As
filed with the Securities and Exchange Commission on May 10, 2024.
Registration
No. 333-
UNITED
STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM
F-1
REGISTRATION
STATEMENT
UNDER
THE SECURITIES ACT OF 1933
NeuroSense
Therapeutics Ltd.
(Exact Name of Registrant as Specified in Its Charter)
Not
Applicable
(Translation of Registrant’s name into English)
| State
of Israel |
|
2834 |
|
Not
Applicable |
(State or Other Jurisdiction
of
Incorporation or Organization) |
|
(Primary Standard Industrial
Classification Code Number) |
|
(I.R.S. Employer
Identification Number) |
11
HaMenofim Street, Building B
Herzliya 4672562 Israel
+972-9-7996183
(Address,
Including Zip Code, and Telephone Number, Including Area Code, of Registrant’s Principal Executive Offices)
Cogency
Global Inc.
122 East 42nd Street,
18th Floor, New York, NY 10168
(212) 947-7200
(Name, Address, Including Zip Code, and Telephone Number, Including Area Code, of Agent For Service)
Copies to:
Robert
L. Grossman, Esq.
Gary
Emmanuel, Esq.
David
Huberman, Esq.
Greenberg
Traurig, P.A.
One
Azrieli Center
Round
Tower, 30th Floor
132
Menachem Begin Rd
Tel
Aviv, Israel 6701101
+1
212 801 9337 |
|
Perry Wildes
Goldfarb Gross
Seligman & Co.
One Azrieli Center
Tel Aviv 6702100, Israel
+972 (3) 607-4444 |
Approximate
date of commencement of proposed sale to the public: As soon as practicable after the effective date of this Registration Statement.
If
any of the securities being registered on this form are to be offered on a delayed or continuous basis pursuant to Rule 415 under
the Securities Act of 1933, check the following box. ☒
If
this form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, check
the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same
offering. ☐
If
this form is a post-effective amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box and
list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
If
this form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and
list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
Indicate
by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933.
Emerging
growth company ☒
If
an emerging growth company that prepares its financial statements in accordance with U.S. GAAP, indicate by check mark if the registrant
has elected not to use the extended transition period for complying with any new or revised financial accounting standards†
provided pursuant to Section 7(a)(2)(B) of the Securities Act. ☐
| † | The
term “new or revised financial accounting standard” refers to any update issued
by the Financial Accounting Standards Board to its Accounting Standards Codification after
April 5, 2012. |
The
Registrant hereby amends this Registration Statement on such date or dates as may be necessary to delay its effective date until the
Registrant shall file a further amendment which specifically states that this Registration Statement shall thereafter become effective
in accordance with Section 8(a) of the Securities Act of 1933, as amended, or until the Registration Statement shall
become effective on such date as the Commission, acting pursuant to such Section 8(a), may determine.
The
information in this preliminary prospectus is not complete and may be changed. These securities may not be sold until the registration
statement filed with the Securities and Exchange Commission is effective. This preliminary prospectus is not an offer to sell these securities
nor does it seek an offer to buy these securities in any jurisdiction where the offer or sale is not permitted.
| PRELIMINARY PROSPECTUS |
|
SUBJECT TO COMPLETION DATED MAY
10, 2024 |

Up to 3,050,964 Ordinary Shares
Ordinary Warrants to Purchase up to 2,980,000
Ordinary Shares
This prospectus relates to
the resale, from time to time, by the selling shareholders identified in this prospectus under the section “Selling Shareholders,”
(each a “selling shareholder” and collectively the “selling shareholders”) of NeuroSense Therapeutics Ltd. (“NeuroSense,”
“we,” “us” or the “Company”) of (i) up to 2,980,000 of our ordinary shares, no par value per share
(the “Shares”), issuable to the selling shareholder identified in this prospectus under the section “Selling Shareholder,”
upon exercise of warrants to purchase ordinary shares at an exercise price of $1.50 per ordinary share (the “Ordinary Warrants”),
acquired by the selling shareholder under a securities purchase agreement (the “Purchase Agreement”), dated April 10, 2024,
by and between us and the purchaser named therein (the “April 2024 Offering”), (ii) the Ordinary Warrants, and
(iii) 70,964 ordinary shares issued to our placement agent in the April 2024 Offering as partial consideration in exchange for their services
(the “PA Shares”). The Shares, Ordinary Warrants and the PA Shares were offered pursuant to the exemptions provided in Section 4(a)(2)
under the Securities Act of 1933, as amended (the “Securities Act”), and Regulation D promulgated thereunder.
The selling shareholders
will receive all of the proceeds from any sales of the resale of Shares, PA Shares and Ordinary Warrants offered hereby. We will not receive
any of the proceeds, but we will incur expenses in connection with such offering. To the extent the Ordinary Warrants are exercised for
cash, if at all, we will receive the exercise price of the Ordinary Warrants.
The selling shareholders
may sell the Shares, the PA Shares and Ordinary Warrants covered by this prospectus through public or private transactions at market prices
prevailing at the time of sale, at negotiated prices or such other prices as such selling shareholder may determine. The timing and amount
of any sale are within the sole discretion of such selling shareholder. Our registration of the Shares, PA Shares, and Ordinary Warrants
for resale covered by this prospectus does not mean that the selling shareholders will offer or sell any of the Shares PA Shares, or Ordinary
Warrants. For further information regarding the possible methods by which the Shares, Ordinary Warrants and/or the PA Shares may be distributed,
see “Plan of Distribution.”
We
are an “emerging growth company” and a “foreign private issuer”, each as defined under federal securities laws,
and, as such, have elected to comply with certain reduced public company reporting requirements for this prospectus and future filings.
See “Prospectus Summary — Implications of Being an Emerging Growth Company” and “Prospectus Summary — Implications
of Being a Foreign Private Issuer” for additional information.
Our ordinary shares and warrants to purchase our ordinary shares are
listed on The Nasdaq Capital Market (“Nasdaq”) under the symbols “NRSN” and “NRSNW.” On May 9, 2024,
the closing price for our ordinary shares of was $1.1738 and the closing price for the warrants to purchase our ordinary shares was $0.18.
Investing in our securities
is highly speculative and involves a high degree of risk. See “Risk Factors” beginning on page 6 to read about factors you
should consider before buying the Shares or the Ordinary Warrants.
Neither
the Securities and Exchange Commission nor any other regulatory body has approved or disapproved of these securities or passed upon the
accuracy or adequacy of the disclosures in this prospectus. Any representation to the contrary is a criminal offense.
The
date of this prospectus is , 2024
TABLE
OF CONTENTS
ABOUT
THIS PROSPECTUS
This prospectus is part of
the registration statement on Form F-1 that we filed with the Securities and Exchange Commission (the “SEC”) for the
offering of the Shares, PA Shares, and Ordinary Warrants by the selling shareholders.
You should not assume that the information contained in, or incorporated
by reference into, this prospectus is accurate on any date subsequent to the date set forth on the front cover of this prospectus, even
though this prospectus is delivered or Shares, PA Shares, and Ordinary Warrants covered by this prospectus are sold or otherwise disposed
of on a later date. It is important for you to read and consider all information contained in, or incorporated by reference into, this
prospectus in making your investment decision. You should also read and consider the information in the documents to which we have referred
you under the caption “Where You Can Find Additional Information” in this prospectus.
Neither we nor the selling shareholders have authorized anyone to provide
any information or to make any representation other than those contained in, or incorporated by reference into, this prospectus. You must
not rely upon any information or representation not contained in, or incorporated by reference into, this prospectus. This prospectus
does not constitute an offer to sell or the solicitation of an offer to buy any of our securities other than the securities covered hereby,
nor does this prospectus constitute an offer to sell or the solicitation of an offer to buy any of our securities in any jurisdiction
to any person to whom it is unlawful to make such offer or solicitation in such jurisdiction.
This
prospectus, including the information incorporated by reference herein, contains forward-looking statements that are subject to a number
of risks and uncertainties, many of which are beyond our control. See “Risk Factors” and “Cautionary Note Regarding
Forward-Looking Statements.”
TRADEMARKS,
SERVICE MARKS AND TRADENAMES
The
NeuroSense Therapeutics logo and other trademarks and service marks of NeuroSense Therapeutics Ltd. appearing in this prospectus or the
information incorporated by reference herein are the property of the Company. Solely for convenience, some of the trademarks, service
marks, logos and trade names referred to in this prospectus are presented without the® and™ symbols,
but such references are not intended to indicate, in any way, that we will not assert, to the fullest extent under applicable law, our
rights to these trademarks, service marks and trade names. This prospectus, including the information incorporated by reference herein,
contains additional trademarks, service marks and trade names of others. All trademarks, service marks and trade names appearing in this
prospectus or the information incorporated by reference herein are, to our knowledge, the property of their respective owners. We do
not intend our use or display of other companies’ trademarks, service marks, copyrights or trade names to imply a relationship
with, or endorsement or sponsorship of us by, any other companies.
MARKET
AND INDUSTRY DATA
This
prospectus, including the information incorporated by reference herein, contains industry, market and competitive position data that
are based on industry publications and studies conducted by third parties as well as our own internal estimates and research. These industry
publications and third-party studies generally state that the information that they contain has been obtained from sources believed to
be reliable, although they do not guarantee the accuracy or completeness of such information. While we believe that each of these publications
and third-party studies is reliable, we have not independently verified the market and industry data obtained from these third-party
sources. Forecasts and other forward-looking information obtained from these sources are subject to the same qualifications and uncertainties
as the other forward-looking statements included in, or incorporated by reference into, this prospectus. While we believe our internal
research is reliable and the definition of our market and industry are appropriate, neither such research nor these definitions have
been verified by any independent source.
CAUTIONARY
NOTE REGARDING FORWARD-LOOKING STATEMENTS
This
prospectus and certain information incorporated by reference herein contain forward-looking statements within the meaning of Section 27A
of the Securities Act and Section 21E of the U.S. Securities Exchange Act of 1934, as amended (the “Exchange Act”),
and other securities laws. Many of the forward-looking statements contained in, or incorporated by reference into, this prospectus can
be identified by the use of forward-looking words such as “anticipate,” “believe,” “could,” “estimate,”
“expect,” “intend,” “may,” “might,” “plan,” “potential,” “should,”
“target,” “would” and other similar expressions that are predictions of or indicate future events and future
trends, although not all forward-looking statements contain these identifying words.
Forward-looking
statements are based on our management’s beliefs and assumptions and on information currently available to our management. Such
statements are subject to substantial risks and uncertainties, and actual results may differ materially from those expressed or implied
in the forward-looking statements due to a variety of factors, including, but not limited to, those identified under the section titled
“Risk Factors” in this prospectus or the documents incorporated herein. These risks and uncertainties include factors relating
to:
| ● | the
going concern reference in our financial statements and our need for substantial additional
financing to achieve our goals; |
| ● | our
limited operating history and history of incurring significant losses and negative cash flows
since our inception, which we anticipate will continue for the foreseeable future; |
| ● | our
dependence on the success of our lead product candidate, PrimeC, including our obtaining
of regulatory approval to market PrimeC in the United States; |
| ● | our
limited experience in conducting clinical trials and reliance on clinical research organizations
and others to conduct them; |
| ● | our
ability to advance our preclinical product candidates into clinical development and through
regulatory approval; |
| ● | the
results of our clinical trials, which may fail to adequately demonstrate the safety and efficacy
of our product candidates; |
| ● | our
ability to achieve the broad degree of physician adoption and use and market acceptance necessary
for commercial success; |
| ● | our
reliance on third parties in marketing, producing or distributing products and research materials
for certain raw materials, compounds and components necessary to produce PrimeC for clinical
trials and to support commercial scale production of PrimeC, if approved; |
| ● | our
receipt of regulatory clarity and approvals for our therapeutic candidates and the timing
of other regulatory filings and approvals; |
| ● | estimates
of our expenses, revenues, capital requirements and our needs for additional financing; |
| ● | our
efforts to obtain, protect or enforce our patents and other intellectual property rights
related to our product candidates and technologies; |
| ● | our
ability to maintain the listing of our ordinary shares on Nasdaq; |
| ● | the
impact of the public health, political and security situation in Israel, the U.S. and other
countries in which we may obtain approvals for our products or our business; |
| ● | the
impacts on our ongoing and planned trials and manufacturing as a result of the war in Israel;
and |
| ● | those factors referred to in our most recent Annual Report on Form
20-F incorporated by reference herein in “Item 3. Key Information — D. Risk Factors,” “Item 4. Information on
the Company,” and “Item 5. Operating and Financial Review and Prospects,” as well as in our most recent Annual Report
on Form 20-F generally, which is incorporated by reference into this prospectus. |
The
preceding list is not intended to be an exhaustive list of all of our risks and uncertainties. As a result of these factors, we cannot
assure you that the forward-looking statements contained in, or incorporated by reference into, this prospectus will prove to be accurate.
Furthermore, if our forward-looking statements prove to be inaccurate, the inaccuracy may be material. In light of the significant uncertainties
in these forward-looking statements, you should not regard these statements as a representation or warranty by us or any other person
that we will achieve our objectives and plans in any specified time frame, or at all.
In
addition, statements that “we believe” and similar statements reflect our beliefs and opinions on the relevant subject. These
statements are based upon information available to us as of the date of this prospectus or any document incorporated herein or therein,
and while we believe such information forms a reasonable basis for such statements, such information may be limited or incomplete, and
our statements should not be read to indicate that we have conducted an exhaustive inquiry into, or review of, all relevant information.
PROSPECTUS
SUMMARY
This summary highlights
selected information about us and the Shares, PA Shares, and the Ordinary Warrants being offered. It may not contain all of the information
that may be important to you. Before investing in the Shares, PA Shares, and Ordinary Warrants, you should read this entire prospectus
and other information incorporated by reference from our other filings with the SEC carefully for a more complete understanding of our
business and this offering, including our consolidated financial statements and the section entitled “Risk Factors” included
or incorporated by reference in this prospectus. Some of the statements in this prospectus and the documents incorporated by reference
herein constitute forward-looking statements that involve risks and uncertainties. See information set forth under the section “Cautionary
Note Regarding Forward-Looking Statements.”
Overview
We
are a clinical-stage biotechnology company focused on discovering and developing treatments for people living with neurodegenerative
diseases, including ALS AD and PD. We believe these diseases represent some of the most significant unmet medical needs of our time,
with limited effective therapeutic options available. The burden of these diseases on both patients and society is substantial. For example,
the average annual cost of ALS alone is $180,000 per patient, and its estimated annual burden on the U.S. healthcare system is greater
than $1 billion. Due to the complexity of neurodegenerative diseases, our strategy is utilizing a combined therapeutic approach
to target multiple disease-related pathways.
Our
lead therapeutic candidate, PrimeC, is a novel extended-release oral formulation, fixed-dose combination of two FDA-approved drugs, ciprofloxacin
and celecoxib. PrimeC is designed to treat ALS by modulating microRNA synthesis, iron accumulation, and neuroinflammation, all of which
are hallmarks of ALS pathology. The U.S. Food and Drug Administration, or the FDA and the European Medicines Agency, or the EMA
have granted PrimeC orphan drug designation for the treatment of ALS. In addition, the EMA has granted PrimeC the Small and Medium-Sized
Enterprise, or SME, status, which offers significant potential benefits leading up to and following drug regulatory approval. We believe
PrimeC’s multifunctional mechanism of action has the potential to significantly prolong lifespan and improve ALS patients’
quality of life, thereby reducing the burden of this debilitating disease on both patients and healthcare systems.
