As filed with the Securities and Exchange
Commission on February 2, 2024.
Registration No. 333-275995
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM S-1
(Amendment No. 2)
REGISTRATION STATEMENT
Under
The Securities Act of 1933
Agile Therapeutics, Inc.
(Exact name of Registrant as specified in its charter)
Delaware
(State or Other Jurisdiction of
Incorporation or Organization) |
2834
(Primary Standard Industrial
Classification Code Number) |
23-2936302
(I.R.S. Employer
Identification Number) |
| |
|
|
500 College Road East, Suite 310
Princeton, New Jersey 08540
(609) 683-1880
(Address, including zip code, and telephone number,
including
area code, of Registrant’s principal executive
offices)
Alfred Altomari
Chief Executive Officer
Agile Therapeutics, Inc.
500 College Road East, Suite 310
Princeton, New Jersey 08540
(609) 683-1880
(Name, address, including zip code, and telephone
number, including area code, of agent for service)
Please
send copies of all communications to:
Steven M. Cohen Bryan
S. Keighery
Morgan, Lewis & Bockius LLP
502 Carnegie Center
Princeton, New Jersey 08540
(609) 919-6600
Approximate
date of commencement of the proposed sale to the public: As soon as practicable after the effective date of this Registration
Statement.
If
any of the securities being registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415
under the Securities Act, check the following box. x
If
this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act,
check the following box and list the Securities Act registration statement number of the earlier effective registration statement for
the same offering. ¨
If
this Form is a post-effective amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box
and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ¨
If
this Form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box
and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ¨
Indicate by check mark whether
the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company or an emerging
growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting
company” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| Large accelerated filer ¨ |
Accelerated filer ¨ |
Non-accelerated filer x |
Smaller reporting company x
Emerging
growth company ¨ |
If
an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying
with any new or revised financial accounting standards provided pursuant to Section 7(a)(2)(b) of the Securities Act. ¨
The Registrant hereby amends
this Registration Statement on such date or dates as may be necessary to delay its effective date until the Registrant shall file a
further amendment which specifically states that this Registration Statement shall thereafter become effective in accordance with Section 8(a) of
the Securities Act of 1933, as amended, or until the Registration Statement shall become effective on such date as the Securities and
Exchange Commission, acting pursuant to said Section 8(a), may determine.
The information in this preliminary
prospectus is not complete and may be changed. We may not sell these securities until the registration statement filed with the Securities
and Exchange Commission is effective. This preliminary prospectus is not an offer to sell these securities and it is not soliciting offers
to buy these securities in any state or other jurisdiction where the offer or sale is not permitted.
SUBJECT TO COMPLETION, DATED
FEBRUARY 2, 2024
Preliminary Prospectus
Agile Therapeutics, Inc.
$15,000,000
Up to
Shares of Common Stock and
Accompanying Series E-1 Warrants to
Purchase up to Shares of Common Stock
and Series E-2 Warrants to Purchase
up to Shares of Common Stock
or
Up to
Series F Pre-funded Warrants to Purchase up to
Shares of Common Stock and Accompanying Series E-1
Warrants to Purchase up to Shares
of Common Stock and Series E-2
Warrants to Purchase up to Shares
of Common Stock
Placement Agent Warrants
to Purchase up to Shares of
Common Stock
Up to Shares
of Common Stock underlying the Series E-1 Warrants, Series E-2 Warrants, Series F Pre-funded
Warrants, and Placement Agent Warrants
We are offering up to
shares of common
stock, together with Series E-1 warrants to purchase up to shares
of common stock (the “Series E-1 warrants”) and Series E-2 warrants to purchase up to shares
of common stock (the “Series E-2 warrants”, and together with the Series E-1 warrants, the “Series E
warrants”) at an assumed combined public offering price of $
per share and Series E warrants, which is equal to the last reported sale price per share of our common stock on the Nasdaq Capital
Market on , 2024 (and the shares issuable from time to time upon exercise of the Series E warrants) pursuant to this prospectus.
The shares of common stock and Series E warrants will be separately issued, but the shares of common stock and Series E warrants
will be issued to purchasers in the ratio of one to one. Each Series E-1 warrant will have an exercise price of $ per
share, will be exercisable upon beginning on the effective date of stockholder approval of the issuance of the shares upon exercise of
the Series E warrants (the “Warrant Stockholder Approval”), provided however, if the Pricing Conditions (as defined
below) are met, the Series E warrants will be exercisable upon issuance (the “Initial Exercise Date”) and will expire
five years from the Initial Exercise Date. Each Series E-2 warrant will have an exercise price of $ per
share, will be exercisable beginning on the Initial Exercise Date and will expire eighteen months from the Initial Exercise Date. As
used herein “Pricing Conditions” means that the combined offering price per share and accompanying Series E warrants
is such that the Warrant Stockholder Approval is not required under Nasdaq rules because either (i) the offering is an at-the-market
offering under Nasdaq rules and such price equals or exceeds the sum of (a) the applicable “Minimum Price” per
share under Nasdaq rule 5635(d) plus (b) $0.125 per whole share of common stock underlying the Series E warrants
or (ii) the offering is a discounted offering where the pricing and discount (including attributing a value of $0.125 per whole
share underlying the Series E warrants) meet the pricing requirements under the Nasdaq rules.
We are also offering up
to pre-funded warrants (the “Series F pre-funded warrants”
and collectively with the Series E warrants, the “warrants”) to those purchasers whose purchase of shares of common
stock in this offering would result in the purchaser, together with its affiliates and certain related parties, beneficially owning more
than 4.99% (or, at the election of the purchaser, 9.99%) of our outstanding common stock following the consummation of this offering
in lieu of the shares of our common stock that would result in ownership in excess of 4.99% (or, at the election of the purchaser, 9.99%).
Each Series F pre-funded warrant will be exercisable for one share of common stock at an exercise price of $0.001 per share. Each
Series F pre-funded warrant is being issued together with the same Series E warrants described above being issued with each
share of common stock. The assumed combined public offering price for each such Series F pre-funded warrant, together with the Series E
warrants, is $ , which is equal to the last reported sale
price of our common stock on the Nasdaq Capital Market on , 2024 less the $0.001 per share exercise price of each such Series F
pre-funded warrant. Each Series F pre-funded warrant will be exercisable upon issuance and may be exercised at any time until all
of the pre-funded warrants are exercised in full. The Series F pre-funded warrants and Series E warrants are immediately separable
and will be issued separately in this offering. We are also registering the shares issuable from time to time upon exercise of the Series F
pre-funded warrants and Series E warrants. For each Series F pre-funded warrant and the accompanying Series E warrants we sell,
the number of shares of common stock and the accompanying Series E warrants we are offering will be decreased on a one-for-one basis.
There is no established public
trading market for the warrants, and we do not expect a market to develop. We do not intend to apply for listing of the warrants on any
securities exchange or other nationally recognized trading system. Without an active trading market, the liquidity of the warrants will
be limited.
This offering will terminate
on , unless we decide to terminate the offering (which we
may do at any time in our discretion) prior to that date. We will have one closing for all the securities purchased in this offering.
The combined public offering price per share of common stock (or Series F pre-funded warrant) and Series E warrants will be
fixed for the duration of this offering.
We have engaged , or the
placement agent, to act as our exclusive placement agent in connection with this offering. The placement agent has agreed to use its
reasonable best efforts to arrange for the sale of the securities offered by this prospectus. The placement agent is not purchasing or
selling any of the securities we are offering and the placement agent is not required to arrange the purchase or sale of any specific
number of securities or dollar amount. We have agreed to pay to the placement agent the placement agent fees set forth in the table below,
which assumes that we sell all of the securities offered by this prospectus. There is no arrangement for funds to be received in escrow,
trust or similar arrangement. There is no minimum offering requirement. We will bear all costs associated with the offering. See “Plan
of Distribution” on page 66 of this prospectus for more information regarding these arrangements.
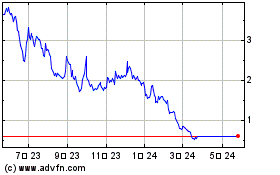
Our common stock is listed
on the Nasdaq Capital Market under the symbol “AGRX.” On , 2024, the last reported sale price of our common stock on the
Nasdaq Capital Market was $ per share.
All share, Series E warrant, and Series F pre-funded warrant numbers are based on an assumed combined public offering price
of $ per share or Series F pre-funded warrant, as applicable,
and Series E warrants.
The combined public offering
price per share and Series E warrants and the combined public offering price per Series F pre-funded warrant and Series E
warrants will be determined between us and investors based on market conditions at the time of pricing, and may be at a discount to the
current market price of our common stock. Therefore, the recent market price used throughout this prospectus may not be indicative of
the actual public offering price.
Investing in the offered
securities involves a high degree of risk. See “Risk Factors” beginning on page 10 of this prospectus and the section
entitled “Risk Factors” included in our most recent Annual Report on Form 10-K, as revised or supplemented by our subsequent
Quarterly Reports on Form 10-Q, which are or will be incorporated herein by reference, for a discussion of information that you should
consider before investing in our securities.
On March 27, 2023,
we received a deficiency letter from the Listing Qualifications Department, or the Staff, of the Nasdaq Stock Market, or Nasdaq, notifying
us that we are not in compliance with the minimum stockholders’ equity requirement for continued listing on the Nasdaq Capital Market.
Nasdaq Listing Rule 5550(b)(1) requires companies listed on the Nasdaq Capital Market to maintain stockholders’ equity
of at least $2,500,000 (the “Stockholders’ Equity Requirement”). In accordance
with Nasdaq rules, we were provided 45 calendar days from the receipt of the Nasdaq notification, or until May 11, 2023, to submit
a plan to regain compliance (the “Compliance Plan”). We submitted that plan on May 11, 2023. On June 2, 2023,
we received a letter (the “June Extension Notice”) from Nasdaq notifying us that we had been granted an additional 180-day
period, or until September 25, 2023, to regain compliance with the Stockholders’ Equity Requirement. On September 27,
2023, we received a notice from the Staff advising us that the Staff had determined that we did not meet the terms of the extension and
that unless we requested an appeal, the Staff would proceed with delisting. We submitted a hearing request to the Nasdaq Hearings Panel
(the “Panel”) on October 4, 2023. The request stayed any delisting action by the Staff at least until the hearing process
concludes and any extension granted by the Panel expires. The Panel hearing took place on November 30, 2023, where we presented a
multi-step plan to regain compliance with the Stockholders’ Equity Requirement. Our common stock will continue to trade on the Nasdaq
Capital Market under the symbol “AGRX” at least pending the ultimate conclusion of the hearing process.
