- Both drugs demonstrated > 50% response for treating
depression. NRX-101 demonstrated a mean 76% reduction in symptoms
of akathisia compared to lurasidone that was sustained over 42
days (Effect Size .37; P=0.025), using prespecified analytic
methodology memorialized in FDA Special Protocol Agreement. Levels
of akathisia with NRX-101 were essentially zero at day 42
- This safety advantage was previously reported in the Company's
published STABIL-B trial
- Akathisia is identified as a life-threatening side effect of
nearly all antidepressants, reported in 10-15% of treated patients
and is closely linked to suicide in FDA black box warning
- Akathisia was seen in 2% of participants treated with NRX-101
vs. 11% treated with lurasidone
- Company plans to seek accelerated approval of NRX-101 for use
in patients with bipolar depression at risk of akathisia while
continuing to broaden the indication to all patients with bipolar
depression and perhaps schizophrenia
- Study will be presented at the American Society of Clinical
Psychopharmacology (ASCP) meeting May 28-31,
2024 (Miami) together with
study investigators, accompanied by a broadcast scientific
presentation on akathisia and antidepressant safety, and investor
Q&A
RADNOR,
Pa., May 6, 2024 /PRNewswire/ -- NRx
Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the
"Company"), a clinical stage pharmaceutical company, today
announced a statistically significant safety advantage of NRX-101
compared to the standard of care comparator in its recently
completed clinical trial in patients with suicidal bipolar
depression. Therefore, the Company believes that demonstration of
reduced akathisia in the setting of comparable antidepressant
efficacy constitutes a basis for Accelerated FDA Approval of
NRX-101. The full clinical trial results will be presented at the
upcoming meeting to the American Society of Clinical
Psychopharmacology held May 28-31,
2024 in Miami. NRx will
gather Key Opinion Leaders to educate the public on the importance
and potentially life-saving implication of this finding.
Last week, the Company released preliminary top-line data as
required by SEC disclosure rules. The Company believes that today's
findings based on mixed model regression analysis as specified in
the Company's Special Protocol Agreement with the FDA, when
combined with the prior STABIL-B trial1, demonstrate a
basis for seeking accelerated drug approval of NRX-101 based on
improved safety related to akathisia and suicidality in the setting
of comparable antidepressant efficacy.
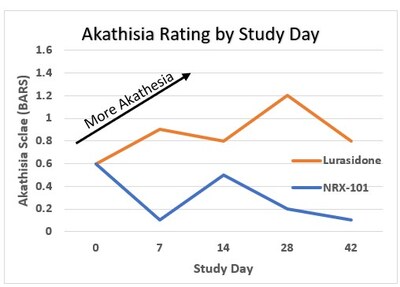
Trial participants had identical mean scores on the Barnes
Akathisia Rating Scale (BARS) at baseline with subsequent decrease
in the NRX-101 treated group versus an increase in the
lurasidone-treated group, yielding a 76% relative mean difference
between the groups. The difference was apparent at the first
post-randomization visit and continued throughout the trial. (Fig
1) Over the 42 days of observation, an effect size of .37 was
identified with a statistically significant P value of 0.025 on the
Mixed Model for Repeated Measures (MMRM) methodology agreed to with
FDA in the 2018 Special Protocol Agreement. Akathisia as
ascertained by a 1 point increase in the BARS was seen in 11% of
participants randomized to lurasidone (comparable to previous
reports in the literature) and seen in only 2% of those treated
with NRX-101, an akathisia level that was previously reported for
the placebo arm of the lurasidone registration trial.
Akathisia was a prespecified key safety endpoint of the
Company's clinical trial. Hence this finding is not a "post-hoc"
observation. As previously noted, this clinical trial of 91
participants with suicidal bipolar depression who were not
pre-treated with ketamine demonstrated that NRX-101 and lurasidone
were comparable in their antidepressant effect. A 33% but
statistically non-significant sustained decrease in suicidality was
also seen favoring NRX-101. As noted above, improved antidepressant
efficacy is not required to seek drug accelerated drug approval
based on a statistically-significant safety benefit.
Based on this safety finding, NRx plans to seek Accelerated
Approval of NRX-101 for treatment of bipolar depression in patients
at risk for akathisia who are at highest risk of suicide, while
continuing to develop evidence to support broader indications both
in treatment of depression and schizophrenia. Should these data be
confirmed in additional large scale trials, the Company believes
that physicians and patients will universally prefer antidepressant
and antipsychotic drugs with a reduced akathisia risk. The NRx
patent portfolio supports the development of a broad range of
combined NMDA/serotonergic drugs for treatment of depression and
psychosis.