PrimeC
is currently being evaluated in PARADIGM (“NST003”), a Phase IIb randomized, multi-center, multinational, prospective,
double-blind, placebo-controlled study, to evaluate safety, tolerability, and efficacy of PrimeC in 68 people living with ALS. Participants
were being administered PrimeC or placebo at a 2:1 ratio, respectively, for the six-month double-blind part. Study participants were
allowed to continue standard of care treatment of approved products. The primary endpoints of the study are an evaluation of ALS-biomarkers
as well as safety and tolerability assessment. Secondary and exploratory endpoints are the evaluation of clinical efficacy (ALS Functional
Rating Scale — Revised, or ALSFRS-R, and slow vital capacity), survival, and improvement in quality of life. All subjects
who completed the six-month double-blind, placebo-controlled dosing period had the opportunity to be transferred to the PrimeC active
arm for a 12-month open label extension. The study completed enrollment in May 2023, enrolling 69 participants, in which 68 are
living with ALS and one participant who was misdiagnosed for ALS and was excluded from the evaluations. Four ALS clinical centers participated
in the study in 3 territories: Israel, Italy, and Canada. In December 2023, we reported that we met the primary safety and tolerability
endpoints and achieved secondary clinical efficacy endpoints in the top-line results of our 6-month double-blind phase of PARADIGM. In May 2024, we announced new positive data analysis from PARADIGM
clinical trial demonstrating statistically significant slowing of disease progression in high-risk ALS patients. We
also expect to report on additional primary ALS-biomarkers endpoint: TDP-43 and Prostagladin2, in the second or third quarter of 2024.
Following
the FDA’s recommendation for additional non-clinical data to support long term use of Ciprofloxacin (as PrimeC is intended for long-term
administration in treating ALS) a long-term tox study was initiated. Results from this study are expected in Q3 towards the initiation
of Phase III study in the US. We plan to have an End of Phase 2 Meeting with the FDA and EMA in the second or third quarter of 2024 and
to commence a pivotal clinical trial for PrimeC in ALS treatment by the end of the year. Additionally, in November 2023, we concluded
a successful Type D meeting with the FDA regarding CMC development plans for the expected Phase 3 pivotal study and subsequent marketing
approval. The FDA endorsed our proposed CMC development plan.
PrimeC
was previously evaluated in a Phase IIa clinical trial (“NST002”) in 15 people living with ALS, conducted at the Tel
Aviv Sourasky Medical Center, Israel. The primary endpoint of the NST002 trial, which was safety and tolerability, was met. In this trial,
the safety profile observed was consistent with known safety profiles of ciprofloxacin and celecoxib. Side effects were mild and transient
in nature. There were no new or unexpected safety signals detected during the trial.
Additionally, we observed positive clinical signals in comparison to
virtual controls, and a serum biomarker analysis showed significant changes following treatment, indicating biological activity of the
drug in comparison to untreated matched ALS patients. All 12 patients who completed the NST002 trial elected to continue into an extension
study with PrimeC, that was conducted as an Investigator Initiated Study. To date, we are still supporting the drug supply for a few of
the participants in this study, which is over than 40 months since NST002 was initiated.
We
completed three additional studies in 2022 as part of our drug development program to further support our future regulatory submissions.
In April 2022, we initiated a pharmacokinetic, or PK, study (“NCT05232461”) of PrimeC. The PK open-label, randomized,
single-dose, three-treatment, three-period crossover study evaluated the effect of food on the bioavailability of PrimeC as compared
to the bioavailability of co-administered ciprofloxacin tablets and celecoxib capsules in adult subjects in the U.S. under an FDA
cleared IND protocol.
In August 2022, we completed enrollment and dosing of all subjects
in a multi-dose PK study (“NCT05436678”). On September 28, 2022, we released the results of the NCT05436678 study. Based
on results, we believe the PK profile of PrimeC supports the formulation’s extended-release properties, as the concentrations of
the active components have been synchronized, aiming to potentially maximize the synergism between the two compounds. In June 2022,
we reported the successful completion of the “in-life” phase of its 90-day GLP toxicology study. In this study, the components
of PrimeC, celecoxib and ciprofloxacin, were administered to rodents at doses 4x the maximal clinical dose. All animals appeared normal,
with no significant findings observed. We intend to present the data from these studies to the FDA as part of PrimeC’s drug development
plan.
We
believe we have a strong patent estate, including patents on method of use, combination, and formulation. We secured U.S. Patent
10,980,780 relating to methods for treatment of ALS using ciprofloxacin and celecoxib, the components of PrimeC, which expires in 2038.
This patent also been issued in the European Patent Office, Canada, Australia, Israel and Japan. We also expect to take advantage of
orphan drug exclusivity for PrimeC, if approved, for seven years in the United States and ten years in the European Union.
In addition, U.S. patent application 16/623,467, which relates to methods of treatment of neurodegenerative disease using combinations
of ciprofloxacin and celecoxib, is currently pending. This patent application is expected to expire on June 20, 2038.
Our
organization is built around a management team with extensive experience in the pharmaceutical industry, with a particular focus on ALS
research and clinical trials. We believe that our leadership team is well-positioned to lead us through clinical development, regulatory
approval and commercialization of our product candidates. Furthermore, we maintain steadfast and extensive communication and collaboration
with patient advocacy groups and associations, underscoring the importance of patient perspectives in advancing therapeutic strategies.
In
addition to PrimeC, we extended our pipeline and conducted research and development efforts for AD and PD, with a similar strategy of
combined products. The following chart represents our current product development pipeline:
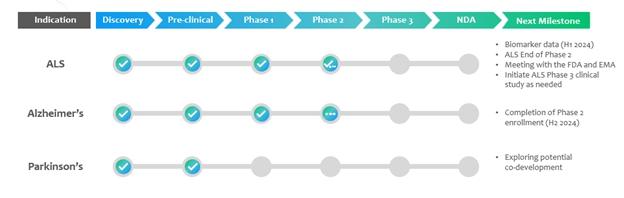
Recent
Developments
On
April 15, 2024, pursuant to the Purchase Agreement, we issued and sold to a selling shareholder in a registered direct offering
(the “Registered Direct Offering”), (i) an aggregate of 1,732,000 ordinary shares, at an offering price of $1.50 per
share; and (ii) an aggregate of 1,248,000 pre-funded warrants, each representing the right to acquire one ordinary share (the
“Pre-Funded Warrants”), at an offering price of $1.4999 per Pre-Funded Warrant, for gross proceeds of approximately
$4.47 million before deducting the placement agent fee and related offering expenses, which included in part the PA Shares. Each
Pre-Funded Warrant represents the right to purchase one ordinary share at an exercise price of $0.0001 per share. The Pre-Funded
Warrants are exercisable immediately and may be exercised at any time until the Pre-Funded Warrants are exercised in full (subject
to certain beneficial ownership limitations). In a concurrent private placement (together with the Registered Direct Offering, the
“Offering”), pursuant to the Purchase Agreement, we issued and sold to such selling shareholder the Ordinary Warrants,
which are exercisable immediately upon issuance at an exercise price of $1.50 per ordinary share, and will expire on the fifth
anniversary of the original issuance date.
Corporate
Information
Our legal and commercial name is NeuroSense Therapeutics Ltd. We were
incorporated on February 13, 2017 and were registered as a private company limited by shares under the laws of the State of Israel.
We completed our initial public offering on the Nasdaq in December 2021. Our ordinary shares and warrants to purchase our ordinary
shares are traded on the Nasdaq under the symbol “NRSN” and “NRSNW,” respectively.
Our
principal executive offices are located at 11 HaMenofim Street, Building B, Herzliya, 4672562 Israel, and our telephone number is +972-9-7996183.
Our website address is www.neurosense-tx.com. The information on our website does not constitute a part of this prospectus.
Our agent for service of process in the United States is Cogency Global Inc., 122 East 42nd Street, 18th Floor,
New York, NY 10168.
Implications
of Being an Emerging Growth Company
As
a company with less than $1.235 billion in revenue during our last fiscal year, we are an “emerging growth company”
as defined in the Jumpstart Our Business Startups Act of 2012 (the “JOBS Act”). As such, we may take advantage
of certain exemptions from various reporting requirements that are applicable to other publicly traded entities that are not emerging
growth companies. These exemptions include:
| ● | not
being required to have our registered independent public accounting firm attest to management’s assessment of our internal control
over financial reporting; |
| |
● |
not being required to comply
with any requirement that may be adopted by the Public Company Accounting Oversight Board (“PCAOB”), regarding mandatory
audit firm rotation or a supplement to the auditor’s report providing additional information about the audit and the financial
statements (i.e., an auditor discussion and analysis); |
| |
● |
not being required to submit
certain executive compensation matters to stockholder advisory votes, such as “say-on-pay,” “say-on-frequency”
and “say-on-golden parachutes”; |
| |
● |
the audit reports of our
independent registered public accounting firm do not require communication of critical audit matters; and |
| |
● |
not being required to disclose
certain executive compensation related items such as the correlation between executive compensation and performance and comparisons
of the chief executive officer’s compensation to median employee compensation. |
As
a result, the information contained in this prospectus and the documents incorporated by reference herein may be different from the information
you receive from other public companies in which you hold shares. We may take advantage of these provisions for up until we are no longer
an emerging growth company. We would cease to be an emerging growth company upon the earliest to occur of: (i) the last day
of the first fiscal year in which our annual gross revenues exceed $1.235 billion; (ii) the date on which we have issued more
than $1 billion in non-convertible debt securities during the previous three years; (iii) the date on which we are deemed
to be a “large accelerated filer” as defined in Rule 12b-2 under the Exchange Act; or (iv) the last day
of the fiscal year following the fifth anniversary of our initial public offering.
Implications
of Being a Foreign Private Issuer
We
are also a non-U.S. company with foreign private issuer status. Even after we no longer qualify as an emerging growth company, as
long as we continue to qualify as a foreign private issuer under the Exchange Act, we will be exempt from certain provisions of
the Exchange Act that are applicable to U.S. domestic public companies, including:
| |
● |
the rules under the Exchange Act
requiring domestic filers to issue financial statements prepared under U.S. generally accepted accounting principles (“U.S. GAAP”); |
| |
● |
the sections of the Exchange Act
regulating the solicitation of proxies, consents or authorizations in respect of a security registered under the Exchange Act; |
| |
● |
the sections of the Exchange Act
requiring insiders to file public reports of their share ownership and trading activities and liability for insiders who profit from
trades made in a short period of time; and |
| |
● |
the rules under the Exchange Act
requiring the filing with the SEC of quarterly reports on Form 10-Q containing unaudited financial statements and other specified
information, and current reports on Form 8-K upon the occurrence of specified significant events. |
Notwithstanding
these exemptions, we will file with the SEC, within four months after the end of each fiscal year, or such applicable time as required
by the SEC, an Annual Report on Form 20-F containing financial statements audited by an independent registered public accounting
firm.
We
may take advantage of these exemptions until such time as we are no longer a foreign private issuer. We would cease to be a foreign private
issuer at such time as more than 50% of our outstanding voting securities are held by U.S. residents and any of the following three
circumstances applies: (i) the majority of our executive officers or directors are U.S. citizens or residents; (ii) more
than 50% of our assets are located in the United States; or (iii) our business is administered principally in the United States.
Both
foreign private issuers and emerging growth companies also are exempt from certain more stringent executive compensation disclosure rules.
Thus, even if we no longer qualify as an emerging growth company, but remain a foreign private issuer, we will continue to be exempt
from the more stringent compensation disclosures required of companies that are neither an emerging growth company nor a foreign private
issuer.
THE
OFFERING
| Ordinary shares offered by the
selling shareholders |
|
Up to (i)
2,980,000 ordinary shares issuable upon exercise of the Ordinary Warrants; and (ii) 70,964 ordinary shares. |
| |
|
|
| Ordinary
Warrants offered by the selling shareholder |
|
Ordinary Warrants to purchase up to 2,980,000 ordinary shares. |
| |
|
|
| Ordinary shares to be outstanding immediately following
this offering |
|
20,385,006 ordinary shares. |
| |
|
|
| Use
of proceeds |
|
All of the Shares, PA Shares and Ordinary Warrants offered by the selling
shareholders for resale pursuant to this prospectus will be sold by the selling shareholders. We will not receive any proceeds from such
sales. See the section titled “Use of Proceeds” in this prospectus for more information. However, to the extent the Ordinary
Warrants are exercised for cash, if at all, we will receive the exercise price of the Ordinary Warrants. The exercise price of the Ordinary
Warrants may exceed the trading price of our ordinary shares. If the price of our ordinary shares is below $1.50, we believe that holder
of the Ordinary Warrants will be unlikely to exercise their warrants, resulting in little to no cash proceeds to us. If all the Ordinary
Warrants were exercised, we would receive gross proceeds of approximately $4.5 million. See the section titled “Use of Proceeds”
in this prospectus for more information. |
| |
|
|
| Offering
price |
|
The exercise price of the Ordinary Warrants is $1.50 per ordinary share.
The Shares, PA Shares and Ordinary Warrants offered by the selling shareholders under this prospectus may be offered and sold at prevailing
market prices, negotiated prices or such other prices as the selling shareholders may determine. See the section titled “Plan of
Distribution” in this prospectus for more information. |
| |
|
|
| Risk
factors |
|
See the section titled “Risk Factors” beginning on page
6 and the other information included in this prospectus for a discussion of factors you should consider before deciding to invest in the
Shares, PA Shares and Ordinary Warrants. |
| |
|
|
| Listing |
|
Our ordinary shares and warrants to purchase ordinary shares are listed
on The Nasdaq Capital Market under the symbols “NRSN” and “NRSNW, respectively.” |
Unless otherwise stated, all information in this prospectus, is based
on 17,405,006 ordinary shares outstanding as of May 9, 2024, and does not include the following as of that date:
| |
● |
1,069,128 ordinary shares
issuable upon the exercise of options outstanding, at a weighted average exercise price of $2.30 per share under our 2018 Employee
Share Option Plan; |
| |
● |
94,000 ordinary shares
issuable upon the vesting of restricted share units outstanding, some of which are under our 2018 Share Incentive Plan; |
| |
● |
233,543 ordinary shares
reserved for issuance and available for future grant under our 2018 Share Incentive Plan; |
| |
● |
7,735,000 ordinary shares
underlying warrants with a weighted average exercise price of $2.54 per share; and |
| |
● |
1,248,000 ordinary shares
underlying the Pre-Funded Warrants, with an exercise price of $0.0001 per share. |
Except as otherwise indicated, all information in this prospectus assumes
no exercise of any Pre-Funded Warrants (as defined below) or the Ordinary Warrants.
RISK
FACTORS
Investing
in our securities involves significant risks. Before making an investment decision, you should carefully consider the risks described
below and under the section titled “Item 3. Key Information—D. Risk Factors” in our Annual Report on Form 20-F for
the year ended December 31, 2023, as well as the information included or incorporated by reference in this prospectus supplement and
the accompanying prospectus, together with all of the other information appearing in this prospectus supplement or the accompanying prospectus
or incorporated by reference herein or therein, including in light of your particular investment objectives and financial circumstances.
The risks so described are not the only risks we face. Additional risks not presently known to us or that we currently deem immaterial
may also impair our business operations and become material. Our business, financial condition and results of operations could be materially
adversely affected by any of these risks. The trading price of our securities could decline due to any of these risks, and you may lose
all or part of your investment. The discussion of risks includes or refers to forward-looking statements; you should read the explanation
of the qualifications and limitations on such forward-looking statements discussed elsewhere in this prospectus supplement under the
caption “Cautionary Statement Regarding Forward-Looking Statements” above.
Risks
Related to this Offering
If
we fail to maintain compliance with NASDAQ’s continued listing requirements, our shares may be delisted from the NASDAQ Capital
Market.
To
continue to be listed on the Nasdaq, we need to satisfy a number of conditions, including a minimum closing bid price per share of $1.00
for 30 consecutive business days (the “Minimum Bid Price Rule”) and shareholders’ equity of at least $2.5 million.