On December 5, 2023,
we received a letter (the “December Extension Notice”) from the Panel notifying us that it had been granted an additional
period, or until February 16, 2024, to regain compliance with the Stockholders’ Equity Requirement. If our securities are delisted,
it could be more difficult to buy or sell our securities and to obtain accurate quotations, and the price of our securities could suffer
a material decline. Delisting could also impair the liquidity of our common stock and could harm our ability to raise capital through
alternative financing sources on terms acceptable to us, or at all, and may result in potential loss of confidence by investors, employees,
and fewer business development opportunities.
| | |
Per Share
and
Series E
Warrants | |
Per Series F
Pre-Funded
Warrant
and Series E
Warrants | |
| Total | |
| Public offering price | |
$ | |
$ | |
$ | | |
| Placement Agent’s fees(1) | |
$ | |
$ | |
$ | | |
| Proceeds, before expenses, to us(2) | |
$ | |
$ | |
$ | | |
| |
(1) |
We have agreed to pay
the placement agent a total cash fee equal to 7.0% of the gross proceeds raised in this offering. We have also agreed to pay the
placement agent a management fee equal to 1.0% of the gross proceeds raised in this offering and to reimburse the placement agent
for its non-accountable expenses in the amount of $10,000 and for its legal fees and expenses and other out-of-pocket expenses in
an amount up to $100,000, and for its clearing expenses in the amount of $15,950. In addition, we have agreed to issue to the placement
agent, or its designees, warrants to purchase a number of shares of our common stock equal to 5.0% of the aggregate number of shares
of common stock and Series F pre-funded warrants being offered at an exercise price equal to 125% of the combined public offering
price per share of common stock and Series E warrants. We refer you to “Plan of Distribution” on page 66 of
this prospectus for additional information regarding placement agent compensation. |
| |
(2) |
Because there is no
minimum number of securities or amount of proceeds required as a condition to closing in this offering, the actual public offering
amount, placement agent fees, and proceeds to us, if any, are not presently determinable and may be substantially less than the total
maximum offering amounts set forth above. We refer you to “Plan of Distribution” on page 66 of this prospectus for
additional information regarding placement agent compensation. |
The placement agent expects
to deliver the shares and warrants to purchasers in the offering on or about ,
2024, subject to satisfaction of certain conditions.
Neither the Securities
and Exchange Commission nor any state securities commission has approved or disapproved of these securities or determined if this prospectus
is truthful or complete. Any representation to the contrary is a criminal offense.
Prospectus dated , 2024
TABLE OF CONTENTS
ABOUT THIS PROSPECTUS
The registration statement
we filed with the Securities and Exchange Commission (the “SEC”) includes exhibits that provide more detail of the matters
discussed in this prospectus. You should read this prospectus, the related exhibits filed with the SEC, and the documents incorporated
by reference herein before making your investment decision. You should rely only on the information provided in this prospectus and the
documents incorporated by reference herein or any amendment thereto. In addition, this prospectus contains summaries of certain provisions
contained in some of the documents described herein, but reference is made to the actual documents for complete information. All of the
summaries are qualified in their entirety by the actual documents. Copies of some of the documents referred to herein have been filed,
will be filed or will be incorporated by reference as exhibits to the registration statement of which this prospectus is a part, and you
may obtain copies of those documents as described below under the heading “Where You Can Find Additional Information.”
We have not, and the placement
agent has not, authorized anyone to provide any information or to make any representations other than those contained in this prospectus,
the documents incorporated by reference herein or in any free writing prospectuses prepared by or on behalf of us or to which we have
referred you. We take no responsibility for, and can provide no assurance as to the reliability of, any other information that others
may give you. The information contained in this prospectus, the documents incorporated by reference herein or in any applicable free writing
prospectus is current only as of its date, regardless of its time of delivery or any sale of our securities. Our business, financial condition,
results of operations and prospects may have changed since that date.
This prospectus is an offer
to sell only the securities offered hereby, and only under circumstances and in jurisdictions where it is lawful to do so. We are not,
and the placement agent is not, making an offer to sell these securities in any state or jurisdiction where the offer or sale is not permitted.
All other trademarks, trade
names and service marks appearing in this prospectus or the documents incorporated by reference herein are the property of their respective
owners. Use or display by us of other parties’ trademarks, trade dress or products is not intended to and does not imply a relationship
with, or endorsements or sponsorship of, us by the trademark or trade dress owner. Solely for convenience, trademarks, tradenames and
service marks referred to in this prospectus appear without the ® and ™ symbols, but those references are not intended to indicate,
in any way, that we will not assert, to the fullest extent under applicable law, our rights or that the applicable owner will not assert
its rights, to these trademarks and trade names.
FORWARD-LOOKING STATEMENTS
This prospectus,
including the information incorporated by reference into this prospectus, contains “forward-looking statements” within the
meaning of Section 27A of the Securities Act of 1933, as amended, or the Securities Act, and Section 21E of the Securities
Exchange Act of 1934, as amended, or the Exchange Act. In some cases, these forward-looking statements can be identified by the use of
forward-looking terminology, including the terms “believes,” “estimates,” “anticipates,” “expects,”
“plans,” “intends,” “may,” “designed,” “could,” “might,” “will,”
“should,” “approximately” or, in each case, their negative or other variations thereon or comparable terminology,
although not all forward-looking statements contain these words. They appear in a number of places throughout this prospectus and include
statements regarding our current intentions, beliefs, projections, outlook, analyses or current expectations concerning, among other
things, our ongoing and planned manufacturing and commercialization of Twirla®, the potential market acceptance and uptake of Twirla®,
including the level of reimbursement from third-party payors, the development of our other potential product candidates, the attractiveness
of our business to potential investors or business partners, the strength and breadth of our intellectual property, our planned clinical
studies, the timing of and our ability to make regulatory filings and obtain and maintain regulatory approvals for our potential product
candidates, the legal and regulatory landscape impacting our business, the degree of clinical utility of our products, particularly in
specific patient populations, expectations regarding study data, our results of operations, financial condition, liquidity, prospects,
growth and strategies, including expense reduction strategies, the length of time that we will be able to continue to fund our operating
expenses and capital expenditures, our expected financing needs and sources of financing, the industry in which we operate and the trends
that may affect the industry or us.
By their nature,
forward-looking statements involve risks and uncertainties because they relate to events, competitive dynamics, and healthcare, regulatory
and scientific developments and depend on the economic circumstances that may or may not occur in the future or may occur on longer or
shorter timelines than anticipated. Although we believe that we have a reasonable basis for each forward-looking statement contained in
this prospectus, we caution you that forward-looking statements are not guarantees of future performance and that our actual results of
operations, financial condition and liquidity, and the development of the industry in which we operate may differ materially from the
forward-looking statements contained in this prospectus. In addition, even if our results of operations, financial condition and liquidity,
and the development of the industry in which we operate are consistent with the forward-looking statements contained in this prospectus,
they may not be predictive of results or developments in future periods.
Some of the factors that we believe
could cause actual results to differ from those anticipated or predicted include:
| |
· |
our available cash and our ability to obtain additional funding to fund our business plan without delay and to continue as a going concern; |
| |
|
|
| |
· |
our ability to come into compliance with the listing requirements of the Nasdaq Capital Market; |
| |
|
|
| |
· |
our ability to successfully maintain and enhance the commercialization of and increase the uptake for Twirla, our only approved product; |
| |
· |
the rate and degree of market acceptance of Twirla by physicians, patients, clinics, institutions, third-party payors and others in the healthcare community; |
| |
· |
our ability to obtain adequate coverage and reimbursement for Twirla in the United States from private and public third-party payors; |
| |
· |
the size and growth of the markets for Twirla and our ability to serve those markets; |
| |
· |
shortages of key materials in the supply chain implicating the manufacture and distribution of Twirla; |
| |
· |
regulatory and legislative developments in the United States and foreign countries, which could include, among other things, a government shutdown or limiting access to prescription contraceptives; |
| |
· |
the accuracy of our estimates regarding expenses, future revenues, capital requirements and needs for additional financing; |
| |
· |
the growth in demand for Twirla and our ability to manage the levels of Twirla inventory, which could result in our having to write off inventory and our inability to meet the minimum requirements under our supply agreement with Corium Innovations, Inc. (“Corium”); |
| |
· |
our ability to timely obtain from our third-party manufacturer, Corium, sufficient quantities or quality of Twirla or other materials required for a clinical trial or other tests and studies; |
| |
· |
the ability of Corium to produce commercial supply in quantities and quality sufficient to satisfy market demand for Twirla; |
| |
· |
the performance and financial condition of Corium or any of the suppliers; |
| |
· |
our ability to design and successfully complete a post-marketing long-term, prospective observational safety study comparing risks for venous thromboembolism, or VTE, and arterial thromboembolism, or ATE, in new users of Twirla to new users of oral combined hormonal contraceptives, or CHCs, and new users of Xulane in U.S. women of reproductive age using CHCs and the outcomes of our discussions with the United States Food and Drug Administration, or FDA, regarding the results of our post-marketing commitment, or PMC, to assess the residual drug content of Twirla after use; |
| |
· |
our ability to maintain regulatory approval of Twirla and the labeling under any approval we obtain; |
| |
· |
our ability to obtain and maintain intellectual property protection for Twirla and our product candidates; |
| |
· |
the success and timing of our clinical trials or other studies, including post-marketing studies for Twirla; |
| |
· |
development of unexpected safety or efficacy concerns related to Twirla; |
| |
· |
our ability to continue to develop and maintain successful sales and marketing capabilities, including our ability to maintain an effective sales force or failure to build-out and implement an effective health care compliance program; |
| |
· |
our ability to retain key employees and recruit the additional personnel we will need to support our commercialization plan for Twirla; |
| |
· |
our ability to successfully implement our strategy; and |
| |
· |
our use of the proceeds from this offering. |
We may not
actually achieve the plans, intentions or expectations disclosed in our forward-looking statements, and you should not place undue reliance
on our forward-looking statements. Actual results or events could differ materially from the plans, intentions and expectations disclosed
in the forward-looking statements we make. We have included important cautionary statements in this prospectus or in the documents incorporated
by reference in this prospectus, particularly in the “Risk Factors” section, that we believe could cause actual results or
events to differ materially from the forward-looking statements that we make. For a summary of such factors, please refer to the section
entitled “Risk Factors” in this prospectus, as updated and supplemented by the discussion of risks and uncertainties under
“Risk Factors” contained in any further supplements to our prospectus and in our most recent Annual Report on Form 10-K,
as revised or supplemented by our subsequent Quarterly Reports on Form 10-Q or our Current Reports on Form 8-K, as well as any
amendments thereto, as filed with the SEC and which are incorporated herein by reference. The information contained in this document is
believed to be current as of the date of this document. We do not intend to update any of the forward-looking statements after the date
of this document to conform these statements to actual results or to changes in our expectations, except as required by law.
