- Collaboration with Bringham Women's Hospital to validate
MRI-compatible surgical robot for deep brain stimulation (DBS) in
Parkinson's patients –
- Successful cadaver trial demonstrates
precise delivery of bilateral DBS leads using real-time MRI
guidance -
- Announced collaboration with Synaptive
Medical to integrate neuro-navigation software, creating an
optimized workflow for unparalleled precision and efficacy in
neurosurgery -
WORCESTER, Mass., May 21, 2024
/PRNewswire/ -- AiM Medical Robotics Inc. (AiM), developer of
MRI-compatible intraoperative robotics for neurosurgery and other
applications, today announced a collaboration with
Brigham and Women's
Hospital (BWH) and the
Surgical Navigation and Robotics (SNR) Lab at
Harvard to validate
their cutting-edge robot for deep brain stimulation in Parkinson's
patients. The study will take place in BWH's
Advanced Multimodality Image-Guided Operating (AMIGO)
Suite, a state-of-the-art clinical research
facility. AiM also reported a successful cadaver trial
at the PracticePoint
facility, demonstrating precise delivery of
bilateral DBS leads using real-time MRI guidance. Prior to AiM's
approach, DBS leads may miss the intended target area due to the
brain shifting from the time of pre-operative imaging and surgery
due to creating access holes in the skull. This demonstration
highlights the robot's ability to account for brain shift.
Additionally, AiM announced a partnership with
Synaptive Medical to integrate
their Modus Nav neuro-navigation software with the robot, creating
an optimized workflow for unparalleled precision and efficiency in
neurosurgery.
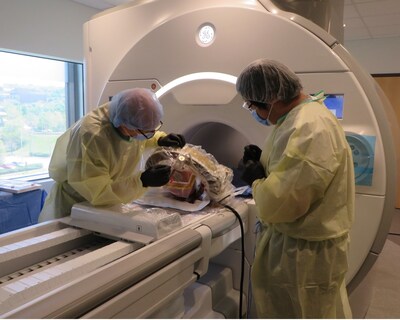
AiM has partnered with the Surgical Navigation and
Robotics (SNR) Lab at Harvard and
entered into a clinical research agreement with the Clinical Trial
Office at Brigham & Women's Hospital (BWH). This collaboration
builds upon the long-standing and successful joint research efforts
of AiM's CEO, Gregory Fischer,
Ph.D., and Pedro Moreira, Ph.D. and
Noby Hata, Ph.D. of the SNR Lab,
with Dr. Moreira as the lead investigator for the trial at BWH. The
team's previous work includes a successful 30-patient trial for
MRI-guided, robot-assisted prostate cancer biopsy. In this new
endeavor, they plan to utilize AiM's stereotactic neurosurgery
robot to accurately deliver deep brain stimulation leads with
real-time MRI guidance for patients with Parkinson's Disease. AiM
is thrilled to be collaborating with Dr. Reese Cosgrove, Head of Epilepsy and Functional
Neurosurgery, on this groundbreaking project.
AiM Medical Robotics is bringing to market a compact,
MRI-compatible surgical robot that facilitates intelligent
intraoperative surgical planning and guidance through real-time
soft tissue imaging. This innovative technology is crucial for
tackling the issue of brain shift, which often leads to
inconsistent and suboptimal outcomes in many procedures, as the
target moves relative to the skull during surgery. It has been
reported that 34% of deep brain stimulation lead placements require
another surgery for removal or revision, largely due to missing the
intended anatomical targets in the brain. AiM's pioneering product
for image-guided stereotactic neurosurgery is the result of
approximately 15 years and $15M of
NIH-funded academic research. The company is poised to introduce
its approach to the $4.3B market,
addressing challenges related to the efficient, accurate, and safe
intracranial placement of neuromodulation, ablation, and drug
delivery devices. By making intraoperative MRI guidance mainstream
and routine, AiM aims to improve hospital throughput, procedural
consistency, and patient outcomes.