On December 21, 2023, we received a notice from the Listing Qualifications staff (the “Staff”) of The Nasdaq Stock Market
LLC indicating that we failed to comply with the continued listing requirement that we maintain either a minimum of $2,500,000 in shareholders’
equity or $35,000,000 market value of listed securities or $500,000 of net income from continuing operations for the most recently completed
fiscal year or two of the three most recently completed fiscal years, as set forth in Nasdaq Listing Rule 5550(b)(1) (the “Minimum
Equity Rule”). On February 5, 2024, we submitted to the Staff a plan to regain compliance with the Minimum Equity Rule. After
submitting a plan to regain compliance, Nasdaq granted us an extension to regain compliance. Under the terms of the extension, we were
required to complete a fundraising of $20 million on or before April 30, 2024, and if we are unable to demonstrate compliance
by then, we were required to advise the Staff in advance of that date to request a further extension. In any case, if we fail to evidence
compliance upon the filing with the SEC of our periodic report for the period ending June 30, 2024, we may be subject to delisting.
Previously on September 5, 2023, we received notice from the Staff that we did not meet the minimum bid requirement for continued
listing however during February 2024, we regained compliance with the Minimum Bid Price Rule.
No assurance can be given
that we will be able to regain compliance with the Minimum Equity Rule or comply with the other standards that we are required to meet
in order to maintain a listing on such exchange, such as the Minimum Bid Price Rule, and no assurance can be given that even if we regain
compliance with the Minimum Equity Rule we will maintain sufficient shareholders’ equity or the price of our ordinary shares will
not again be in violation of Nasdaq’s Minimum Bid Price Rule in the future. Our failure to meet these requirements may result in
our securities being delisted from Nasdaq.
If our ordinary shares are
delisted from Nasdaq, we may seek to list them on other markets or exchanges or our ordinary shares may trade on the pink sheets. In the
event of such delisting, our shareholders’ ability to trade, or obtain quotations of the market value of, our ordinary shares would
be severely limited because of lower trading volumes and transaction delays. These factors could contribute to lower prices and larger
spreads in the bid and ask prices for our securities. In addition, the substantially decreased trading in our ordinary shares and decreased
market liquidity of our ordinary shares as a result of the loss of market efficiencies associated with Nasdaq and the loss of federal
preemption of state securities laws, which could materially adversely affect our ability to obtain financing on acceptable terms, if at
all, and may result in the potential loss of confidence by investors, suppliers, customers and employees and fewer business development
opportunities. Additionally, the market price of our ordinary shares may decline further and shareholders may lose some or all of their
investment. There can be no assurance that our ordinary shares, if delisted from the Nasdaq in the future, would be listed on another
national or international securities exchange or on a national quotation service, the Over-The-Counter Markets or the pink sheets.
The sale of a substantial amount of our
ordinary shares in the public market, including resale of the shares issued to the selling shareholders, could adversely affect the prevailing
market price of our Ordinary Shares and PA Shares.
We are registering for resale 2,980,000 Shares and 70,964 PA Shares
held by the selling shareholders and the Ordinary Warrants held by one of the selling shareholders. Sales of substantial amounts of our
ordinary shares in the public market, or the perception that such sales might occur, could adversely affect the market price of our ordinary
shares, and the market value of our other securities. We cannot predict if and when a selling shareholder may sell such shares in the
public markets. Furthermore, in the future, we may issue additional ordinary shares or other equity or debt securities convertible into
ordinary shares. Any such issuance could result in substantial dilution to our existing shareholders and could cause our share price to
decline.
The
price of our ordinary shares may be volatile.
The market price of our ordinary
shares has fluctuated in the past. Consequently, the current market price of our ordinary shares may not be indicative of future market
prices, and we may be unable to sustain or increase the value of your investment in our ordinary shares.
There
is no guarantee that the Ordinary Warrants will be in the money, and they may expire worthless.
The exercise price of the Ordinary Warrants is $1.50 per ordinary share,
subject to adjustment. The exercise price of the Ordinary Warrants has at times exceeded the market price of our ordinary shares. To the
extent the price of our ordinary shares remains below $1.50, we believe that holders of the Ordinary Warrants will be unlikely to exercise
their warrants, resulting in little to no cash proceeds to us. There is no guarantee the exercise price of the Ordinary Warrants will
ever exceed the market price of our ordinary shares in the future and, as such, the Ordinary Warrants may expire worthless.
We
do not intend to apply for any listing of the Ordinary Warrants on any exchange or nationally recognized trading system, and we do not
expect a market to develop for the Ordinary Warrants.
We
do not intend to apply for any listing of the Ordinary Warrants on Nasdaq or any other securities exchange or nationally recognized trading
system, and we do not expect a market to develop for the Ordinary Warrants. Without an active market, the liquidity of the Ordinary Warrants
will be limited. Further, the existence of the Ordinary Warrants may act to reduce both the trading volume and the trading price of our
ordinary shares.
Holders of Ordinary Warrants will have no
rights as shareholders of ordinary shares until such holders exercise their Ordinary Warrants and acquire ordinary shares.
The Ordinary Warrants do
not confer any rights of ordinary share ownership on their holders, such as voting rights or the right to receive dividends, but rather
merely represent the right to acquire ordinary shares at a fixed price. A holder of an Ordinary Warrant may exercise the right to acquire
an ordinary share and pay an exercise price of $1.50 at any time. Upon exercise of the Ordinary Warrants, the holders thereof will be
entitled to exercise the rights of a holder of our ordinary shares only as to matters for which the record date occurs after the exercise
date.
We
conduct some of our operations in Israel. Conditions in Israel, including the recent attack by Hamas and other terrorist organizations
from the Gaza Strip and Israel’s war against them, may affect our operations.
Our
corporate headquarters is located in Herzliya, Israel. Because we are incorporated under the laws of the State of Israel, and most of
our officers and fourteen out of sixteen of our employees are residents of Israel, our business and operations are directly affected
by economic, political, geopolitical and military conditions in Israel. Since the establishment of the State of Israel in 1948, a number
of armed conflicts have occurred between Israel and its neighboring countries and terrorist organizations active in the region. These
conflicts have involved missile strikes, hostile infiltrations and terrorism against civilian targets in various parts of Israel, which
have negatively affected business conditions in Israel.
In
October 2023, Hamas terrorists infiltrated Israel’s southern border from the Gaza Strip and conducted a series of attacks on civilian
and military targets. Hamas also launched extensive rocket attacks on Israeli population and industrial centers located along Israel’s
border with the Gaza Strip and in other areas within the State of Israel. These attacks resulted in thousands of deaths and injuries,
and Hamas additionally kidnapped many Israeli civilians and soldiers. Following the attack, Israel’s security cabinet declared
war against Hamas and a military campaign against these terrorist organizations commenced in parallel to their continued rocket and terror
attacks. In parallel, border clashes between Israel and the Hezbollah terrorist group on Israel’s northern border with Lebanon
intensified and may escalate into a greater regional conflict.
In
addition, since the commencement of these events, there have been continued hostilities along Israel’s northern border with
Lebanon (with the Hezbollah terror organization) and southern border (with the Houthi movement in Yemen). It is possible that
hostilities with Hezbollah in Lebanon will continue escalate, and that other terrorist organizations, including Palestinian military
organizations in the West Bank as well as other hostile countries will join the hostilities. In addition, Iran (in concert with
other regional actors) recently launched a direct attack on Israel involving hundreds of drones and missiles and has threatened to
continue to attack Israel and is widely believed to be developing nuclear weapons. Iran is also believed to have a strong influence
among extremist groups in the region, such as Hamas in Gaza, Hezbollah in Lebanon, the Houthi movement in Yemen and various rebel
militia groups in Syria and Iraq. These situations may potentially escalate in the future to more violent events which may affect
Israel and us. Additionally, Yemeni rebel group, the Houthis, launched series of attacks on global shipping routes in the Red Sea,
causing disruptions of supply chain. These geopolitical developments may adversely affect our ability to continue carrying out
various administrative, research, operational and commercial functions and activities both in Israel and globally. Furthermore,
as a result of the war, the international rating agency, Moody’s and S&P, has cut Israel’s credit rating and
has also lowered Israel’s outlook from stable to negative, stating that it sees a possible further downgrade in the future.
Lowered credit rating of Israel could materially impact our ability to raise capital and ability to secure loans, if needed, in
each case on reasonable terms.
Any
hostilities, armed conflicts, terrorist activities involving Israel or the interruption or curtailment of trade between Israel and its
trading partners, or any political instability in the region could directly or indirectly adversely affect business conditions and our
results of operations and could make it more difficult for us to raise capital and could adversely affect the market price of our ordinary
share. An escalation of tensions or violence might result in a significant downturn in the economic or financial condition of Israel,
which could have a material adverse effect on our operations in Israel and our business. Parties with whom we do business have sometimes
declined to travel to Israel during periods of heightened unrest or tension, forcing us to make alternative arrangements when necessary
in order to meet our business partners face to face. In addition, the political and security situation in Israel may result in parties
with whom we have agreements involving performance in Israel claiming that they are not obligated to perform their commitments under
those agreements pursuant to force majeure provisions in such agreements.
The
Israel Defense Force, or IDF, the national military of Israel, is a conscripted military service, subject to certain exceptions.
Since October 7, 2023, the IDF has called up several hundred thousand of its reserve forces to serve. Fourteen out of our current 16
employees are resident in Israel. Three of our five executive officers and four out of 11 other employees who we believe are
performing critical Company functions, reside in Israel. Two of our non-management employees in Israel who do not perform critical
Company functions have been called up, and additional employees may be called for service in the current or future wars or other
armed conflicts with Hamas, Hezbollah or other regional threat actors, and such persons may be absent for an extended period of
time. As a result, our operations in Israel may be disrupted by such absences, which disruption may materially and adversely affect
our business, prospects, financial condition and results of operations.
Since
the war broke out on October 7, 2023, our operations have not been adversely affected by this situation, and we have not experienced
disruptions to our clinical studies. As such, our clinical and business development activities remain on track. However, the
intensity and duration of Israel’s current war against Hamas and Hezbollah is difficult to predict at this stage, as are such
war’s economic implications on our business and operations and on Israel’s economy in general. If the war extends for a
long period of time or expands to other fronts, such as Iran, Lebanon, Syria and the West Bank, our operations may be adversely
affected.
All
of our clinical and pre-clinical research and development is currently being conducted outside of Israel, other
than our 12-month open label-extension, or OLE, study of the PARADIGM trial, partially conducted in Tel Aviv, and
a planned Phase 2 trial with PrimeC for AD that we plan to conduct in Haifa, Israel. The OLE has not been affected
by the war, although the quality of the study may be adversely affected if as a result of the war patients are unable to visit the study
center or the study coordinator is not able to conduct home visits and monitor the patients. In addition, in the event of a significant
escalation of hostilities in northern Israel, there may be a delay in the planned AD trial. We do not believe the planned
AD trial will be materially affected by the war and do not anticipate that any such delay as a result of the war would have a material
impact on us. We may also elect to set up a site in Israel for a Phase 3 pivotal ALS trial of PrimeC, but this would be in addition to
numerous other sites in Europe and the United States, and as a result we do not expect the timeline or quality of this trial to be adversely
affected by the war. Our manufacturing is conducted in India. We do not currently anticipate any disruption to the supply chain relevant
to our ongoing clinical trials and believe there are alternative sources of supply from whom we could obtain the necessary finished drug
product to conduct our clinical trials. In addition, we believe we have sufficient finished product in inventory to continue our ongoing
clinical trials for at least the next few months.
Our
commercial insurance does not cover losses that may occur as a result of an event associated with the security situation in the Middle
East. Although the Israeli government is currently committed to covering the reinstatement value of direct damages that are caused by
terrorist attacks or acts of war, there can be no assurance that this government coverage will be maintained, or if maintained, will
be sufficient to compensate us fully for damages incurred. Any losses or damages incurred by us could have a material adverse effect
on our business, financial condition and results of operations.
Finally,
political conditions within Israel may affect our operations. Israel has held five general elections between 2019 and 2022, and prior
to October 2023, the Israeli government pursued extensive changes to Israel’s judicial system, which sparked extensive political
debate and unrest. To date, these initiatives have been substantially put on hold. Actual or perceived political instability in Israel
or any negative changes in the political environment, may individually or in the aggregate adversely affect the Israeli economy and,
in turn, our business, financial condition, results of operations and growth prospects.
THE
PRIVATE PLACEMENT
On April 10, 2024, we agreed,
(i) pursuant to the Purchase Agreement, to issue to a selling shareholder Ordinary Warrants exercisable for an aggregate of 2,980,000 ordinary
shares at an exercise price of $1.50 per share (the “Private Placement”); and (ii) to issue 70,964 ordinary shares to our
placement agent as partial consideration in exchange for their services, in connection with the Purchase Agreement. Each Ordinary Warrant
became exercisable on the date of issuance and will remain exercisable until the fifth anniversary of the original issuance date. The
Shares, the PA Shares and Ordinary Warrants were offered pursuant to the exemption provided in Section 4(a)(2) under the Securities
Act, and Rule 506(b) promulgated thereunder.
Pursuant to the terms of
the Purchase Agreement, we agreed to use commercially reasonable efforts to cause this registration statement on Form F-1 providing
for the resale by holders of ordinary shares issuable upon the exercise of the Ordinary Warrants, within 60 days, or within 90 days of
the filing of the registration statement in the event of a full review by the SEC, and to keep such registration statement effective as
provided in the securities purchase agreement.
The
foregoing descriptions of the form of Purchase Agreement and the form of Ordinary Warrant are not complete and are subject to and qualified
in their entirety by reference to the form of Purchase Agreement and the form of Ordinary Warrant, respectively, copies of which are
attached as Exhibits 10.2 and 10.3 respectively, to our Report on Form 6-K filed with the SEC on April 12, 2024, and are incorporated
herein by reference.
USE
OF PROCEEDS
All of the Shares, PA Shares
and Ordinary Warrants offered by the selling shareholders for resale pursuant to this prospectus will be sold by the selling shareholders.
We will not receive any of the proceeds from such sales. The selling shareholders will receive all of the proceeds from any sales of the
Shares, PA Shares and Ordinary Warrants offered hereby. However, we will incur expenses in connection with the registration of the Shares,
PA Shares and Ordinary Warrants offered hereby.
We will receive the exercise price upon any exercise of the Ordinary
Warrant, to the extent exercised on a cash basis. If all the Ordinary Warrants were exercised, we would receive gross proceeds of approximately
$4.5 million. However, the holders of the Ordinary Warrants are not obligated to exercise the Ordinary Warrants, and we cannot predict
whether or when, if ever, the holders of the Ordinary Warrants will choose to exercise the Ordinary Warrants, in whole or in part. The
exercise price of the Ordinary Warrants may exceed the trading price of our ordinary shares. If the price of our ordinary shares is below
$1.50, we believe that holder of the Ordinary Warrants will be unlikely to exercise their warrants, resulting in little to no cash proceeds
to us. Accordingly, we currently intend to use the proceeds received upon such exercise, if any, for general corporate purposes and working
capital.
MARKET
FOR ORDINARY SHARES AND DIVIDEND POLICY
Our ordinary shares are traded on the Nasdaq Capital Market under the
symbol “NRSN.” The last reported sale price of our ordinary shares on May 9, 2024 on the Nasdaq Capital Market was $1.1738
per share. We do not plan on applying to list the Ordinary Warrants on any exchange or nationally recognized trading system.
We
have never declared or paid any cash dividends on our ordinary shares, and we anticipate that, for the foreseeable future, we will retain
any future earnings to support operations and to finance the growth and development of our business. Therefore, we do not expect to pay
cash dividends for at least the next several years.
The
distribution of dividends may also be limited by the Israeli Companies Law, 5759-1999 (the “Companies Law”), which permits
the distribution of dividends only out of retained earnings or earnings derived over the two most recent fiscal years, whichever
is higher, provided that there is no reasonable concern that payment of a dividend will prevent a company from satisfying its existing
and foreseeable obligations as they become due. As of December 31, 2023, we did not have distributable earnings pursuant to the
Companies Law. Dividend distributions may be determined by our board of directors, as our amended and restated articles of association
do not provide that such distributions require shareholder approval.
CAPITALIZATION
The
following table sets forth our cash and cash equivalents and capitalization as of December 31, 2023 as follows:
The
following table should be read in conjunction with “Use of Proceeds,” our financial statements and related notes that are
incorporated by reference into this prospectus and the other financial information included or incorporated by reference into this prospectus.