In light of
these assumptions, risks and uncertainties, the results and events discussed in the forward-looking statements contained in this prospectus
or in any document incorporated herein by reference might not occur. Investors are cautioned not to place undue reliance on the forward-looking
statements, which speak only as of the date of this prospectus or the date of the document incorporated by reference in this prospectus.
We are not under any obligation, and we expressly disclaim any obligation, to update or alter any forward-looking statements, whether
as a result of new information, future events or otherwise. All subsequent forward-looking statements attributable to us or to any person
acting on our behalf are expressly qualified in their entirety by the cautionary statements contained or referred to in this section.
PROSPECTUS SUMMARY
This summary highlights
information contained in other parts of this prospectus. Because it is only a summary, it does not contain all of the information that
you should consider before investing in shares of our common stock and it is qualified in its entirety by, and should be read in conjunction
with, the more detailed information appearing elsewhere in this prospectus, any applicable free writing prospectus and the documents incorporated
by reference herein and therein. You should read all such documents carefully, especially the risk factors and our financial statements
and the related notes included or incorporated by reference herein or therein, before deciding to buy shares of our common stock. Unless
the context requires otherwise, references in this prospectus to “Agile,” “we,” “us” and “our”
refer to Agile Therapeutics, Inc.
Company Overview
We are a women’s healthcare
company dedicated to fulfilling the unmet health needs of today’s women. We are committed to innovating in women’s healthcare
where there continues to be unmet needs — not only in contraception — but also in other meaningful women’s health therapeutic
areas. We are focused on our advancement as a commercial company and the growth of our first and only product, Twirla, a once-weekly prescription
combination hormonal contraceptive patch.
Twirla is a once-weekly prescription
combination hormonal contraceptive patch. It exposes patients to an estrogen dose consistent with commonly prescribed combined hormonal
contraceptives, or CHCs, and is lower than the estrogen dose found in other marketed contraceptive patches. We believe there is a market
need for a contraceptive patch that is designed to deliver hormonal exposure equivalent to 30 mcg of estrogen and 120 mcg of progestin
in a convenient once-weekly dosage form that may support compliance in a noninvasive fashion. Twirla leverages our proprietary transdermal
patch technology called Skinfusion®. Skinfusion is designed to allow drug delivery through the skin while promoting patch adhesion
and patient comfort and wearability, which may help support compliance.
We are focused on our
advancement as a commercial company. During 2024, we plan to continue implementing our commercialization plan for Twirla, with the
goal of establishing a growing position in the hormonal contraceptive market. We believe we can achieve this goal by focusing our
growth strategy in the states with the highest Twirla reimbursement potential, which we estimate will allow us to reach
approximately 45% of U.S. women between the ages of 18-24. We also believe we can increase uptake of Twirla in the United States by
growing our telemedicine presence through our partnerships and our existing partnership with Nurx®, and driving
growth in the non-retail channel through our collaboration with Afaxys, which provides us access to some of the largest Planned
Parenthood organizations in the country. In addition to growing Twirla, we also plan to continue pursuing opportunities to broaden
our portfolio to address areas of unmet medical need in women’s health.
Recent Developments
Perceptive Credit Agreement Amendment
On October 30, 2023, we entered into a seventh
amendment (the “Seventh Amendment”) to that certain Credit Agreement and Guaranty, dated February 10, 2020, as amended
(the “Perceptive Credit Agreement”), with Perceptive Credit Holdings III, LP (“Perceptive”). The Seventh Amendment:
(1) amends our obligations to comply with certain financial covenants relating to minimum revenue requirements, (2) amends and
waives our obligations to comply with certain financial covenants relating to minimum liquidity through December 31, 2023, and (3) requires
us to make principal payments on its outstanding loan balance of $150,000 per month beginning on December 1, 2023. The Company will
make monthly payments of $150,000 until February 10, 2024 (“Maturity Date”), at which time all remaining principal amount
outstanding is due.
Warrant Amendments
On December 3, 2023, we entered into a Warrant
Amendment and Additional Issuance Agreement (“Warrant Amendment and Additional Issuance Agreement”) relating to the amendment
of warrants to purchase shares of our common stock that were issued in transactions on March 14, 2022, April 25, 2022, and May 25,
2023 (collectively, the “Warrants”). Collectively, the Warrants represent the right to purchase approximately 3.8 million
shares of our common stock. Under the terms of the Warrant Amendment and Additional Issuance Agreement, the holder agreed to revise provisions
related to the use of a Black-Scholes model to value the Warrants in the event of a change of control transaction. The holder also agreed
to revise provisions related to the cashless exercise of the Warrants. In exchange for the holder’s agreement to amend the Warrants,
we agreed to issue an additional new warrant (the “New Warrant”) to purchase 1,005,560 shares of our common stock. The New
Warrant has an exercise price of $2.09 per share. The New Warrant is exercisable six months after issuance and will expire five years
from the date that the New Warrant is initially exercisable. The exercise price of the New Warrant is subject to adjustment for stock
splits, reverse splits, and similar capital transactions as described in the New Warrant.
We intend to use the revisions to the Warrants
to support the reclassification of the Warrants from a liability and to account for the Warrants as a component of stockholders’
equity on our balance sheet. We also intend to use the reclassification of the Warrants as part of our plan to regain compliance with
the Nasdaq listing requirements relating to minimum shareholders’ equity (as discussed below).
Risks Associated with this Offering
This
offering is subject to numerous risks and uncertainties, including those highlighted in the section entitled “Risk Factors”
immediately following this prospectus summary and the section entitled “Risk Factors” included in our Annual Report on Form 10-K for the fiscal year ended December 31, 2022 as revised or supplemented by our Quarterly Reports on Form 10-Q for the
quarters ended March 31, 2023, June 30, 2023 and September 30, 2023, which are incorporated herein by reference. These
risks include, but are not limited to, the following:
| |
· |
we are not in compliance with the Nasdaq continued listing requirements. If we are unable to comply with the continued listing requirements of the Nasdaq Capital Market, our common stock could be delisted, which could affect our common stock’s market price and liquidity and reduce our ability to raise capital; |
| |
· |
the price of our common stock may be volatile and fluctuate substantially, and you may not be able to resell your shares at or above the price you paid for them; |
| |
· |
we have identified a material weakness in our internal control over financial reporting. This material weakness could continue to adversely affect our results of operations and financial condition. In the future, we may identify additional material weaknesses or otherwise fail to maintain an effective system of internal control over financial reporting or adequate disclosure controls and procedures, which may result in material errors in our financial statements or cause us to fail to meet our period reporting obligations; |
| |
· |
there is no public market for the warrants being offered in this offering; |
| |
· |
holders of warrants purchased in this offering will have no rights as common stockholders until such holders exercise their warrants and acquire our common stock; |
| |
· |
the warrants being offered may not have value; |
| |
· |
certain of our outstanding common stock purchase warrants contain price protection provisions (anti-dilution protection) in the event that we sell our securities at prices lower than the current exercise price of such warrants, which may have a negative impact on the trading price of our common stock or impair our ability to raise capital; |
| |
· |
management will have broad discretion as to the use of the proceeds from this offering, and we may not use the proceeds effectively; and |
| |
· |
we have incurred operating losses in each year since our inception and expect to continue to incur substantial losses for the foreseeable future. Management has concluded that these factors raise substantial doubt about our ability to continue as a going concern. |
Corporate Information
Information concerning our
business is contained in the documents that we file with the SEC as a reporting company under the Exchange Act, which are accessible at
www.sec.gov, and on our website at www.agiletherapeutics.com. The information contained on, or that can be accessed through, our website
is not a part of this prospectus. Investors should not rely on any such information in deciding whether to purchase our common stock.
We have included our website address in this prospectus solely as an inactive textual reference.
Our principal executive offices
are located at 500 College Road East, Suite 310, Princeton, New Jersey 08540, and our telephone number is (609) 683-1880.
