders may not be able to sell their shares of common stock on any such substitute
market in the quantities, at the times, or at the prices that could potentially be available on a more liquid trading market. As a result
of these factors, if our common stock is delisted from Nasdaq, the value and liquidity of our common stock, warrants and pre-funded warrants
would likely be significantly adversely affected. A delisting of our common stock from Nasdaq could also adversely affect our ability
to obtain financing for our operations and/or result in a loss of confidence by investors, employees and/or business partners.
The Company continues to actively pursue numerous
capital raising transactions with the objective of obtaining sufficient bridge funding to meet the Company’s existing capital needs
as well as more substantial capital raises to meet the Company’s longer-term needs.
In addition, factors such as stock price, volatility,
trading volume, market conditions, demand and regulatory requirements may adversely affect the Company’s ability to raise capital
in an efficient manner. Because of these factors, the Company believes that this creates substantial doubt with the Company’s ability
to continue as a going concern.
In addition to the shelf registration, the Company
has the ability to raise capital from equity or debt through private placements or public offerings pursuant to a registration statement
on Form S-1. We may also secure loans from related parties.
The financial statements included in this report
do not include any adjustments to reflect the possible future effects on the recoverability and classification of assets or the amounts
and classification of liabilities that may result from the matters discussed herein. The Company’s ability to continue as a going
concern is dependent upon the ability to complete clinical studies and implement the business plan, generate sufficient revenues and
to control operating expenses. In addition, the Company is consistently focused on raising capital, strategic acquisitions and alliances,
and other initiatives to strengthen the Company.
NOTE 3 – SUMMARY OF SIGNIFICANT ACCOUNTING
POLICIES
Basis of Presentation
The accompanying unaudited condensed consolidated
financial statements have been prepared in accordance with accounting principles generally accepted in the United States of America (“U.S.
GAAP”) for interim financial information and the rules and regulations of the Securities and Exchange Commission (“SEC”).
In the opinion of the Company’s management, the accompanying condensed consolidated financial statements reflect all adjustments,
consisting of normal, recurring adjustments, considered necessary for a fair presentation of the results for the interim periods ended
March 31, 2024 and March 31, 2023. Although management believes that the disclosures in these unaudited condensed consolidated financial
statements are adequate to make the information presented not misleading, certain information and footnote disclosures normally included
in condensed consolidated financial statements that have been prepared in accordance U.S. GAAP have been omitted pursuant to the rules
and regulations of the SEC.
The accompanying unaudited condensed consolidated
financial statements should be read in conjunction with the Company’s financial statements and notes related thereto included in
the Company’s Annual Report on Form 10-K for the year ended December 31, 2023, filed with the SEC on April 16, 2024. The interim
results for the three months ended March 31, 2024 are not necessarily indicative of the results to be expected for the year ended December
31, 2024 or for any future interim periods.
Principles of Consolidation
The consolidated financial statements include
the accounts of Aditxt, Inc., its wholly owned subsidiaries and, one majority owned subsidiary. All significant intercompany balances
and transactions have been eliminated in the consolidated financial statements.
Use of Estimates
The preparation of financial statements in conformity
with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure
of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenue and expense during the
reporting period. Actual results could differ from those estimates. Significant estimates underlying the financial statements include
the collectability of notes receivable, the reserve on insurance billing, value of preferred shares issued, our investments in preferred
shares, estimation of discounts on non-interest bearing borrowing, and the fair value of stock options and warrants.
Fair Value Measurements and Fair Value
of Financial Instruments
The Company adopted Financial Accounting Standards
Board (“FASB”) Accounting Standards Codification (“ASC”) Topic 820, Fair Value Measurements. ASC Topic 820 clarifies
the definition of fair value, prescribes methods for measuring fair value, and establishes a fair value hierarchy to classify the inputs
used in measuring fair value as follows:
| |
Level 1 |
- |
Inputs are unadjusted quoted
prices in active markets for identical assets or liabilities available at the measurement date. |
| |
Level 2 |
- |
Inputs are unadjusted quoted
prices for similar assets and liabilities in active markets, quoted prices for identical or similar assets and liabilities in markets
that are not active, inputs other than quoted prices that are observable, and inputs derived from or corroborated by observable market
data. |
| |
Level 3 |
- |
Inputs are unobservable
inputs which reflect the reporting entity’s own assumptions on what assumptions the market participants would use in pricing
the asset or liability based on the best available information. |
The Company did not identify any assets or liabilities
that are required to be presented on the balance sheets at fair value in accordance with ASC Topic 820.
Due to the short-term nature of all financial
assets and liabilities, their carrying value approximates their fair value as of the balance sheet dates. (See Note 9)
Concentrations of Credit Risk
Financial instruments that potentially subject
the Company to concentrations of credit risk consist primarily of cash and cash equivalents and accounts receivable.
The Company maintains its cash accounts at financial
institutions which are insured by the Federal Deposit Insurance Corporation. At times, the Company may have deposits in excess of federally
insured limits.
Substantially all the Company’s accounts
receivable are with companies in the healthcare industry, individuals, and the U.S. government. However, concentration of credit risk
is mitigated due to the Company’s number of customers. In addition, for receivables due from U.S. government agencies, the Company
does not believe the receivables represent a credit risk as these are related to healthcare programs funded by the U.S. government and
payment is primarily dependent upon submitting the appropriate documentation.
Cash and Cash Equivalents
Cash and cash equivalents include short-term,
liquid investments.
Inventory
Inventory consists of laboratory materials and
supplies used in laboratory analysis. We capitalize inventory when purchased. Inventory is valued at the lower of cost or net realizable
value on a first-in, first-out basis. We periodically perform obsolescence assessments and write off any inventory that is no longer
usable.
Fixed Assets
Fixed assets are stated at cost less accumulated
depreciation. Cost includes expenditures for furniture, office equipment, laboratory equipment, and other assets. Maintenance and repairs
are charged to expense as incurred. When assets are sold, retired, or otherwise disposed of, the cost and accumulated depreciation are
removed from the accounts and any resulting gain or loss is reflected in operations. The costs of fixed assets are depreciated using
the straight-line method over the estimated useful lives or lease life of the related assets.
Useful lives assigned to fixed assets are as
follows:
| Computers |
|
Three years to five years |
| Lab Equipment |
|
Seven to ten years |
| Office Furniture |
|
Five to ten years |
| Other fixed assets |
|
Five to ten years |
| Leasehold Improvements |
|
Shorter of estimated useful life or remaining lease
term |
Intangible Assets
Intangible assets are stated at cost less accumulated
amortization. For intangible assets that have finite lives, the assets are amortized using the straight-line method over the estimated
useful lives of the related assets. For intangible assets with indefinite lives, the assets are tested periodically for impairment.
Investments
The following table sets forth a summary of the
changes in equity investments. This investment has been recorded at cost in accordance with ASC 321.
| | |
For the
three months
ended
March 31,
2024 | |
| | |
| |
| As of December 31, 2023 | |
| 22,711,221 | |
| Purchase of equity investments | |
| - | |
| Unrealized gains | |
| - | |
| As of March 31, 2024 | |
$ | 22,711,221 | |
This investment is included in its own line item
on the Company’s consolidated balance sheets.
Non-marketable equity investments (for which
we do not have significant influence or control) are investments without readily determinable fair values that are recorded based on
initial cost minus impairment, if any, plus or minus adjustments resulting from observable price changes in orderly transactions for
identical or similar securities, if any. All gains and losses on investments in non-marketable equity securities, realized and unrealized,
are recognized in investment and other income (expense), net.
We monitor equity method and non-marketable equity
investments for events or circumstances that could indicate the investments are impaired, such as a deterioration in the investee’s
financial condition and business forecasts and lower valuations in recently completed or anticipated financings, and recognize a charge
to investment and other income (expense), net for the difference between the estimated fair value and the carrying value. For equity
method investments, we record impairment losses in earnings only when impairments are considered other-than-temporary.
Accounts Receivable and Allowance for Doubtful
Accounts
Accounts receivable are stated at the amount
management expects to collect from outstanding balances. The Company generally does not require collateral to support customer receivables.
The Company determines if receivables are past due based on days outstanding, and amounts are written off when determined to be uncollectible
by management. As of March 31, 2024 and December 31, 2023, there was an allowance for doubtful accounts of zero and zero,
respectively. Accounts receivable is made up of billed and unbilled of $273,802 and $153,242 as of March 31, 2024 and $236,605 and $171,721
as of December 31, 2023, respectively.
Income Taxes
Deferred tax assets and liabilities are recognized
for the future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities
and their respective tax bases and operating loss and tax credit carry forwards. Deferred tax assets and liabilities are measured using
enacted tax rates expected to apply to taxable income in the years in which those temporary differences are expected to be recovered
or settled. The effect on deferred tax assets and liabilities of a change in tax rates is recognized in income in the period that includes
the enactment date. At March 31, 2024 and December 31, 2023, the Company had a full valuation allowance against its deferred tax assets.
Offering Costs
Offering costs incurred in connection with equity
are recorded as a reduction of equity and offering costs incurred in connection with debt are recorded as a reduction of debt as a debt
discount. Equity instruments issued as offering costs have zero net effect on the Company’s equity.
Revenue Recognition
In accordance with ASC 606 (Revenue From Contracts
with Customers), revenue is recognized when a customer obtains control of promised services. The amount of revenue recognized reflects
the consideration to which the Company expects to be entitled to receive in exchange for these services. To achieve this core principle,
the Company applies the following five steps:
| |
1) |
Identify
the contract with a customer |
| |
2) |
Identify the performance
obligations in the contract |
| |
3) |
Determine the transaction
price |
| |
4) |
Allocate the transaction
price to performance obligations in the contract |
| |
5) |
Recognize revenue when
or as the Company satisfies a performance obligation |
Revenues reported from services relating to the
AditxtScoreTM are recognized when the AditxtScoreTM report is delivered to the customer. The services performed
include the analysis of specimens received in the Company’s CLIA laboratory and the generation of results which are then delivered
upon completion.
The Company recognizes revenue in the following
manner for the following types of customers:
Client Payers:
Client payers include physicians or other entities
for which services are billed based on negotiated fee schedules. The Company principally estimates the allowance for credit losses for
client payers based on historical collection experience and the period of time the receivable has been outstanding.
Cash Pay:
Customers are billed based on established patient
fee schedules or fees negotiated with physicians on behalf of their patients. Collection of billings is subject to credit risk and the
ability of the patients to pay.
Insurance:
Reimbursements from healthcare insurers are based
on fee for service schedules. Net revenues recognized consist of amounts billed net of contractual allowances for differences between
amounts billed and the estimated consideration the Company expects to receive from such payers, collection experience, and the terms
of the Company’s contractual arrangements.
Leases
Under Topic 842 (Leases), operating lease expense
is generally recognized evenly over the term of the lease. The Company has operating leases consisting of office space, laboratory space,
and lab equipment.
Leases with an initial term of twelve months
or less are not recorded on the balance sheet. We combine the lease and non-lease components in determining the lease liabilities and
right of use (“ROU”) assets.
Stock-Based Compensation
The Company accounts for stock-based compensation
costs under the provisions of ASC 718, Compensation—Stock Compensation, which requires the measurement and recognition of compensation
expense related to the fair value of stock-based compensation awards that are ultimately expected to vest. Stock-based compensation expense
recognized includes the compensation cost for all stock-based payments granted to employees, officers, and directors based on the grant
date fair value estimated in accordance with the provisions of ASC 718. ASC 718 is also applied to awards modified, repurchased, or cancelled
during the periods reported. Stock-based compensation is recognized as expense over the employee’s requisite vesting period and
over the nonemployee’s period of providing goods or services.
Patents
The Company incurs fees from patent licenses,
which are reflected in research and development expenses, and are expensed as incurred. During the three months ended March 31, 2024
and 2023, the Company incurred patent licensing fees of $61,666 and $60,000, respectively.
Research and Development
We incur research and development costs during
the process of researching and developing our technologies and future offerings. We expense these costs as incurred unless such costs
qualify for capitalization under applicable guidance. During the three months ended March 31, 2024 and 2023, the Company incurred research
and development costs of $8,145,266 and $1,387,541, respectively.
Non-controlling Interest in Subsidiary
Non-controlling interests represent the Company’s
subsidiary’s cumulative results of operations and changes in deficit attributable to non-controlling shareholders. During the three
months ended March 31, 2024 and 2023, the Company recognized $138,967 and $0 in net loss attributable to non-controlling interest in
Pearsanta. The Company owns approximately 90.2% of Pearsanta, Inc., as of March 31, 2024.
Basic and Diluted Net Loss per Common Share
Basic loss per common share is computed by dividing
the net loss by the weighted average number of shares of common stock outstanding for each period. Diluted loss per share is computed
by dividing the net loss attributable of common stockholders by the weighted average number of shares of common stock outstanding plus
the dilutive effect of shares issuable through the common stock equivalents. The weighted-average number of common shares outstanding
excludes common stock equivalents because their inclusion would be anti-dilutive. As of March 31, 2024, 45,572 stock options, 0 unvested restricted
stock units, 5,097,080 warrants, 22,280 shares of preferred series A-1 stock, 6,000 shares of preferred series B-1, and 2,625 shares
of preferred series B-2 stock were excluded from dilutive earnings per share as their effects were anti-dilutive.
| Instrument | |
Quantity Issued and Outstanding as
of
March 31,
2024 | | |
Common Stock Equivalent | |
| Series A Preferred Stock | |
| - | | |
| - | |
| Preferred Series A-1 Stock | |
| 22,280 | | |
| 5,018,019 | |
| Series B Preferred Stock | |
| - | | |
| - | |
| Preferred Series B-1 Stock | |
| 6,000 | | |
| 1,477,833 | |
| Preferred Series B-2 Stock | |
| 2,625 | | |
| 557,325 | |
| Series C Preferred Stock | |
| - | | |
| - | |
| Warrants | |
| 5,097,080 | | |
| 5,097,080 | |
| Options | |
| 45,572 | | |
| 45,572 | |
| Total Common Stock Equivalent | |
| | | |
| 12,195,829 | |
As of March 31, 2023, 44,710 stock options,
4,267 unvested restricted stock units, and 5,079,348 warrants were excluded from dilutive earnings per share as their effects were anti-dilutive.
Recent Accounting Pronouncements
The FASB issues ASUs to amend the authoritative
literature in ASC. There have been several ASUs to date, including those above, that amend the original text of ASC. Management believes
that those issued to date either (i) provide supplemental guidance, (ii) are technical corrections, (iii) are not applicable to us or
(iv) are not expected to have a significant impact on our financial statements.