Our historical results do not necessarily indicate our expected results for any future periods.
| | |
As of
December 31, 2023 | |
| | |
(in thousands) | |
| Cash | |
$ | 2,640 | |
| Liability in respect to warrants and pre-funded warrants | |
| 1,518 | |
| Shareholders’ equity: | |
| | |
| Ordinary shares, no par value per share | |
| — | |
| Share premium and capital reserve | |
| 30,192 | |
| Accumulated deficit | |
| (32,066 | ) |
| Total shareholders’ equity | |
| (1,874 | ) |
| | |
| | |
| Total capitalization | |
$ | (356 | ) |
SELLING
SHAREHOLDERS
The ordinary shares being
offered by the selling shareholders pursuant to this prospectus are those issuable to such selling shareholder upon exercise of the Ordinary
Warrants, the Ordinary Warrants and the PA Shares. For additional information regarding the issuance of the Ordinary Warrants and PA Shares,
see “The Private Placement” above. The PA Shares being offered by the selling shareholder for resale pursuant to this prospectus
are those which were issued to such selling shareholder as partial consideration for placement agent services in connection with the April
2024 Offering. We are registering the Shares, PA Shares and Ordinary Warrants in order to permit the selling shareholders to offer the
Shares, PA Shares and Ordinary Warrants for resale from time to time. Except for the ownership of securities issued pursuant to prior
financings and other than in the case of the placement agent which acted as a placement agent in a prior financing in June 2023, to our
knowledge, the selling shareholders have not had any material relationship with us or our affiliates within the past three years.
Our knowledge is based on information provided by the selling shareholders in connection with the filing of this prospectus.
The table below lists the
selling shareholders and other information regarding the beneficial ownership of the Shares, Ordinary Warrants and the PA Shares by the
selling shareholders. The second column lists the number of our ordinary shares beneficially owned by the selling shareholder, based on
its ownership of our ordinary shares, as of May 9, 2024, and assuming exercise of all of the warrants held by the selling shareholder
on that date, without regard to any limitations on exercises. The third column lists the maximum number of our ordinary shares that may
be sold or otherwise disposed of by the selling shareholders pursuant to the registration statement of which this prospectus forms a part.
The selling shareholders may sell or otherwise dispose of some, all or none of their Shares, Ordinary Warrants or PA Shares in this offering.
Pursuant to the rules of the SEC, beneficial ownership includes any of the ordinary shares as to which a shareholder has sole or shared
voting power or investment power, as well as any ordinary shares that a selling shareholder has the right to acquire within 60 days
of May 9, 2024. The percentage of beneficial ownership for the selling shareholders is based on 17,405,006 ordinary shares outstanding
as of May 9, 2024 and the number of ordinary shares issuable upon exercise or conversion of convertible securities that are currently
exercisable or convertible or are exercisable or convertible within 60 days of May 9, 2024 beneficially owned by the applicable selling
shareholder. The fourth column assumes the sale of all of the Shares, Ordinary Warrants and PA Shares offered by the selling shareholders
pursuant to this prospectus.
Under the terms of the Ordinary Warrants, the selling shareholder may
not exercise the Ordinary Warrants to the extent such exercise would cause the selling shareholder, together with its affiliates and attribution
parties, to beneficially own a number of ordinary shares which would exceed 4.99% of our then outstanding ordinary shares immediately
after giving effect to the issuance of Shares upon exercise of the Ordinary Warrants held by the selling shareholder. Furthermore, under
the terms of the Pre-Funded Warrants, the selling shareholder may not exercise the Pre-Funded Warrants to the extent such exercise would
cause the selling shareholder, together with its affiliates and attribution parties, to beneficially own a number of ordinary shares which
would exceed 9.99% of our then outstanding ordinary shares immediately after giving effect to the issuance of ordinary shares upon exercise
of the Pre-Funded Warrants held by the selling shareholder. The number of ordinary shares in the second and fifth columns do not reflect
these limitations. See “Plan of Distribution.”
| Name of Selling Shareholder |
|
Ordinary
Shares
Beneficially
Owned Prior
to the
Offering(1) |
|
|
Percentage of
Outstanding
Ordinary
Shares(1) |
|
|
Maximum
Number of
Ordinary
Shares and PA
Shares
To Be Sold
Pursuant
to this
Prospectus |
|
|
Number of Ordinary Shares Beneficially Owned
After the
Offering(2) |
|
|
Percentage of
Outstanding
Ordinary
Shares after the
Offering(2) |
|
| Armistice Capital Master Fund Ltd. |
|
|
8,444,000 |
(3) |
|
|
34.3 |
% |
|
|
2,980,000 |
|
|
|
5,464,000 |
|
|
|
26.8 |
% |
| A.G.P./Alliance Global Partners |
|
|
70,964 |
(4) |
|
|
0.4 |
% |
|
|
70,964 |
|
|
|
- |
|
|
|
- |
|
| (1) | Assumes all warrants held by the selling shareholder on May 9, 2024
are exercised. |
| (2) |
Assumes
that (i) all of the Shares covered by the registration statement of which this prospectus is a part are sold in this offering and (ii)
the selling shareholders do not acquire additional ordinary shares after the date of this prospectus and prior to completion of this
offering. The percentage of beneficial ownership after the offering is based on 20,385,006 ordinary shares outstanding, consisting of
(a) 17,405,006 ordinary shares outstanding on May 9, 2024, and (b) the 2,980,000 ordinary shares offered for resale under this prospectus.
The number and percentage of ordinary shares listed do not take into account any limitations on exercise of warrants preventing the Master
Fund (as defined below) from exercising any portion of such warrants if such exercise would result in the Master Fund owning greater
than 4.99% or 9.99%, as applicable, of the outstanding ordinary shares following such exercise. |
| (3) | Consists of (i) 1,216,000 ordinary shares, (ii) 1,248,000 ordinary
shares issuable upon exercise of the Pre-Funded Warrants, and (iii) 5,980,000 ordinary shares issuable upon the exercise of warrants,
including the Ordinary Warrants. The securities are directly held by Armistice Capital Master Fund Ltd., a Cayman Islands exempted company
(the “Master Fund”), and may be deemed to be beneficially owned by: (i) Armistice Capital, LLC (“Armistice Capital”),
as the investment manager of the Master Fund; and (ii) Steven Boyd, as the Managing Member of Armistice Capital. The warrants are subject
to a beneficial ownership limitation of 4.99% or 9.99%, as applicable, which such limitation restricts the selling shareholder from exercising
that portion of the warrants that would result in the selling stockholder and its affiliates owning, after exercise, a number of ordinary
shares in excess of the beneficial ownership limitation. The number of ordinary shares set forth in the above table does not reflect the
application of this limitation. The address of Armistice Capital Master Fund Ltd. is c/o Armistice Capital, LLC, 510 Madison Avenue, 7th
Floor, New York, NY 10022. |
| (4) | Consists of 70,964 ordinary shares issued to the selling shareholder,
a registered broker-dealer, in consideration for placement agent and certain investment banking services rendered in connection with the
April 2024 Offering. The business address of the selling shareholder is 590 Madison Avenue, 28th Floor, New York, NY 10022. |
PLAN
OF DISTRIBUTION
The
selling shareholders and any of their pledgees, assignees and successors-in-interest may, from time to time, sell any or all of their
securities covered hereby on the Nasdaq Capital Market or any other stock exchange, market or trading facility on which the securities
are traded or in private transactions. These sales may be at fixed or negotiated prices. A selling shareholder may use any one or more
of the following methods when selling securities:
| ● | ordinary
brokerage transactions and transactions in which the broker-dealer solicits purchasers; |
| ● | block
trades in which the broker-dealer will attempt to sell the securities as agent but may position
and resell a portion of the block as principal to facilitate the transaction; |
| ● | purchases
by a broker-dealer as principal and the subsequent resale by the broker-dealer for its account; |
| ● | an
exchange distribution in accordance with the rules of the applicable exchange; |
| ● | an
over-the-counter distribution in accordance with the rules of Nasdaq; |
| ● | through
trading plans entered into by the selling shareholders pursuant to Rule 10b5-1 under
the Exchange Act that are in place at the time of an offering pursuant to this prospectus
and any applicable prospectus supplement hereto that provide for periodic sales of their
securities on the basis of parameters described in such trading plans; |
| ● | transactions
other than on such exchanges or in the over-the-counter market; |
| ● | directly
to purchasers, including through a specific bidding, auction or other process or in privately
negotiated transactions; |
| ● | settlement
of short sales; |
| ● | in
transactions through broker-dealers that agree with the selling shareholders to sell a specified
number of such securities at a stipulated price per security; |
| ● | through
the writing or settlement of options or other hedging transactions, whether through an options
exchange or otherwise; |
| ● | a
combination of any such methods of sale; or |
| ● | any
other method permitted pursuant to applicable law. |
The
selling shareholders may also sell securities under Rule 144 or any other exemption from registration under the Securities Act, if
available, rather than under this prospectus.
Broker-dealers
engaged by the selling shareholders may arrange for other brokers-dealers to participate in sales. Broker-dealers may receive commissions
or discounts from the selling shareholders (or, if any broker-dealer acts as agent for the purchaser of securities, from the purchaser)
in amounts to be negotiated, but, except as set forth in a supplement to this prospectus, in the case of an agency transaction not in
excess of a customary brokerage commission in compliance with FINRA Rule 2121; and in the case of a principal transaction a markup
or markdown in compliance with FINRA Rule 2121.
In
connection with the sale of the securities or interests therein, the selling shareholders may enter into hedging transactions with broker-dealers
or other financial institutions, which may in turn engage in short sales of the securities in the course of hedging the positions they
assume. The selling shareholders may also sell securities short and deliver these securities to close out their short positions, or loan
or pledge the securities to broker-dealers that in turn may sell these securities. The selling shareholders may also enter into option
or other transactions with broker-dealers or other financial institutions or create one or more derivative securities which require the
delivery to such broker-dealer or other financial institution of securities offered by this prospectus, which securities such broker-dealer
or other financial institution may resell pursuant to this prospectus (as supplemented or amended to reflect such transaction).
The selling shareholders
and any broker-dealers or agents that are involved in selling the securities may be deemed to be “underwriters” within the
meaning of the Securities Act in connection with such sales. In such event, any commissions received by such broker-dealers or agents
and any profit on the resale of the securities purchased by them may be deemed to be underwriting commissions or discounts under the Securities
Act. The selling shareholders have informed us that they do not have any written or oral agreement or understanding, directly or indirectly,
with any person to distribute the securities.
We are required to pay certain
fees and expenses incurred by us incident to the registration of the resale of the Shares, Ordinary Warrants and PA Shares covered by
this prospectus. We have agreed to indemnify the selling shareholders against certain losses, claims, damages and liabilities, including
liabilities under the Securities Act.
DESCRIPTION
OF OUR SECURITIES BEING REGISTERED
The securities to be registered
for resale on this registration statement on Form F-1 of which this prospectus forms a part include up to an aggregate amount of
(i) 2,980,000 ordinary shares issuable upon exercise of Ordinary Warrants at the exercise price of $1.50, acquired by the selling
shareholder under the Purchase Agreement, (ii) the Ordinary Warrants, and (ii) 70,964 ordinary shares issued to our placement agent as
partial consideration in exchange for their services, in connection with the April 2024 Offering.
Our Ordinary
Shares
Our authorized share capital
consists of 60,000,000 ordinary shares, no par value per share, of which 17,405,006 ordinary shares were issued and outstanding as
of May 9, 2024.
All of the outstanding ordinary
shares are validly issued, fully paid and non-assessable. Our ordinary shares are not redeemable and do not have any pre-emptive rights.
Voting
Rights and Conversion
All
ordinary shares will have identical voting and other rights in all respects. None of our major shareholders have different voting rights
than our other shareholders.
Transfer
of Shares
Fully paid ordinary shares
are issued in registered form and may be freely transferred under our amended and restated articles of association, unless the transfer
is restricted or prohibited by another instrument, applicable law or the rules of a stock exchange on which the shares are listed for
trading. The ownership or voting of ordinary shares by non-residents of Israel is not restricted in any way by the amended and restated
articles of association or the laws of the State of Israel, except for ownership by nationals of some countries that are, or have been,
in a state of war with Israel.
Liability
to Further Capital Calls
Our board of directors may
make, from time to time, such calls as it may deem fit upon shareholders with respect to any sum unpaid with respect to ordinary shares
held by such shareholders, which is not payable at a fixed time. Such shareholder shall pay the amount of every call so made upon him.
Unless otherwise stipulated by our board of directors, each payment in response to a call shall be deemed to constitute a pro rata payment
on account of all shares with respect to which such call was made. A shareholder shall not be entitled to his rights as shareholder, including
the right to dividends, unless such shareholder has fully paid all the notices of call delivered to him, or which according to our amended
and restated articles of association are deemed to have been delivered to him, together with interest, linkage and expenses, if any, unless
otherwise determined by our board of directors.
Business
Combinations
Under our amended and restated
articles of association, we may not engage in any “business combinations” with any “interested shareholder” for
a three-year period following the time that such shareholder became an interested shareholder, unless:
| ● | prior
to the time that such shareholder became an interested shareholder, our board of directors
approved either the business combination or the transaction which resulted in the shareholder
becoming an interested shareholder; |
| ● | upon
consummation of the transaction which resulted in the shareholder becoming an interested
shareholder, the interested shareholder owned at least 85% of our voting shares outstanding
at the time the transaction commenced, excluding for purposes of determining our voting shares
outstanding (but not the outstanding voting shares owned by the interested shareholder)
those shares owned by persons who are directors and also officers; or |
| ● | at
the time that such shareholder became an interested shareholder, or subsequent to such time,
the business combination is approved by our board of directors and authorized at a general
meeting of shareholders by the affirmative vote of at least 662/3% of our voting shares outstanding
that are not owned by the interested shareholder. |
Generally,
a “business combination” includes any merger, consolidation, sale or other transaction resulting in a financial benefit to
the interested shareholder. Subject to certain exceptions, an “interested shareholder” is any person (other than us and any
of our direct or indirect majority-owned subsidiaries) who, together with such person’s affiliates and associates, owns, or within
the previous three years owned, 15% or more of our voting shares.
Under
certain circumstances, this provision will make it more difficult for a person who would be an “interested shareholder” to
effect various business combinations with a corporation for a three-year period. This provision may encourage companies interested in
acquiring us to negotiate in advance with our board of directors because the shareholder approval requirement would be avoided if our
board of directors approves either the business combination or the transaction which results in the shareholder becoming an interested
shareholder. These provisions also may have the effect of preventing changes in our board of directors and may make it more difficult
to accomplish transactions which shareholders may otherwise deem to be in their best interests.
Election
of Directors
Our
ordinary shares do not have cumulative voting rights for the election of directors. As a result, the holders of a majority of the voting
power represented at a shareholders meeting have the power to elect our directors, subject to the special approval requirements for external
directors under the Companies Law. Under our amended and restated articles of association, the number of directors on our board of directors
must be no less than three and no more than nine, including any external directors required to be appointed under the Companies Law.
The minimum and maximum number of directors may be changed, at any time and from time to time, by a special vote of the holders of at
least 662/3 of the outstanding shares.
Other
than external directors, for whom special election requirements apply under the Companies Law, the vote required to appoint a director
is a simple majority vote. In addition, under our amended and restated articles of association, our board of directors may elect new
directors to fill vacancies (whether such vacancy is due to a director no longer serving or due to the number of directors serving being
less than the maximum required in our amended and restated articles of association), provided that the total number of directors shall
not, at any time, exceed nine directors and provided that our board of directors may not elect external directors. Our amended and restated
articles of association provide that the term of a director appointed by our board of directors to fill any vacancy will be for the remaining
term of office of the director(s) whose office(s) have been vacated. Furthermore, under our amended and restated articles of
association, our directors, other than external directors, are divided into three classes with staggered three-year terms. Each class
of directors consists, as nearly as possible, of 1/3 of the total number of directors constituting the entire board of directors (other
than the external directors).