THE OFFERING
| Securities
we are offering |
Up
to shares of our
common stock, Series E-1 warrants to purchase up to shares
of common stock, and Series E-2 warrants to purchase up to
shares of common stock or up to
Series F pre-funded warrants to purchase shares of common stock, Series E-1 warrants to purchase up to shares
of common stock, and Series E-2 warrants to purchase up to shares
of common stock in each case, at a combined public offering price of $ per
share (or Series F pre-funded warrant in lieu thereof) and Series E warrants. The shares, or Series F pre-funded
warrants, and Series E warrants will be separately transferable immediately upon issuance, but the shares, or Series F
pre-funded warrants, and Series E warrants will be issued to purchasers in the ratio of one to one. |
| |
|
| Description
of Series E warrants |
Each
Series E-1 warrant will have an exercise price of $ per share,
will be exercisable beginning on the Initial Exercise Date and will expire five years from the Initial Exercise Date. Each Series E-2
warrant will have an exercise price of $ per share, will be exercisable
beginning on the Initial Exercise Date and will expire eighteen months from the Initial Exercise Date. This prospectus also relates
to the offering of the shares of common stock issuable upon exercise of these Series E warrants. |
| |
|
| Description of Series F pre-funded warrants |
If the issuance of shares of our common stock to a purchaser in this offering would result in such purchaser, together with its affiliates and certain related parties, beneficially owning more than 4.99% (or, at the election of the purchaser, 9.99%) of our outstanding common stock following the consummation of this offering, then such purchaser may purchase, if they so choose, in lieu of the shares of our common stock that would result in such excess ownership, a Series F pre-funded warrant to purchase shares of our common stock for a purchase price per share of common stock subject to such Series F pre-funded warrant equal to the per share public offering price for the common stock in this offering less $0.0001. Each Series F pre-funded warrant will have an exercise price of $0.0001 per share, will be exercisable upon issuance and may be exercised at any time until all of the pre-funded warrants are exercised in full. Purchasers of Series F pre-funded warrants will also receive Series E warrants as if such purchasers were buying shares of our common stock in this offering. This prospectus also relates to the offering of the shares of common stock issuable upon exercise of these Series F pre-funded warrants. |
| |
|
| |
For each Series F pre-funded warrant and the accompanying Series E warrants we sell, the number of shares of common stock and the accompanying Series E warrants we are offering will be decreased on a one-for-one basis. |
| |
|
| Common
stock outstanding before offering: |
shares
of common stock. |
| |
|
| Common
stock outstanding after this offering |
shares
of common stock, assuming no sale of Series F pre-funded warrants in this offering and no exercise of the Series E warrants
being issued in this offering and assuming a combined public offering price of $ per
share and Series E warrants, which is equal to the last reported sale price per share of our common stock on the Nasdaq Capital
Market on , 2024. |
| |
|
| Use of proceeds |
We intend to use the net proceeds from this offering for working capital, business development activities, repayment of certain indebtedness and general corporate purposes. See “Use of Proceeds” on page 17 of this prospectus. |
| |
|
| Risk factors |
See “Risk Factors” beginning on page 10 of this prospectus and the section entitled “Risk Factors” included in our Annual Report on Form 10-K for the fiscal year ended December 31, 2022 as revised or supplemented by our Quarterly Reports on Form 10-Q for the quarters ended March 31, 2023, June 30, 2023, and September 30, 2023, which are incorporated herein by reference, and the other information included or incorporated by reference elsewhere in this prospectus for a discussion of factors you should carefully consider before deciding to invest in our securities. |
| Nasdaq Capital Market Symbol |
Our common stock is listed on the Nasdaq Capital Market under the symbol “AGRX.” There is no established trading market for the Series E warrants or the Series F pre-funded warrants, and we do not expect a trading market to develop. We do not intend to list the Series E warrants or the Series F pre-funded warrants on any securities exchange or other trading market. Without a trading market, the liquidity of the Series E warrants or Series F pre-funded warrants will be extremely limited. |
The number of shares of
our common stock to be outstanding after this offering is based on 2,963,657 shares of our common stock outstanding as of December 31,
2023, and excludes:
| |
· |
43,043
shares of common stock issuable upon the exercise of outstanding options to purchase common stock as of December 31, 2023 at a weighted
average exercise price of $548.70 per share; |
| |
· |
173,517 shares of common
stock issuable upon the vesting of outstanding restricted stock units as of December 31, 2023; |
| |
· |
6,200 shares of common
stock reserved for future issuance under our 2023 Equity Incentive Plan as of December 31, 2023; and |
| |
· |
5,589,637
shares of common stock issuable upon the exercise of outstanding warrants as of December 31, 2023 at a weighted average exercise
price of $8.55 per share. |
Unless otherwise indicated,
all information contained in this prospectus assumes:
| |
· |
no exercise of the outstanding options or warrants described in the bullets above; |
| |
|
|
| |
· |
no exercise of the Series E warrants or the placement agent warrants issued in this offering; and |
| |
· |
no sale of Series F pre-funded warrants in this offering. |
All information included
in this prospectus, where relevant, has been adjusted to reflect a 1-for-50 reverse stock split of our common stock effective on April 11,
2023.
RISK FACTORS
An investment in our common
stock involves a high degree of risk. Before deciding whether to invest in our common stock, you should carefully consider the risks described
below and those discussed under the Section captioned “Risk Factors” contained in our Annual Report on Form 10-K for the year ended December 31, 2022, our Quarterly Report on Form 10-Q for the quarter ended March 31, 2023, our Quarterly Report on Form 10-Q for the quarter ended June 30, 2023, our Quarterly Report on Form 10-Q for the quarter ended September 30, 2023, or our current reports on Form 8-K, each as filed with the SEC and which are incorporated by reference in this prospectus,
together with other information in this prospectus, the information and documents incorporated by reference herein and therein, and in
any free writing prospectus that we have authorized for use in connection with this offering. If any of these risks actually occurs, our
business, financial condition, results of operations or cash flow could be seriously harmed. This could cause the trading price of our
common stock to decline, resulting in a loss of all or part of your investment. Please also read carefully the section above entitled
“Forward-Looking Statements.”
Risks Related to This Offering
We
have incurred operating losses in each year since our inception and we incurred losses every quarter in 2023. We
may continue to incur substantial losses in the future if we are unable to generate positive cash flow from operations. Management has
concluded that these factors raise substantial doubt about our ability to continue as a going concern.
We
have incurred losses in each year since our inception in December 1997. Our net loss was $10.0 million and $21.5 million
for the nine months ended September 30, 2023 and 2022, respectively, and $25.4 million, $71.1 million and $51.9 million for the years
ended December 31, 2022, 2021 and 2020, respectively. As of September 30, 2023, we had an accumulated deficit of approximately
$418.7 million. As of December 31, 2023, our unaudited cash and cash equivalents were approximately $2.5 million, which will not be sufficient
to fund our current and planned operations through December 31, 2024, which raises substantial doubt about our ability to continue as
a going concern. Substantial doubt about our ability to continue as a going concern may create negative reactions to the price of our
common stock and we may have a more difficult time obtaining financing in the future.
Specialty
pharmaceutical product development is a speculative undertaking, involves a substantial degree of risk and is a capital-intensive business.
We may incur expenses without corresponding revenues until we are able to sell Twirla in significant quantities, which may not happen.
We have devoted most of our financial resources to research and development, including our non-clinical development activities and clinical
trials. We will require additional capital to fund our operating needs beyond January 2024, including among other items, the commercialization
of Twirla and advancing the development of our other potential product candidates. We may not be able to obtain sufficient additional
funding to continue our operations at planned levels and be forced to reduce, or even terminate, our operations. To date, we have financed
our operations primarily through sales of common stock, convertible preferred stock and convertible promissory notes and to a lesser extent,
through term loans and government grants.
We expect
that our expenses will increase as we continue to commercialize Twirla. As a result, we may continue to incur substantial losses for the
foreseeable future if we are unable to generate positive cash flow from operations. We are uncertain when or if we will be able to achieve
or sustain profitability. If we achieve profitability in the future, we may not be able to sustain profitability in subsequent periods.
Any failure to become and remain profitable could impair our ability to sustain operations and adversely affect the price of our common
stock and our ability to raise additional capital. We are significantly dependent on the success of Twirla, and if we do not achieve the
commercial success of Twirla and/or are unable to obtain additional funding, we will need to reassess our operating capital needs and
may be unable to continue our operations at planned levels and be forced to reduce, or even terminate, our operations.
Our forecast
of the period of time through which our financial resources will be adequate to support our operating requirements is a forward-looking
statement and involves risks and uncertainties, and actual results could vary as a result of a number of factors, including the factors
discussed elsewhere in this “Risk Factors” section and Part I, Item 1A “Risk Factors” of our Annual
Report on Form 10-K filed with the SEC on March 23, 2023, as revised or supplemented by our quarterly reports on Form 10-Q
and current reports on Form 8-K, each as filed with the SEC. We have based this estimate on a number of assumptions that may prove
to be wrong, and changing circumstances beyond our control may cause us to consume capital more rapidly than we currently anticipate.
Our inability to obtain additional funding when we need it could seriously harm our business.
This offering is being made on a best efforts
basis and we may sell fewer than all of the securities offered hereby and may receive significantly less in net proceeds from this offering,
which will provide us only limited working capital.
This offering is being made on a best efforts
basis and we may sell fewer than all of the securities offered hereby and may receive significantly less in net proceeds from this offering.
Assuming that we receive net proceeds of approximately $ million from this offering
(assuming an offering with gross proceeds of $ million), we believe that the net proceeds
from this offering, together with our existing cash and cash equivalents, will meet our capital needs for the next months
under our current business plan. Assuming that we receive net proceeds of approximately $ million
from this offering (assuming an offering with gross proceeds of $ million), we
believe that the net proceeds from this offering, together with our existing cash and cash equivalents, will satisfy our capital needs
for the next months under our current business plan. Assuming that we receive
net proceeds of approximately $ million from this offering (assuming an offering
with gross proceeds of $ million), we believe that the net proceeds from this
offering, together with our existing cash and cash equivalents, will satisfy our capital needs for the next months
under our current business plan. Without giving effect to the receipt of any proceeds from this offering, we currently estimate that
our existing cash and cash equivalents are sufficient to fund business operations into ,
.
We are not in compliance
with the Nasdaq continued listing requirements. If we are unable to comply with the continued listing requirements of the Nasdaq Capital
Market, our common stock could be delisted, which could affect our common stock’s market price and liquidity and reduce our ability
to raise capital.
On March 27, 2023, we
received a deficiency letter from Nasdaq notifying us that we are not in compliance with the minimum stockholders’ equity requirement
for continued listing on the Nasdaq Capital Market. Nasdaq Listing Rule 5550(b)(1) (the “Rule”) requires companies
listed on the Nasdaq Capital Market to maintain stockholders’ equity of at least $2,500,000 (the “Stockholders’ Equity
Requirement”). Our Annual Report on Form 10-K for the fourth quarter and year ended December 31, 2022 reported stockholders’
equity of $(5,545,000), which is below the Stockholders’ Equity Requirement for continued listing on the Nasdaq Capital Market.