NOTE 4 – FIXED ASSETS
The Company’s fixed assets include the
following on March 31, 2024:
| | |
Cost Basis | | |
Accumulated
Depreciation | | |
Net | |
| Computers | |
$ | 378,480 | | |
$ | (342,303 | ) | |
$ | 36,177 | |
| Lab Equipment | |
| 2,711,525 | | |
| (949,414 | ) | |
| 1,762,111 | |
| Office Furniture | |
| 56,656 | | |
| (15,282 | ) | |
| 41,374 | |
| Other Fixed Assets | |
| 148,605 | | |
| (25,632 | ) | |
| 122,973 | |
| Leasehold Improvements | |
| 120,440 | | |
| (61,315 | ) | |
| 59,125 | |
| Total Fixed Assets | |
$ | 3,415,706 | | |
$ | (1,393,946 | ) | |
$ | 2,021,760 | |
The Company’s fixed assets include the
following on December 31, 2023
| | |
Cost Basis | | |
Accumulated
Depreciation | | |
Net | |
| Computers | |
$ | 378,480 | | |
$ | (320,473 | ) | |
$ | 58,007 | |
| Lab Equipment | |
| 2,585,077 | | |
| (859,612 | ) | |
| 1,725,465 | |
| Office Furniture | |
| 56,656 | | |
| (13,866 | ) | |
| 42,790 | |
| Other Fixed Assets | |
| 8,605 | | |
| (2,084 | ) | |
| 6,521 | |
| Leasehold Improvements | |
| 120,440 | | |
| (54,980 | ) | |
| 65,460 | |
| Total Fixed Assets | |
$ | 3,149,258 | | |
$ | (1,251,015 | ) | |
$ | 1,898,243 | |
Depreciation expense was $142,932 and $109,896
for the three months ended March 31, 2024 and 2023, respectively. As of March 31, 2024 and December 31, 2023, the fixed assets that serve
as collateral subject to the financed asset liability have a carrying value of $1,006,741 and $1,316,830, respectively.
Fixed asset activity for the three months ended
March 31, 2024 consisted of the following:
| | |
For the
three months
ended
March 31,
2024 | |
| As of December 31, 2023 | |
| 3,149,258 | |
| Brain Scientific Asset Purchase | |
| 266,448 | |
| Additions | |
| - | |
| As of March 31, 2024 | |
$ | 3,415,706 | |
Financed Assets:
In October 2020, the Company purchased two pieces
of lab equipment and financed them for a period of twenty-four months with a monthly payment of $19,487, with an interest rate of 8%.
As of March 31, 2024, the Company has one payment in arrears.
In January of 2021, the Company purchased one
piece of lab equipment and financed it for a period of twenty-four months with a monthly payment of $9,733, with an interest rate of 8%.
As of March 31, 2024, the Company has one payment in arrears.
In March of 2021, the Company purchased five
pieces of lab equipment and financed them for a period of twenty-four months with a monthly payment of $37,171, with an interest rate
of 8%. As of March 31, 2024, the Company has four payments in arrears.
As of March 31, 2024, all lab equipment financing
agreements have matured and are in default status.
NOTE 5 – INTANGIBLE ASSETS
The Company’s intangible assets include
the following on March 31, 2024:
| | |
Cost Basis | | |
Accumulated
Amortization | | |
Net | |
| Proprietary Technology | |
$ | 321,000 | | |
$ | (321,000 | ) | |
$ | - | |
| Intellectual property | |
| 10,000 | | |
| (1,389 | ) | |
| 8,611 | |
| Total Intangible Assets | |
$ | 331,000 | | |
$ | (322,389 | ) | |
$ | 8,611 | |
The Company’s intangible assets include
the following on December 31, 2023:
| | |
Cost Basis | | |
Accumulated
Amortization | | |
Net | |
| Proprietary Technology | |
$ | 321,000 | | |
$ | (321,000 | ) | |
$ | - | |
| Intellectual property | |
| 10,000 | | |
| (556 | ) | |
| 9,444 | |
| Total Intangible Assets | |
$ | 331,000 | | |
$ | (321,556 | ) | |
$ | 9,444 | |
Amortization expense was $833 and $26,750 for
the three months ended March 31, 2024 and 2023, respectively. The Company’s proprietary technology is being amortized over its
estimated useful life of three years.
| | |
For the
three months
ended
March 31,
2024 | |
| As of December 31, 2023 | |
| 321,000 | |
| Additions | |
| - | |
| As of March 31, 2024 | |
$ | 321,000 | |
NOTE 6 – RELATED PARTY TRANSACTIONS
On November 30, 2023, Amro Albanna, the Chief
Executive Officer of the Company, loaned $10,000 to the Company. The loan was evidenced by an unsecured promissory note (the “November
Note”). Pursuant to the terms of the November Note, it will accrue interest at a rate of eight and a half percent (8.50%) per annum,
the Prime rate on the date of signing, and is due on the earlier of May 30, 2024 or an event of default, as defined therein. As of March
31, 2024, the note was fully paid off.
On December 6, 2023, Amro Albanna, the Chief
Executive Officer of the Company, loaned $200,000 to the Company. The loan was evidenced by an unsecured promissory note (the “First
December Note”). Pursuant to the terms of the First December Note, it will accrue interest at a rate of eight and a half percent
(8.50%) per annum, the Prime rate on the date of signing, and is due on the earlier of June 6, 2024 or an event of default, as defined
therein. As of March 31, 2024, the note was fully paid off.
On December 20, 2023, Amro Albanna, the Chief
Executive Officer of the Company, loaned $165,000 to the Company. The loan was evidenced by an unsecured promissory note (the “Second
December Note”). Pursuant to the terms of the Second December Note, it will accrue interest at a rate of eight and a half percent
(8.50%) per annum, the Prime rate on the date of signing, and is due on the earlier of June 20, 2024 or an event of default, as defined
therein. As of March 31, 2024, the note was fully paid off.
On February 7, 2024, Amro Albanna, the Chief
Executive Officer of the Company loaned $30,000 to the Company. The loan was evidenced by an unsecured promissory note (the “February
7th Note”). Pursuant to the terms of the February 7th Note, it will accrue interest at the Prime rate of eight and one-half percent
(8.5%) per annum and is due on the earlier of August 7, 2024 or an event of default, as defined therein. As of March 31, 2024 the note
has an outstanding principal balance of $30,000.
On February 15, 2024, Amro Albanna, the Chief
Executive Officer of the Company loaned $205,000 to the Company. The loan was evidenced by an unsecured promissory note (the “February
15th Note”). Pursuant to the terms of the February 15th Note, it will accrue interest at the Prime rate of eight and one-half percent
(8.5%) per annum and is due on the earlier of August 15, 2024 or an event of default, as defined therein. As of March 31, 2024 the note
has an outstanding principal balance of $205,000.
On February 29, 2024, Amro Albanna, the Chief
Executive Officer of the Company, and Shahrokh Shabahang, the Chief Innovation Officer of the Company, loaned $117,000 and $115,000,
respectively, to the Company. The loans were evidenced by an unsecured promissory note (the “February 29th Notes”). Pursuant
to the terms of the February 29th Notes, it will accrue interest at the Prime rate of eight and one-half percent (8.5%) per annum and
is due on the earlier of August 29, 2024 or an event of default, as defined therein. As of March 31, 2024 these notes have an outstanding
principal balance of $232,000.
See Note 12 for additional loans incurred or
paid subsequent to March 31, 2024.
NOTE 7 – NOTES PAYABLE
On October 5, 2023, the Company entered into
an Agreement for the Purchase and Sale of Future Receipts (the “October MCA Agreement”) pursuant to which the existing funder
(the “Funder”) increased the existing outstanding amount to $4,470,000 (the “October MCA Purchased Amount”) for
gross proceeds to the Company of $3,000,000, less origination fees of $240,000 and the outstanding balance under the existing agreement
of $1,234,461, resulting in net proceeds to the Company of $1,525,539. Pursuant to the October MCA Agreement, the Company granted the
Funder a security interest in all of the Company’s present and future accounts receivable in an amount not to exceed the October
MCA Purchased Amount. The October MCA Purchased Amount shall be repaid by the Company in 30 weekly installments of $149,000. The October
Purchased Amount may be prepaid by the Company via a payment of $3,870,000 if repaid within 30 days, $4,110,000 if repaid within 60 days
and $4,230,000 if repaid within 90 days. On January 24, 2024, the October MCA Agreement was restructured in connection with the January
Loan Agreement, as defined below.
On November 7, 2023, the Company entered into
a Business Loan and Security Agreement (the “November Loan Agreement”) with the lender (the “Lender”), pursuant
to which the Company obtained a loan from the Lender in the principal amount of $2,100,000, which satisfied the outstanding balance on
the August Loan of $1,089,000 and includes origination fees of $140,000 (the “November Loan”). Pursuant to the November Loan
Agreement, the Company granted the Lender a continuing secondary security interest in certain collateral (as defined in the November
Loan Agreement). The total amount of interest and fees payable by us to the Lender under the November Loan will be $3,129,000, which
will be repaid in 34 weekly installments ranging from $69,000 - $99,000. As of March 31, 2024 the November Loan has an outstanding principal
balance of $1,752,827, an unamortized debt discount of $57,647, and accrued interest of $469,892. As of December 31, 2023 the November
Loan has an outstanding principal balance of $1,990,699. The November Loan Agreement is currently in default status.
On November 24, 2023, the Company entered into
a loan with a principal of $53,099. The loan was evidenced by an unsecured promissory note (the “Second November Note”).
Pursuant to the terms of the Second November Note, it will accrue interest at a rate of eight and a half percent (8.50%) per annum, the
Prime rate on the date of signing, and is due on the earlier of May 24, 2024 or an event of default, as defined therein. As of March
31, 2024, the note was fully paid off. As of December 31, 2023, there was a remaining principal balance of $53,099 on the Second December
Loan and accrued interest of $458.
On January 24, 2024, the Company entered
into a Business Loan and Security Agreement (the “January Loan Agreement”) with a commercial funding source (the “Lender”),
pursuant to which the Company obtained a loan from the Lender in the principal amount of $3,600,000, which includes origination fees
of $252,000 (the “January Loan”). Pursuant to the January Loan Agreement, the Company granted the Lender a continuing secondary
security interest in certain collateral (as defined in the January Loan Agreement). The total amount of interest and fees payable by
the Company to the Lender under the January Loan will be $5,364,000, which will be repayable by the Company in 30 weekly installments
of $178,800. The Company received net proceeds from the January Loan of $814,900 following repayment of the outstanding balance on the
October Purchased Amount of $2,533,100. As of March 31, 2024, there was a remaining principal balance of $3,519,577, an unamortized debt
discount of $176,400, and accrued interest of $714,826. The January Loan Agreement is currently in default status.
On March 7, 2024, Sixth Borough Capital Fund,
LP loaned $300,000 to the Company. The loan was evidenced by an unsecured promissory note (the “Sixth Borough Note”). Pursuant
to the terms of the Sixth Borough Note, it will accrue interest at the Prime rate of eight and one-half percent (8.5%) per annum and
is due on the earlier of March 31, 2024 or an event of default, as defined therein. As of March 31, 2024 the note has an outstanding
principal balance of $300,000. The Sixth Borough Note was subsequently converted into Series C-1 Preferred Stock in connection with the
Private Placement (as defined below). (See note 12)
Evofem Merger
In connection with the Agreement and Plan of
Merger (the “Merger Agreement”) with Adicure, Inc., a Delaware corporation and wholly owned subsidiary of the Company (“Merger
Sub”) and Evofem Biosciences, Inc., a Delaware corporation (“Evofem”), the Company, Evofem and the holders (the “Holders”)
of certain senior indebtedness (the “Notes”) entered into an Assignment Agreement dated December 11, 2023 (the “Assignment
Agreement”), pursuant to which the Holders assigned the Notes to the Company in consideration for the issuance by the Company of
(i) an aggregate principal amount of $5 million in secured notes of the Company due on January 2, 2024 (the “January 2024 Secured
Notes”), (ii) an aggregate principal amount of $8 million in secured notes of the Company due on September 30, 2024 (the “September
2024 Secured Notes”), (iii) an aggregate principal amount of $5 million in ten-year unsecured notes (the “Unsecured Notes”),
and (iv) payment of $154,480 in respect of net sales of Phexxi in respect of the calendar quarter ended September 30, 2023, which amount
is due and payable on December 14, 2023. The January 2024 Secured Notes are secured by certain intellectual property assets of the Company
and its subsidiaries pursuant to an Intellectual Property Security Agreement (the “IP Security Agreement”) entered into in
connection with the Assignment Agreement. The September 2024 Secured Notes are secured by the Notes and certain associated security documents
pursuant to a Security Agreement (the “Security Agreement”) entered into in connection with the Assignment Agreement. As
of March 31, 2024, there was a remaining principal balance of $250,000 on the Notes.
Subject to the terms and conditions set forth
in the Merger Agreement, at the effective time of the Merger (the “Effective Time”), (i) all issued and outstanding shares
of common stock, par value $0.0001 per share of Evofem (“Evofem Common Stock”), other than any shares of Evofem Common Stock
held by the Company or Merger Sub immediately prior to the Effective Time, will be converted into the right to receive an aggregate of
610,000 shares of the Company’s common stock, par value $0.001 per share (“Company Common Stock”); and (ii) all issued
and outstanding shares of Series E-1 Preferred Stock, par value $0.0001 of Evofem (the “Evofem Unconverted Preferred Stock”),
other than any shares of Evofem Unconverted Preferred Stock held by the Company or Merger Sub immediately prior to the Effective Time,
will be converted into the right to receive an aggregate of 2,327 shares of Series A-1 Preferred Stock, par value $0.001 of the Company
(the “Company Preferred Stock”), having such rights, powers, and preferences set forth in the form of Certificate of Designation
of Series A-1 Preferred Stock, the form of which is attached as Exhibit C to the Merger Agreement.
The respective obligations of each of the Company,
Merger Sub and Evofem to consummate the closing of the Merger (the “Closing”) are subject to the satisfaction or waiver,
at or prior to the closing of certain conditions, including but not limited to, the following:
| |
(i) |
approval
by the Company’s shareholders and Evofem shareholders; |
| |
(ii) |
the registration statement
on Form S-4 pursuant to which the shares of the Company Common Stock issuable in the Merger being declared effective by the U.S.