External
directors are elected for an initial term of three years, may be elected for additional three-year terms, and may be removed from
office pursuant to the terms of the Companies Law.
Dividend
and Liquidation Rights
We
may declare a dividend to be paid to the holders of our ordinary shares in proportion to their respective shareholdings. Under the Companies
Law, dividend distributions are determined by our board of directors and do not require the approval of the shareholders of a company
unless our articles of association provide otherwise. Our amended and restated articles of association do not require shareholder approval
of a dividend distribution and provide that dividend distributions may be determined by our board of directors.
Pursuant
to the Companies Law, subject to certain exceptions with the respect to the buyback by us of our ordinary shares, the distribution amount
is limited to the greater of retained earnings or earnings generated over the previous two years, according to our then last reviewed
or audited financial statements (less the amount of previously distributed dividends, if not reduced from the earnings), provided that
the end of the period to which the financial statements relate is not more than six months prior to the date of the distribution.
If we do not meet such criteria, then the we may distribute dividends only with court approval. In each case, we are only permitted to
distribute a dividend if our board of directors and, if applicable, the court determines that there is no reasonable concern that payment
of the dividend will prevent us from satisfying our existing and foreseeable obligations as they become due. In the event of our liquidation,
after satisfaction of liabilities to creditors, our assets will be distributed to the holders of our ordinary shares in proportion to
their shareholdings. This right, as well as the right to receive dividends, may be affected by the grant of preferential dividend or
distribution rights to the holders of a class of shares with preferential rights that may be authorized in the future.
Exchange
Controls
There
are currently no Israeli currency control restrictions on remittances of dividends on our ordinary shares, proceeds from the sale of
ordinary shares or interest or other payments to non-residents of Israel, except for shareholders who are subjects of certain countries
that are, or have been, in a state of war with Israel at such time.
Shareholder
Meetings
Under
Israeli law, we are required to hold an annual general meeting of our shareholders once every calendar year that must be held no later
than 15 months after the date of the previous annual general meeting. All general meetings other than the annual meeting of shareholders
are referred to in our amended and restated articles of association as special meetings. Our board of directors may call special meetings
whenever it sees fit, at such time and place, within or outside of Israel, as it may determine. In addition, the Companies Law provides
that our board of directors is required to convene a special general meeting upon the written request of (i) any two of our directors
or one-quarter of the members of our board of directors or (ii) one or more shareholders holding, in the aggregate, either (a) 5%
or more of our outstanding issued shares and 1% or more of our outstanding voting power or (b) 5% or more of our outstanding voting
power.
Under
Israeli law, one or more shareholders holding at least 1% of the voting rights at the general meeting may request that the board of directors
include a matter in the agenda of a general meeting to be convened in the future, such as nominating a director candidate, provided that
it is appropriate to discuss such a matter at the general meeting. Our amended and restated articles of association contain procedural
guidelines and disclosure items with respect to the submission of shareholder proposals for shareholders meetings.
Subject
to the provisions of the Companies Law and the regulations promulgated thereunder, shareholders entitled to participate and vote at general
meetings are the shareholders of record on a date to be decided by the board of directors, which may be between four and 40 days
prior to the date of the meeting. Furthermore, the Companies Law requires that resolutions regarding, among other things, the following
matters must be passed at a general meeting of our shareholders:
| ● | amendments
to our amended and restated articles of association; |
| ● | appointment
or termination of our auditors; |
| ● | election
of directors, including external directors (unless otherwise determined in our amended and
restated articles of association); |
| ● | approval
of certain related party transactions; |
| ● | increases
or reductions of our authorized share capital; |
| ● | the
exercise of our board of directors’ powers by a general meeting, if our board of directors
is unable to exercise its powers and the exercise of any of its powers is required for our
proper management. |
Under
our amended and restated articles of association, we are not required to give notice to our registered shareholders pursuant to the Companies
Law, unless otherwise required by law. The Companies Law requires that a notice of any annual general meeting or special general meeting
be provided to shareholders at least 21 days prior to the meeting and if the agenda of the meeting includes the appointment or removal
of directors, the approval of transactions with office holders or interested or related parties, or an approval of a merger, or as otherwise
required under applicable law, notice must be provided at least 35 days prior to the meeting. Under the Companies Law, shareholders
of a public company are not permitted to take action by written consent in lieu of a meeting.
Voting
Rights
Quorum
Requirements
Pursuant
to our amended and restated articles of association, holders of our ordinary shares have one vote for each ordinary share held on all
matters submitted to a vote before the shareholders at a general meeting. Under our amended and restated articles of association, the
quorum required for general meetings of shareholders must consist of at least two shareholders present in person or by proxy (including
by voting deed) holding 25% or more of our voting rights. A meeting adjourned for lack of a quorum will generally be adjourned to the
same day of the following week at the same time and place, or to such other day, time or place as indicated by our board of
directors if so specified in the notice of the meeting. At the reconvened meeting, any number of shareholders present in person or by
proxy shall constitute a lawful quorum.
Vote
Requirements
Our
amended and restated articles of association provide that all resolutions of our shareholders require a simple majority vote, unless
otherwise required by the Companies Law or by our amended and restated articles of association. Our amended and restated articles of
association provide that all resolutions of our board of directors require a simple majority vote of the directors present and voting
at such meeting, unless otherwise required by the Companies Law or by our amended and restated articles of association. Pursuant to our
amended and restated articles of association, in the event the vote is tied, the chair of the board of directors will have a casting
vote.
Pursuant
to our amended and restated articles of association, an amendment to our amended and restated articles of association regarding any change
of the composition or election procedures of our directors will require a special majority vote (66⅔%). In addition, any change
to the rights and privileges of the holders of any class of our shares requires a simple majority of the class so affected (or otherwise
in accordance with the terms of such class), in addition to the simple majority vote of all classes of shares voting together as a single
class at a shareholder meeting.
Under
the Companies Law, each of (i) the approval of an extraordinary transaction with a controlling shareholder and (ii) the terms
of employment or other engagement of the controlling shareholder of the company or such controlling shareholder’s relative (even
if not extraordinary) requires the approval of the compensation committee, board of directors, and shareholders, subject to limited exceptions.
Certain transactions with respect to remuneration of our office holders and directors require approvals of the compensation committee,
board of directors, and in the case of the chief executive officer and directors, shareholders, subject to limited exceptions Another
exception to the simple majority vote requirement is a resolution for the voluntary winding up, or an approval of a scheme of arrangement
or reorganization, of the company pursuant to Section 350 of the Companies Law, which requires the approval of holders of 75% of
the voting rights represented at the meeting, in person or by proxy and voting on the resolution.
Access
to Corporate Records
Under
the Companies Law, shareholders are provided access to: minutes of our general meetings; our shareholders register and material shareholders
register; our amended and restated articles of association; our financial statements; and any document that we are required by law to
file publicly with the Israeli Companies Registrar (the “ISA”). In addition, shareholders may request to be provided with
any document related to an action or transaction requiring shareholder approval under the related party transaction provisions of the
Companies Law. We may deny this request if we believe it has not been made in good faith or if such denial is necessary to protect our
interest or protect a trade secret or patent.
Modification
of Class Rights
Under
the Companies Law and our amended and restated articles of association, the rights attached to any class of share, such as voting, liquidation
and dividend rights, may be amended by adoption of a resolution by the holders of a majority of the shares of that class present at a
separate class meeting, or otherwise in accordance with the terms of such class of shares, in addition to the simple majority vote of
all classes of shares voting together as a single class at a shareholder meeting, as set forth in our amended and restated articles of
association.
Acquisitions
Under Israeli Law
Full
Tender Offer. A person wishing to acquire shares of an Israeli public company and who would as a result hold over 90% of the target
company’s issued and outstanding share capital is required by the Companies Law to make a tender offer to all of the company’s
shareholders for the purchase of all of the issued and outstanding shares of the company. A person wishing to acquire shares of an Israeli
public company and who would as a result hold over 90% of the issued and outstanding share capital of a certain class of shares is required
to make a tender offer to all of the shareholders who hold shares of the relevant class for the purchase of all of the issued and outstanding
shares of that class. If the shareholders who do not accept the offer hold less than 5% of the issued and outstanding share capital of
the company or of the applicable class, and more than half of the shareholders who do not have a personal interest in the offer accept
the offer, all of the shares that the acquirer offered to purchase will be transferred to the acquirer by operation of law. However,
a tender offer will also be accepted if the shareholders who do not accept the offer hold less than 2% of the issued and outstanding
share capital of the company or of the applicable class of shares.
Upon
a successful completion of such a full tender offer, any shareholder that was an offeree in such tender offer, whether such shareholder
accepted the tender offer or not, may, within six months from the date of acceptance of the tender offer, petition an Israeli court
to determine whether the tender offer was for less than fair value and that the fair value should be paid as determined by the court.
However, under certain conditions, the offeror may include in the terms of the tender offer that an offeree who accepted the offer will
not be entitled to petition the Israeli court as described above.
If
(a) the shareholders who did not respond or accept the tender offer hold at least 5% of the issued and outstanding share capital
of the company or of the applicable class or the shareholders who accept the offer constitute less than a majority of the offerees that
do not have a personal interest in the acceptance of the tender offer or (b) the shareholders who did not accept the tender offer
hold 2% or more of the issued and outstanding share capital of the company (or of the applicable class), the acquirer may not acquire
shares of the company that will increase its holdings to more than 90% of the company’s issued and outstanding share capital or
of the applicable class from shareholders who accepted the tender offer.
Special
Tender Offer. The Companies Law provides that an acquisition of shares of an Israeli public company must be made by means of a special
tender offer if as a result of the acquisition the purchaser would become a holder of 25% or more of the voting rights in the company.
Similarly, the Companies Law provides that an acquisition of shares in a public company must be made by means of a special tender offer
if as a result of the acquisition the purchaser would become a holder of more than 45% of the voting rights in the company. These requirements
do not apply if, in general, the acquisition (1) was made in a private placement that received shareholder approval, (2) was
from a 25% or greater shareholder of the company which resulted in the acquirer becoming a 25% or greater shareholder of the company,
or (3) was from a 45% or greater shareholder of the company which resulted in the acquirer becoming a 45% or greater shareholder
of the company.
A
special tender offer must be extended to all shareholders of a company but the offeror is not required to purchase shares representing
more than 5% of the voting power attached to the company’s outstanding shares, regardless of how many shares are tendered by shareholders.
A special tender offer may be consummated only if (i) at least 5% of the voting power attached to the company’s outstanding
shares will be acquired by the offeror and (ii) the number of shares tendered in the offer exceeds the number of shares whose holders
objected to the offer (excluding the purchaser and its controlling shareholders, holders of 25% or more of the voting rights in the company
or any person having a personal interest in the acceptance of the tender offer or any other person acting on their behalf, including
the relatives and entities under such person’s control). If a special tender offer is accepted, then the purchaser or any person
or entity controlling it or under common control with the purchaser or such controlling person or entity may not make a subsequent tender
offer for the purchase of shares of the target company and may not enter into a merger with the target company for a period of one year
from the date of the offer, unless the purchaser or such person or entity undertook to effect such an offer or merger in the initial
special tender offer.
Merger.
The Companies Law permits merger transactions if approved by each party’s board of directors and, unless certain requirements
described under the Companies Law are met, by a majority vote of each party’s shares, and, in the case of the target company, a
majority vote of each class of its shares, voted on the proposed merger at a shareholders meeting.
For
purposes of the shareholder vote, unless a court rules otherwise, the merger will not be deemed approved if a majority of the votes of
shares represented at the shareholders meeting that are held by parties other than the other party to the merger, or by any person (or
group of persons acting in concert) who holds (or hold, as the case may be) 25% or more of the voting rights or the right to appoint
25% or more of the directors of the other party, vote against the merger. If, however, the merger involves a merger with a company’s
own controlling shareholder or if the controlling shareholder has a personal interest in the merger, then the merger is instead subject
to the same special majority approval that governs all extraordinary transactions with controlling shareholders. See “Management — Approval
of Related Party Transactions under Israeli Law — Fiduciary Duties of Directors and Officers — Disclosure
of Personal Interests of an Office Holder and Approval of Certain Transactions.”
If
the transaction would have been approved by the shareholders of a merging company but for the separate approval of each class or the
exclusion of the votes of certain shareholders as provided above, a court may still approve the merger upon the request of holders of
at least 25% of the voting rights of a company, if the court holds that the merger is fair and reasonable, taking into account the value
to the parties to the merger and the consideration offered to the shareholders of the company.
Upon
the request of a creditor of either party to the proposed merger, the court may delay or prevent the merger if it concludes that there
exists a reasonable concern that, as a result of the merger, the surviving company will be unable to satisfy the obligations of the merging
entities, and may further give instructions to secure the rights of creditors.
In
addition, a merger may not be consummated unless at least 50 days have passed from the date on which a proposal for approval of
the merger was filed by each party with the IRC and at least 30 days have passed from the date on which the merger was approved
by the shareholders of each party.
Israeli
tax law treats some acquisitions, such as share-for-share exchanges between an Israeli company and a foreign company, less favorably
than U.S. tax laws. For example, Israeli tax law may, under certain circumstances, subject a shareholder who exchanges his ordinary
shares for shares in another corporation to taxation prior to the sale of the shares received in such share-for-share swap.
Anti-Takeover
Measures Under Israeli Law
The
Companies Law allows us to create and issue shares having rights different from those attached to ordinary shares, including shares providing
certain preferred rights with respect to voting, distributions or other matters and shares having pre-emptive rights. As of the date
of this prospectus, no preferred shares were authorized under our amended and restated articles of association. In the future, if we
do authorize, create and issue a specific class of preferred shares, such class of shares, depending on the specific rights that may
be attached to it, may have the ability to frustrate or prevent a takeover or otherwise prevent our shareholders from realizing a potential
premium over the market value of their ordinary shares. The authorization and designation of a class of preferred shares will require
an amendment to our amended and restated articles of association, which requires the prior approval of the holders of a majority of the
voting power attaching to our issued and outstanding shares at a general meeting. The convening of the meeting, the shareholders entitled
to participate and the majority vote required to be obtained at such a meeting will be subject to the requirements set forth in the Companies
Law as described above in “— Voting Rights.”
Borrowing
Powers
Pursuant
to the Companies Law and our amended and restated articles of association, our board of directors may exercise all powers and take all
actions that are not required under law or under our amended and restated articles of association to be exercised or taken by our shareholders,
including the power to borrow money for company purposes.
Changes
in Capital
Our
amended and restated articles of association enable us to increase or reduce our share capital. Any such changes are subject to the provisions
of the Companies Law and must be approved by a resolution duly adopted by our shareholders at a general meeting. In addition, transactions
that have the effect of reducing capital, such as the declaration and payment of dividends in the absence of sufficient retained earnings
or profits, require the approval of both our board of directors and an Israeli court.
Establishment
We
were incorporated under the laws of the State of Israel on February 13, 2017. We are registered with the IRC.
Exclusive
Forum
Our
amended and restated articles of association provide that unless we
consent in writing to the selection of an alternative forum, the federal district courts of the United States shall be the exclusive
forum for the resolution of any complaint asserting a cause of action arising under the Securities Act. This choice of forum provision
may limit a shareholder’s ability to bring a claim in a judicial forum that it finds favorable for disputes with us or our directors,
officers or other employees and may increase the costs associated with such lawsuits, which may discourage such lawsuits against us and
our directors, officers and employees.
This
exclusive forum provision would not apply to claims brought pursuant to the Securities Act or the Exchange Act or any other claim
for which U.S. federal courts would have exclusive jurisdiction. To the extent that any such claims may be based upon federal law
claims, Section 27 of the Exchange Act creates exclusive federal jurisdiction over all suits brought to enforce any duty or
liability created by the Exchange Act or the rules and regulations thereunder.
Alternatively,
if a court were to find these provisions of our amended and restated articles of association inapplicable to, or unenforceable in respect of, one or more of the specified types of actions or proceedings, we may incur
additional costs associated with resolving such matters in other jurisdictions, which could adversely affect our business and financial
condition.