As of the date of this filing, the Company does not have a market value of listed securities of $35 million, or net income from continued
operations of $500,000 in the most recently completed fiscal year or in two of the last three most recently completed fiscal years, the
alternative quantitative standards for continued listing on the Nasdaq Capital Market. In accordance with Nasdaq rules, we were provided
45 calendar days from the receipt of the Nasdaq notification, or until May 11, 2023, to submit a plan to regain compliance (the “Compliance
Plan”). We submitted that plan on May 11, 2023.
On June 2, 2023, we
received a letter (the “June Extension Notice”) from Nasdaq notifying us that we had been granted an additional 180-day
period, or until September 25, 2023, to regain compliance with Nasdaq Listing Rule 5550(b)(1). On September 27, 2023, we
received a notice from the Staff advising us that the Staff had determined that we did not meet the terms of the extension and that unless
we requested an appeal, the Staff would proceed with delisting. We submitted a hearing request to the Nasdaq Hearings Panel (the “Panel”)
on October 4, 2023. The request stayed any delisting action by the Staff at least until the hearing process concludes and any extension
granted by the Panel expires.
The hearing before the Panel
took place on November 30, 2023. At the hearing, we presented a multi-step plan to regain compliance with the Stockholders’
Equity Requirement, which included relying on the proceeds of this Offering to improve stockholder equity. Our common stock will continue
to trade on the Nasdaq Capital Market under the symbol “AGRX” at least pending the ultimate conclusion of the hearing process.
On December 5, 2023,
we received a letter (the “December Extension Notice”) from the Panel notifying us that it had been granted an additional
period, or until February 16, 2024, to regain compliance with the Stockholders’ Equity Requirement.
There is no guarantee that
the Panel will grant this or any extension. In addition, even if an extension is granted, if we fail to complete this Offering, or any
part of our compliance plan presented to the Panel, we may be unable to regain compliance with the Stockholders’ Equity Requirement
and may be delisted. There can be no assurance that following the closing of this Offering, that we will be able to regain compliance
with the Stockholders’ Equity Requirement or otherwise maintain compliance any of the other listing requirements, which could lead
to the delisting of our securities.
If our securities are delisted,
it could be more difficult to buy or sell our securities and to obtain accurate quotations, and the price of our securities could suffer
a material decline. Delisting could also impair the liquidity of our common stock and could harm our ability to raise capital through
alternative financing sources on terms acceptable to us, or at all, and may result in potential loss of confidence by investors, employees,
and fewer business development opportunities.
If we do not raise
sufficient funds in this offering, it will be difficult for us to come into compliance with the Stockholders’ Equity Requirement
by the deadline of February 16, 2024.
If we do not receive
sufficient net proceeds in this offering, we may not come into compliance with the Stockholders’ Equity Requirement discussed above.
There are limited ways we can improve our financial condition by February 16, 2024 in order to demonstrate compliance with the Stockholders’
Equity Requirement. We may attempt to raise further capital, on less favorable terms, which may result in further dilution to you and
our current stockholders. We may be unsuccessful in raising further capital, or otherwise satisfying the Stockholders’ Equity Requirement,
and Nasdaq may take steps to delist our common stock. Such a delisting would likely have a negative effect on the price of our common
stock and would impair your ability to sell or purchase our common stock when you wish to do so. In the event of a delisting, we can
provide no assurance that we will be able to continue as a going concern or that any action taken by us to restore compliance with listing
requirements would allow our common stock to become listed again, stabilize the market price or improve the liquidity of our common stock,
prevent our common stock from dropping below the Nasdaq minimum bid price requirement or prevent future non-compliance with Nasdaq’s
listing requirements.
The price of our common
stock may be volatile and fluctuate substantially, and you may not be able to resell your shares at or above the price you paid for them.
The shares
sold in this offering, if any, will be sold from time to time at various prices. The market price for shares of our common stock may be
subject to wide fluctuations in response to many risk factors, including:
| |
· |
Our failure to commercialize Twirla or develop and commercialize additional potential product candidates; |
| |
· |
Our ability to successfully enhance the commercialization and increase the uptake for Twirla, our only approved product; |
| |
· |
Unanticipated efficacy, safety or tolerability concerns related to the use of Twirla; |
| |
· |
Regulatory actions with respect to Twirla; |
| |
· |
The growth in demand for Twirla and our ability to manage the levels of Twirla inventory, which could result in our having to write off inventory and our inability to meet the minimum requirements under our supply agreement with Corium; |
| |
· |
Inability to obtain adequate product supply of Twirla or inability to do so at acceptable prices; |
| |
· |
Adverse results or delays in our clinical trials for our potential product candidates; |
| |
· |
Changes in laws or regulations applicable to Twirla or any future potential product candidates, including but not limited to clinical trial requirements for approvals, post-approval requirements, and product marketing, advertising, and promotional requirements and limitations; |
| |
|
|
| |
· |
Coverage and reimbursement policies with respect to Twirla or our product candidates, if approved; |
| |
· |
Our ability to maintain compliance with the listing requirements of the Nasdaq Capital Market; |
| |
· |
Actual or anticipated fluctuations in our financial condition and operating results; |
| |
· |
Actual or anticipated changes in our growth rate relative to our competitors; |
| |
· |
Competition from existing products or new products that may emerge; |
| |
· |
Announcements by us, our collaborators or our competitors of significant acquisitions, strategic partnerships, joint ventures, collaborations or capital commitments; |
| |
· |
Failure to meet or exceed financial estimates and projections of the investment community or that we provide to the public; |
| |
· |
Issuance of new or updated research or reports by securities analysts; |
| |
· |
Fluctuations in the valuation of companies perceived by investors to be comparable to us; |
| |
· |
Share price and volume fluctuations attributable to inconsistent trading volume levels of our shares; |
| |
· |
Additions or departures of key personnel; |
| |
· |
Disputes or other developments related to proprietary rights, including patents, litigation matters and our ability to obtain and retain patent protection for our technologies; |
| |
· |
Announcement or expectation of additional debt or equity financing efforts; |
| |
· |
Sales of our common stock by us, our insiders or our other stockholders; and |
| |
· |
General economic and market conditions. |
In addition,
the stock market has experienced significant volatility, particularly with respect to pharmaceutical and other life sciences company stocks.
The volatility of such stocks often does not relate to individual company performance. As we operate in a single industry, we are especially
vulnerable to these factors to the extent that they affect our industry or our product candidates or, to a lesser extent, our markets.
In the past, securities class-action litigation has often been instituted against companies following periods of volatility in their stock
price. We may face securities class-action litigation if we fail to commercialize Twirla or cannot obtain regulatory approvals for our
product candidates. Such litigation, if instituted against us, could cause us to incur substantial costs to defend such claims and divert
management’s attention and resources, which could materially harm our financial condition and results of operations.
There is no public market
for the warrants being offered in this offering.
There is no established trading
market for the warrants being offered in this offering, and we do not expect a market to develop. In addition, we do not intend to apply
to list the warrants on any securities exchange or nationally recognized trading system, including the Nasdaq Capital Market. Without
an active market, the liquidity of the warrants will be limited.
Holders of warrants purchased
in this offering will have no rights as common stockholders until such holders exercise their warrants and acquire our common stock, except
as provided in the warrants.
Until holders of the warrants
being offered in this offering acquire shares of our common stock upon exercise of such warrants, the holders will have no rights with
respect to the shares of our common stock underlying such warrants. Upon exercise of the warrants, the holders will be entitled to exercise
the rights of a common stockholder only as to matters for which the record date occurs after the exercise.
The warrants being offered
may not have value.
The warrants being offered
by us in this offering have an exercise price of $ per share, subject to
certain adjustments, and will expire years from the date of issuance, after which
date any unexercised warrants will have no further value. In the event that the market price of our common stock does not exceed the
exercise price of the warrants during the period when they are exercisable, the warrants may not have any value.
The Series E warrants are not exercisable
until stockholder approval, provided however, if the Pricing Conditions are met, the Series E warrants will be exercisable upon issuance.
The Series E
warrants will have an exercise price of $ per share and
will be exercisable beginning on the effective date of the Warrant Stockholder Approval, provided however, if the Pricing Conditions
are met, the Series E warrants will be exercisable upon issuance. The Series E-1 warrants will expire on the five-year anniversary
of the Initial Exercise Date. The Series E-2 warrants will expire on the eighteen-month anniversary of the Initial Exercise Date.
While
we intend to promptly seek Warrant Stockholder Approval, there is no guarantee that the Warrant Stockholder Approval will ever
be obtained. If we are unable to obtain the Warrant Stockholder Approval, the Series E warrants may have no value.
Purchasers who purchase our securities in
this offering pursuant to a securities purchase agreement may have rights not available to purchasers that purchase without the benefit
of a securities purchase agreement.
In addition to rights and
remedies available to all purchasers in this offering under federal securities and state law, the purchasers that enter into a securities
purchase agreement will also be able to bring claims of breach of contract against us. The ability to pursue a claim for breach of contract
provides those investors with the means to enforce the covenants uniquely available to them under the securities purchase agreement including:
(i) timely delivery of shares; (ii) agreement to not enter into variable rate financings for one year from closing, subject
to an exception; (iii) agreement to not enter into any financings for days from closing, subject to certain exceptions; and (iv) indemnification
for breach of contract.
Our operating activities may be restricted as
a result of covenants related to the outstanding indebtedness under our loan agreement and we may be required to repay the outstanding
indebtedness in an event of default, which could have a materially adverse effect on our business.
In February 2020,
we entered into the Perceptive Credit Agreement, the terms of which are described in more detail throughout this prospectus. The Perceptive
Credit Agreement, as amended, subjects us to various customary affirmative and negative covenants. Our business may be adversely affected
by these restrictions on our ability to operate our business. The Perceptive Credit Agreement also subjects us to financial covenants
in respect of minimum liquidity and minimum product revenue.
The loans
provided under the Perceptive Credit Agreement are secured by substantially all of our assets. The Perceptive Credit Agreement contains
certain customary events of default, which include, among others, non-payment of principal, violation of covenants, inaccuracy of representations
and warranties, bankruptcy and insolvency events, material judgments, certain regulatory-related events and events constituting a Change
of Control (as defined in the Perceptive Credit Agreement). The facility will mature on February 10, 2024 (“Maturity Date”),
at which time all remaining principal outstanding is due. We may not have enough available cash or be able to raise additional funds through
equity or debt financings to repay such indebtedness by the Maturity Date or at the time any such event of default occurs. We have received
a waiver of certain financial covenants through December 31, 2023, see “Recent Developments” on page 5 of this
prospectus.