Securities and Exchange Commission; |
| |
(iii) |
the entry into a voting
agreement by the Company and certain members of Evofem management; |
| |
(iv) |
all preferred stock of
Evofem other than the Evofem Unconverted Preferred Stock shall have been converted to Evofem Common Stock; |
| |
(v) |
Evofem shall have received
agreements (the “Evofem Warrant Holder Agreements”) from all holders of Evofem warrants which provide: |
a. waivers with respect to any fundamental
transaction, change in control or other similar rights that such warrant holder may have under any such Evofem warrants, and (b) an agreement
to such Evofem warrants to exchange such warrants for not more than an aggregate (for all holders of Evofem warrants) of 551 shares of
Company Preferred Stock;
| |
(vi) |
Evofem shall have cashed
out any other holder of Evofem warrants who has not provided an Evofem Warrant Holder Agreement; and |
| |
(vii) |
Evofem shall have obtained
waivers from the holders of the convertible notes of Evofem (the “Evofem Convertible Notes”) with respect to any fundamental
transaction rights that such holder may have under the Evofem Convertible Notes, including any right to vote, consent, or otherwise
approve or veto any of the transactions contemplated under the Merger Agreement. |
The obligations of the Company and Merger Sub
to consummate the Closing are subject to the satisfaction or waiver, at or prior to the Closing of certain conditions, including but
not limited to, the following:
| |
(i) |
the Company shall have
obtained agreements from the holders of Evofem Convertible Notes and purchase rights they hold to exchange such Convertible Notes
and purchase rights for not more than an aggregate (for all holders of Evofem Convertible Notes) of 86,153 shares of Company Preferred
Stock; |
| |
(ii) |
the Company shall have
received waivers form the holders of certain of the Company’s securities which contain prohibitions on variable rate transactions;
and |
| |
(iii) |
the Company, Merger Sub
and Evofem shall work together between the Execution Date and the Effective Time to determine the tax treatment of the Merger and
the other transactions contemplated by the Merger Agreement. |
The obligations of the Company to consummate
the Closing are subject to the satisfaction or waiver, at or prior to the Closing of certain conditions, including but not limited to,
the following:
| |
(i) |
the Company shall have
regained compliance with the stockholders’ equity requirement in Nasdaq Listing Rule 5550(b)(1) and shall meet all other applicable
criteria for continued listing, subject to any panel monitor imposed by Nasdaq. |
As the January 2024 Secured Notes and September
2024 Secured Notes did not contain a stated interest rate, the Company calculated an imputed interest rate of 26.7% based on the Company’s
weighted average cost of capital for the period in which the January 2024 Secured Notes and September 2024 Secured Notes were outstanding.
This amounted to approximately $1.8 million which was recorded as a discount to be amortized over the life of the January 2024 Secured
Notes and September 2024 Secured Notes.
Secured Notes Amendments and Assignment
On January 2, 2024, the Company and certain holders
of the secured notes (the “Holders”) entered into amendments to the January 2024 Secured Notes (“Amendment No. 1 to
January 2024 Secured Notes”), pursuant to which the maturity date of the January 2024 Notes was extended to January 5, 2024.
On January 5, 2024, the Company and the Holders
entered into amendments to the January 2024 Secured Notes (“Amendment No. 2 to January 2024 Secured Notes”) and amendments
to the September 2024 Secured Notes (“Amendment No. 1 to September 2024 Secured Notes”), pursuant to which the Company and
the Holders agreed that in consideration of a principal payment in the aggregate amount of $1 million on the January 2024 Secured Notes
and in increase in the aggregate principal balance of $250,000 on the September 2024 Secured Notes, that the maturity date of the January
2024 Secured Notes would be further extended to January 31, 2024.
On January 31, 2024, the Company and the Holders
entered into amendments to the January 2024 Secured Notes (“Amendment No. 3 to January 2024 Secured Notes”), pursuant to
which the maturity date of the January 2024 Notes was extended to February 29, 2024. In addition, on January 31, 2024, the Company and
the Holders entered into amendments to the September 2024 Secured Notes (“Amendment No. 2 to September 2024 Secured Notes”),
pursuant to which the Company and the Holders agreed that in consideration of a principal payment in the aggregate amount of $1.25 million
on the January 2024 Secured Notes and in increase in the aggregate principal balance of $300,000 on the September 2024 Secured Notes.
Pursuant to Amendment No. 3 to the January 2024
Secured Notes, the Company was required to make the Additional Consideration payment no later than February 9, 2024. As a result of the
Company’s failure to make the Additional Consideration payment by February 9, 2023, the January 2024 Secured Notes and the September
2024 Secured Notes were in default and the entire principal balance of the January 2024 Secured Notes and the September 2024 Secured
Notes, without demand or notice, were due and payable.
As a result of the defaults on the January 2024
Secured Notes and the September 2024 Secured Notes, the Company was in default on the Business Loan and Security Agreement dated January
24, 2024 (the January Business Loan”), which had a current balance of approximately $5.2 million, and the Business Loan and Security
Agreement dated November 7, 2023 (the “November Business Loan”) which had a current balance of approximately $2.7 million.
On February 26, 2024, the Company and the Holders
entered into an Assignment Agreement (the “February Assignment Agreement”), pursuant to which the Company assigned all remaining
amounts due under the January 2024 Secured Notes, the September 2024 Secured Notes and the Unsecured Notes (collectively, the “Notes”)
back to the Holders. The Company recognized a $208,670 loss on the transfer of these notes. In connection with the February Assignment
Agreement, the Company and the Holders entered into a payoff letter (the “Payoff Letter”) and amendments to the January 2024
Secured Notes (“Amendment No. 4 to January 2024 Secured Notes”), pursuant to which the maturity date of the January 2024
Secured Notes was extended to March 31, 2024 and the outstanding balance under the Notes, after giving effect to the transactions contemplated
by the February Assignment Agreement as applied pursuant to the Payoff Letter, was adjusted to $250,000. On April 15, 2024, the Company
repaid the $250,000.
NOTE 8 – LEASES
Our lease agreements generally do not provide
an implicit borrowing rate; therefore, an internal incremental borrowing rate is determined based on information available at lease commencement
date for purposes of determining the present value of lease payments. We used the incremental borrowing rate on March 31, 2024 and December
31, 2023 for all leases that commenced prior to that date. In determining this rate, which is used to determine the present value of
future lease payments, we estimate the rate of interest we would pay on a collateralized basis, with similar payment terms as the lease
and in a similar economic environment.
Our corporate headquarters is located in Richmond,
Virginia, where we lease approximately 25,000 square feet. The lease expires in August 31, 2026, subject to extension.
As of March 31, 2024 the Company is 4.75 months in arrears on this lease.
On March 6, 2024, the Company received correspondence
from 532 Realty Associates, LLC (the “Landlord”) that the Company is in default under that certain Agreement of Lease dated
November 3, 2021 by and between the Landlord and the Company (the “New York Lease”) for failure to pay Basic Rent and Additional
Rent (as each term is defined in the New York Lease) in the aggregate amount of $40,707 (the “Past Due Rent”).
We also lease approximately 5,810 square
feet of laboratory and office space in Mountain View, California. The lease expires in August 31, 2024, subject to extension. As
of March 31, 2024 the Company is 4 months in arrears on this lease.
Additionally, we lease approximately 3,150 square
feet of office space in Melville, New York. The lease expires in December 31, 2025, subject to extension. As of March 31, 2024 the
Company is 4 months in arrears on this lease.
The overdue amounts represent a payable of $413,300
which are included in accounts payable and accrued liabilities on the Company’s condensed consolidated balance sheet.
Lease Costs
| | |
Three Months Ended
March 31,
2024 | | |
Three Months Ended
March 31,
2023 | |
| Components of total lease costs: | |
| | |
| |
| Operating lease expense | |
$ | 305,049 | | |
$ | 297,091 | |
| Total lease costs | |
$ | 305,049 | | |
$ | 297,091 | |
Lease Positions as of March 31, 2024 and December
31, 2023
ROU lease assets and lease liabilities for our
operating leases are recorded on the balance sheet as follows:
| | |
March 31,
2024 | | |
December 31,
2023 | |
| Assets | |
| | |
| |
| Right of use asset –
long term | |
$ | 1,940,076 | | |
$ | 2,200,299 | |
| Total right of use asset | |
$ | 1,940,076 | | |
$ | 2,200,299 | |
| | |
| | | |
| | |
| Liabilities | |
| | | |
| | |
| Operating lease liabilities – short term | |
$ | 900,979 | | |
$ | 999,943 | |
| Operating lease liabilities –
long term | |
| 891,747 | | |
| 1,041,744 | |
| Total lease liability | |
$ | 1,792,726 | | |
$ | 2,041,687 | |
Lease Terms and Discount Rate as of March
31, 2024
| Weighted average remaining lease term (in years) – operating leases | |
| 1.79 | |
| Weighted average discount rate – operating leases | |
| 8.00 | % |
Maturities of leases are as follows:
| 2024 (remaining) | |
$ | 718,751 | |
| 2025 | |
| 710,546 | |
| 2026 | |
| 423,930 | |
| Total lease payments | |
$ | 1,853,227 | |
| Less imputed interest | |
| (60,501 | ) |
| Less current portion | |
| (900,979 | ) |
| Total maturities, due beyond one year | |
$ | 891,747 | |
NOTE 9 – COMMITMENTS & CONTINGENCIES
License Agreement with Loma Linda University
On March 15, 2018, as amended on July 1, 2020,
we entered into a LLU License Agreement directly with Loma Linda University.
Pursuant to the LLU License Agreement, we obtained
the exclusive royalty-bearing worldwide license in and to all intellectual property, including patents, technical information, trade
secrets, proprietary rights, technology, know-how, data, formulas, drawings, and specifications, owned or controlled by LLU and/or any
of its affiliates (the “LLU Patent and Technology Rights”) and related to therapy for immune-mediated inflammatory diseases
(the ADI™ technology). In consideration for the LLU License Agreement, we issued 13 shares of common stock to LLU.
Pursuant to the LLU License Agreement, we are
required to pay an annual license fee to LLU. Also, we paid LLU $455,000 in July 2020 for outstanding milestone payments and license
fees. We are also required to pay to LLU milestone payments in connection with certain development milestones. Specifically, we are required
to make the following milestone payments to LLU: $175,000 on March 31, 2022; $100,000 on March 31, 2024; $500,000 on March 31, 2026;
and $500,000 on March 31, 2027. In lieu of the $175,000 milestone payment due on March 31, 2023, the Company paid LLU an extension fee
of $100,000. The Company did not make the March 31, 2024 payment; the Company intends to obtain an extension for this payment. Upon payment
of this extension fee, an additional year will be added for the March 31, 2023 milestone. Additionally, as consideration for prior expenses
incurred by LLU to prosecute, maintain and defend the LLU Patent and Technology Rights, we made the following payments to LLU: $70,000
at the end of December 2018, and a final payment of $60,000 at the end of March 2019. We are required to defend the LLU Patent and Technology
Rights during the term of the LLU License Agreement. Additionally, we will owe royalty payments of (i) 1.5% of Net Product Sales (as
such terms are defined under the LLU License Agreement) and Net Service Sales on any Licensed Products (defined as any finished pharmaceutical
products which utilizes the LLU Patent and Technology Rights in its development, manufacture or supply), and (ii) 0.75% of Net Product
Sales and Net Service Sales for Licensed Products and Licensed Services (as such terms are defined under the LLU License Agreement) not
covered by a valid patent claim for technology rights and know-how for a three (3) year period beyond the expiration of all valid patent
claims. We also are required to produce a written progress report to LLU, discussing our development and commercialization efforts, within
45 days following the end of each year. All intellectual property rights in and to LLU Patent and Technology Rights shall remain with
LLU (other than improvements developed by or on our behalf).
The LLU License Agreement shall terminate on
the last day that a patent granted to us by LLU is valid and enforceable or the day that the last patent application licensed to us is
abandoned. The LLU License Agreement may be terminated by mutual agreement or by us upon 90 days written notice to LLU. LLU may terminate
the LLU License Agreement in the event of (i) non-payments or late payments of royalty, milestone and license maintenance fees not cured
within 90 days after delivery of written notice by LLU, (ii) a breach of any non-payment provision (including the provision that requires
us to meet certain deadlines for milestone events (each, a “Milestone Deadline”)) not cured within 90 days after delivery
of written notice by LLU and (iii) LLU delivers notice to us of three or more actual breaches of the LLU License Agreement by us in any
12-month period. Additional Milestone Deadlines include: (i) the requirement to have regulatory approval of an IND application to initiate
first-in-human clinical trials on or before March 31, 2023, which will be extended to March 31, 2024 with a payment of a $100,000 extension
fee, (ii) the completion of first-in-human (phase I/II) clinical trials by March 31, 2024, (iii) the completion of Phase III clinical
trials by March 31, 2026 and (iv) biologic licensing approval by the FDA by March 31, 2027. The Company has not initiated clinical trials
to date and the Company intends to obtain an extension to commence human trials by March 31, 2025.
License Agreement with Leland Stanford Junior University
On February 3, 2020, we entered into an exclusive
license agreement (the “February 2020 License Agreement”) with Stanford regarding a patent concerning a method for detection
and measurement of specific cellular responses. Pursuant to the February 2020 License Agreement, we received an exclusive worldwide license
to Stanford’s patent regarding use, import, offer, and sale of Licensed Products (as defined in the agreement). The license to
the patented technology is exclusive, including the right to sublicense, beginning on the effective date of the agreement, and ending
when the patent expires. Under the exclusivity agreement, we acknowledged that Stanford had already granted a non-exclusive license in
the Nonexclusive Field of Use, under the Licensed Patents in the Licensed Field of Use in the Licensed Territory (as those terms are
defined in the February 2020 License Agreement”). However, Stanford agreed to not grant further licenses under the Licensed Patents
in the Licensed Field of Use in the Licensed Territory. On December 29, 2021, we entered into an amendment to the February 2020 License
Agreement which extended our exclusive right to license the technology deployed in AditxtScoreTM and securing worldwide
exclusivity in all fields of use of the licensed technology.
We were obligated to pay and paid a fee of $25,000
to Stanford within 60 days of February 3, 2020. We also issued 10 shares of the Company’s common stock to Stanford.
An annual licensing maintenance fee is payable by us on the first anniversary of the February 2020 License Agreement in the amount of
$40,000 for 2021 through 2024 and $60,000 starting in 2025 until the license expires upon the expiration of the patent. The Company is
required to pay and has paid $25,000 for the issuances of certain patents. The Company will pay milestone fees of $50,000 on the first
commercial sales of a licensed product and $25,000 at the beginning of any clinical study for regulatory clearance of an in vitro diagnostic
product developed and a potential licensed product. The Company paid a milestone fee for a clinical study for regulatory clearance of
an in vitro diagnostic product developed and a potential licensed product of $25,000 in March of 2022. We are also required to: (i) provide
a listing of the management team or a schedule for the recruitment of key management positions by March 31, 2020 (which has been completed),
(ii) provide a business plan covering projected product development, markets and sales forecasts, manufacturing and operations, and financial
forecasts until at least $10,000,000 in revenue by June 30, 2020 (which has been completed), (iii) conduct validation studies by September
30, 2020 (which has been completed), (iv) hold a pre-submission meeting with the FDA by September 30, 2020 (which has been completed),
(iv) submit a 510(k) application to the FDA, Emergency Use Authorization (“EUA”), or a Laboratory Developed Test (“LDT”)
by March 31, 2021 (which has been completed), (vi) develop a prototype assay for human profiling by December 31, 2021 (which has been
completed), (vii) execute at least one partnership for use of the technology for transplant, autoimmunity, or infectious disease purposes
by March 31, 2022 (which has been completed) and (viii) provided further development and commercialization milestones for specific fields
of use in writing prior to December 31, 2022.
In addition to the annual license maintenance
fees outlined above, we will pay Stanford royalties on Net Sales (as such term is defined in the February 2020 License Agreement) during
the of the term of the agreement as follows: 4% when Net Sales are below or equal to $5 million annually or 6% when Net Sales are above
$5 million annually. The February 2020 License Agreement may be terminated upon our election on at least 30 days advance notice to Stanford,
or by Stanford if we: (i) are delinquent on any report or payment; (ii) are not diligently developing and commercializing Licensed Product;
(iii) miss certain performance milestones; (iv) are in breach of any provision of the February 2020 License Agreement; or (v) provide
any false report to Stanford. Should any events in the preceding sentence occur, we have a thirty (30) day cure period to remedy such
violation.