Today, AiM announced that on May
10, 2024, the company conducted a cadaver trial in
collaboration with the clinical team from Brigham and Women's Hospital (BWH) at the
PracticePoint Medtech Accelerator facility located at
Worcester Polytechnic Institute (WPI),
where AiM is headquartered. During the trial, the team showcased
AiM's robot in a fully direct MRI-guided procedure for bilateral
deep brain stimulation lead placement in a human cadaver, with the
surgery performed by neurosurgeon collaborator, Dr. Cosgrove. The
AiM platform demonstrated success in completing a full procedure
entirely within the MRI suite, achieving high levels of efficiency
and accuracy. Notably, the trial highlighted the system's ability
to identify and account for intraoperative brain shift, a
phenomenon that frequently occurs during these surgeries and can
impact outcomes.
AiM Medical Robotics also announced its collaboration with
Synaptive Medical, Inc. ("Synaptive"), a Toronto-based Medtech company that provides
solutions for surgical, imaging, and data challenges. Through this
partnership, AiM and Synaptive have integrated Synaptive's highly
capable Modus Nav neuro-navigation software platform with AiM's
stereotactic neurosurgery robot, enabling enhanced visualization,
navigation, and control. The platform utilizes intraoperative MRI
updates to ensure precise targeting and MRI-based localization and
tracking of AiM's robot. By combining AiM's MRI-compatible robotic
stereotactic frame, Synaptive's advanced navigation software, and
intraoperative MRI, the two companies have created an optimized
workflow that delivers unparalleled precision and efficiency for
deep brain interventions. This collaboration marks a significant
step forward in improving patient outcomes and advancing the field
of neurosurgery.
About AiM Medical Robotics
AiM Medical Robotics is a privately held medical device company
based in Worcester, MA. It is
focused on the development of MRI-compatible advanced robotics that
are precision-focused, portable, and can be readily used with any
MRI scanner. Follow AiM at www.aimmedrobotics.com and on
LinkedIn.
Forward-Looking Statement
This press release includes forward-looking statements based
upon AiM's current expectations which may constitute
forward-looking statements for the purposes of the safe harbor
provisions under the Private Securities Litigation Reform Act of
1995 and other federal securities laws, and are subject to
substantial risks, uncertainties and assumptions. These statements
concern AiM's business strategies; the timing of regulatory
submissions; the ability to obtain and maintain regulatory
approvals of AiM's products; market acceptance of AiM's products;
AiM's intellectual property; AiM's reliance on third party
organizations; AiM's competitive position; AiM's industry
environment; AiM's anticipated financial and operating results,
including anticipated sources of revenues; AiM's assumptions
regarding the size of the available market, benefits of AiM's
products, product pricing and timing of product launches;
management's expectation with respect to future acquisitions;
statements regarding AiM's goals, intentions, plans and
expectations, including the introduction of new products and
markets; and AiM's cash needs and financing plans. There are a
number of factors that could cause actual events to differ
materially from those indicated by such forward-looking statements.
You should not place undue reliance on these forward-looking
statements, which include words such as "could," "believe,"
"anticipate," "intend," "estimate," "expect," "may," "continue,"
"predict," "potential," "project" or similar terms, variations of
such terms or the negative of those terms. Although AiM believes
that the expectations reflected in the forward-looking statements
are reasonable, AiM cannot guarantee such outcomes. AiM may not
realize its expectations, and its beliefs may not prove correct.
Actual results may differ materially from those indicated by these
forward-looking statements as a result of various factors. All such
statements speak only as of the date of this press release.
Consequently, forward-looking statements should be regarded solely
as AiM's current plans, estimates, and beliefs. AiM cannot
guarantee future results, events, levels of activity, performance
or achievements. AiM does not undertake and specifically declines
any obligation to update or revise any forward-looking statements
to reflect new information, future events or circumstances or to
reflect the occurrences of unanticipated events, except as may be
required by applicable law.
Contacts:
Gregory
Fischer, Ph.D.
Founder & Chief Executive Officer
AiM Medical Robotics, Inc.
LinkedIn:
https://www.linkedin.com/in/gregfischer/
Phone: (617) 203-5560
Email: info@aimmedrobotics.com
Investors:
Investor Relations
IR@aimmedrobotics.com
 View original content to download
multimedia:https://www.prnewswire.com/news-releases/aim-medical-robotics-partners-with-brigham-and-womens-hospital-for-a-first-human-clinical-study-for-mri-guided-device-placement-in-the-brain-302151595.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/aim-medical-robotics-partners-with-brigham-and-womens-hospital-for-a-first-human-clinical-study-for-mri-guided-device-placement-in-the-brain-302151595.html
SOURCE AiM Medical Robotics