In the event
we do not pay off our indebtedness to Perceptive, we may need to negotiate for a waiver of our obligations to comply with certain financial
covenants. If we cannot repay our indebtedness and do not receive a waiver, we may be required to delay, limit, reduce or terminate our
pipeline development or commercialization efforts or grant to others rights to develop and market product candidates that we would otherwise
prefer to develop and market ourselves. Perceptive could also exercise its rights as collateral agent to take possession and dispose of
the collateral securing the loan for its benefit, which collateral includes substantially all of our property. Our business, financial
condition and results of operations could be materially adversely affected as a result of any of these events.
Management will have broad
discretion as to the use of the proceeds from this offering, and we may not use the proceeds effectively.
Our management
will have broad discretion with respect to the use of proceeds of this offering, including for any of the purposes described in the section
of this prospectus entitled “Use of Proceeds.” You will be relying on the judgment of our management regarding the application
of the proceeds of this offering. The results and effectiveness of the use of proceeds are uncertain, and we could spend the proceeds
in ways that you do not agree with or that do not improve our results of operations or enhance the value of our common stock. Our failure
to apply these funds effectively could have a material adverse effect on our business, delay the development of our product candidates
and cause the price of our common stock to decline.
We have identified a material
weakness in our internal control over financial reporting. This material weakness could adversely affect our results of operations and
financial condition. In the future, we may identify additional material weaknesses or otherwise fail to maintain an effective system of
internal control over financial reporting or adequate disclosure controls and procedures, which may result in material errors in our financial
statements or cause us to fail to meet our period reporting obligations.
Our management is responsible
for establishing and maintaining adequate internal control over financial reporting, evaluating the effectiveness of our internal controls
and disclosing any changes or material weaknesses identified through such evaluation. A material weakness is a deficiency, or a combination
of deficiencies, in internal control over financial reporting, such that there is a reasonable possibility that a material misstatement
of our annual or interim financial statements will not be prevented or detected on a timely basis.
In February 2023, we
determined that we incorrectly classified certain warrants that were issued to investors in connection with a public offering of our common
stock in October 2021, a registered direct offering of our preferred stock in March 2022, and a public offering of our common
stock in July 2022. Our management subsequently concluded that a material weakness existed and our internal control over financial
reporting was not effective as of October 2021.
As a result, we determined
that there were material errors in the financial statements that required a restatement of the December 31, 2021 financial statements
included in the Annual Report on Form 10-K for the year ended December 31, 2021 and our Forms 10-Q for the quarterly periods
ended March 31, 2022, June 30, 2022 and September 30, 2022. This was due to the inadequate design and implementation of
controls to evaluate the accounting for warrant classification between liability and equity.
Management has implemented
enhanced internal controls to remediate the material weakness. Specifically, we expanded and improved our review process for complex securities
and related accounting standards. We plan to further improve this process by enhancing access to accounting literature and identification
of third-party accounting professionals with whom to consult regarding complex accounting applications. The elements of our remediation
plan can only be accomplished over time, and we can offer no assurance that these initiatives will ultimately have the intended effects.
Effective internal controls
over financial reporting are necessary for us to provide reliable financial reports and, together with adequate disclosure controls and
procedures, are designed to prevent fraud. Any failure to implement required new or improved controls, or difficulties encountered in
their implementation, could cause us to fail to meet our reporting obligations. If we are not able to comply with the requirements of
the Sarbanes-Oxley Act or if we are unable to maintain effective internal control over financial reporting, we may not be able to produce
timely and accurate financial statements or guarantee that information required to be disclosed by us in the reports that we file with
the SEC, is recorded, processed, summarized, and reported within the time periods specified in SEC rules and forms. Any failure of
our internal control over financial reporting or disclosure controls and procedures could cause our investors to lose confidence in our
publicly reported information, cause the market price of our stock to decline, expose us to sanctions or investigations by the SEC or
other regulatory authorities, or impact our results of operations.
Raising additional capital
may cause dilution to our existing stockholders or restrict our operations.
We will need to seek additional
capital through a combination of private and public equity offerings, debt financings and strategic collaborations. The sale of additional
equity or convertible debt securities could result in the issuance of additional shares of our capital stock and could result in dilution
to our stockholders. The incurrence of indebtedness would result in increased fixed payment obligations and could also result in certain
restrictive covenants, such as limitations on our ability to incur additional debt, limitations on our ability to acquire or license intellectual
property rights and other operating restrictions that could adversely impact our ability to conduct our business. We cannot guarantee
that future financing will be available in sufficient amounts or on terms acceptable to us, if at all. If we are unable to raise additional
capital in sufficient amounts or on terms acceptable to us, we will be prevented from pursuing research and development efforts, and could
be forced to limit funding of our efforts to commercialize Twirla. This could harm our business, operating results and financial condition
and cause the price of our common stock to fall.
This is a best efforts offering,
no minimum amount of securities is required to be sold, and we may not raise the amount of capital we believe is required for our business
plans, including our near-term business plans.
The placement agent has agreed
to use its reasonable best efforts to solicit offers to purchase the securities in this offering. The placement agent has no obligation
to buy any of the securities from us or to arrange for the purchase or sale of any specific number or dollar amount of the securities.
There is no required minimum number of securities that must be sold as a condition to completion of this offering. Because there is no
minimum offering amount required as a condition to the closing of this offering, the actual offering amount, placement agent fees and
proceeds to us are not presently determinable and may be substantially less than the maximum amounts set forth above. We may sell fewer
than all of the securities offered hereby, which may significantly reduce the amount of proceeds received by us, and investors in this
offering will not receive a refund in the event that we do not sell an amount of securities sufficient to support our continued operations,
including our near-term continued operations. Thus, we may not raise the amount of capital we believe is required for our operations in
the short-term and may need to raise additional funds to complete such short-term operations. Such additional fundraises may not be available
or available on terms acceptable to us.
The exercise of outstanding common stock
purchase warrants and stock options will have a dilutive effect on the percentage ownership of our capital stock by existing stockholders.
As of December 31, 2023,
we had outstanding warrants to acquire 5,589,637 shares of our common stock, and stock options to purchase 43,043 shares of our common
stock. A number of such warrants have exercise prices that exceed the recent trading prices of our common stock, but such warrants contain
a cashless exercise provision that permit exercise on a cashless basis at any time where there is no effective registration statement
under the Securities Act of 1933, as amended, covering the issuance of the underlying shares. If a significant number of such warrants
and stock options are exercised by the holders, the percentage of our common stock owned by our existing stockholders will be diluted.
If you purchase our securities in this offering
you may experience future dilution as a result of future equity offerings or other equity issuances.
We will likely offer and issue
additional shares of our common stock or other equity or convertible debt securities in order to raise additional capital. Future equity
offerings or other equity issuances may be at a price per share that is equal to or greater than the price per share paid by investors
in this offering. Future investors in such offerings may have rights superior to existing stockholders, and the price per share at which
we sell additional shares of common stock or other equity or convertible debt securities in future transactions may be at a higher or
lower price per share than the price per share in this offering.
You will likely experience immediate and
substantial dilution in the net tangible book value per share of the common stock you purchase.
Since the public offering
price for our common stock in this offering will likely be substantially higher than the net tangible book value per share of our common
stock outstanding prior to this offering, you will likely suffer immediate and substantial dilution in the net tangible book value of
the common stock you purchase in this offering. The shares sold in this offering, if any, will be sold from time to time at various prices.
After giving effect to the sale of our common stock in the maximum aggregate offering amount of $ at
an assumed offering price of $ per share (the last reported sale price of our common stock on the Nasdaq Capital Market on , 2024), and
after deducting estimated offering commissions and expenses payable by us, you would suffer immediate dilution of $ per
share in the net tangible book value of the common stock. See the section entitled “Dilution” below for a more detailed discussion
of the dilution you will incur if you purchase shares in this offering.
FINRA
sales practice requirements may limit a stockholder’s ability to buy and sell our securities.
Effective June 30, 2020, the
SEC implemented Regulation Best Interest requiring that “A broker, dealer, or a natural person who is an associated person of a
broker or dealer, when making a recommendation of any securities transaction or investment strategy involving securities (including account
recommendations) to a retail customer, shall act in the best interest of the retail customer at the time the recommendation is made, without
placing the financial or other interest of the broker, dealer, or natural person who is an associated person of a broker or dealer making
the recommendation ahead of the interest of the retail customer.” This is a significantly higher standard for broker-dealers to
recommend securities to retail customers than before under FINRA suitability rules. FINRA suitability rules do still apply to institutional
investors and require that in recommending an investment to a customer, a broker-dealer must have reasonable grounds for believing that
the investment is suitable for that customer. Prior to recommending securities to their customers, broker-dealers must make reasonable
efforts to obtain information about the customer’s financial status, tax status, investment objectives and other information, and
for retail customers determine the investment is in the customer’s “best interest” and meet other SEC requirements.
Both SEC Regulation Best Interest and FINRA’s suitability requirements may make it more difficult for broker-dealers to recommend
that their customers buy speculative, low-priced securities. They may affect investing in our common stock, which may have the effect
of reducing the level of trading activity in our securities. As a result, fewer broker-dealers may be willing to make a market in our
common stock, reducing a stockholder’s ability to resell shares of our common stock.