Asset Purchase Agreement
MDNA Lifesciences, Inc.
On January 4, 2024 (the “Closing Date”),
the Company completed its acquisition of certain assets and issued to MDNA Lifesciences, Inc. (“MDNA”): 50,000 shares of
the Company’s Common Stock, 50,000 shares of the Company’s Warrants, and 5,000 shares of the Pearsanta Preferred Stock. The
Company accounted for this transaction as an asset acquisition.
On January 4, 2024, the Company, Pearsanta and
MDNA entered into a First Amendment to Asset Purchase Agreement (the “First Amendment to Asset Purchase Agreement”), pursuant
to which the parties agreed to: (i) the removal of an upfront working capital payment, (ii) the removal of a Closing Working Capital
Payment (as defined in the Purchase Agreement”), and (iii) to increase the maximum amount of payments to be made by Aditxt under
the Transition Services Agreement (as defined below) from $2.2 million to $3.2 million.
On January 4, 2024, Pearsanta and MDNA entered
into a Transition Services Agreement (the “Transition Services Agreement”), pursuant to which MDNA agreed that it would perform,
or cause certain of its affiliates or third parties to perform, certain services as described in the Transition Services Agreement for
a term of three months in consideration for the payment by Pearsanta of certain fees as provided in the Transition Services Agreement,
in an amount not to exceed $3.2 million.
As part of this transaction, the Company acquired
$1,008,669 in patents which was expensed to R&D. The fair market value of this transaction was determined by the purchase price paid
in the transaction of 50,000 shares of the Company’s Common Stock which had a value of $256,000 based on the trading price of the
common stock, 50,000 shares of the Company’s Warrants which had a value of $252,669 using a Black Sholes valuation, and 5,000 shares
of the Pearsanta Preferred Stock which had a value of $500,000 based on the stated value of Pearsanta’s Preferred Stock of $5,000
per share.
Brain Scientific, Inc.
On January 24, 2024, the Company entered into
an Assignment and Assumption Agreement (the “Brain Assignment Agreement”) with the agent (the “Agent”) of certain
secured creditors (the “Brain Creditors”) of Brain Scientific, Inc., a Nevada corporation (“Brain Scientific”)
and Philip J. von Kahle (the “Brain Seller”), as assignee of Brain Scientific and certain affiliated entities (collectively,
the “Brain Companies”) under an assignment for the benefit of creditors pursuant to Chapter 727 of the Florida Statutes.
Pursuant to the Brain Assignment Agreement, the Agent assigned its rights under that certain Asset Purchase and Settlement Agreement
dated October 31, 2023 between the Seller and the Agent (the “Brain Asset Purchase Agreement”) to the Company in consideration
for the issuance by the Company of an aggregate of 6,000 shares of a new series of convertible preferred stock of the Company, designated
as Series B-1 Convertible Preferred Stock, $0.001 par value (the “Series B-1 Preferred Stock”). The shares of Series B-1
Preferred Stock were issued pursuant to a Securities Purchase Agreement entered into by and between the Company and each of the purchasers
signatory thereto (the “Brain Purchase Agreement”). (See Note 10)
In connection with the Brain Assignment Agreement,
on January 24, 2024, the Company entered into a Patent Assignment with the Brain Seller (the “Brain Patent Assignment”),
pursuant to which the Seller assigned all of its rights, titles and interests in certain patents and patent applications that were previously
held by the Brain Companies to the Company.
As part of this transaction, the Company acquired
$5,703,995 in patents which was expensed to R&D and $266,448 in fixed assets. The fair market value of this transaction was determined
by the purchase price paid in the transaction of 6,000 shares of the Company’s Series B-1 Preferred Stock which had a value of
$5,970,443 based on stated value of the Series B-1 Preferred Stock of $1,000 per share.
Contingent Liability
On September 7, 2023, the Company received a
demand letter from the holder of certain warrants issued by the Company in April 2023. The demand letter alleged that the investor suffered
more than $2 million in damages as a result of the Company failing to register the shares of the Company’s common stock underlying
the warrants as required under the securities purchase agreement.
On January 3, 2024, the Company entered into
a settlement agreement and general release with an investor (the “Settlement Agreement”), pursuant to which the Company and
the investor agreed to settle an action filed in the United States District Court in the Southern District of New York by an investor
against the Company (the “Action”) in consideration of the issuance by the Company of shares of the Company’s Common
Stock (the “Settlement Shares”). The number of Settlement Shares to be issued will be equal to $1.6 million divided by the
closing price of the Company’s Common Stock on the day prior to court approval of the joint motion. Following the issuance of the
Settlement Shares, the Investor will file a dismissal stipulation in the Action.
On January 17, 2024, the Company issued 296,296
Settlement Shares to the investor. The Settlement Shares were issued pursuant to an exemption from registration pursuant to Section 3(a)(10)
under the Securities Act of 1933, as amended.
EvoFem Merger Agreement
On December 11, 2023 (the “Execution Date”),
Aditxt, Inc., a Delaware corporation (the “Company”) entered into an Agreement and Plan of Merger (the “Merger Agreement”)
with Adicure, Inc., a Delaware corporation and wholly owned subsidiary of the Company (“Merger Sub”) and Evofem Biosciences,
Inc., a Delaware corporation (“Evofem”), pursuant to which, Merger Sub will be merged into and with Evofem (the “Merger”),
with Evofem surviving the Merger as a wholly owned subsidiary of the Company.
In connection with the Merger Agreement the Company
assumed $13.0 million in notes payable held by Evofem (see Note 7) and assumed a payable for $154,480 (see Note 7). These items were
capitalized on the Company’s balance sheet to deposit on acquisition as of March 31, 2024. The Company recognized a debt discount
of $1,826,250. As of March 31, 2024, there was an unamortized discount of $0. During the three months ended March 31, 2024 and 2023,
the Company recognized an amortization of debt discount of $571,904 and $0.
Subject to the terms and conditions set forth
in the Merger Agreement, at the effective time of the Merger (the “Effective Time”), (i) all issued and outstanding shares
of common stock, par value $0.0001 per share of Evofem (“Evofem Common Stock”), other than any shares of Evofem Common Stock
held by the Company or Merger Sub immediately prior to the Effective Time, will be converted into the right to receive an aggregate of
610,000 shares of the Company’s common stock, par value $0.001 per share (“Company Common Stock”); and (ii) all issued
and outstanding shares of Series E-1 Preferred Stock, par value $0.0001 of Evofem (the “Evofem Unconverted Preferred Stock”),
other than any shares of Evofem Unconverted Preferred Stock held by the Company or Merger Sub immediately prior to the Effective Time,
will be converted into the right to receive an aggregate of 2,327 shares of Series A-1 Preferred Stock, par value $0.001 of the Company
(the “Company Preferred Stock”), having such rights, powers, and preferences set forth in the form of Certificate of Designation
of Series A-1 Preferred Stock.
On December 11, 2023 the Company entered into
an Agreement and Plan of Merger (the “Merger Agreement”) with Adicure, Inc., a Delaware corporation and wholly owned subsidiary
of the Company (“Merger Sub”) and Evofem Biosciences, Inc., a Delaware corporation (“Evofem”), pursuant to which,
Merger Sub will be merged into and with Evofem (the “Merger”), with Evofem surviving the Merger as a wholly owned subsidiary
of the Company.
On January 8, 2024, the Company, Adicure, Inc.,
a Delaware corporation and wholly owned subsidiary of the Company (“Merger Sub”), and Evofem Biosciences, Inc., a Delaware
corporation (“Evofem”) entered into the First Amendment (the “First Amendment to Merger Agreement”), to the Agreement
and Plan of Merger (the “Merger Agreement”) pursuant to which the parties agreed to extend the date by which the joint proxy
statement would be filed with the SEC until February 14, 2024.
On January 30, 2024, the Company, Adicure and
Evofem entered into the Second Amendment to the Merger Agreement (the “Second Amendment to Merger Agreement”) to amend (i)
the date of the Parent Loan (as defined in the Merger Agreement) to Evofem to be February 29, 2024, (ii) to change the date by which
Evofem may terminate the Merger Agreement for failure to receive the Parent Loan to be February 29, 2024, and (iii) to change the filing
date for the Joint Proxy Statement (as defined in the Merger Agreement) to April 1, 2024.
On February 29, 2024, the Company, Adicure and
Evofem entered into the Third Amendment to the Merger Agreement (the “Third Amendment to Merger Agreement”) in order to (i)
make certain conforming changes to the Merger Agreement regarding the Notes, (ii) extend the date by which the Company and Evofem will
file the joint proxy statement until April 30, 2024, and (iii) remove the requirement that the Company make the Parent Loan (as defined
in the Merger Agreement) by February 29, 2024 and replace it with the requirement that the Company make an equity investment into Evofem
consisting of (a) a purchase of 2,000 shares of Evofem Series F-1 Preferred Stock for an aggregate purchase price of $2.0 million on
or prior to April 1, 2024, and (b) a purchase of 1,500 shares of Evofem Series F-1 Preferred Stock for an aggregate purchase price of
$1.5 million on or prior to April 30, 2024. As of the date of this filing the Company has not purchased the 2,000 shares of EvoFem Series
F-1 Preferred Stock.
Engagement Letter with Dawson James Securities,
Inc.
On February 16, 2024, the “Company”
entered into an engagement letter (the “Dawson Engagement Letter”) with Dawson James Securities, Inc.(“Dawson”),
pursuant to which the Company engaged Dawson to serve as financial advisor with respect to one or more potential business combinations
involving the Company for a term of twelve months. Pursuant to the Dawson Engagement Letter, the Company agreed to pay Dawson an initial
fee of $1.85 million (the “Dawson Initial Fee”), which amount is payable on the later of (i) the closing of an offering resulting
in gross proceeds to the Company of greater than $4.9 million, or (ii) five days after the execution of the Dawson Engagement Letter.
At the Company’s option, the Dawson Initial Fee may be paid in securities of the Company. In addition, with respect to any business
combination (i) that either is introduced to the Company by Dawson following the date of the Dawson Engagement Letter or (ii) that with
respect to which the Company hereafter requests Dawson to provide M&A advisory services, the Company shall compensate Dawson in an
amount equal to 5% of the Total Transaction Value (as defined in the Engagement Letter) with respect to the first $20.0 million in Total
Transaction Value plus 10.0% of the Total Transaction Value that is in excess of $20.0 million (the “Transaction Fee”). The
Transaction Fee is payable upon the closing of a business combination transaction.
Advance on Private Placement
On March 5, 2024, the Company received a $1,000,000
deposit for an ongoing Private Placement (as defined below), of which $400,000 was attributed to offering costs in connection with the
Private Placement. As of March 31, 2024 the Private Placement had not yet closed. The Private Placement closed subsequent to the quarter
and the net advance amount was converted into Series C-1 Preferred Stock. (See note 12)
NOTE 10 – STOCKHOLDERS’ EQUITY
Common Stock
On May 24, 2021, the Company increased the number
of authorized shares of the Company’s common stock, par value $0.001 per share, from 27,000,000 to 100,000,000 (the
“Authorized Shares Increase”) by filing a Certificate of Amendment (the “Certificate of Amendment”) to its Amended
and Restated Certificate of Incorporation with the Secretary of State of the State of Delaware. In accordance with the General Corporation
Law of the State of Delaware, the Authorized Shares Increase and the Certificate of Amendment were approved by the stockholders of the
Company at the Company’s Annual Meeting of Stockholders on May 19, 2021. On September 13, 2022, the Company effectuated a
1 for 50 reverse stock split (the “2022 Reverse Split”). The Company’s stock began trading at the 2022 Reverse
Split price effective on the Nasdaq Stock Market on September 14, 2022. There was no change to the number of authorized shares of the
Company’s common stock. On August 17, 2023, the Company effectuated a 1 for 40 reverse stock split (the “2023 Reverse Split”). The
Company’s stock began trading at the 2023 Reverse Split price effective on the Nasdaq Stock Market on August 17, 2023. There was
no change to the number of authorized shares of the Company’s common stock.
Formed in January 2023, our majority owned subsidiary
Pearsanta™, Inc. (“Pearsanta”) seeks to take personalized medicine to a new level by delivering “Health by the
Numbers.” On November 22, 2023, Pearsanta entered into an assignment agreement with FirstVitals LLC, an entity controlled by Pearsanta’s
CEO, Ernie Lee (“FirstVitals”), pursuant to which FirstVitals assigned its rights in certain intellectual property and website
domain to Pearsanta in consideration of the issuance of 500,000 shares of Pearsanta common stock to FirstVitals. On December 18, 2023,
the board of directors of Pearsanta adopted the Pearsanta 2023 Omnibus Equity Incentive Plan (the “Pearsanta Omnibus Incentive
Plan”), pursuant to which it reserved 15 million shares of common stock of Pearsanta for future issuance under the Pearsanta Omnibus
Incentive Plan and the Pearsanta 2023 Parent Service Provider Equity Incentive Plan (the “Pearsanta Parent Service Provider Plan”)
and approved the issuance of 9.32 million options, exercisable into shares of Pearsanta common stock under the Pearsanta Parent Service
Provider Plan and the issuance of 4.0 million options, exercisable into shares of Pearsanta common stock, subject to vesting, and 1.0
million restricted common stock shares under the Pearsanta Omnibus Incentive Plan.
During the three months ended March 31, 2024,
the Company issued 50,000 shares of common stock as part of the MDNA asset purchase agreement. (See Note 9) During the three
months ended March 31, 2024, the Company issued 296,296 shares of common stock as part of a settlement agreement. (See Note 9)
During the three months ended March 31, 2023,
the Company issued 4,675 shares of common stock and recognized expense of $168,300 in stock-based compensation for consulting
services. The stock-based compensation for consulting services is calculated by the number shares multiplied by the closing price on
the effective date of the contract. During the three months ended March 31, 2023, 44 Restricted Stock Units vested which
resulted in the issuance of shares. The Company recognized expense of $111,187 in stock-based compensation for the three months
ended March 31, 2023. The stock-based compensation for shares issued or RSU’s granted during the period were valued based on the
fair market value on the date of grant.
Closing of Private Placement
On December 29, 2023, the Company entered into
a securities purchase agreement (the “Purchase Agreement”) with an institutional investor (“the “December Purchaser”)
for the issuance and sale in a private placement (the “December Private Placement”) of (i) pre-funded warrants (the “December
Pre-Funded Warrants”) to purchase up to 1,237,114 shares of the Company’s Common Stock, par value $0.001 at an exercise price
of $0.001 per share, and (ii) warrants (the “December Common Warrants”) to purchase up to 2,474,228 shares of the Company’s
Common Stock, at a purchase price of $4.85 per share.
