- NRX-101 is first oral antidepressant to show 33% advantage in
sustained remission in suicidality (not statistically
significant at this sample size) and 75% advantage in relief from
Akathisia relative to lurasidone - never previously shown with
an oral antidepressant. Suicidality signal met the study's
promising zone criteria and the akathisia signal approached
statistical significance (P=0.076)
- Both NRX-101 and lurasidone, an accepted standard of
care in Bipolar Depression, demonstrated approximately 50%
reduction in symptoms of depression
- These data are comparable to previous statistically-significant
finding of reduced suicidality and in the published STABIL-B
trial and support an approval pathway via a 300-person
registrational trial with sustained remission in suicidality as the
primary endpoint
- Company believes that an oral antidepressant that demonstrates
reduction in suicidality has potential to become standard of
care for treatment of bipolar depression.
- Data from this study expand the potential utility of NRx-101 to
treat both patients with suicidal bipolar depression (who will
require prior use of ketamine) and those without subacute
suicidality (nearly 7 million patients in the US).
RADNOR,
Pa., April 30, 2024 /PRNewswire/ -- NRx
Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the
"Company"), a clinical stage pharmaceutical company today that its
Breakthrough Therapy designated investigational drug NRX-101 vs
lurasidone demonstrated a promising, though not yet statistically
significant 33% reduction in suicidality together with a 70%
reduction (P=.076) reduction in symptoms of akathisia – a side
effect of antidepressants that is closely linked to suicide and
considered a medical emergency. Because of the high-risk
nature of these patients, a placebo group could not be employed,
and NRX-101, a fixed dose combination of D-cycloserine (DCS) and
lurasidone, was compared to lurasidone alone (the standard of
care). In the Company's previously published STABIL-B trial
(STABIL-B), NRX-101 was demonstrated to be superior to lurasidone
in reducing both depression and suicidality after ketamine while
showing a trend towards reducing akathisia (a side effect involving
restlessness and agitation that is considered a warning sign of
impending suicide). In this trial, without prior use of ketamine,
NRX-101 and lurasidone were comparable in their effect on
depression. The trial was a randomized, prospective, double-blind
study conducted at multiple sites in the Unites States whose
protocol and statistical analysis plan may be viewed on
www.clinicaltrials.gov (NCT03395392).
"We are gratified by these results, which extend the findings of
the STABIL-B trial in suggesting that NRX-101 has the potential to
be the first oral antidepressant to decrease potential for suicide,
whereas all currently approved oral antidepressants are known to
increase the risk of suicide," said Prof. Jonathan Javitt, MD, MPH, the Company's Chairman
and Chief Scientist. "Should these findings be confirmed in a
registrational trial of 300 patients, NRX-101 has the potential to
represent a paradigm-changing blockbuster drug. The finding of a
dramatic difference in akathisia was also seen in the STABIL-B
trial and provides important mechanistic support for the difference
seen on the Columbia Suicide Severity Rating Scale. Many of the
patients who tragically die from suicide in bipolar depression are
taking traditional antidepressants at the time of their death, a
tragedy we have seen within the families of our investors and board
members, as well as the many patients we have known. If today's
findings are replicated in a registration-sized trial, we will
change the world for patients who currently have a 50% lifetime
risk of suicide attempt, a 20% lifetime risk of death by suicide,
and whose only approved treatment option today is electroshock
therapy."
"These findings are consistent with our original Phase 2
objectives and promising zone methodology in terms of a
demonstrable advantage of NRX-101 compared to the standard of care
in treating patients with bipolar depression who are known to be at
high risk of suicide. We originally proposed to test suicidality,
rather than depression as the primary endpoint for this trial and
took the advice of senior FDA leadership that demonstrating a
difference in suicidality might be too challenging. Today's
findings demonstrate that differences in suicidality and akathisia
can be demonstrated compared to best available antidepressant
therapy in a properly sized registration trial and that superiority
over placebo on the depression scale may readily be demonstrated in
a less acute patient population where it would be safe to do so,"
said Dr. Philip Lavin, the study's
Lead Methodologist. Dr. Lavin is one of the world's most widely
published statisticians who has led the approval/clearance of more
than 80 drugs, devices, and biologics.
In the current study, without prior use of ketamine, NRX-101 and
lurasidone exhibited comparable antidepressant effects, each
reducing depression (the primary endpoint) on the Montgomery Asberg
Depression Rating Scale (MADRS) by about 50% from baseline.
Lurasidone is known to reduce symptoms of depression by
approximately 4 points in multiple registration trials compared to
placebo.
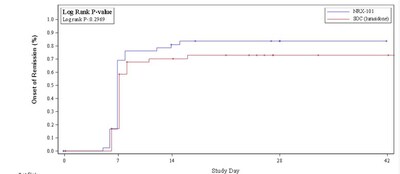
Analysis of suicidality using the Columbia Suicide Severity
Rating Scale (C-SSRS) demonstrated a sustained 33% advantage in
remission from suicidality favoring NRX-101 (see figure). This
difference was not statistically significant at the phase 2 sample
size but met the study's original promising zone criteria and, if
sustained in a registration trial of 300 or more patients, would be
powered to yield a statistically significant result. The reduction
in suicidality is comparable to that demonstrated after ketamine,
both in the Company's STABIL-B trial and in an independently
conducted trial comparing DCS to placebo after ketamine (Chen, et.
al.). A meaningful remission in suicidality has not been
demonstrated with any prior oral antidepressant drug – indeed,
antidepressant drugs carry a Black Box warning of increased suicide
risk.