USE OF PROCEEDS
We estimate that the net
proceeds from this offering will be approximately $
million based on the sale of shares of common stock, Series E-1 warrants to purchase
shares of common stock and Series E-2 warrants to purchase shares of
common stock at a combined public offering price of $ per share
of common stock and Series E warrants, after deducting the placement agent fees and estimated offering expenses payable by us, and
assuming no sale of Series F pre-funded warrants in this offering and no exercise of the Series E warrants being issued in
this offering. However, because this is a best efforts offering and there is no minimum offering amount required as a condition to the
closing of this offering, the actual offering amount, the placement agent’s fees and net proceeds to us are not presently determinable
and may be substantially less than the maximum amounts set forth on the cover page of this prospectus. The table below depicts
how we plan to utilize the proceeds in the event that 25%, 50%, 75%, and 100% of the securities in this offering are sold, after deducting
estimated offering expenses payable by us.
| Use of Proceeds |
|
|
100% |
|
|
|
75% |
|
|
|
50% |
|
|
|
25% |
|
| Working Capital/General Corporate Purposes |
|
$ |
|
|
|
$ |
|
|
|
$ |
|
|
|
$ |
|
|
| Total: |
|
$ |
|
|
|
$ |
|
|
|
$ |
|
|
|
$ |
|
|
These estimates exclude
the proceeds, if any, from the exercise of Series E warrants issued in this offering. If all of the Series E warrants issued
in this offering were to be exercised in cash at an assumed exercise price of $ per
share of common stock, we would receive additional proceeds of approximately $
million. We cannot predict when or if these Series E warrants will be exercised. It is possible that these Series E warrants
may expire and may never be exercised. Additionally, the Series E warrants contain a cashless exercise provision that permit exercise
of Series E warrants on a cashless basis at any time where there is no effective registration statement under the Securities Act
of 1933, as amended, covering the issuance of the underlying shares.
We intend to use the net
proceeds of this offering for working capital and other general corporate purposes, as well as to repay all or a portion of the Credit
Agreement with Perceptive. The interest rate under the Perceptive Credit Agreement is an annual rate equal to the Secured Overnight Financing
Rate, or SOFR, plus 10.25%, provided that SOFR shall not be less than 1.5%. Pursuant to the Perceptive Credit Agreement, beginning on
August 31, 2022, we made monthly principal payments in an amount equal to $75,000. Beginning on December 1, 2023 and pursuant
to the Seventh Amendment, we began making monthly payments of $150,000, which will continue until February 10, 2024, at which time
all remaining principal amount outstanding is due, and the Credit Agreement will mature. We have used borrowings under the Perceptive
Credit Agreement for working capital purposes. See “Risk Factors” beginning on page 10 of this prospectus for more information.
Our operating activities may be restricted as a result of covenants related to the outstanding indebtedness under the Perceptive Credit
Agreement and we may be required to repay the outstanding indebtedness in an event of default, which could have a materially adverse
effect on our business. We may also use a portion of the net proceeds of this offering to invest in or acquire businesses or technologies
that we believe are complementary to our own, although we have no current plans, commitments or agreements with respect to any acquisitions
as of the date of this prospectus supplement. Pending these uses, we plan to invest these net proceeds in investment-grade, interest
bearing securities.
These expected uses represent
our intentions based upon our current plans and business conditions, which could change in the future as our plans and business conditions
evolve. The amounts and timing of our actual expenditures may vary significantly depending on numerous factors, including the progress
of our development as a commercial company, the implementation of our commercial plan for Twirla, the status of and results from future
clinical trials, and any unforeseen cash needs. As a result, our management will have broad discretion in the application of the net proceeds
from this offering, and investors will be relying on the judgment of our management regarding the application of the net proceeds from
this offering. The timing and amount of our actual expenditures will be based on many factors, including cash flows from operations and
the anticipated growth of our business.
CAPITALIZATION
The following table sets forth
our cash and cash equivalents and our capitalization as of September 30, 2023:
| |
· |
On an as adjusted basis to
give further effect to the sale of up to shares
of common stock and accompanying Series E warrants in this offering at an assumed public offering
price of $ per share and accompanying Series E warrants, the last reported sale price of Common
Stock on the Nasdaq Capital Market on , 2024, assuming no sale of any Series F pre-funded warrant
and accompanying warrants, after deducting the estimated placement agent fees and estimate offering
expenses payable by us, and excluding the proceeds, if any, from the exercise of the warrants issued
pursuant to this offering |
You should read this table
together with our financial statements and the related notes incorporated by reference in this prospectus supplement and the sections
entitled “Management’s Discussion and Analysis of Financial Condition and Results of Operations” included in our most
recent Annual Report on Form 10-K and Quarterly Report on Form 10-Q, each as incorporated by reference herein.
| |
|
As of September 30, 2023 |
|
| |
|
Actual |
|
|
As
Adjusted(1) |
|
| |
|
(in thousands,
except share
and per share data)
(unaudited) |
|
| Cash and cash equivalents |
|
$ |
2,873 |
|
|
$ |
|
|
| |
|
|
|
|
|
|
|
|
| Stockholders’ equity: |
|
|
|
|
|
|
|
|
| Preferred
stock, $0.0001 par value; 10,000,000 shares authorized, 4,850 shares issued and no shares outstanding actual and as adjusted. |
|
|
- |
|
|
|
|
|
Common
stock, $0.0001 par value; 300,000,000 shares authorized, 2,277,657 shares issued and outstanding,
actual, shares issued and outstanding,
as adjusted. |
|
|
- |
|
|
|
- |
|
| Additional paid-in capital |
|
|
406,288 |
|
|
|
|
|
| Accumulated other comprehensive income |
|
|
- |
|
|
|
|
|
| Accumulated deficit |
|
|
(418,699 |
) |
|
|
|
|
| Total stockholders’
equity (deficit) |
|
$ |
(12,411 |
) |
|
$ |
|
|
| Total capitalization |
|
$ |
(12,411 |
) |
|
$ |
|
|
| (1) |
The
as adjusted balance sheet data reflects the sale of shares
of common stock and Series E Warrants to purchase shares
of common stock offered by us in this offering at the public offering price of $ per
share and accompanying Series E Warrants, after deducting the underwriting discounts and commissions and estimated offering
expenses payable by us. |
The discussion and table above
assume no exercise of the warrants sold in this offering.
The foregoing table and calculations
are based on 2,277,657 shares of our common stock outstanding as of September 30, 2023, and excludes:
| |
· |
43,510
shares of common stock issuable upon the exercise of outstanding options to purchase common stock as of September 30, 2023 at a weighted
average exercise price of $550.67
per share; |
| |
· |
135,037 shares of common stock issuable upon the vesting of outstanding restricted stock units as of September 30, 2023; |
| |
· |
6,074 shares of common stock reserved for future issuance under our 2023 Equity Incentive Plan as of September 30, 2023; and |
| |
· |
686,000 shares of common stock issuable upon the exercise of outstanding pre-funded warrants as of September 30, 2023; and |
| |
· |
4,484,077 shares of common stock issuable upon the exercise of outstanding warrants as of September 30,
2023 at a weighted average exercise price of $10.10 per share. |
Each $0.25 increase or
decrease in the assumed public offering price per share of common stock and accompanying Series E warrants of $
, the last reported sale price of our common stock on the Nasdaq Capital Market on , 2024, would increase or decrease the net proceeds
to us from this offering by $ million, assuming
that the number of shares of common stock and accompanying Series E warrants offered by us, as set forth on the cover page of
this prospectus, remains the same, assuming no sale of any Series F pre-funded warrants and accompanying Series E warrants,
after deducting the estimated placement agent fees and estimated offering expenses payable by us, and excluding the proceeds, if any,
from the exercise of the warrants issued pursuant to this offering. We may also increase or decrease the number of shares of common stock
and accompanying Series E warrants offered in this offering. Each increase or decrease of 1,000,000 shares of common stock and accompanying
Series E warrants offered by us would increase or decrease the net proceeds to us by approximately $ million,
assuming the assumed public offering price per share of common stock and accompanying Series E warrants of $ remains
the same, assuming no sale of any Series F pre-funded warrants and accompanying Series E warrants, after deducting the estimated
placement agent fees and estimated offering expenses payable by us, and excluding the proceeds, if any, from the exercise of the warrants
issued pursuant to this offering. The as adjusted information discussed above is illustrative only and will be adjusted based on the
actual public offering price and other terms of this offering as determined between us and the placement agent at pricing.
DILUTION
If you invest in our common
stock and warrants in this offering, your ownership interest will be diluted to the extent of the difference between the effective public
offering price per share of common stock included in the share of common stock and accompanying Series E warrants or issuable upon
the exercise of the Series F pre-funded warrants and the as adjusted net tangible book value per share of common stock after this
offering. As of September 30, 2023, our historical net tangible book value was $(12.4) million, or $(5.45) per share, based on 2,277,657
shares of common stock outstanding as of September 30, 2023. Our historical net tangible book value per share represents the amount
of our total tangible assets reduced by the amount of our total liabilities, divided by the total number of shares of common stock outstanding
as of September 30, 2023.
After giving further effect
to our sale in this offering of shares of common stock and accompanying
Series E warrants at an assumed public offering price per share of common stock and accompanying Series E warrants of $ , the
last reported sale price of common stock on the Nasdaq Capital Market on , 2024, assuming no sale of any Series F pre-funded warrants
in this offering, and after deducting the estimated placement agent fees and estimated offering expenses payable by us, and excluding
the proceeds, if any, from the exercise of the warrants issued in this offering, our as adjusted net tangible book value as of September 30,
2023 would have been $ million, or $ per
share. This represents an immediate increase of net tangible book value of $ per
share to our existing stockholders and an immediate dilution of net tangible book value of $
per share to investors purchasing shares of common stock and accompanying Series E warrants in this offering. The following table
illustrates this per share dilution:
| Assumed
public offering price per share of common stock and accompanying Series E warrants |
|
|
|
|
$ |
|
|
| Historical
net tangible book value (deficit) per share as of September 30, 2023 |
$ |
(5.45 |
) |
|
|
|
|
| Increase
in net tangible book value per share attributable to investors purchasing in this offering |
$ |
|
|
|
|
|
|
| As
adjusted net tangible book value per share as of September 30, 2023 after this offering |
|
|
|
|
$ |
|
|
| Dilution
per share to investors purchasing in this offering |
|
|
|
|
$ |
|
|
Each $0.25 increase or
decrease in the assumed public offering price per share of common stock and accompanying Series E warrants of $ , the last reported
sale price of our common stock on the Nasdaq Capital Market on , 2024, would increase or decrease the net proceeds to us from this
offering by $ million, assuming that the number of shares of common
stock and accompanying Series E warrants offered by us, as set forth on the cover page of this prospectus, remains the same,
assuming no sale of any Series F pre-funded warrants and accompanying Series E warrants, after deducting the estimated placement
agent fees and estimated offering expenses payable by us, and excluding the proceeds, if any, from the exercise of the warrants issued
pursuant to this offering. We may also increase or decrease the number of shares of common stock and accompanying Series E warrants
offered in this offering. Each increase or decrease of 1,000,000 share of common stock and accompanying Series E warrants offered
by us would increase or decrease the net proceeds to us by approximately $
million, assuming the assumed public offering price per share of common stock and accompanying Series E warrants of $
remains the same, assuming no sale of any Series F pre-funded warrants and accompanying Series E warrants, after deducting
the estimated placement agent fees and estimated offering expenses payable by us, and excluding the proceeds, if any, from the exercise
of the warrants issued pursuant to this offering. The as adjusted information discussed above is illustrative only and will be adjusted
based on the actual public offering price and other terms of this offering as determined between us and the placement agent at pricing.