Pursuant to the Purchase Agreement, the Company
agreed to reduce the exercise price of certain outstanding warrants to purchase Common Stock of the Company (“Certain Outstanding
Warrants”) held by the Purchaser to $4.60 per share in consideration for the cash payment by the December Purchaser of $0.125 per
share of Common Stock underlying the Certain Outstanding Warrants, effective immediately.
The December Private Placement closed on January
4, 2024. The net proceeds to the Company from the December Private Placement were approximately $5.5 million, after deducting placement
agent fees and expenses and estimated offering expenses payable by the Company.
In addition, the Company agreed to pay H.C. Wainwright
& Co., LLC (“Wainwright”) certain expenses and issued to Wainwright or its designees warrants (the “December Placement
Agent Warrants”) to purchase up to an aggregate of 74,227 shares of Common Stock at an exercise price equal to $6.0625 per share.
The December Placement Agent Warrants are exercisable immediately upon issuance and have a term of exercise equal to three years from
the date of issuance.
Preferred Stock
The Company is authorized to issue 3,000,000 shares
of preferred stock, par value $0.001 per share. There were 30,905 and 24,905 shares of preferred stock outstanding as
of March 31, 2024 and December 31, 2023, respectively.
| Aditxt Preferred Share Class | |
Quantity
Issued and
Outstanding
as of
March 31,
2024 | |
| Series A Preferred Stock | |
| - | |
| Series A-1 Convertible Preferred Stock | |
| 22,280 | |
| Series B Preferred Stock | |
| - | |
| Series B-1 Convertible Preferred Stock | |
| 6,000 | |
| Series B-2 Convertible Preferred Stock | |
| 2,625 | |
| Series C Preferred Stock | |
| - | |
| Total Aditxt Preferred Shares Outstanding | |
| 30,905 | |
Issuance of Series A-1 Preferred Stock:
On December 11, 2023 (the “Execution Date”),
the Company entered into an Agreement and Plan of Merger (the “Merger Agreement”) with Adicure, Inc., a Delaware corporation
and wholly owned subsidiary of the Company (“Merger Sub”) and Evofem Biosciences, Inc., a Delaware corporation (“Evofem”),
pursuant to which, Merger Sub will be merged into and with Evofem (the “Merger”), with Evofem surviving the Merger as a wholly
owned subsidiary of the Company.
Subject to the terms and conditions set forth
in the Merger Agreement, at the effective time of the Merger (the “Effective Time”), (i) all issued and outstanding shares
of common stock, par value $0.0001 per share of Evofem (“Evofem Common Stock”), other than any shares of Evofem Common Stock
held by the Company or Merger Sub immediately prior to the Effective Time, will be converted into the right to receive an aggregate of
610,000 shares of the Company’s common stock, par value $0.001 per share (“Company Common Stock”); and (ii) all issued
and outstanding shares of Series E-1 Preferred Stock, par value $0.0001 of Evofem (the “Evofem Unconverted Preferred Stock”),
other than any shares of Evofem Unconverted Preferred Stock held by the Company or Merger Sub immediately prior to the Effective Time,
will be converted into the right to receive an aggregate of 2,327 shares of Series A-1 Preferred Stock, par value $0.001 of the Company
(the “Company Preferred Stock”), having such rights, powers, and preferences set forth in the form of Certificate of Designation
of Series A-1 Preferred Stock. See Series A-1 Preferred Stock certificate of designation incorporated by reference to this document.
On December 22, 2023, the Company entered into
an Exchange Agreement (the “Exchange Agreement”) with the holders (the “Holders”) of an aggregate of 22,280 shares
of Series F-1 Convertible Preferred Stock of Evofem (the “Evofem Series F-1 Preferred Stock”) agreed to exchange their respective
shares of Evofem Series F-1 Preferred Stock for an aggregate of 22,280 shares of a new series of convertible preferred stock of the Company
designated as Series A-1 Convertible Preferred Stock, $0.001 par value, (the “Series A-1 Preferred Stock”).
The following is only a summary of the Series
A-1 Certificate of Designations, and is qualified in its entirety by reference to the full text of the Series A-1 Certificate of Designations,
a copy of which is filed as Exhibit 3.1 to our Current Report on Form 8-K filed on December 26, 2023 and is incorporated by reference
herein.
Designation, Amount, and Par Value: The number
of Series A-1 Preferred Stock designated is 22,280 shares. The shares of Series A-1 Preferred Stock have a par value of $0.001 per share
and a stated value of $1,000 per share.
Conversion Price: The Series A-1 Preferred Stock
will be convertible into shares of Common Stock at an initial conversion price of $4.44 (subject to adjustment pursuant to the Series
A-1 Certificate of Designations) (the “Conversion Price”). The Certificate of Designations also provides that in the event
of certain Triggering Events (as defined below) any holder may, at any time, convert any or all of such holder’s Series A-1 Preferred
Stock at an alternate conversion rate equal to the product of (i) the Alternate Conversion Price (as defined below) and (ii) the quotient
of (x) the 25% redemption premium multiplied by (y) the amount of Series A-1 Preferred Stock subject to such conversion. “Triggering
Events” include, among others, (i) a suspension of trading or the failure to be traded or listed on an eligible market for five
consecutive days or more, (ii) the failure to remove restrictive legends when required, (iii) the Company’s default in payment
of indebtedness in an aggregate amount of $500,000 or more (the Company is currently in default for payments greater than $500,000),
(iv) proceedings for a bankruptcy, insolvency, reorganization or liquidation, which are not dismissed with 30 days, (v) commencement
of a voluntary bankruptcy proceeding, and (viii) final judgments against the Company for the payment of money in excess of $100,000.
“Alternate Conversion Price” means the lowest of (i) the applicable conversion price the in effect, (ii) the greater of (x)
$0.888 (the “Floor Price”) and (y) 80% of the volume weighted average price (“VWAP”) of the Common Stock on the
trading day immediately preceding the delivery of the applicable conversion notice. Further, the Series A-1 Certificate of Designations
provides that if on any of the 90th and 180th day after each of the occurrence of any Stock Combination Event (as defined in the Series
A-1 Certificate of Designations) and the Applicable Date (as defined in the Series A-1 Certificate of Designations), the conversion price
then in effect is greater than the market price then in effect (the “Adjustment Price”), on such date then the conversion
price shall automatically lower to the Adjustment Price.
Dividends: Holders of the Series A-1 Preferred
Stock shall be entitled to receive dividends when and as declared by the Board, from time to time, in its sole discretion, which Dividends
shall be paid by the Company out of funds legally available therefor, payable, subject to the conditions and other terms hereof, in cash,
in securities of the Company or any other entity, or using assets as determined by the Board on the Stated Value of such Preferred Share.
Liquidation: In the event of a Liquidation Event
(as defined in the Series A-1 Certificate of Designation), the holders the Series A-1 Preferred Stock shall be entitled to receive in
cash out of the assets of the Company, before any amount shall be paid to the holders of any other shares of capital stock of the Company,
equal to the greater of (A) 125% of the Conversion Amount (as defined in the Series A-1 Certificate of Designation) on the date of such
payment and (B) the amount per share such holder of Series A-1 Preferred Stock would receive if they converted such share of Series A-1
Preferred Stock into Common Stock immediately prior to the date of such payment
Company Redemption: The Company may redeem all,
or any portion, of the Series A-1 Preferred Stock for cash, at a price per share of Series A-1 Preferred Stock equal to 115% of the greater
of (i) the Conversion Amount (as defined in the Series A-1 Certificate of Designation)being redeemed as of the Company Optional Redemption
Date (as defined in the Series A-1 Certificate of Designation) and (ii) the product of (1) the Conversion Rate (as defined in the Series
A-1 Certificate of Designation) with respect to the Conversion Amount being redeemed as of the Company Optional Redemption Date multiplied
by (2) the greatest Closing Sale Price (as defined in the Certificate of Designation) of the Common Stock on any Trading Day during the
period commencing on the date immediately preceding such Company Optional Redemption Notice Date (as defined in the Certificate of Designation)
and ending on the Trading Day immediately prior to the date the Company makes the entire payment required to be made under the Certification
of Designation.
Maximum Percentage: Holders of Series A-1 Preferred
Stock are prohibited from converting shares of Series A-1 Preferred Stock into shares of Common Stock if, as a result of such conversion,
such holder, together with its affiliates, would beneficially own in excess of 4.99% (the “Maximum Percentage”) of the total
number of shares of Common Stock issued and outstanding immediately after giving effect to such conversion.
Voting Rights: The holders of the Series A-1
Preferred Stock shall have no voting power and no right to vote on any matter at any time, either as a separate series or class or together
with any other series or class of share of capital stock, and shall not be entitled to call a meeting of such holders for any purpose
nor shall they be entitled to participate in any meeting of the holders of Common Stock, except as expressly provided in the Certificate
of Designations and where required by the DGCL.
Issuance of Series B Preferred Stock:
On July 19, 2022, the Company entered into a
Subscription and Investment Representation Agreement with its Chief Executive Officer (the “Purchaser”), pursuant to which
the Company agreed to issue and sell one (1) share of the Company’s Series B Preferred Stock (the “Preferred Stock”),
par value $0.001 per share, to the Purchaser for $20,000 in cash.
On July 19, 2022, the Company filed a certificate
of designation (the “Certificate of Designation”) with the Secretary of State of Delaware, effective as of the time of filing,
designating the rights, preferences, privileges and restrictions of the share of Preferred Stock. The Certificate of Designation provides
that the share of Preferred Stock will have 250,000,000 votes and will vote together with the outstanding shares of the Company’s
common stock as a single class exclusively with respect to any proposal to amend the Company’s Restated Certificate of Incorporation
to effect a reverse stock split of the Company’s common stock. The Preferred Stock will be voted, without action by the holder,
on any such proposal in the same proportion as shares of common stock are voted. The Preferred Stock otherwise has no voting rights except
as otherwise required by the General Corporation Law of the State of Delaware.
The Preferred Stock is not convertible into,
or exchangeable for, shares of any other class or series of stock or other securities of the Company. The Preferred Stock has no rights
with respect to any distribution of assets of the Company, including upon a liquidation, bankruptcy, reorganization, merger, acquisition,
sale, dissolution or winding up of the Company, whether voluntarily or involuntarily. The holder of the Preferred Stock will not be entitled
to receive dividends of any kind. See Series B Preferred Stock certificate of designation incorporated by reference to this document.
The outstanding share of Preferred Stock shall
be redeemed in whole, but not in part, at any time (i) if such redemption is ordered by the Board of Directors in its sole discretion
or (ii) automatically upon the effectiveness of the amendment to the Certificate of Incorporation implementing a reverse stock split.
Upon such redemption, the holder of the Preferred Stock will receive consideration of $20,000 in cash.
Redemption of Series B Preferred Stock
On October 7, 2022, the Company paid $20,000 in
consideration for the one share of Preferred Stock which was redeemed on September 13, 2022.
Series B-1 Preferred Stock Certificate of
Designation
On January 24, 2024, the Company filed a Certificate
of Designations for its Series B-1 Preferred Stock with the Secretary of State of Delaware (the “Series B-1 Certificate of Designations”).
The following is only a summary of the Series B-1 Certificate of Designations, and is qualified in its entirety by reference to the full
text of the Series B-1 Certificate of Designations, a copy of which is filed as Exhibit 3.1 to this Current Report on Form 8-K and is
incorporated by reference herein.
Designation, Amount, and Par Value: The
number of Series B-1 Preferred Stock designated is 6,000 shares. The shares of Series B-1 Preferred Stock have a par value of $0.001
per share and a stated value of $1,000 per share.
Conversion Price: The Series B-1 Preferred Stock
will be convertible into shares of Common Stock at an initial conversion price of $4.06 (subject to adjustment pursuant to the Series
B-1 Certificate of Designations) (the “Conversion Price”). The Series B-1 Certificate of Designations also provides
that in the event of certain Triggering Events (as defined below) any holder may, at any time, convert any or all of such holder’s
Series B-1 Preferred Stock at an alternate conversion rate equal to the product of (i) the Alternate Conversion Price (as defined
below) and (ii) the quotient of (x) the 125% redemption premium multiplied by (y) the amount of Series B-1 Preferred Stock
subject to such conversion. “Triggering Events” include, among others, (i) a suspension of trading or the failure to be traded
or listed on an eligible market for five consecutive days or more, (ii) the failure to remove restrictive legends when required, (iii)
the Company’s default in payment of indebtedness in an aggregate amount of $500,000 or more, (iv) proceedings for a bankruptcy,
insolvency, reorganization or liquidation, which are not dismissed with 30 days, (v) commencement of a voluntary bankruptcy proceeding,
and (viii) final judgments against the Company for the payment of money in excess of $500,000. “Alternate Conversion Price”
means the lowest of (i) the applicable conversion price the in effect, (ii) the greater of (x) $0.9420 (the “Floor Price”)
and (y) 80% of the lowest volume weighted average price (“VWAP”) of the Common Stock during the five consecutive trading
day period ending and including the trading day immediately preceding the delivery of the applicable conversion notice. Further, the
Series B-1 Certificate of Designations provides that if on any of the 90th and 180th day after each
of the occurrence of any Stock Combination Event (as defined in the Series B-1 Certificate of Designations) and the Applicable
Date (as defined in the Series B-1 Certificate of Designations), the conversion price then in effect is greater than the market
price then in effect (the “Adjustment Price”), on such date then the conversion price shall automatically lower to the Adjustment
Price.
Dividends: Holders of the Series B-1 Preferred
Stock shall be entitled to receive dividends when and as declared by the Board, from time to time, in its sole discretion, which Dividends
shall be paid by the Company out of funds legally available therefor, payable, subject to the conditions and other terms hereof, in cash,
in securities of the Company or any other entity, or using assets as determined by the Board on the Stated Value of such Preferred Share.
Liquidation: In the event
of a Liquidation Event (as defined in the Series B-1 Certificate of Designations), the holders the Series B-1 Preferred Stock shall be
entitled to receive in cash out of the assets of the Company, before any amount shall be paid to the holders of any other shares of capital
stock of the Company, equal to the greater of (A) 125% of the Conversion Amount (as defined in the Series B-1 Certificate of Designation)
on the date of such payment and (B) the amount per share such holder of Series B-1 Preferred Stock would receive if they converted such
share of Series B-1 Preferred Stock into Common Stock immediately prior to the date of such payment.
Company Redemption: The Company may redeem
all, or any portion, of the Series B-1 Preferred Stock for cash, at a price per share of Series B-1 Preferred Stock equal to 115% of
the greater of (i) the Conversion Amount (as defined in the Series B-1 Certificate of Designations) being redeemed as of the Company
Optional Redemption Date (as defined in the Series B-1 Certificate of Designations) and (ii) the product of (1) the Conversion Rate (as
defined in the Series B-1 Certificate of Designations) with respect to the Conversion Amount being redeemed as of the Company Optional
Redemption Date multiplied by (2) the greatest Closing Sale Price (as defined in the Series B-1 Certificate of Designations) of the Common
Stock on any Trading Day during the period commencing on the date immediately preceding such Company Optional Redemption Notice Date
(as defined in the Series B-1 Certificate of Designations) and ending on the Trading Day immediately prior to the date the Company makes
the entire payment required to be made under the Certification of Designation.