There is a recent regulatory precedent for the approval of
psychiatry drugs that demonstrate comparable efficacy with improved
safety. A combination of olanzapine and samidorphan (LYBALVI®) was
approved based on comparable effect on schizophrenia symptoms with
evidence of less weight gain favoring LYBALVI. Thus, public
assertions by journalists and short-sellers that NRx has no path to
market based on the finding of comparable efficacy in this trial
are utterly baseless and may be designed to mislead investors.
"More than 7 million Americans suffer from bipolar depression
with a potential market opportunity in excess of $20 billion. No prior drug to treat bipolar
depression has demonstrated superiority on side effects most
closely linked to suicide. Patients with bipolar depression
have a 50% lifetime risk of a suicidal attempt and a 20% lifetime
risk of dying from suicide. On this basis of these superiority
findings, previously seen in the STABIL-B trial, we plan to seek
Accelerated Approval from FDA for treatment of patients with
bipolar depression who are at risk for akathisia, as we and our
partners continue to broaden the indication to the treatment of all
patients with bipolar depression." said Dr. Jonathan Javitt, NRx's Chairman and Chief
Scientist. "Patients and key opinion leaders alike have told us
clearly that an antidepressant with comparable antidepressant
effect and reduced risk of akathisia and other risk factors for
suicidality would be unambiguously preferred in the
marketplace."
Background on Akathisia
Akathisia is a
known extrapyramidal side effect of all
serotonin-targeted antidepressants and antipsychotic
drugs. It can be demonstrated in laboratory models, using
rodent behavioral data.2 Prof.
Daniel Javitt, the inventor of NRX-101 first
discovered the ability of NMDA antagonist drugs to
reduce akathisia in 2009, which forms the basis of the
Company's composition of matter patent portfolio. In his 2012
patent application Javitt stated
3
A major limitation in use of antipsychotic
and antidepressant medications is the liability to produce
behavioral side effects, especially anxiety, agitation, and
akathisia, all of which are associated with generating or
exacerbating suicidality in psychotic or depressed patients. These
behavioral side effects can be differentiated from symptoms of the
illness by consideration of both time course and specific patterns
of symptoms.
The side effect is clearly recognized in an FDA-mandated black
box warning applied to lurasidone and all medications in its class
of drugs, with a specific warning about suicidality as follows:
"The following symptoms, anxiety, agitation,
panic attacks, insomnia, irritability, hostility, aggressiveness,
impulsivity, akathisia (psychomotor restlessness), hypomania, and
mania have been reported in adult and pediatric patients being
treated with antidepressants for major depressive disorder as well
as for other indications, both psychiatric and non-psychiatric.
Although a causal link between the emergence of such symptoms and
either the worsening of depression and/or the emergence of suicidal
impulses has not been established, there is concern that such
symptoms may represent precursors to emerging
suicidality"4.
In most clinical trials, safety endpoints are not a basis for
drug approval, although adverse safety findings may prevent drug
approval. Typically, new drugs are compared to placebo in order to
prove a difference in efficacy. In this case, the trial was
conducted against the standard of care drug, not against placebo
and, therefore, safety differences are highly important. Akathisia
is a labeled negative side-effect of lurasidone and all similar
drugs, and is closely linked to suicidal
behavior.56 It is a side effect about which
patients must be warned[7] and has figured prominently in class
action lawsuits brought against manufacturers of serotonin-targeted
antidepressants and antipsychotics. Currently, there is no
FDA-approved treatment for akathisia and it is considered a medical
emergency by many treating psychiatrists. Akathisia causes extreme
anxiety to patients, inability to control motor movement, and
impulsive acts, all to often leading to suicide and may be the key
driver of the observed increase in suicidal ideation and behavior
associated with all serotonin-targeted antidepressants and
antipsychotic drugs.
"These studies support our original observation that
D-cycloserine reverses the akathisia-like behaviors induced by
lurasidone in rodent models. It is exciting to see those findings
demonstrated in a second trial with unequivocal statistical
significance," said Prof. Daniel
Javitt, co-founder of NRx Pharmaceuticals. "Hopefully, this
opens a path to bringing a lifesaving drug to millions of patients
with suicidal depression whose only approved long-term treatment is
electroconvulsive therapy."
About NRx Pharmaceuticals
NRx Pharmaceuticals is a clinical-stage biopharmaceutical
company developing therapeutics based on its NMDA platform for the
treatment of central nervous system disorders, specifically
suicidal bipolar depression, chronic pain and PTSD. The Company is
developing NRX-101, an FDA-designated investigational Breakthrough
Therapy for suicidal treatment-resistant bipolar depression and
chronic pain. NRx has partnered with Alvogen and Lotus around the
development and marketing of NRX-101 for the treatment of suicidal
bipolar depression. NRX-101 additionally has potential to act as a
non-opioid treatment for chronic pain, as well as a treatment for
complicated UTI.