Our
amended and restated articles of association also provide that unless we consent in writing to the selection of an alternative forum,
the competent courts in Tel Aviv, Israel shall be the exclusive forum for any derivative action or proceeding brought on our behalf,
any action asserting a breach of a fiduciary duty owed by any of our directors, officers or other employees to us or our shareholders
or any action asserting a claim arising pursuant to any provision of the Companies Law or the Israeli Securities Law.
Any
person or entity purchasing or otherwise acquiring any interest in our share capital shall be deemed to have notice of and to have consented
to these choice of forum provisions.
History
of Share Capital
Since
January 1, 2021, our issued share capital has changed as provided below.
In
February, May, June and July 2021, we entered into Simple Agreements for Future Equity (each, a “SAFE”) with four separate
investors for aggregate proceeds of $800 thousand. Pursuant to the terms of each SAFE, upon consummation of an equity financing, we were
required to issue to each investor the number of ordinary shares equal to the purchase amount divided by the SAFE price, which was defined
as the price per share equal to 80% of the equity financing valuation (to be no less than $25,000 thousand). The SAFE Agreements also
provide the investor the right to automatically receive ordinary shares in the case of a liquidity event, defined as a change of control
event or an initial public offering. In the case of a liquidity event the investors are entitled to the number of ordinary shares equal
to the investment amount divided by the liquidity price. The liquidity price is defined as the price per share equal to our valuation
at the time of the liquidity event, multiplied by 80%, and divided by our capitalization (to be no less than $25,000 thousand). As part
of our initial public offering, the SAFEs were converted at a price per share of $2.892 into 276,672 ordinary shares.
In
May and October 2021, we issued 240,000 and 45,000 ordinary shares, respectively, pursuant to the exercise of options at an
exercise price of $0.033 per share.
In
August and September 2021, we issued 1,837,500 ordinary shares pursuant to the exercise of warrants at a weighted average price
of $0.67 per share.
On
November 9, 2021, we implemented a 1-for-3 split of our share capital, resulting in an increase of the issued and outstanding ordinary
shares.
On
December 13, 2021, we completed our initial public offering and issued 2,000,000 units, each consisting of one ordinary share
and a warrant representing the right to purchase one ordinary share with an exercise price of $6.00 per share, at an initial public offering
price of $6.00 per unit.
In
connection with the closing of our initial public offering, on December 13, 2021, we issued 961,440 ordinary shares pursuant
to the exercise of options at a weighted average exercise price of $0.079 per share.
In
March 2022, we issued 645,000 ordinary shares upon exercise of warrants issued in our initial public offering at a price per
share of $6.00.
In
October 2022, we issued 35,980 ordinary shares pursuant to vesting of restricted stock units (“RSUs”) that were
granted to one of our officers in October 2021.
In
December 2022, we issued 72,000 ordinary shares pursuant to vesting of RSUs that were granted to our officers in November 2021.
In
2022, we issued 85,449 ordinary shares to certain consultants in exchange for their services. One of the consultants received 44,000
shares of restricted stock which were issued in 4 equal installments and subject to 2 year lock-up period. The other consultant’s
received ordinary shares subject to a 6 month lock-up.
In
March and June 2023, we issued 36,000 ordinary shares pursuant to vesting of RSUs that were granted to our officers in November 2021.
In
May 2023, we issued 3,600 ordinary shares for gross proceeds of $7,200 in connection with our at-the-market offering program.
The ordinary shares were sold at market prices at the time of sale.
In
May and June we issued 320,479 ordinary shares pursuant to vesting of RSUs that were granted to our officers in March 202.
In
June 2023, we issued 25,000 ordinary shares pursuant to vesting of RSUs that were granted to our employees in March 2023.
In
June 2023, we issued 126,000 ordinary shares pursuant to the exercise of options at an exercise price of $0.033 per share.
In
June 2023, we issued 1,330,000 ordinary shares pursuant to a registered direct offering at a price per share of $1.50.
In
October 2023, we issued 1,670,000 ordinary shares pursuant to the exercise of pre-funded warrants that were granted pursuant to a registered
direct offering at a price per share of $0.0001.
In
April 2024, we issued 1,732,000 ordinary shares pursuant to a registered direct offering at a price per share of $1.50.
In
April 2024, we issued 70,964 ordinary shares to our placement agent as partial consideration in exchange for their services, in
connection with a registered direct offering.
Transfer
Agent and Registrar of ordinary shares
The
transfer agent and registrar for our ordinary shares is American Stock Transfer & Trust Company, LLC. Its address is 6201 15th Avenue,
Brooklyn, New York 11219, and its telephone number is (800) 937-5449.
EXPENSES
The
following are the estimated expenses of this offering payable by us with respect to the issuance and distribution of the securities covered
by the registration statement of which this prospectus forms a part. With the exception of the SEC registration fee, all amounts are
estimates and may change:
| SEC
registration fee | |
$ | 673.34 | |
| Legal
fees and expenses | |
| 20,000 | |
| Accountants’
fees and expenses | |
| 5,000 | |
| Miscellaneous | |
| 5,000 | |
| Total | |
$ | 30,673.34 | |
LEGAL
MATTERS
Certain
legal matters with respect to Israeli law and with respect to the validity of the offered securities under Israeli law will be passed
upon for us by Goldfarb Gross Seligman & Co. Certain legal matters with respect to U.S. law will be passed upon for us
by Greenberg Traurig, P.A.
EXPERTS
The
consolidated financial statements of NeuroSense Therapeutics Ltd. as of December 31, 2023 and 2022, and for each of the years
in the three-year period ended December 31, 2023, have been incorporated by reference herein in reliance upon the report of Somekh
Chaikin, a member firm of KPMG International, independent registered public accounting firm, incorporated by reference herein, and upon
the authority of said firm as experts in accounting and auditing. The audit report covering the December 31, 2023 consolidated financial
statements contains an explanatory paragraph that states that our recurring losses and our expectation to incur significant additional
losses raise substantial doubt about our ability to continue as a going concern. The consolidated financial statements do not include
any adjustments that might result from the outcome of that uncertainty.
ENFORCEMENT
OF CIVIL LIABILITIES
We
are incorporated under the laws of the State of Israel. Service of process upon us and upon our directors and officers and the Israeli
experts named in this prospectus, substantially all of whom reside outside the United States, may be difficult to obtain within
the United States. Furthermore, because substantially all of our assets and substantially all of our directors and officers are
located outside the United States, any judgment obtained in the United States against us or any of our directors and officers
may not be collectible within the United States.
We
have irrevocably appointed Cogency Global Inc. as our agent to receive service of process in any action against us in any U.S. federal
or state court arising out of this offering or any purchase or sale of securities in connection with this offering. The address of our
agent is 122 East 42nd Street, 18th Floor, New York, NY 10168.
We
have been informed by our legal counsel in Israel, Goldfarb Gross Seligman & Co., that it may be difficult to initiate an action
with respect to U.S. securities law in Israel. Israeli courts may refuse to hear a claim based on an alleged violation of U.S. securities
laws reasoning that Israel is not the most appropriate forum to hear such a claim. In addition, even if an Israeli court agrees to hear
a claim, it may determine that Israeli law and not U.S. law is applicable to the claim. If U.S. law is found to be applicable,
the content of applicable U.S. law must be proved as a fact by expert witnesses which can be a time-consuming and costly process.
Certain matters of procedure may also be governed by Israeli law.
Subject
to certain time limitations and legal procedures, Israeli courts may enforce a U.S. judgment in a civil matter which, subject to
certain exceptions, is non-appealable, including judgments based upon the civil liability provisions of the Securities Act and the Exchange Act
and including a monetary or compensatory judgment in a non-civil matter, provided that:
| ● | the
judgment was rendered by a court which was, according to the laws of the state of the court,
competent to render the judgment; |
| ● | the
obligation imposed by the judgment is enforceable according to the rules relating to the
enforceability of judgments in Israel and the substance of the judgment is not contrary to
public policy; and |
| ● | the
judgment is executory in the state in which it was given. |
| ● | Even
if these conditions are met, an Israeli court will not declare a foreign civil judgment enforceable
if: |
| ● | the
judgment was given in a state whose laws do not provide for the enforcement of judgments
of Israeli courts (subject to exceptional cases); |
| ● | the
enforcement of the judgment is likely to prejudice the sovereignty or security of the State
of Israel; |
| ● | the
judgment was obtained by fraud; |
| ● | the
opportunity given to the defendant to bring its arguments and evidence before the court was
not reasonable in the opinion of the Israeli court; |
| ● | the
judgment was rendered by a court not competent to render it according to the laws of private
international law as they apply in Israel; |
| ● | the
judgment is contradictory to another judgment that was given in the same matter between the
same parties and that is still valid; or |
| ● | at
the time the action was brought in the foreign court, a lawsuit in the same matter and between
the same parties was pending before a court or tribunal in Israel. |
If
a foreign judgment is enforced by an Israeli court, it generally will be payable in Israeli currency, which can then be converted into
non-Israeli currency and transferred out of Israel. The usual practice in an action before an Israeli court to recover an amount in a
non-Israeli currency is for the Israeli court to issue a judgment for the equivalent amount in Israeli currency at the rate of exchange
in force on the date of the judgment, but the judgment debtor may make payment in foreign currency. Pending collection, the amount of
the judgment of an Israeli court stated in Israeli currency ordinarily will be linked to the Israeli consumer price index plus interest
at the annual statutory rate set by Israeli regulations prevailing at the time. Judgment creditors must bear the risk of unfavorable
exchange rates.
WHERE
CAN YOU FIND MORE INFORMATION
This
prospectus is part of the registration statement on Form F-1 we have filed with the SEC under the Securities Act, with respect to
the securities offered by this prospectus. However, as is permitted by the rules and regulations of the SEC, this prospectus, which constitutes
part of the registration statement, omits certain non-material information, exhibits, schedules and undertakings set forth in the registration
statement. For further information about us, and the securities offered by this prospectus, please refer to the registration statement
and the exhibits and schedules filed as a part of the registration statement. You should rely only on the information contained in this
prospectus or incorporated by reference. We have not authorized anyone else to provide you with different information. We are not making
an offer of these securities in any state where the offer is not permitted.
We
are subject to the reporting requirements of the Exchange Act that are applicable to a foreign private issuer. In accordance with
the Exchange Act, we file reports, including annual reports on Form 20-F by April 30th of each year. We also
furnish to the SEC under cover of Form 6-K material information required to be made public in Israel, filed with and made public
by any stock exchange or distributed by us to our shareholders.
The
SEC maintains an internet site that contains reports, proxy and information statements, and other information regarding issuers, such
as us, that file electronically with the SEC (http://www.sec.gov).
As
a foreign private issuer, we are exempt from the rules under the Exchange Act prescribing the furnishing and content of proxy statements
to shareholders and our officers, directors and principal shareholders are exempt from the “short-swing profits” reporting
and liability provisions contained in Section 16 of the Exchange Act and related Exchange Act rules.
INCORPORATION
BY REFERENCE
We
file annual and special reports and other information with the SEC (File Number 001-41084). These filings contain important information
which does not appear in this prospectus. The SEC allows us to “incorporate by reference” information into this prospectus,
which means that we can disclose important information to you by referring you to other documents which we have filed or will file with
the SEC. We are incorporating by reference in this prospectus the documents listed below and all amendments or supplements we may
file to such documents:
| ● | our Annual Report on Form 20-F for the fiscal year ended December 31, 2023, filed with the SEC on April 4, 2024; |
| ● | the description of our ordinary shares contained under the heading “Item 1. Description
of Registrant’s Securities to be Registered” in our registration statement on
Form
8-A, as filed with the SEC on November 18, 2021, including any subsequent amendment
or any report filed for the purpose of updating such description. |
Certain
statements in and portions of this prospectus update and replace information in the above listed documents incorporated by reference.
Likewise, statements in or portions of a future document incorporated by reference in this prospectus may update and replace statements
in and portions of this prospectus or the above listed documents.
We
will provide each person, including any beneficial owner to whom a prospectus is delivered, without charge, upon a written or oral request,
a copy of any of the documents incorporated by reference in this prospectus, other than exhibits to such documents which are not specifically
incorporated by reference into such documents. Please direct your written or telephone requests to NeuroSense Therapeutics Ltd., 11 HaMenofim
Street, Building B, Herzliya, 4672562, Israel, Attn: Or Eisenberg, telephone number +972587531153. You may also obtain information about
us by visiting our website at www.neurosense-tx.com. Information contained in our website is not part of this prospectus.
Up to 3,050,964 Ordinary Shares
Ordinary Warrants to Purchase up to 2,980,000
Ordinary Shares

______________________
Prospectus
______________________
The
date of this prospectus is , 2024.
Part II
INFORMATION
NOT REQUIRED IN THE PROSPECTUS
Item 6.
Indemnification of Directors and Officers
Exculpation,
Insurance and Indemnification of Directors and Officers
Under
the Companies Law, a company may not exculpate an office holder from liability for a breach of the duty of loyalty. An Israeli company
may exculpate an office holder in advance from liability to the company, in whole or in part, for damages caused to the company as a
result of a breach of duty of care but only if a provision authorizing such exculpation is included in its articles of association. Our
amended and restated articles of association include such a provision. A company may not exculpate in advance a director from liability
arising from a breach of his or her duty of care in connection with a prohibited dividend or distribution to shareholders.
As
permitted under the Companies Law, our amended and restated articles of association provide that we may indemnify an office holder in
respect of the following liabilities, payments and expenses incurred for acts performed by him or her as an office holder, either in
advance of an event or following an event:
| ● | a
monetary liability incurred by or imposed on the office holder in favor of another person
pursuant to a court judgment, including pursuant to a settlement confirmed as judgment or
arbitrator’s decision approved by a competent court. However, if an undertaking to
indemnify an office holder with respect to such liability is provided in advance, then such
an undertaking must be limited to events which, in the opinion of the board of directors,
can be foreseen based on the company’s activities when the undertaking to indemnify
is given, and to an amount or according to criteria determined by the board of directors
as reasonable under the circumstances, and such undertaking shall detail the abovementioned
foreseen events and amount or criteria; |
| ● | reasonable
litigation expenses, including reasonable attorneys’ fees, which were incurred by the
office holder as a result of an investigation or proceeding filed against the office holder
by an authority authorized to conduct such investigation or proceeding, provided that such
investigation or proceeding was either (i) concluded without the filing of an indictment
against such office holder and without the imposition on him of any monetary obligation in
lieu of a criminal proceeding, (ii) concluded without the filing of an indictment against
the office holder but with the imposition of a monetary obligation on the office holder in
lieu of criminal proceedings for an offense that does not require proof of criminal intent
or (iii) in connection with a monetary sanction; |
| ● | reasonable
litigation expenses, including attorneys’ fees, incurred by the office holder or which
were imposed on the office holder by a court (i) in a proceeding instituted against
him or her by the company, on its behalf, or by a third party, (ii) in connection with
criminal indictment of which the office holder was acquitted or (iii) in a criminal
indictment for which the office holder was convicted of an offense that does not require
proof of criminal intent; |
| ● | expenses
he or she incurs as a result of administrative proceedings that may be instituted against
him or her under Israeli securities laws, if applicable, and payments made to injured persons
under specific circumstances thereunder; and |
| ● | any
other matter in respect of which it is permitted or will be permitted under applicable law
to indemnify an office holder in the company. |
As
permitted under the Companies Law, our amended and restated articles of association provide that we may insure an office holder against
the following liabilities incurred for acts performed by him or her as an office holder:
| ● | a
breach of the duty of loyalty to the company, provided that the office holder acted in good
faith and had a reasonable basis to believe that the act would not harm the company; |
| ● | a
breach of duty of care to the company or to another person, to the extent such a breach arises
out of the negligent conduct of the office holder; |
| ● | a
monetary liability imposed on the office holder in favor of a third party; |
| ● | expenses
he or she incurs as a result of administrative proceedings that may be instituted against
him or her under the Israeli securities laws if applicable, and payments made to injured
persons under specific circumstances thereunder; and |
| ● | any
other matter in respect of which it is permitted or will be permitted under applicable law
to insure the liability of an office holder in the company. |
Under
the Companies Law, a company may not indemnify, exculpate or insure an office holder against any of the following:
| ● | a
breach of the duty of loyalty, except for indemnification and insurance for a breach of the
duty of loyalty to the company to the extent that the office holder acted in good faith and
had a reasonable basis to believe that the act would not prejudice the company; |
| ● | a
breach of duty of care committed intentionally or recklessly, excluding a breach arising
out of the negligent conduct of the office holder; |
| ● | an
act or omission committed with intent to derive illegal personal benefit; or |
| ● | a
fine or forfeit levied against the office holder. |
Under
the Companies Law, exculpation, indemnification and insurance of office holders must be approved by the compensation committee and the
board of directors and, with respect to directors or controlling shareholders, their relatives and third parties in which controlling
shareholders have a personal interest, also by the shareholders.