Maximum Percentage: Holders of Series B-1 Preferred
Stock are prohibited from converting shares of Series B-1 Preferred Stock into shares of Common Stock if, as a result of such conversion,
such holder, together with its affiliates, would beneficially own in excess of 4.99% (the “Maximum Percentage”) of the total
number of shares of Common Stock issued and outstanding immediately after giving effect to such conversion.
Voting Rights: The holders
of the Series B-1 Preferred Stock shall have no voting power and no right to vote on any matter at any time, either as a separate series
or class or together with any other series or class of share of capital stock, and shall not be entitled to call a meeting of such holders
for any purpose nor shall they be entitled to participate in any meeting of the holders of Common Stock, except as expressly provided
in the Series B-1 Certificate of Designations and where required by the DGCL.
Issuance of Series B-2 Preferred Stock:
On December 29, 2023, the Company entered into
an Exchange Agreement (the “Note Exchange Agreement”) with the Noteholder, pursuant to which the Noteholder agreed, subject
to the terms and conditions set forth therein, to exchange the Note, including all accrued but unpaid interest thereon, for an aggregate
of 2,625 shares of a new series of convertible preferred stock of the Company, designated as Series B-2 Convertible Preferred Stock,
$0.001 par value (the “Series B-2 Preferred Stock”). See Series B-2 Preferred Stock certificate of designation incorporated
by reference to this document.
The following is only a summary of the Series
B-2 Certificate of Designations, and is qualified in its entirety by reference to the full text of the Series B-2 Certificate of Designations,
a copy of which is filed as an exhibit to our Current Report on Form 8-K filed with the SEC on January 2, 2024.
Designation, Amount, and Par Value: The number
of Series B-2 Preferred Stock designated is 2,625 shares. The shares of Series B-2 Preferred Stock have a par value of $0.001 per share
and a stated value of $1,000 per share.
Conversion Price: The Series B-2 Preferred Stock
will be convertible into shares of Common Stock at an initial conversion price of $4.71 (subject to adjustment pursuant to the Series
B-2 Certificate of Designations) (the “Conversion Price”). The Series B-2 Certificate of Designations also provides that
in the event of certain Triggering Events (as defined below) any holder may, at any time, convert any or all of such holder’s Series
B-2 Preferred Stock at an alternate conversion rate equal to the product of (i) the Alternate Conversion Price (as defined below) and
(ii) the quotient of (x) the 125% redemption premium multiplied by (y) the amount of Series B-2 Preferred Stock subject to such conversion.
“Triggering Events” include, among others, (i) a suspension of trading or the failure to be traded or listed on an eligible
market for five consecutive days or more, (ii) the failure to remove restrictive legends when required, (iii) the Company’s default
in payment of indebtedness in an aggregate amount of $500,000 or more(the Company is currently in default for payments greater than $500,000),
(iv) proceedings for a bankruptcy, insolvency, reorganization or liquidation, which are not dismissed with 30 days, (v) commencement
of a voluntary bankruptcy proceeding, and (viii) final judgments against the Company for the payment of money in excess of $500,000.
“Alternate Conversion Price” means the lowest of (i) the applicable conversion price the in effect, (ii) the greater of (x)
$0.9420 (the “Floor Price”) and (y) 80% of the lowest volume weighted average price (“VWAP”) of the Common Stock
during the five consecutive trading day period ending and including the trading day immediately preceding the delivery of the applicable
conversion notice. Further, the Series B-2 Certificate of Designations provides that if on any of the 90th and 180th day after each of
the occurrence of any Stock Combination Event (as defined in the Series B-2 Certificate of Designations) and the Applicable Date (as
defined in the Series B-2 Certificate of Designations), the conversion price then in effect is greater than the market price then in
effect (the “Adjustment Price”), on such date then the conversion price shall automatically lower to the Adjustment Price.
Dividends: Holders of the Series B-2 Preferred
Stock shall be entitled to receive dividends when and as declared by the Board, from time to time, in its sole discretion, which Dividends
shall be paid by the Company out of funds legally available therefor, payable, subject to the conditions and other terms hereof, in cash,
in securities of the Company or any other entity, or using assets as determined by the Board on the Stated Value of such Preferred Share.
Liquidation: In the event of a Liquidation Event
(as defined in the Series B-2 Certificate of Designations), the holders the Series B-2 Preferred Stock shall be entitled to receive in
cash out of the assets of the Company, before any amount shall be paid to the holders of any other shares of capital stock of the Company,
equal to the greater of (A) 125% of the Conversion Amount (as defined in the Series B-2 Certificate of Designation) on the date of such
payment and (B) the amount per share such holder of Series B-2 Preferred Stock would receive if they converted such share of Series B-2
Preferred Stock into Common Stock immediately prior to the date of such payment.
Company Redemption: The Company may redeem all,
or any portion, of the Series B-2 Preferred Stock for cash, at a price per share of Series B-2 Preferred Stock equal to 115% of the greater
of (i) the Conversion Amount (as defined in the Series B-2 Certificate of Designations) being redeemed as of the Company Optional Redemption
Date (as defined in the Series B-2 Certificate of Designations) and (ii) the product of (1) the Conversion Rate (as defined in the Series
B-2 Certificate of Designations) with respect to the Conversion Amount being redeemed as of the Company Optional Redemption Date multiplied
by (2) the greatest Closing Sale Price (as defined in the Series B-2 Certificate of Designations) of the Common Stock on any Trading
Day during the period commencing on the date immediately preceding such Company Optional Redemption Notice Date (as defined in the Series
B-2 Certificate of Designations) and ending on the Trading Day immediately prior to the date the Company makes the entire payment required
to be made under the Certification of Designation.
Maximum Percentage: Holders of Series B-2 Preferred
Stock are prohibited from converting shares of Series B-2 Preferred Stock into shares of Common Stock if, as a result of such conversion,
such holder, together with its affiliates, would beneficially own in excess of 4.99% (the “Maximum Percentage”) of the total
number of shares of Common Stock issued and outstanding immediately after giving effect to such conversion.
Voting Rights: The holders of the Series B-2
Preferred Stock shall have no voting power and no right to vote on any matter at any time, either as a separate series or class or together
with any other series or class of share of capital stock, and shall not be entitled to call a meeting of such holders for any purpose
nor shall they be entitled to participate in any meeting of the holders of Common Stock, except as expressly provided in the Series B-2
Certificate of Designations and where required by the DGCL.
Series C Preferred Stock
On July 11, 2023, the Company filed a certificate
of designation (the “Certificate of Designation”) with the Secretary of State of Delaware, effective as of the time of filing,
designating the rights, preferences, privileges and restrictions of the share of Preferred Stock. The Certificate of Designation provides
that the share of Preferred Stock will have 250,000,000 votes and will vote together with the outstanding shares of the Company’s
common stock as a single class exclusively with respect to any proposal to amend the Company’s Amended and Restated Certificate
of Incorporation to effect a reverse stock split of the Company’s common stock. The Preferred Stock will be voted, without action
by the holder, on any such proposal in the same proportion as shares of common stock are voted. The Preferred Stock otherwise has no
voting rights except as otherwise required by the General Corporation Law of the State of Delaware.
The Preferred Stock is not convertible into,
or exchangeable for, shares of any other class or series of stock or other securities of the Company. The Preferred Stock has no rights
with respect to any distribution of assets of the Company, including upon a liquidation, bankruptcy, reorganization, merger, acquisition,
sale, dissolution or winding up of the Company, whether voluntarily or involuntarily. The holder of the Preferred Stock will not be entitled
to receive dividends of any kind.
The outstanding share of Preferred Stock shall
be redeemed in whole, but not in part, at any time (i) if such redemption is ordered by the Board of Directors in its sole discretion
or (ii) automatically upon the effectiveness of the amendment to the Certificate of Incorporation implementing a reverse stock split.
Upon such redemption, the holder of the Preferred Stock will receive consideration of $1,000 in cash. As of December 31, 2023, the share
has been redeemed and the consideration has been paid.
On July 11, 2023, the Company entered into a
Subscription and Investment Representation Agreement (the “Subscription Agreement”) with Amro Albanna, its Chief Executive
Officer, who is an accredited investor (the “Purchaser”), pursuant to which the Company agreed to issue and sell one (1)
share of the Company’s Series C Preferred Stock, par value $0.001 per share (the “Preferred Stock”), to the Purchaser
for $1,000 in cash. The sale closed on July 11, 2023. The Subscription Agreement contains customary representations and warranties and
certain indemnification rights and obligations of the parties. See Series C Preferred Stock certificate of designation incorporated by
reference to this document. On August 17, 2023, the share was redeemed.
Stock-Based Compensation
In October 2017, our Board of Directors adopted
the Aditx Therapeutics, Inc. 2017 Equity Incentive Plan (the “2017 Plan”). The 2017 Plan provides for the grant of equity
awards to directors, employees, and consultants. The Company is authorized to issue up to 2,500,000 shares of our common
stock pursuant to awards granted under the 2017 Plan. The 2017 Plan is administered by our Board of Directors, and expires ten years
after adoption, unless terminated earlier by the Board of Directors. All shares of our common stock pursuant to awards under the
2017 Plan have been awarded.
On February 24, 2021, our Board of Directors
adopted the Aditx Therapeutics, Inc. 2021 Omnibus Equity Incentive Plan (the “2021 Plan”). The 2021 Plan provides for grants
of nonqualified stock options, incentive stock options, stock appreciation rights, restricted stock and restricted stock units, and other
stock-based awards (collectively, the “Awards”). Eligible recipients of Awards include employees, directors or independent
contractors of the Company or any affiliate of the Company. The Compensation Committee of the Board of Directors (the “Committee”)
administers the 2021 Plan. A total of 60,000 shares of common stock, par value $0.001 per share, of the Company may be
issued pursuant to Awards granted under the 2021 Plan. The exercise price per share for the shares to be issued pursuant to an exercise
of a stock option will be no less than one hundred percent (100%) of the Fair Market Value (as defined in the 2021 Plan) of a share of
Common Stock on the date of grant. The 2021 Plan was submitted and approved by the Company’s stockholders at the 2021 annual meeting
of stockholders, held on May 19, 2021.
During the three months ended March 31, 2024
and 2023, the Company granted no new options.
The Company recognizes option forfeitures as
they occur, as there is insufficient historical data to accurately determine future forfeitures rates.
The following is an analysis of the stock option
grant activity under the Plan:
| Vested and Nonvested Stock Options | |
Number | | |
Weighted
Average
Exercise
Price | | |
Weighted
Average
Remaining
Life | |
| Outstanding December 31, 2023 | |
| 45,572 | | |
$ | 173.12 | | |
| 9.74 | |
| Granted | |
| - | | |
| - | | |
| - | |
| Exercised | |
| - | | |
| - | | |
| - | |
| Expired or forfeited | |
| - | | |
| - | | |
| - | |
| Outstanding March 31, 2024 | |
| 45,572 | | |
$ | 173.12 | | |
| 9.49 | |
| Nonvested Stock Options | |
Number | | |
Weighted-
Average
Exercise
Price | |
| Nonvested on December 31, 2023 | |
| - | | |
$ | - | |
| Granted | |
| - | | |
| - | |
| Vested | |
| - | | |
| - | |
| Forfeited | |
| - | | |
| - | |
| Nonvested on March 31, 2024 | |
| - | | |
$ | - | |
As of March 31, 2024 there were 45,572 exercisable
options; these options had a weighted average exercise price $173.12.
On December 18, 2023, our Board of Directors
adopted the Pearsanta, Inc. 2023 Omnibus Equity Incentive Plan (the “Pearsanta 2023 Plan”) and the 2023 Parent Service Provider
Equity Incentive Plan (the “Pearsanta Parent 2023 Plan”), collectively (the “Pearsanta Plans”). The Pearsanta
Plans provides for grants of nonqualified stock options, incentive stock options, stock appreciation rights, restricted stock and restricted
stock units, and other stock-based awards (collectively, the “Pearsanta Awards”). Eligible recipients of Pearsanta Awards
include employees, directors or independent contractors of the Company or any affiliate of the Company. The Board of Directors administers
the Pearsanta Plans. The Pearsanta 2023 Plan consists of a total of 15,000,000 shares of Pearsanta common stock, par value
$0.001 per share, which may be issued pursuant to Pearsanta Awards granted under the Pearsanta 2023 Plan. The Pearsanta Parent 2023
Plan consists of a total of 9,320,000 shares of Pearsanta common stock, par value $0.001 per share, which may be issued
pursuant to Pearsanta Awards granted under the Pearsanta Parent 2023 Plan. The exercise price per share for the shares to be issued pursuant
to an exercise of a stock option will be no less than one hundred percent (100%) of the Fair Market Value (as defined in the Pearsanta
Plans) of a share of Common Stock on the date of grant.
During the three months ended March 31, 2024
and 2023, Pearsanta granted no new options under the Pearsanta 2023 Plan.
The following is an analysis of the stock option
grant activity under the Pearsanta Plans:
| Vested and Nonvested Stock Options | |
Number | | |
Weighted
Average
Exercise
Price | | |
Weighted
Average
Remaining
Life | |
| Outstanding December 31, 2023 | |
| 13,320,000 | | |
$ | 0.02 | | |
| 9.97 | |
| Granted | |
| - | | |
| - | | |
| - | |
| Exercised | |
| - | | |
| - | | |
| - | |
| Expired or forfeited | |
| - | | |
| - | | |
| - | |
| Outstanding March 31, 2024 | |
| 13,320,000 | | |
$ | 0.02 | | |
| 9.72 | |
| Nonvested Stock Options | |
Number | | |
Weighted-
Average
Exercise
Price | |
| Nonvested on December 31, 2023 | |
| 4,000,000- | | |
$ | 0.02 | |
| Granted | |
| - | | |
| - | |
| Vested | |
| (1,334,000 | ) | |
| 0.02 | |
| Forfeited | |
| - | | |
| - | |
| Nonvested on March 31, 2024 | |
| 2,666,000 | | |
$ | 0.02 | |
As of March 31, 2024, there were 10,654,000 exercisable
options; these options had a weighted average exercise price $0.02.
The Company recognized stock-based compensation
expense related to all options granted and vesting expense of $24,572 during the three months ended March 31, 2024, of which $24,572 is
included in general and administrative expenses in the accompanying statements of operations. The remaining value to be expensed is $53,240 as
of March 31, 2024. The weighted average vesting term is 1.92 years as of March 31, 2024. The Company recognized stock-based
compensation expense related to all options granted and vesting expense of $59,964 during the three months ended March 31, 2023,
of which $24,429 is included in general and administrative expenses and $35,535 is included in research and development expenses in the
accompanying statements of operations.