Reduction in akathisia was first identified in the laboratory as
a distinguishing feature of DCS and is the basis of the approved
claims in the Company's Composition of Matter patents. Akathisia is
often characterized as a state of agitation and motor restlessness
that is associated with particularly impulsive and tragically
effective attempts at suicide, such as hanging, shooting, jumping
from buildings and in front of vehicles and trains. In this trial,
a 75% relative difference was seen on the Barnes Akathisia Rating
Scale (BARS), with two-sided P=0.076, which would be expected to
achieve significance in a properly powered registration-sized
trial. While reduction in akathisia is not proposed as a
primary labeled indication, continued finding of a statistically
significant reduction in this side effect would be highly
supportive of a demonstrated primary endpoint of reduced
suicidality and would provide clinical corroboration.
Based on these findings and widespread adoption of ketamine as
initial treatment for suicidal depression, the Company believes
that NRX-101 may become the drug of choice for potentiating the
effect of ketamine in patients with acute and subacute suicidality.
The FDA recently affirmed to the Company that the Special Protocol
Agreement for this indication remains in place, subject to the
Company filing a New Drug Approval for ketamine, which is expected
by July 2024. Moreover the Company
aims to explore the role of NRX-101 as primary treatment for the
much larger population (approximately 7 million in the US) of
patients with bipolar depression who do not have active suicidality
and, therefore, do not require prior treatment with intravenous
ketamine.
About NRx Pharmaceuticals
NRx Pharmaceuticals is a clinical-stage biopharmaceutical
company developing therapeutics based on its NMDA platform for the
treatment of central nervous system disorders, specifically
suicidal bipolar depression, chronic pain and PTSD. The Company is
developing NRX-101, an FDA-designated investigational Breakthrough
Therapy for suicidal treatment-resistant bipolar depression and
chronic pain. NRx has partnered with Alvogen and Lotus around the
development and marketing of NRX-101 for the treatment of suicidal
bipolar depression. NRX-101 additionally has potential to act as a
non-opioid treatment for chronic pain, as well as a treatment for
complicated UTI.
NRx has recently announced plans to submit a New Drug
Application for HTX-100 (IV ketamine), through Hope Therapeutics,
in the treatment of suicidal depression, based on results of
well-controlled clinical trials conducted under the auspices of the
US National Institutes of Health and newly obtained data from
French health authorities, licensed under a data sharing agreement.
NRx was awarded Fast Track Designation for development of ketamine
(NRX-100) by the US FDA as part of a protocol to treat patients
with acute suicidality.
About HOPE Therapeutics, Inc.
HOPE Therapeutics, Inc. (www.hopetherapeutics.com) is a
Specialty Pharmaceutical Company, wholly-owned by NRX
Pharmaceuticals focused on development and marketing of an
FDA-approved form of intravenous ketamine for the treatment of
acute suicidality and depression together with a digital
therapeutic-enabled platform designed to augment and preserve the
clinical benefit of NMDA-targeted drug therapy.
Notice Regarding Forward-Looking Statements
The information contained herein includes forward-looking
statements within the meaning of Section 21E of the Securities
Exchange Act of 1934, as amended, and Section 27A of the Securities
Act of 1933, as amended. These statements include, among others,
statements regarding the proposed public offering and the timing
and the use of the proceeds from the offering. Forward-looking
statements generally include statements that are predictive in
nature and depend upon or refer to future events or conditions, and
include words such as "may," "will," "should," "would," "expect,"
"plan," "believe," "intend," "look forward," and other similar
expressions among others. These statements relate to future events
or to the Company's future financial performance, and involve known
and unknown risks, uncertainties and other factors that may cause
the Company's actual results to be materially different from any
future results, levels of activity, performance or achievements
expressed or implied by these forward-looking statements. You
should not place undue reliance on forward-looking statements since
they involve known and unknown risks, uncertainties and other
factors which are, in some cases, beyond the Company's control and
which could, and likely will, materially affect actual results,
levels of activity, performance or achievements. Any
forward-looking statement reflects the Company's current views with
respect to future events and is subject to these and other risks,
uncertainties and assumptions relating to the Company's operations,
results of operations, growth strategy and liquidity. More detailed
information about the Company and the risk factors that may affect
the realization of forward-looking statements is set forth in the
Company's most recent Annual Report on Form 10-K and other filings
with the Securities and Exchange Commission. Investors and security
holders are urged to read these documents free of charge on the
SEC's website at http://www.sec.gov. Except as may be required
by applicable law, The Company assumes no obligation to publicly
update or revise these forward-looking statements for any reason,
or to update the reasons actual results could differ materially
from those anticipated in these forward-looking statements, whether
as a result of new information, future events or otherwise.

 View original content to download
multimedia:https://www.prnewswire.com/news-releases/nrx-pharmaceuticals-nasdaqnrxp-announces-promising-findings-in-phase-2b3-clinical-trial-of-nrx-101-vs-lurasidone-for-treatment-of-suicidal-bipolar-depression-302131482.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/nrx-pharmaceuticals-nasdaqnrxp-announces-promising-findings-in-phase-2b3-clinical-trial-of-nrx-101-vs-lurasidone-for-treatment-of-suicidal-bipolar-depression-302131482.html
SOURCE NRx Pharmaceuticals