Our
amended and restated articles of association permit us to exculpate, indemnify and insure our office holders to the fullest extent permitted
or to be permitted by law. Our office holders are currently covered by a directors’ and officers’ liability insurance policy.
As of the date of this prospectus, no claims for directors’ and officers’ liability insurance have been filed under this
policy and we are not aware of any pending or threatened litigation or proceeding involving any of our office holders, including our
directors, in which indemnification is sought.
We
have entered into agreements with each of our current office holders exculpating them from a breach of their duty of care to us to the
fullest extent permitted by law, subject to limited exceptions, and undertaking to indemnify them to the fullest extent permitted by
law, including, with respect to liabilities resulting from this offering, to the extent that these liabilities are not covered by insurance.
This indemnification is limited, with respect to any monetary liability imposed in favor of a third party, to events determined as foreseeable
by the board of directors based on our activities. The maximum aggregate amount of indemnification that we may pay to our office holders
based on such indemnification agreement is the greater of (i) an amount equal to 25% of our shareholders’ equity on a consolidated
basis, based on our most recent financial statements made publicly available before the date on which the indemnity payment is made,
and (ii) $25 million. Such indemnification amounts are in addition to any insurance amounts. These indemnification agreements
supersede all previous letters of indemnification that we have provided to him or her in the past, if any. However, in the opinion of
the SEC, indemnification of office holders for liabilities arising under the Securities Act is against public policy and therefore unenforceable.
Item 7.
Recent Sales of Unregistered Securities
The
following sets forth information regarding all unregistered securities sold since January 2021. These issuances did not result in
changes to the voting rights attached to the ordinary shares.
Issuance
of Capital Stock
| ● | In
June 2023, we issued Ordinary Warrants to purchase up to 3,000,000 ordinary
shares issuable upon exercise of Ordinary Warrants. |
| ● | In
April 2024, we issued Ordinary Warrants to purchase up to 2,980,000 ordinary shares issuable
upon exercise of Ordinary Warrants and 70,964 ordinary shares. |
The
offers, sales and issuances of the securities described in the preceding paragraphs were exempt from registration either (a) under
Section 4(a)(2) of the Securities Act and the rules and regulations promulgated thereunder (including Regulation D and
Rule 506), in that the transactions were between an issuer and sophisticated investors or members of its senior executive management
and did not involve any public offering within the meaning of Section 4(a)(2), (b) under Regulation S promulgated under
the Securities Act in that offers, sales and issuances were not made to persons in the United States and no directed selling efforts
were made in the United States, (c) under Rule 144A under the Securities Act in that the shares were offered and sold
by the initial purchasers to qualified institutional buyers or (d) under Rule 701 promulgated under the Securities Act in that
the transactions were under compensatory benefit plans and contracts relating to compensation.
Item 8.
Exhibits and Financial Statement Schedules
(a) The following documents are filed as part of this registration statement:
| Exhibit no. |
|
Exhibit |
| 1.1# |
|
Amended
and Restated Articles of Association (incorporated by reference to Exhibit 3.2 to the Registration Statement on Form F-1/A, filed
with the SEC on December 6, 2021) |
| 2.1 |
|
Description
of the Registrant’s Securities Registered Pursuant to Section 12 of the Securities Exchange Act of 1934 (incorporated by
reference to Exhibit 2.1 of the Registrant’s Annual Report on Form 20-F filed with the SEC on April 4, 2024) |
| 4.1 |
|
Form
of Indemnification Agreement (incorporated by reference to Exhibit 10.1 to the Registration Statement on Form F-1/A, filed with the
SEC on December 6, 2021) |
| 4.2 |
|
2018
Employee Share Option Plan (incorporated by reference to Exhibit 10.1 to the Registration Statement on Form S-8, filed with the SEC
on February 2, 2022) |
| 4.3 |
|
Compensation
Policy (incorporated by reference to Exhibit 10.3 to the Registration Statement on Form F-1/A, filed with the SEC on December 6,
2021) |
| 4.4 |
|
Form
of Share Purchase Agreement (incorporated by reference to Exhibit 4.1 to the Registration Statement on Form F-1/A, filed with the
SEC on December 6, 2021) |
| 4.5 |
|
Registrant’s
Specimen Certificate for ordinary shares (incorporated by reference to Exhibit 4.4 to the Registration Statement on Form F-1/A, filed
with the SEC on December 6, 2021) |
| 4.6 |
|
Form
of Underwriter’s Warrant (incorporated by reference to Exhibit 4.5 to the Registration Statement on Form F-1/A, filed with
the SEC on December 6, 2021) |
| 4.7 |
|
Form
of Ordinary Share Purchase Warrant (incorporated by reference to Exhibit 4.6 to the Registration Statement on Form F-1/A, filed with
the SEC on December 6, 2021) |
| 4.8 |
|
Form
of Warrant Agency Agreement between American Stock Transfer & Trust Company. LLC and NeuroSense Therapeutics Ltd. (incorporated
by reference to Exhibit 4.7 to the Registration Statement on Form F-1/A, filed with the SEC on December 6, 2021) |
| 4.9 |
|
Sales
Agreement, dated April 14, 2023, by and between NeuroSense Therapeutics Ltd. and A.G.P./Alliance Global Partners (incorporated by
reference to Exhibit 10.1 to the Registrant’s Report on Form 6-K filed with the SEC on April 14, 2023) |
| 4.10 |
|
Placement
Agent Agreement, dated as of June 22, 2023, by and between NeuroSense Therapeutics Ltd. and A.G.P./Alliance Global Partners (incorporated
by reference to Exhibit 1.1 to the Registrant’s Report on Form 6-K filed with the SEC on June 23, 2023) |
| 4.11! |
|
Form
of Securities Purchase Agreement, dated as of June 22, 2023, by and between NeuroSense Therapeutics Ltd. and the purchaser identified
on the signature page thereto (incorporated by reference to Exhibit 10.1 to the Registrants Report on Form 6-K filed with the SEC
on June 23, 2023) |
| 4.12 |
|
Form
of Ordinary Warrant (incorporated by reference to Exhibit 4.1 to the Registrant’s Report on Form 6-K filed with the SEC on
June 23, 2023) |
| 4.13 |
|
Form
of Pre-Funded Warrant (incorporated by reference to Exhibit 4.2 to the Registrant’s Report on Form 6-K filed with the SEC on
June 23, 2023) |
| 4.14 |
|
Placement Agent Agreement, dated as of April 10, 2024, by and between the Registrant and A.G.P./Alliance Global Partners (incorporated by reference to Exhibit 10.1 to the Registrant’s Report on Form 6-K filed with the SEC on April 12, 2024). |
| 4.15! |
|
Form of Securities Purchase Agreement, dated as of April 10, 2024, by and between the Registrant and the purchaser identified on the signature page thereto (incorporated by reference to Exhibit 10.2 to the Registrant’s Report on Form 6-K filed with the SEC on April 12, 2024). |
| 4.16 |
|
Form of Ordinary Warrant (incorporated by reference to Exhibit 10.3 to the Registrant’s Report on Form 6-K filed with the SEC on April 12, 2024) |
| 4.17 |
|
Form of Pre-Funded Warrant (incorporated by reference to Exhibit 4.1 to the Registrant’s Report on Form 6-K filed with the SEC on April 12, 2024) |
| 5.1* |
|
Opinion of Goldfarb Gross Seligman & Co., Israeli counsel to the Registrant. |
| 5.2* |
|
Opinion of Greenberg Traurig, P.A., U.S. counsel to the Registrant. |
| 8.1 |
|
List
of Subsidiaries of NeuroSense Therapeutics Ltd. (incorporated by reference to Exhibit 8.1 of the Registrant’s Annual Report
on Form 20-F filed with the SEC on April 4, 2024) |
| 23.1* |
|
Consent of Somekh Chaikin (Member firm of KPMG International), Independent Registered Public Accounting Firm |
| 23.2 |
|
Consent of Goldfarb Gross Seligman & Co. (included in Exhibit 5.1). |
| 23.3 |
|
Consent of Greenberg Traurig, P.A., U.S. counsel to the Registrant (included in Exhibit 5.2). |
| 24.1 |
|
Powers of Attorney (included on signature page to Registration Statement). |
| 107* |
|
Filing Fee Table. |
| * | Filed
herewith |
| ! | Annexes,
schedules and exhibits have been omitted pursuant to Item 601(b)(2) of Regulation S-K. The
registrant agrees to furnish supplementally a copy of any omitted attachment to the Securities
and Exchange Commission on a confidential basis upon request. |
| # | English
translation of original Hebrew document. |
Item 9.
Undertakings
| (1) | The
undersigned registrant hereby undertakes: |
| (a) | to
file, during any period in which offers or sales are being made, a post-effective amendment
to this Registration Statement: |
| (i) | to
include any prospectus required by Section 10(a)(3) of the Securities Act of 1933
(“the Securities Act”); |
| (ii) | to
reflect in the prospectus any facts or events arising after the effective date of this Registration
Statement (or the most recent post-effective amendment thereof) which, individually or in
the aggregate, represent a fundamental change in the information set forth in this Registration
Statement. Notwithstanding the foregoing, any increase or decrease in volume of securities
offered (if the total dollar value of securities offered would not exceed |
| | that
which was registered) and any deviation from the low or high end of the estimated maximum
offering range may be reflected in the form of prospectus filed with the Securities and Exchange
Commission pursuant to Rule 424(b) if, in the aggregate, the changes in volume
and price represent no more than a 20% change in the maximum aggregate offering price set
forth in the “Calculation of Registration Fee” table in the effective registration
statement; and |
| (iii) | to
include any material information with respect to the plan of distribution not previously
disclosed in the Registration Statement or any material change to such information in this
Registration Statement. |
| (b) | that,
for the purpose of determining any liability under the Securities Act, each such post-effective
amendment shall be deemed to be a new registration statement relating to the securities offered
therein, and the offering of such securities at that time shall be deemed to be the initial
bona fide offering thereof. |
| (c) | to
remove from registration by means of a post-effective amendment any of the securities being
registered which remain unsold at the termination of the offering. |
| (d) | to
file a post-effective amendment to the registration statement to include any financial statements
required by Item 8.A. of Form 20-F at the start of any delayed offering or throughout
a continuous offering. Financial statements and information otherwise required by Section 10(a)(3) of
the Securities Act need not be furnished, provided that the registrant includes in the prospectus,
by means of a post-effective amendment, financial statements required pursuant to this paragraph
(1)(d) and other information necessary to ensure that all other information in the prospectus
is at least as current as the date of those financial statements. |
| (e) | that,
for the purpose of determining any liability under the Securities Act to any purchaser each
prospectus filed pursuant to Rule 424(b) as part of a registration statement relating
to an offering, other than registration statements relying on Rule 430B or other than
prospectuses filed in reliance on Rule 430A, shall be deemed to be part of and included
in the registration statement as of the date it is first used after effectiveness. Provided,
however, that no statement made in a registration statement or prospectus that is part of
the registration statement or made in a document incorporated or deemed incorporated by reference
into the registration statement or prospectus that is part of the registration statement
will, as to a purchaser with a time of contract of sale prior to such first use, supersede
or modify any statement that was made in the registration statement or prospectus that was
part of the registration statement or made in any such document immediately prior to such
date of first use. |
| (f) | that,
for the purpose of determining liability of a registrant under the Securities Act to any
purchaser in the initial distribution of the securities, the undersigned registrant undertakes
that in a primary offering of securities of the undersigned registrant pursuant to this registration
statement, regardless of the underwriting method used to sell the securities to the purchaser,
if the securities are offered or sold to such purchaser by means of any of the following
communications, the undersigned registrant will be a seller to the purchaser and will be
considered to offer or sell such securities to such purchaser: |
| (i) | any
preliminary prospectus or prospectus of the undersigned registrant to the offering required
to be filed pursuant to Rule 424; |
| (ii) | any
free writing prospectus relating to the offering prepared by or on behalf of the undersigned
registrant or used or referred to by an undersigned registrant; |
| (iii) | the
portion of any other free writing prospectus relating to the offering containing material
information about the undersigned registrant or its securities provided by or on behalf of
the undersigned registrant; and |
| (iv) | any
other communication that is an offer in the offering made by the undersigned registrant to
the purchaser. |
| (2) | Insofar
as indemnification for liabilities arising under the Securities Act may be permitted to directors,
officers and controlling persons of the registrant pursuant to the foregoing provisions,
or otherwise, the registrant has been advised that in the opinion of the SEC such indemnification
is against public policy as expressed in the Securities Act and is, therefore, unenforceable.
In the event that a claim for indemnification against such liabilities (other than the payment
by the registrant of expenses incurred or paid by a director, officer or controlling person
of the registrant in the successful defense of any action, suit or proceeding) is asserted
by such director, officer or controlling person in connection with the securities being registered,
the registrant will, unless in the opinion of its counsel the matter has been settled by
controlling precedent, submit to a court of appropriate jurisdiction the question whether
such indemnification by it is against public policy as expressed in the Securities Act and
will be governed by the final adjudication of such issue. |
SIGNATURES
Pursuant
to the requirements of the Securities Act, the Registrant certifies that it has reasonable grounds to believe that it complies with all
of the requirements for filing on Form F-1 and has duly caused this registration statement to be signed on its behalf by the undersigned,
thereunto duly authorized, in Herzliya, Israel on May 10, 2024.
| |
NEUROSENSE
THERAPEUTICS LTD. |
| |
|
| |
By: |
/s/
Or Eisenberg |
| |
Name: |
Or
Eisenberg |
| |
Title: |
Chief
Financial Officer |
KNOW
ALL PERSONS BY THESE PRESENTS, each director and officer whose signature appears below constitutes and appoints, Mr. Alon Ben-Noon
and Mr. Or Eisenberg, and each of them, individually, as his or her true and lawful attorney-in-fact and agent, with full power
of substitution and re-substitution, to sign in any and all capacities any and all amendments or post-effective amendments to this registration
statement on Form F-1, and to sign any and all additional registration statements relating to the same offering of securities of
the Registration Statement that are filed pursuant to Rule 462(b) of the Securities Act, and to file the same with all exhibits
thereto and other documents in connection therewith with the Securities and Exchange Commission, granting such attorney-in-fact and agent
full power and authority to do all such other acts and execute all such other documents as he may deem necessary or desirable in connection
with the foregoing, as fully as the undersigned may or could do in person, hereby ratifying and confirming all that such attorney-in-fact
and agent may lawfully do or cause to be done by virtue hereof.