Warrants
For the three months ended March 31, 2024, the
fair value of each warrant granted was estimated using the assumption and/or factors in the Black-Scholes Model as follows:
| Exercise price | |
$ | 5.23 | |
| Expected dividend yield | |
| 0 | % |
| Risk free interest rate | |
| 3.97 | % |
| Expected life in years | |
| 5.0 | |
| Expected volatility | |
| 219 | % |
The risk-free interest rate assumption for warrants
granted is based upon observed interest rates on the United States Government Bond Equivalent Yield appropriate for the expected term
of warrants.
The Company determined the expected volatility
assumption for warrants granted using the historical volatility of comparable public companies’ common stock. The Company will
continue to monitor peer companies and other relevant factors used to measure expected volatility for future warrant grants, until such
time that the Company’s common stock has enough market history to use historical volatility.
The dividend yield assumption for warrants granted
is based on the Company’s history and expectation of dividend payouts. The Company has never declared nor paid any cash dividends
on its common stock, and the Company does not anticipate paying any cash dividends in the foreseeable future.
The Company recognizes warrant forfeitures as
they occur, as there is insufficient historical data to accurately determine future forfeitures rates.
A summary of warrant issuances are as follows:
| Vested and Nonvested Warrants | |
Number | | |
Weighted
Average
Exercise
Price | | |
Weighted
Average
Remaining
Life | |
| Outstanding December 31, 2023 | |
| 5,047,450 | | |
$ | 14.11 | | |
| 2.73 | |
| Granted | |
| 50,000 | | |
| 5.23 | | |
| 4.77 | |
| Exercised | |
| - | | |
| - | | |
| - | |
| Expired or forfeited | |
| (400 | ) | |
| 400.00 | | |
| - | |
| Outstanding March 31, 2024 | |
| 5,097,050 | | |
$ | 13.63 | | |
| 2.56 | |
| Nonvested Warrants | |
Number | | |
Weighted-
Average
Exercise
Price | |
| Nonvested on December 31, 2023 | |
| - | | |
$ | - | |
| Granted | |
| 50,000 | | |
| 5.23 | |
| Vested | |
| (50,000 | ) | |
| 5.23 | |
| Forfeited | |
| - | | |
| - | |
| Nonvested on March 31, 2024 | |
| - | | |
$ | - | |
Restricted Stock Units
A summary of Restricted Stock Units (“RSUs”)
issuances are as follows:
| Nonvested RSUs | |
Number | | |
Weighted
Average
Price | |
| Nonvested December 31, 2023 | |
| - | | |
$ | - | |
| Granted | |
| - | | |
| - | |
| Vested | |
| - | | |
| - | |
| Forfeited | |
| - | | |
| - | |
| Nonvested March 31, 2024 | |
| - | | |
$ | - | |
The Company recognized stock-based compensation
expense related to RSUs granted and vesting expense of zero and $111,187 during the three months ended March 31, 2024 and March
31, 2023, respectively. Of the $111,187, $81,586 is included in general and administrative, $27,098 is included in research
and development, and $2,503 is included in sales and marketing in the accompanying Statements of Operations. The remaining value to be
expensed is $0 with a weighted average vesting term of 0 years as of March 31, 2024.
During the three months ended March 31, 2024,
the Company granted a total of zero RSUs. During the three months ended March 31, 2023, 44 RSUs vested and the Company
issued 44 shares of common stock for the 44 vested RSUs.
NOTE 11 – INCOME TAXES
The Company has incurred losses since inception.
During the three months ended March 31, 2023, the Company did not provide any provision for income taxes as the Company incurred losses
during such period. The Company accounts for income taxes using the asset and liability method in accordance with ASC 740, “Accounting
for Income Taxes”. The asset and liability method provides that deferred tax assets and liabilities are recognized for the expected
future tax consequences of temporary differences between the financial reporting and tax bases of assets and liabilities and for operating
loss and tax credit carry forwards. Deferred tax assets and liabilities are measured using the currently enacted tax rates and laws that
will be in effect when the differences are expected to reverse. In assessing the need for a valuation allowance, the Company has considered
both positive and negative evidence related to the likelihood of realization of deferred tax assets using a “more likely than not”
standard. In making such assessment, more weight was given to evidence that could be objectively verified, including recent cumulative
losses. Based on the Company’s review of this evidence, the Company has recorded a full valuation allowance for its net deferred
tax assets as of March 31, 2023.
As of March 31, 2023, the Company did not have
any amounts recorded pertaining to uncertain tax positions.
NOTE 12 – SUBSEQUENT EVENTS
The Company has evaluated subsequent events through
June 5, 2024, the issuance date of these consolidated financial statements.
Appili Arrangement Agreement
On April 1, 2024 (the “Execution Date”),
the Company, entered into an Arrangement Agreement (the “Arrangement Agreement”), subject to various closing conditions,
with Adivir, Inc., a Delaware corporation and wholly owned subsidiary of the Company (“Adivir” or the “Buyer”),
and Appili Therapeutics, Inc., a Canadian corporation (“Appili”), pursuant to which, Adivir will acquire all of the issued
and outstanding Class A common shares of Appili (the “Appili Shares”) on the terms and subject to the conditions set forth
therein. The acquisition of the Appili Shares (the “Arrangement”) will be completed by way of a statutory plan of arrangement
under the Canada Business Corporation Act.
At the effective time of the Arrangement (the
“Effective Time”), each Appili Share outstanding immediately prior to the Effective Time (other than Appili Shares held by
a registered holder of Appili Shares who has validly exercised such holder’s dissent rights) will be deemed to be assigned and
transferred by the holder thereof to the Buyer in exchange for (i) $0.0467 in cash consideration per share for an aggregate cash payment
of $5,668,222 (the “Cash Consideration”) and (ii) 0.002745004 of a share of common stock of Aditxt or an aggregate of 332,876
shares (the “Consideration Shares” and together with the Cash Consideration, the “Transaction Consideration”).
In connection with the transaction, each outstanding option and warrant of Appili will be cashed-out based on the implied in-the-money
value of the Transaction Consideration, which is expected to result in an additional aggregate cash payment of approximately $341,000
(based on the number of issued and outstanding options and warrants and exchange rates as of the date of the Arrangement Agreement).
Promissory Note
On April 10, 2024, Sixth Borough Capital Fund,
LP (“Sixth Borough) loaned $230,000 to the Company. The loan was evidenced by an unsecured promissory note (the “April Sixth
Borough Note”). Pursuant to the terms of the April Sixth Borough Note, it will accrue interest at the Prime rate of eight and one-half
percent (8.5%) per annum and is due on the earlier of April 19, 2024 or an event of default, as defined therein.
On May 9, 2024, at which point the balance of
the loan was $35,256, Sixth Borough loaned an additional $20,000 to the Company bringing the balance of the loan to $55,256.03. Additionally,
the maturity date of the April Sixth Borough Note was extended to June 9, 2024.
EvoFem Reinstatement and Fourth Amendment
to the Merger Agreement
On April 26, 2024, the Company received notice
from Evofem (the “Termination Notice”) that Evofem was exercising its right to terminate the Merger Agreement as a result
of the Company’s failure to provide the Initial Parent Equity Investment (as defined in the Merger Agreement, as amended).
On May 2, 2024, the Company, Adifem, Inc. f/k/a
Adicure, Inc. and Evofem Biosciences, Inc. (“Evofem”) entered into the Reinstatement and Fourth Amendment to the Merger Agreement
(the “Fourth Amendment”) in order to waive and amend, among other things, the several provisions listed below.
Amendments to Article VI: Covenants and Agreement
Article VI of the Merger Agreement is amended
to:
| ● | reinstate
the Merger Agreement, as amended by the Fourth Amendment, as if never terminated; |
| |
● |
reflect the Company’s
payment to Evofem, in the amount of $1,000,000 (the “Initial Payment”), via wire initiated by May 2, 2024; |
| |
● |
delete Section 6.3, which
effectively eliminates the “no shop” provision, and the several defined terms used therein; |
| |
● |
add a new defined term
“Company Change of Recommendation;” and |
| |
● |
revise section 6.10 of
the Merger Agreement such that, after the Initial Payment, and upon the closing of each subsequent capital raise by the Company (each
a “Parent Subsequent Capital Raise”), the Company shall purchase that number of shares of Evofem’s Series F-1 Preferred
Stock, par value $0.0001 per share (the “Series F-1 Preferred Stock”), equal to forty percent (40%) of the gross proceeds
of such Parent Subsequent Capital Raise divided by 1,000, up to a maximum aggregate amount of $2,500,000 or 2,500 shares of Series
F-1 Preferred Stock. A maximum of$1,500,000 shall be raised prior to June 17, 2024 and $1,000,000 prior to July 1, 2024 (the “Parent
Capital Raise”). |
Amendments to Article VIII: Termination
Article VIII of the Merger Agreement is amended
to:
| |
● |
extend the date after which
either party may terminate from May 8, 2024 to July 15, 2024; |
| |
● |
revise Section 8.1(d) in
its entirety to allow Company to terminate at any time after there has been a Company Change of Recommendation, provided that Aditxt
must receive ten day written notice and have the opportunity to negotiate a competing offer in good faith; and |
| |
● |
amend and restate Section
8.1(f) in its entirety, granting the Company the right to terminate the agreement if (a) the full $1,000,000 Initial Payment required
by the Fourth Amendment has not been paid in full by May 3, 2024 (b) $1,500,000 of the Parent Capital Raise Amount has not been paid
to the Company by June 17, 2024, (c) $1,000,000 of the Parent Capital Raise Amount has not been paid to the Company by July 1, 2024,
or (d) Aditxt does not pay any portion of the Parent Equity Investment within five calendar days after each closing of a Parent Subsequent
Capital Raise. |
Equity Line of Credit
On May 2, 2024, the Company entered into a Common
Stock Purchase Agreement (the “ELOC Purchase Agreement”) with an equity line investor (the “ELOC Investor”),
pursuant to which the ELOC Investor has agreed to purchase from the Company, at the Company’s direction from time to time, in its
sole discretion, from and after the date effective date of the Registration Statement (as defined below) and until the termination of
the ELOC Purchase Agreement in accordance with the terms thereof, shares of the Company’s common stock having a total maximum aggregate
purchase price of $150,000,000 (the “ELOC Purchase Shares”), upon the terms and subject to the conditions and limitations
set forth in the ELOC Purchase Agreement.
In connection with the ELOC Purchase Agreement,
the Company also entered into a Registration Rights Agreement with the Investor (the “ELOC Registration Rights Agreement”),
pursuant to which the Company agreed to file a registration statement with the Securities and Exchange Commission covering the resale
of the shares of common stock issued to the ELOC Investor pursuant to the ELOC Purchase Agreement (the “Registration Statement”)
by the later of (i) the 30th calendar day following the closing date, and (ii) the second business day following Stockholder Approval
(defined below).
The Company may, from time to time and at its
sole discretion, direct the ELOC Investor to purchase shares of its common stock upon the satisfaction of certain conditions set forth
in the ELOC Purchase Agreement at a purchase price per share based on the market price of the Company’s common stock at the time
of sale as computed under the ELOC Purchase Agreement. There is no upper limit on the price per share that the ELOC Investor could be
obligated to pay for common stock under the ELOC Purchase Agreement. The Company will control the timing and amount of any sales of its
common stock to the ELOC Investor, and the ELOC Investor has no right to require us to sell any shares to it under the ELOC Purchase
Agreement. Actual sales of shares of common stock to the ELOC Investor under the ELOC Purchase Agreement will depend on a variety of
factors to be determined by the Company from time to time, including (among others) market conditions, the trading price of its common
stock and determinations by the Company as to available and appropriate sources of funding for the Company and its operations. The ELOC
Investor may not assign or transfer its rights and obligations under the ELOC Purchase Agreement.
Under the applicable Nasdaq rules, in no event
may the Company issue to the ELOC Investor under the ELOC Purchase Agreement more than 332,876 shares of common stock, which number of
shares is equal to 19.99% of the shares of the common stock outstanding immediately prior to the execution of the ELOC Purchase Agreement
(the “Exchange Cap”), unless (i) the Company obtains stockholder approval to issue shares of common stock in excess of the
Exchange Cap in accordance with applicable Nasdaq rules (“Stockholder Approval”), or (ii) the average price per share paid
by the Investor for all of the shares of common stock that the Company directs the ELOC Investor to purchase from the Company pursuant
to the ELOC Purchase Agreement, if any, equals or exceeds the official closing sale price on the Nasdaq Capital Market immediately preceding
the delivery of the applicable purchase notice to the Investor and (B) the average of the closing sale prices of the Company’s
common stock on the Nasdaq Capital market for the five business days immediately preceding the delivery of such purchase notice.
In all cases, the Company may not issue or sell
any shares of common stock to the ELOC Investor under the ELOC Purchase Agreement which, when aggregated with all other shares of the
Company’s common stock then beneficially owned by the ELOC Investor and its affiliates, would result in the ELOC Investor beneficially
owning more than 4.99% of the outstanding shares of the Company’s common stock.
The net proceeds under the ELOC Purchase Agreement
to the Company will depend on the frequency and prices at which the Company sells shares of its stock to the ELOC Investor. The Company
expects that any proceeds received by it from such sales to the Investor will be used for working capital and general corporate purposes.
As consideration for the ELOC Investor’s
commitment to purchase shares of common stock at the Company’s direction upon the terms and subject to the conditions set forth
in the ELOC Purchase Agreement, the Company shall pay the Investor a commitment fee as outlined in the ELOC Purchase Agreement, which
is payable on the later of (i) January 2, 2025 and (ii) the trading day following the date on which Stockholder Approval is obtained.
The ELOC Purchase Agreement contains customary
representations, warranties and agreements of the Company and the ELOC Investor, limitations and conditions regarding sales of ELOC Purchase
Shares, indemnification rights and other obligations of the parties.
There are no restrictions on future financings,
rights of first refusal, participation rights, penalties or liquidated damages in the ELOC Purchase Agreement other than a prohibition
(with certain limited exceptions) on entering into a dilutive securities transaction during certain periods when the Company is selling
common stock to the ELOC Investor under the Purchase Agreement. The ELOC Investor has agreed that it will not engage in or effect, directly
or indirectly, for its own account or for the account of any of its affiliates, any short sales of the Company’s common stock or
hedging transaction that establishes a net short position in the Company’s common stock during the term of the ELOC Purchase Agreement.
The Company has the right to terminate the ELOC
Purchase Agreement at any time after the Commencement Date (as defined in the ELOC Purchase Agreement), at no cost or penalty, upon three
trading days’ prior written notice to the Investor. The Company and the ELOC Investor may also agree to terminate the ELOC Purchase
Agreement by mutual written consent, provided that no termination of the ELOC Purchase Agreement will be effective during the pendency
of any purchase that has not then fully settled in accordance with the ELOC Purchase Agreement. Neither the Company nor the ELOC Investor
may assign or transfer the Company’s respective rights and obligations under the ELOC Purchase Agreement.
May Private Placement
On May 2, 2024, the Company entered into a Securities
Purchase Agreement (the “May PIPE Purchase Agreement”) with certain accredited investors, pursuant to which the Company agreed
to issue and sell to such investors in a private placement (the “Private Placement”) (i) an aggregate of 4,186 shares of
the Company’s Series C-1 Convertible Preferred Stock (the “Series C-1 Preferred Stock”), (ii) an aggregate of 4,186
shares of the Company’s Series D-1 Preferred Stock (the “Series D-1 Preferred Stock”), and (iii) warrants (the “May
PIPE Warrants”) to purchase up to an aggregate of 1,613,092 shares of the Company’s common stock.