NRx has recently announced plans to submit a New Drug
Application for HTX-100 (IV ketamine), through Hope Therapeutics,
in the treatment of suicidal depression, based on results of
well-controlled clinical trials conducted under the auspices of the
US National Institutes of Health and newly obtained data from
French health authorities, licensed under a data sharing agreement.
NRx was awarded Fast Track Designation for development of ketamine
(NRX-100) by the US FDA as part of a protocol to treat patients
with acute suicidality.
About HOPE Therapeutics, Inc.
HOPE Therapeutics, Inc. (www.hopetherapeutics.com) is a
Specialty Pharmaceutical Company, wholly-owned by NRX
Pharmaceuticals focused on development and marketing of an
FDA-approved form of intravenous ketamine for the treatment of
acute suicidality and depression together with a digital
therapeutic-enabled platform designed to augment and preserve the
clinical benefit of NMDA-targeted drug therapy.
Notice Regarding Forward-Looking Statements
The information contained herein includes forward-looking
statements within the meaning of Section 21E of the Securities
Exchange Act of 1934, as amended, and Section 27A of the Securities
Act of 1933, as amended. These statements include, among others,
statements regarding the proposed public offering and the timing
and the use of the proceeds from the offering. Forward-looking
statements generally include statements that are predictive in
nature and depend upon or refer to future events or conditions, and
include words such as "may," "will," "should," "would," "expect,"
"plan," "believe," "intend," "look forward," and other similar
expressions among others. These statements relate to future events
or to the Company's future financial performance, and involve known
and unknown risks, uncertainties and other factors that may cause
the Company's actual results to be materially different from any
future results, levels of activity, performance or achievements
expressed or implied by these forward-looking statements. You
should not place undue reliance on forward-looking statements since
they involve known and unknown risks, uncertainties and other
factors which are, in some cases, beyond the Company's control and
which could, and likely will, materially affect actual results,
levels of activity, performance or achievements. Any
forward-looking statement reflects the Company's current views with
respect to future events and is subject to these and other risks,
uncertainties and assumptions relating to the Company's operations,
results of operations, growth strategy and liquidity. More detailed
information about the Company and the risk factors that may affect
the realization of forward-looking statements is set forth in the
Company's most recent Annual Report on Form 10-K and other filings
with the Securities and Exchange Commission. Investors and security
holders are urged to read these documents free of charge on the
SEC's website at http://www.sec.gov. Except as may be required
by applicable law, The Company assumes no obligation to publicly
update or revise these forward-looking statements for any reason,
or to update the reasons actual results could differ materially
from those anticipated in these forward-looking statements, whether
as a result of new information, future events or otherwise.
|
1
Nierenberg A, Lavin P, Javitt DC, et. al. NRX-101 vs lurasidone for
the maintenance of initial stabilization after ketamine in patients
with severe bipolar depression with acute suicidal ideation and
behavior; a randomized prospective phase 2 trial. Int J Bipolar Dis
2023;11:28-38, doi.org/10.1186/s40345-023-00308-5
|
|
2
Walf, A. A., & Frye, C. A. (2007). The use of the elevated plus
maze as an assay of anxiety-related behavior in rodents. Nature
Protocols, 2(2), 322-328.
https://doi.org/10.1038/nprot.2007.44
|
|
3
Javitt DC. Composition and method for treatment of depression and
psychosis in humans. US Patent 10583138B2. Granted March 10,
2020.
|
|
4
Trivedi et al., J Clin Psychiatry, 72:765-774, 2011
|
|
5
Chow LC, Kahouh NK, Bostwick JR, et. al., Akathisia and newer
second-generation antipsychotic drugs: A review of current
evidence. Pharmacotherapy 202;40(6):565-574 doi:
10.1002/phar.2404
|
|
6
Uwai Y, Nabekura T. Risk factors for suicidal behavior/ideation and
hostility/aggression in patients with bipolar disorders: An
analysis using the Japanese adverse drug event report database. J
Psychiatric Res 2022;153:99-103.
https://doi.org/10.1016/j.jpsychires.2022.07.005
|
|
7
https://www.webmd.com/schizophrenia/side-effects-of-lurasidon
|

 View original content to download
multimedia:https://www.prnewswire.com/news-releases/nrx-pharmaceuticals-nasdaqnrxp-announces-final-clinical-trial-results-superior-safety-combined-with-similar-efficacy-in-the-trial-of-nrx-101-compared-to-lurasidone-in-suicidal-bipolar-depression-302136430.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/nrx-pharmaceuticals-nasdaqnrxp-announces-final-clinical-trial-results-superior-safety-combined-with-similar-efficacy-in-the-trial-of-nrx-101-compared-to-lurasidone-in-suicidal-bipolar-depression-302136430.html
SOURCE NRx Pharmaceuticals, Inc.