Pursuant
to the requirements of the Securities Act, this registration statement has been signed below by the following persons on the dates and
in the capacities indicated.
| Signature |
|
Title |
|
|
| |
|
|
|
|
| /s/
Mark Leuchtenberger |
|
Chairman
of the Board of Directors |
|
May
10, 2024 |
| Mark
Leuchtenberger |
|
|
|
|
| |
|
|
|
|
| /s/
Alon Ben-Noon |
|
Chief
Executive Officer and Director |
|
May
10, 2024 |
| Alon
Ben-Noon |
|
(Principal
Executive Officer and Director) |
|
|
| |
|
|
|
|
| /s/
Or Eisenberg |
|
Chief Financial Officer (Principal Financial Officer |
|
May
10, 2024 |
| Or
Eisenberg |
|
and
Principal Accounting Officer) |
|
|
| |
|
|
|
|
| /s/
Caren Deardorf |
|
Director |
|
May
10, 2024 |
| Caren
Deardorf |
|
|
|
|
| |
|
|
|
|
| /s/
Revital Mandil-Levin |
|
Director |
|
May
10, 2024 |
| Revital
Mandil-Levin |
|
|
|
|
| |
|
|
|
|
| /s/
Cary Claiborne |
|
External
Director |
|
May
10, 2024 |
| Cary
Claiborne |
|
|
|
|
| |
|
|
|
|
| /s/
Christine Pellizzari |
|
External
Director |
|
May
10, 2024 |
| Christine
Pellizzari |
|
|
|
|
Signature
of authorized representative in the United States
Pursuant
to the requirements of the Securities Act of 1933, as amended, the Registrant’s duly authorized representative has signed
this Registration Statement on Form F-1 on this 10th day of May, 2024.
| |
By: |
/s/
Colleen A. De Vries |
| |
Name: |
Colleen
A. De Vries |
| |
Title: |
Sr.
Vice-President, Authorized
U.S. Representative — Cogency Global |
II-7
Exhibit 5.1

Tel Aviv, May 10, 2024
NeuroSense Therapeutics Ltd.
Building B
11 HaMenofim Street
Herzliya 4672562
Israel
Ladies and Gentlemen:
We have acted as Israeli
counsel for NeuroSense Therapeutics Ltd., a company organized under the laws of Israel (the “Company”), in connection
with the registration statement on Form F-1 (the “Registration Statement”) filed by the Company with the United States
Securities and Exchange Commission (the “SEC”) on the date hereof pursuant to the United States Securities Act of 1933,
as amended (the “Securities Act ”), which registers, from time to time, (i) the resale by the selling shareholder
of up to 2,980,000 ordinary shares, no par value, of the Company (“Ordinary Shares”), issuable upon exercise of warrants
to purchase Ordinary Shares (the “Warrant Shares”) acquired by the selling shareholder named therein (the “Ordinary
Warrants”) issued pursuant to a securities purchase agreement, dated April 10, 2024 (the “Securities Purchase Agreement”)
by and between the Company and the purchaser set forth therein, (ii) the resale by the selling shareholder named therein of the Ordinary
Warrants, and (iii) the resale by the selling shareholder named therein of 70,964 Ordinary Shares (the “PA Shares”)
issued pursuant to a placement agent agreement, dated April 10, 2024, and related side letter by and between the Company and the placement
agent set forth therein (the “Placement Agent Agreement”).
This opinion letter is
furnished to you at your request to enable you to fulfill the requirements of Item 601(b)(5) of Regulation S-K under the Securities Act,
in connection with the filing of the Registration Statement.
In connection herewith, we have examined the originals,
or photocopies or copies, certified or otherwise identified to our satisfaction, of: (i) the executed Ordinary Warrant; (ii) a copy of
the Amended and Restated Articles of Association of the Company, as currently in effect; (iii) the executed Securities Purchase Agreement
and Placement Agent Agreement; (iv) minutes of meetings, or written resolutions adopted in lieu of meetings, of the board of directors,
whereby the Company’s entry into the Securities Purchase Agreement, the Ordinary Warrant, and Placement Agent Agreement and all
related actions were approved; and (v) such other corporate records, agreements, documents and other instruments, and such certificates
or comparable documents of public officials and of officers and representatives of the Company as we have deemed relevant and necessary
as a basis for the opinions hereafter set forth. We have also made inquiries of such officers and representatives as we have deemed relevant
and necessary as a basis for the opinions hereafter set forth.


For purposes of our opinions, we have assumed
(i) the authenticity of original documents and the genuineness of all signatures; (ii) the conformity to the original documents of all
documents submitted to us as copies; (iii) the truth, accuracy and completeness of the information, representations and warranties contained
in the corporate records, documents, certificates and instruments we have reviewed; (iv) the due execution and delivery of all documents
where due execution and delivery are a prerequisite to the effectiveness thereof; and (v) the legal capacity of natural persons.
Based upon and subject to the foregoing,
we are of the opinion that (i) the Ordinary Warrant, the Warrant Shares and the PA Shares have been duly authorized, (ii) when the Warrant
Shares are issued, sold and delivered by the Company against receipt of the exercise price therefor pursuant to the terms of the Ordinary
Warrants, in accordance with and in the manner described therein and in the Registration Statement, the Warrant Shares will be validly
issued, fully paid and non-assessable, and (iii) the PA Shares have been validly issued and are fully paid and non-assessable.
Members of our firm are admitted to the Bar in
the State of Israel, and we do not express any opinion as to the laws of any other jurisdiction. This opinion is limited to the matters
stated herein and no opinion is implied or may be inferred beyond the matters expressly stated.
We consent to the filing of this opinion as an
exhibit to the Registration Statement and to the reference to our firm appearing under the caption “Legal Matters” in the
prospectus forming part of the Registration Statement. In giving this consent, we do not thereby admit that we are within the category
of persons whose consent is required under Section 7 of the Securities Act, the rules and regulations of the SEC promulgated thereunder,
or Item 509 of the SEC’s Regulation S-K promulgated under the Securities Act.
This opinion letter is rendered as of the date
hereof and we disclaim any obligation to advise you of facts, circumstances, events or developments that may be brought to our attention
after the effective date of the Registration Statement that may alter, affect or modify the opinions expressed herein. This opinion may
not be relied upon by you for any other purpose or furnished to, or relied upon by, any other person, firm or other entity.
| |
Very truly yours, |
| |
|
| |
/s/ Goldfarb, Gross, Seligman & Co. |
Exhibit 5.2

May 10, 2024
NeuroSense Therapeutics Ltd.
11 HaMenofim Street, Building B
Herzliya 4672562 Israel
| Re: |
Registration Statement on Form F-1 |
Ladies and Gentlemen:
This opinion is furnished
to you in connection with a Registration Statement on Form F-1 (as amended to date, the “Registration Statement”) filed by
NeuroSense Therapeutics Ltd., an Israeli company (the “Company”), with the Securities and Exchange Commission (the “Commission”)
under the Securities Act of 1933, as amended (the “Securities Act”), relating to the registration for resale under the Registration
Statement (i) up to 2,980,000 (the “Investor Shares”) of the Company’s ordinary shares, no par value (the “Ordinary
Shares”) issuable upon exercise of the Warrants (as defined below) by the selling shareholder identified in the Registration Statement,
(ii) the resale of 2,980,000 warrants issued to the selling shareholder identified in the Registration Statement, each representing the
right to purchase one Ordinary Share (the “Warrants”), and (iii) the resale of 70,964 Ordinary Shares (the “PA Shares
and together with the Investor Shares and Warrants, the “Securities”) issued to the selling shareholder identified in the
Registration Statement. The Investor Shares and Warrants were issued pursuant to a securities purchase agreement, dated April 10, 2024
(the “Securities Purchase Agreement”) by and between the Company and the purchaser set forth therein. The PA Shares were issued
pursuant to a placement agent agreement, dated April 10, 2024, and related side letter by and between the Company and the placement agent
set forth therein (the “Placement Agent Agreement”).
We are acting as U.S. securities
counsel for the Company in connection with the Registration Statement. In connection with this opinion, we have examined the Registration
Statement, the Securities Purchase Agreement, a specimen Warrant certificate in the form of Exhibit 4.16 to the Registration Statement,
the Placement Agent Agreement and have also examined and relied upon minutes of meetings of the Board of Directors of the Company as provided
to us by the Company, the articles of association of the Company, as restated and/or amended to date, and such other documents as we have
deemed necessary for purposes of rendering the opinion hereinafter set forth.
In our examination of the
foregoing documents, we have assumed the legal capacity of all natural persons, the genuineness of all signatures, the authenticity of
all documents submitted to us as originals, the conformity to original documents of all documents submitted to us as copies, the authenticity
of the originals of such latter documents and the legal competence of all signatories to such documents. Other than our examination of
the documents indicated above, we have made no other examination in connection with this opinion. Because the Warrants contain provisions
stating that they are to be governed by the laws of the State of New York, we are rendering this opinion as to New York law.
We have assumed further that
the Securities Purchase Agreement and Placement Agent Agreement have been duly authorized, executed and delivered by the Company and the
applicable selling shareholder and constitute the valid and binding obligations of the selling shareholders enforceable against them in
accordance with their respective terms. We have assumed further that the Company has duly authorized the Securities. We have assumed further
that the Company is a company duly organized, validly existing and in good standing under the laws of the jurisdiction in which it is
organized and has all requisite power, authority and legal right to issue the Securities.
We are admitted to practice
in the State of New York, and we express no opinion as to any matters governed by any law other than the law of the State of New York.
You are separately reviewing an opinion from Goldfarb, Gross, Seligman & Co. with respect to the corporate proceedings and due authorization
relating to the issuance of the Securities under Israeli law. For purposes of our opinion, we have assumed that the Securities have been
duly authorized and that the Investor Shares, PA Shares and the Ordinary Shares underlying the Warrants have been duly and validly issued,
fully paid and non-assessable.
Greenberg
Traurig, P.A. | Attorneys at Law
Azrieli
Center, Round Tower | 132 Menachem Begin Road, 30th Floor | Tel Aviv, Israel 6701101 | T +1 +972 (0)
3 636 6000 | F +1 +972 (0) 3 636 6010 |
| www.gtlaw.com |
Based upon the foregoing and
subject to the assumptions and qualifications set forth herein, we are of the opinion that, when the Registration Statement has become
effective under the Securities Act, the Warrants, insofar as they are governed by the law of the State of New York, constitute valid and
binding obligations of the Company enforceable against the Company in accordance with their terms
The opinion set forth herein
is rendered as of the date hereof, and we assume no obligation to update such opinion to reflect any facts or circumstances which may
hereafter come to our attention or any changes in the law which may hereafter occur (which may have retroactive effect). In addition,
the foregoing opinions are qualified to the extent that (a) enforceability may be limited by and be subject to general principles of equity,
regardless of whether such enforceability is considered in a proceeding in equity or at law (including, without limitation, concepts of
notice and materiality), and by bankruptcy, insolvency, reorganization, moratorium and other similar laws affecting creditors’ and debtors’
rights generally (including, without limitation, any state or federal law in respect of fraudulent transfers); and (b) no opinion is expressed
herein as to compliance with or the effect of federal or state securities or blue sky laws.
This opinion is rendered to
you in connection with the Registration Statement. This opinion may not be relied upon for any other purpose, or furnished to, quoted
or relied upon by any other person, firm or corporation for any purpose, without our prior written consent, except that (A) this opinion
may be furnished or quoted to judicial or regulatory authorities having jurisdiction over you, and (B) this opinion may be relied upon
by holders of the Warrants currently entitled to rely on it pursuant to applicable provisions of federal securities law.
We hereby consent to the filing
of this opinion as Exhibit 5.2 to the Registration Statement and to the reference to this firm under the caption “Legal Matters”
in the Registration Statement and in any Registration Statement pursuant to Rule 462(b) under the Securities Act. In giving such consent,
we do not admit that we are in the category of persons whose consent is required under Section 7 of the Securities Act or the rules and
regulations of the Commission. This opinion letter is limited to the matters expressly set forth herein and no opinion is implied or may
be inferred beyond the matters expressly so stated. This opinion letter is given as of the date hereof and we do not undertake any liability
or responsibility to inform you of any change in circumstances occurring, or additional information becoming available to us, after the
date hereof which might alter the opinions contained herein.
| |
Very truly yours, |
| |
|
| |
/s/ Greenberg Traurig, P.A. |
| |
Greenberg Traurig, P.A. |
Greenberg
Traurig, P.A. | Attorneys at Law
Azrieli
Center, Round Tower | 132 Menachem Begin Road, 30th Floor | Tel Aviv, Israel 6701101 | T +1 +972 (0)
3 636 6000 | F +1 +972 (0) 3 636 6010 |
| www.gtlaw.com |
Exhibit 23.1
Consent of Independent Registered Public Accounting
Firm
We consent to the use of our report dated April 4, 2024, with respect
to the consolidated financial statements of NeuroSense Therapeutics Ltd., incorporated herein by reference and to the reference to our
firm under the heading “Experts” in the prospectus.
/s/ Somekh Chaikin
Somekh Chaikin
Member Firm of KPMG International
Tel Aviv, Israel
May 10, 2024
Exhibit 107
Calculation of Filing Fee Table
Form F-1
(Form Type)
NeuroSense Therapeutics Ltd.
(Exact Name of Registrant as Specified in its Charter)
Table 1: Newly Registered Securities
| |
|
Security
Type |
|
Security
Class Title |
|
Fee
Calculation
Rule |
|
Amount
Registered(1) |
|
|
Proposed
Maximum
Offering
Price Per
Share |
|
|
Maximum
Aggregate
Offering
Price |
|
|
Fee
Rate |
|
|
Amount
of
Registration
Fee |
|
| Fees to be Paid |
|
Equity |
|
Ordinary
shares, no par value per share, underlying Ordinary Warrants (2) |
|
457(g) |
|
|
2,980,000 |
|
|
$ |
1.50 |
(3) |
|
$ |
4,470,000 |
|
|
|
0.0001476 |
|
|
$ |
659.78 |
|
| |
|
Equity |
|
Ordinary
Warrants(4) |
|
457(g) |
|
|
2,980,000 |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
| |
|
Equity |
|
Ordinary shares, no par value(5) |
|
457(c) |
|
|
70,964 |
|
|
$ |
1.295 |
|
|
$ |
91,898.38 |
|
|
|
0.0001476 |
|
|
$ |
13.56 |
|
| |
|
Total Offering
Amounts |
$ |
4,561,898.38 |
|
|
|
|
|
|
$ |
673.34 |
|
| |
|
Total
Fees Previously Paid |
|
|
|
|
|
|
|
|
|
$ |
— |
|
| |
|
Total
Fee Offsets |
|
|
|
|
|
|
|
|
|
$ |
— |
|
| |
|
Net
Fee Due |
|
|
|
|
|
|
|
|
|
$ |
673.34 |
|
| (1) | Pursuant to Rule 416(a) under the U.S. Securities Act of
1933, as amended (the “Securities Act”), the securities being registered hereunder also include such indeterminable number
of additional securities as may from time to time be issued after the date hereof as a result of stock splits, stock dividends, recapitalizations
or similar transactions. |
| (2) | Represents the ordinary shares, no par value per share (the
“Ordinary Shares”), of NeuroSense Therapeutics Ltd. (the “Company”), issuable upon the exercise of warrants to
purchase Ordinary Shares at an exercise price of $1.50 per Ordinary Share (the “Ordinary Warrants”) issued in a private placement
to the selling shareholder identified in the registration of which this exhibit forms a part. |
| (3) | Calculated pursuant to Rule 457(g) promulgated under the
Securities Act and solely for the purpose of calculating the registration fee, based on the exercise price of the Ordinary Warrants of
$1.50. |
| (4) | In accordance with Rule 457(g) and consistent with the response
to Question 240.06 of the Securities Act Rules Compliance and Disclosure Interpretations, the entire registration fee for the Ordinary
Warrants is allocated to the Ordinary Shares underlying the Ordinary Warrants, and no separate fee is payable for the Ordinary Warrants. |
| (5) | Estimated solely for the purpose of calculating the registration
fee pursuant to Rule 457(c) promulgated under the Securities Act of 1933, as amended, based on the average of the equivalent high and
low sales prices of the ordinary shares on the Nasdaq Capital Market on May 8, 2024 |
NeuroSense Therapeutics (NASDAQ:NRSNW)
過去 株価チャート
から 4 2024 まで 5 2024

NeuroSense Therapeutics (NASDAQ:NRSNW)
過去 株価チャート
から 5 2023 まで 5 2024