The May PIPE Warrants are exercisable commencing
six months following the initial issuance date at an initial exercise price of $2.47 per share and expire five years from the date of
issuance.
On May 2, 2024, in connection with the Purchase
Agreement, the Company entered into a Registration Rights Agreement with the investors (the “May PIPE Registration Rights Agreement”),
pursuant to which the Company agreed to prepare and file with the Securities and Exchange Commission (the “SEC”) a registration
statement on Form S-3 (the “May PIPE Registration Statement”) covering the resale of the shares of the Company’s common
stock, par value $0.001 (the “Common Stock”) issuable upon conversion of the Series C-1 Preferred Stock (the “Conversion
Shares”) and upon exercise of the May PIPE Warrants (the “May PIPE Warrant Shares”) (i) on the later of (x) the 30th
calendar day after the closing date, or (y) the 2nd business day following the Stockholder Approval Date (as defined in the May PIPE
Purchase Agreement), with respect to the initial registration statement and (ii) on the date on which the Company is required to file
any additional May PIPE Registration Statement pursuant to the terms of the May PIPE Registration Rights Agreement with respect to any
additional Registration Statements that may be required to be filed by the Company (the “Filing Deadline”). Pursuant to the
Registration Rights Agreement, the Company is required to have the initial May PIPE Registration Statement declared effective by the
SEC on the earlier of (x) the 60th calendar day after the Filing Deadline (or the 90th calendar day after the Filing Deadline if subject
to a full review by the SEC), and (y) the 2nd business day after the date the Company is notified by the SEC that such May PIPE Registration
Statement will not be reviewed. In the event that the Company fails to file the May PIPE Registration Statement by the Filing Deadline,
have it declared effective by the Effectiveness Deadline, or the prospectus contained therein is not available for use or the investor
is not otherwise able to sell its May PIPE Warrant Shares pursuant to Rule 144, the Company shall be required to pay the investor an
amount equal to 2% of such investor’s Purchase Price (as defined in the May PIPE Purchase Agreement) on the date of such failure
and on every thirty date anniversary until such failure is cured.
In connection with the Private Placement, the
Sixth Borough Note (Note 7) was converted into Series C-1 Preferred Stock.
The Private Placement closed on May 6, 2024.
The gross proceeds from the Private Placement were approximately $4.2 million, prior to deducting the placement agent’s fees and
other offering expenses payable by the Company. The Company intends to use $1.0 million of the net proceeds to fund certain obligations
under its merger agreement with Evofem Biosciences, Inc. and the remainder of the net proceeds from the offering for working capital
and other general corporate purposes.
Dawson James Securities (“Dawson James”)
served as the Company’s exclusive placement agent in connection with the Private Placement, pursuant to that certain engagement
letter, dated as of May 2, 2024, between the Company and Dawson James (the “Engagement Letter”). Pursuant to the Engagement
Letter, the Company paid Dawson James (i) a total cash fee equal to 7% of the aggregate gross proceeds of the Private Placement. In addition,
the Company agreed to pay Dawson James certain expenses and issued to Dawson James or its designees warrants (the “May PIPE Placement
Agent Warrants”) to purchase 5% of the number of securities sold in the Private Placement. The May PIPE Placement Agent Warrants
are exercisable at an exercise price of $3.24375 per share commencing six months following issuance and have a term of exercise equal
to five years from the date of issuance.
Series C-1 Preferred Stock Certificate of
Designation
On May 2, 2024, the Company filed a Certificate
of Designation for its Series C-1 Preferred Stock with the Secretary of State of Delaware (the “Series C-1 Certificate of Designations”).
The following is only a summary of the Series C-1 Certificate of Designations, and is qualified in its entirety by reference to the full
text of the Series C-1 Certificate of Designations.
Designation, Amount, and Par Value. The number
of Series C-1 Preferred Stock designated is 10,853 shares. The shares of Series C-1 Preferred Stock have a par value of $0.001 per share
and a stated value of $1,000 per share.
Conversion Price: The Series C-1 Preferred Stock
will be convertible into shares of Common Stock at an initial conversion price of $2.595 (subject to adjustment pursuant to the Series
C-1 Certificate of Designations) (the “Series C-1 Conversion Price”). The Series C-1 Certificate of Designations also provides
that in the event of certain Triggering Events (as defined below) any holder may, at any time, convert any or all of such holder’s
Series C-1 Preferred Stock at an alternate conversion rate equal to the product of (i) the Alternate Conversion Price (as defined below)
and (ii) the quotient of (x) the 25% redemption premium multiplied by (y) the amount of Series C-1 Preferred Stock subject to such conversion.
“Triggering Events” include, among others, (i) a suspension of trading or the failure to be traded or listed on an eligible
market for five consecutive days or more, (ii) the failure to remove restrictive legends when required, (iii) the Company’s default
in payment of indebtedness in an aggregate amount of $500,000 or more, (iv) proceedings for a bankruptcy, insolvency, reorganization
or liquidation, which are not dismissed with 30 days, (v) commencement of a voluntary bankruptcy proceeding, and (viii) final judgments
against the Company for the payment of money in excess of $500,000. “Alternate Conversion Price” means the lowest of (i)
the applicable conversion price the in effect, (ii) the greater of (x) $0.519 (the “Floor Price”) and (y) 80% of the volume
weighted average price (“VWAP”) of the Common Stock on the trading day immediately preceding the delivery of the applicable
conversion notice. Further, the Series C-1 Certificate of Designations provides that if on any of the 90th and 180th day after each of
the occurrence of any Stock Combination Event (as defined in the Series C-1 Certificate of Designations) and the Applicable Date (as
defined in the Series C-1 Certificate of Designations), the conversion price then in effect is greater than the market price then in
effect (the “Adjustment Price”), on such date then the conversion price shall automatically lower to the Adjustment Price.
Dividends: Holders of the Series C-1 Preferred
Stock shall be entitled to receive dividends when and as declared by the Board, from time to time, in its sole discretion, which Dividends
shall be paid by the Company out of funds legally available therefor, payable, subject to the conditions and other terms hereof, in cash,
in securities of the Company or any other entity, or using assets as determined by the Board on the Stated Value of such Preferred Share.
Liquidation: In the event of a Liquidation Event
(as defined in the Series C-1 Certificate of Designation), the holders the Series C-1 Preferred Stock shall be entitled to receive in
cash out of the assets of the Company, before any amount shall be paid to the holders of any other shares of capital stock of the Company,
equal to the greater of (A) 125% of the Conversion Amount (as defined in the Series C-1 Certificate of Designation) on the date of such
payment and (B) the amount per share such holder of Series C-1 Preferred Stock would receive if they converted such share of Series C-1
Preferred Stock into Common Stock immediately prior to the date of such payment
Company Redemption: The Company may redeem all,
or any portion, of the Series C-1 Preferred Stock for cash, at a price per share of Series C-1 Preferred Stock equal to 115% of the greater
of (i) the Conversion Amount (as defined in the Series C-1 Certificate of Designations) being redeemed as of the Company Optional Redemption
Date (as defined in the Series C-1 Certificate of Designation) and (ii) the product of (1) the Conversion Rate (as defined in the Series
C-1 Certificate of Designation) with respect to the Conversion Amount being redeemed as of the Company Optional Redemption Date multiplied
by (2) the greatest Closing Sale Price (as defined in the Certificate of Designation) of the Common Stock on any Trading Day during the
period commencing on the date immediately preceding such Company Optional Redemption Notice Date (as defined in the Certificate of Designation)
and ending on the Trading Day immediately prior to the date the Company makes the entire payment required to be made under the Certification
of Designation.
Maximum Percentage: Holders of Series C-1 Preferred
Stock are prohibited from converting shares of Series C-1 Preferred Stock into shares of Common Stock if, as a result of such conversion,
such holder, together with its affiliates, would beneficially own in excess of 4.99% (the “Maximum Percentage”) of the total
number of shares of Common Stock issued and outstanding immediately after giving effect to such conversion.
Voting Rights. The holders of the Series C-1
Preferred Stock shall have no voting power and no right to vote on any matter at any time, either as a separate series or class or together
with any other series or class of share of capital stock, and shall not be entitled to call a meeting of such holders for any purpose
nor shall they be entitled to participate in any meeting of the holders of Common Stock, except as expressly provided in the Certificate
of Designations and where required by the General Corporation Law of the State of Delaware (the “DGCL”).
Series D-1 Preferred Stock Certificate of
Designation
On May 2, 2024, the Company filed a Certificate
of Designation for its Series D-1 Preferred Stock with the Secretary of State of Delaware (the “Series D-1 Certificate of Designations”).
The following is only a summary of the Series D-1 Certificate of Designations, and is qualified in its entirety by reference to the full
text of the Series D-1 Certificate of Designations, a copy of which is filed as Exhibit 3.1 to this Current Report on Form 8-K and is
incorporated by reference herein.
The Series D-1 Certificate of Designations provides
that the share of Preferred Stock will have 418,600,000 votes and will vote together with the outstanding shares of the Company’s
Common Stock as a single class exclusively with respect to any proposal to amend the Company’s Amended and Restated Certificate
of Incorporation to increase the number of shares of Common Stock that the Company is authorized to issue. The Series D-1Preferred Stock
will be voted, without action by the holder, on any such proposal in the same proportion as shares of the Company’s Common Stock
are voted. The Series D-1 Preferred Stock otherwise has no voting rights except as otherwise required by the DGCL.
The Series D-1 Preferred Stock is not convertible
into, or exchangeable for, shares of any other class or series of stock or other securities of the Company. The Series D-1 Preferred
Stock has no rights with respect to any distribution of assets of the Company, including upon a liquidation, bankruptcy, reorganization,
merger, acquisition, sale, dissolution or winding up of the Company, whether voluntarily or involuntarily. The holders of Series D-1
Preferred Stock will not be entitled to receive dividends of any kind.
The outstanding share of Series D-1 Preferred
Stock shall be redeemed in whole, but not in part, at any time (i) if such redemption is ordered by the Board of Directors in its sole
discretion or (ii) automatically upon the effectiveness of the amendment to increase the number of shares of Common Stock that the Company
is authorized to issue. Upon such redemption, the holder of the Preferred Stock will receive consideration of $0.01 per share in cash.
LS Biotech Eight Default
On May 10, 2024, the Company received written
notice (the “2024 Default Notice”) from LS Biotech Eight, LLC (the “Landlord”) that the Company was in violation
of its obligation to (i) pay Base Rent (as defined in the Lease) and Additional Rent (as defined in the Lease) in the amount of $431,182.31
in the aggregate, together with administrative charges and interest, as well as (ii) replenish the Security Deposit (as defined in the
Lease) in the amount of $159,375.00, all as required under that certain Lease Agreement dated as of May 4, 2021 by and between the Landlord
and the Company (the “Lease”). Pursuant to the Notice, the Landlord has demanded that a payment of $590,557.31 plus administrative
charges and interest, which shall accrue at the Default Rate (as defined in the Lease) be made no later than May 17, 2024.
The Company is working with the Landlord to come
to an amicable resolution. However, no assurance can be given that the parties will reach an amicable resolution on a timely basis, on
favorable terms, or at all.
May Senior Notes
On
May 20, 2024, we issued and sold a senior note (the “First May Senior Note”)
to an accredited investor (the “May Holder”) in the original principal
amount of $93,918.75 for a purchase price of $75,135.00, reflecting an original issue discount of $18,783.75. Unless earlier redeemed,
the First May Senior Note will mature on August 18, 2024 (the “First May Senior Note Maturity Date”),
subject to extension at the option of the May Holder in certain circumstances as provided in the First May Senior Note. The First May
Senior Note bears interest at a rate of 8.5% per annum, which is compounded each calendar month and is payable in arrears on the First
May Senior Note Maturity Date. The Rirst May Senior Note contains certain standard events of default, as defined in the First May Senior
Note (each, an “First May Senior Note Event of Default”). Upon the occurrence
of an First May Senior Note Event of Default, the interest rate shall be increased to 18% per annum and the May Holder may require the
Company to redeem the First May Senior Note, subject to an additional 5% redemption premium. In addition, if we sell any shares of our
common stock pursuant to any equity line of credit, we are required to redeem in cash a portion of the First May Senior Note equal to
the lesser of (i) the outstanding amount of the First May Senior Note, and (ii) 80% of 30% of such equity line proceeds, at a redemption
price calculated based upon $1.20 for each $1.00 of outstanding amount of the First May Senior Note. The First May Senior Note also contains
an exchange right, which permits the May Holder, in its discretion, to exchange the First May Senior Note, in whole or in part, for securities
to be sold by us in a subsequent placement, subject to certain exceptions and an additional 20% premium of the amount of the First May
Senior Note exchanged. The First May Senior Note is a senior, unsecured obligation of the Company, ranking senior to all other unsecured
indebtedness of the Company.
On May 24, 2024, we
entered into a Securities Purchase Agreement (the “May Securities Purchase Agreement”) with certain accredited investors
pursuant to which we issued and sold senior notes in the aggregate principal amount of $986,379.68 (the “Second May Senior Notes”)
maturing on August 22, 2024, which included the exchange of a previously issued First May Senior Note in the principal amount of $93,918.75.
The Company received cash proceeds of $775,000 from the sale of the Second May Senior Notes.
Upon an Event of Default
(as defined in the Second May Senior Note), the Second May Senior Note will bear interest at a rate of 14% per annum and the holder shall
have the right to require the Company to redeem the Second May Senior Note at a redemption premium of 125%. In addition, while the Second
May Senior Note is outstanding, the Company is required to utilize 100% of the proceeds from any offering of securities to redeem the
Second May Senior Note. Pursuant to the Purchase Agreement, the Company agreed to use commercially reasonable efforts, including the
filing of a registration statement with the U.S. Securities and Exchange Commission (the “SEC”) for a public offering,
to pursue and consummate a financing transaction within 90 days of the closing date. In connection with the issuance of the Second May
Senior Notes, the Company issued an aggregate of 328,468 shares of its common stock (the “Commitment Shares”) as a
commitment fee to the investors. Pursuant to the Purchase Agreement, the Company also agreed to file a registration statement with the
SEC covering the resale of the Commitment Shares as soon as practicable following notice from an investor, and to cause such registration
statement to become effective within 60 days following the filing thereof.
Aditxt, Inc.
3,785,569 Shares of Common Stock
PRELIMINARY PROSPECTUS
June 13, 2024
Aditxt (NASDAQ:ADTX)
過去 株価チャート
から 5 2024 まで 6 2024
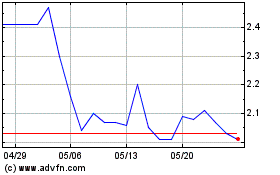
Aditxt (NASDAQ:ADTX)
過去 株価チャート
から 6 2023 まで 6 2024