The discussion and table above
assume no exercise of the warrants sold in this offering.
The foregoing table and calculations
are based on 2,277,657 shares of our common stock outstanding as of September 30, 2023, and exclude:
| |
· |
43,510 shares of common stock issuable upon the exercise of outstanding options to purchase common stock as of September 30, 2023 at a weighted average exercise price of $550.67 per share; |
| |
· |
135,037 shares of common stock issuable upon the vesting of outstanding restricted stock units as of September 30, 2023; |
| |
· |
6,074 shares of common stock reserved for future issuance under our 2023 Equity Incentive Plan as of September 30, 2023; and |
| |
· |
686,000 shares of common stock issuable upon the exercise of outstanding pre-funded warrants as of September 30, 2023; and |
| |
· |
4,484,077 shares of common stock issuable upon the exercise of outstanding warrants as of September 30, 2023 at a weighted average exercise price of $10.10 per share. |
DIVIDEND POLICY
We have never declared or
paid a cash dividend on our capital stock. We currently intend to retain all available funds and any future earnings for use in the operation
of our business and do not anticipate paying any dividends on our common stock in the foreseeable future. In addition, our Credit Agreement
and Guaranty, as amended, among us, the guarantors from time to time party thereto, the lenders from time to time party thereto and Perceptive
Credit Holdings III, LP, as a lender and as Administrative Agent for the lenders, contains, and any other loan facilities that we may
enter into may contain, restrictions on our ability to pay dividends. Subject to such restrictions, any future determination to declare
dividends will be made at the discretion of our board of directors and will depend on our financial condition, operating results, capital
requirements, general business conditions and other factors that our board of directors may deem relevant.
CORPORATE GOVERNANCE
Corporate Governance Materials and Practices
Our board of directors (the
“Board”) structure reflects our Corporate Governance Principles and commitment to good governance. The Board believes the
governance structure we have created among the Board, its committees, the Lead Independent Director, the Board chairperson and Chief
Executive Officer, and management is supporting the growth of the Company.
Our written corporate governance
materials, including our Bylaws, Corporate Governance Guidelines, Code of Business Conduct and Ethics, Audit Committee Charter, Compensation
Committee Charter, Nominating and Corporate Governance Committee Charter, and Science and Technology Committee Charter are posted on
our website at www.agiletherapeutics.com under the heading “Investor Relations—Corporate Governance.” Our corporate
governance practices include the following:
| |
● |
Our Board has a Lead Independent Director, and all
of our non-employee directors and committee members are independent. |
| |
● |
Our Board oversees succession planning for executive
officers, including the Chief Executive Officer. |
| |
● |
Directors have access to all levels of management
and are provided with opportunities to meet with members of management on a regular basis. |
| |
● |
Directors may retain their own independent advisors
at our expense. |
| |
● |
Our Board and each committee thereof conduct self-evaluations
at least once per year to assess their performance and ways in which performance could be improved. |
| |
● |
Our Board addresses the importance of incorporating
new viewpoints through the director evaluation and nomination process. Our director composition reflects a mix of tenure on our Board
(ranging from 1 year to 17 years), which we believe provides an effective balance of historical perspective and an understanding
of the evolution of the Company with fresh perspectives and insights. |
Board Diversity Matrix
The tables below provide certain highlights of
the composition of our Board as of December 31, 2023. Each of the categories listed in the tables below has the meaning as it is used
in Nasdaq Rule 5605(f).
| Board
Diversity Matrix |
| Total
Number of Directors |
6 |
| |
Female |
Male |
Non-Binary |
Did Not Disclose
Gender |
| Part
I: Gender Identity |
| Directors |
3 |
3 |
0 |
0 |
| Part
II: Demographic Background |
| African
American or Black |
0 |
0 |
0 |
0 |
| Alaskan
Native or Native American |
0 |
0 |
0 |
0 |
| Asian |
0 |
0 |
0 |
0 |
| Hispanic
or Latinx |
0 |
0 |
0 |
0 |
| Native
Hawaiian or Pacific Islander |
0 |
0 |
0 |
0 |
| White |
3 |
2 |
0 |
0 |
| Two
or More Races or Ethnicities |
0 |
0 |
0 |
0 |
| LGBTQ+ |
0 |
| Did
Not Disclose Demographic Background |
1 |
Environmental, Social and Governance Considerations
We are a women’s healthcare
company dedicated to fulfilling the unmet needs of today’s women, and we take our responsibility to patients, employees, the medical
community, and the communities in which we live and work very seriously. We are an emerging company that is focused on growing as a commercial
organization. We recognize that as we grow, we have an opportunity to positively influence a variety of environmental, social and governance
(“ESG”) considerations and partner with companies that reflect our values as a company and our commitment to our larger community.
Our Board helps to shape and monitor management’s approach to ESG. We plan to seek ways to incorporate these ESG considerations
into our business plans with a strategic focus on the following areas:
Diversity, Equity and Inclusion:
We are focused on promoting diversity in our workforce and to taking steps to support equity and inclusion for all. We believe that these
values provide us with a competitive advantage that can drive creative and innovative thinking and ultimately enhance our business. We
have a diverse Board and leadership team supporting our commercial operations. Overall, of the last seven (7) openings on our Board
and management team, six (6) have been filled with women candidates. We continue to strive to enhance our Board and workforce diversity
by ensuring we have a gender-balanced and diverse slate of qualified candidates for all open positions, advancing the development of
diverse talent, and pursuing diverse succession plans both in our employee workforce and our Board.
Patient
Support: Advancing women’s health is at the core of our mission. Our current product and product candidates are designed
to offer women more freedom and flexibility through additional contraceptive options. The key phase 3 clinical trial we conducted to
support the approval of Twirla, our transdermal contraceptive product, included a broad and diverse population of women representative
of the U.S. female population seeking hormonal contraception. The trial included women across a broad range of demographic characteristics
such as age, race, ethnicity and weight and was one of the first trials to generate data on the relationship between body mass index,
or BMI, and the safety and efficacy of a hormonal contraceptive. Our goal is to make our products as accessible as possible in all communities,
and we are working with numerous stakeholders to enable this access as appropriate. We seek to support this objective with a comprehensive
series of programs that include educational, product replacement and co-pay support (implemented in a compliant way). We also have engaged
more broadly with women’s health advocacy groups and other advisory groups to understand the needs of the patients we seek to help.
Employee Wellness: We believe
that our employees are a key to our success. We are dedicated to investing in our employees and workplace culture. As part of this effort,
we have put in place several financial wellness programs to help empower our workforce including offering a 40l(k) match program,
short-term and long-term disability insurance, and educational programs to help employees understand how to manage their finances. We
also offer several programs to promote the physical and mental wellbeing of our employees, including an employee assistance program,
disease management and prevention programs and financial incentives to encourage physical and emotional health.
Environmental Impact: We are
cognizant of our responsibility to our broader environment and our need to grow in a sustainable way. Prior to the pandemic and working
remotely, we supported several green measures in an effort to reduce our Company's carbon footprint in our corporate office. We continue
to evaluate ways to enhance these and other sustainability measures as we make plans to build out our business.
Governance: We have adopted
a Code of Business Conduct and Ethics (the “Code”) which applies to all of our employees, officers and directors and outlines
our commitment to ethical and compliant business practices. We promote compliance with all relevant government laws, rules and regulations;
honest and ethical conduct; protection of company assets; and fair dealings and timely and accurate disclosures in all company reporting.
All employees are required to understand, comply with and report any suspected violations of the Code, and the Code is reviewed periodically
to ensure it is appropriate in scope and achieves the objectives above.
Community Service: We are committed
to giving back to our communities. We partake in several employee-led community service initiatives. Through our internal community outreach
program – Agile Cares – and we host from time to time team-building events to support causes that are meaningful to our employees
and our overall mission.
Code of Business Conduct and Ethics
Our Board has adopted a code
of business conduct and ethics. The code of business conduct and ethics (“CBCE”) applies to all of our employees, officers
and directors. The CBCE provides guidance on fundamental ethical and compliance-related principles and practices such as: accurate record
keeping and reporting; avoiding conflicts of interest; protection and proper use of property and information; and compliance with legal
and regulatory requirements. The full text of our code of business conduct and ethics is posted on our website at www.agiletherapeutics.com.
We intend to disclose, to the extent required by applicable rules and regulations, future amendments to, or waivers of, our code
of business conduct and ethics, at the same location on our website identified above or in public filings we will make with the SEC.
Information contained on our website is not incorporated by reference, and you should not consider information contained on our website
to be part of this prospectus.
Hedging and Pledging Policies
We have adopted an insider
trading policy that includes a provision that restricts our directors, officers and employees from engaging in hedging or monetization
transactions involving our securities, and from engaging in short sales of our securities. Our insider trading policy also prohibits
our directors, officers and employees from holding our securities in margin accounts or otherwise pledging our securities as collateral
for loans without prior written approval.
Board Composition
Our business affairs are
managed under the direction of our Board, which is currently composed of six (6) members. Five (5) of our six (6) directors are independent
within the meaning of the listing rules of the Nasdaq Capital Market, or Nasdaq. Our Board is divided into three classes with staggered
three-year terms. At each annual meeting of stockholders, the successors to directors whose terms then expire will be elected to
serve from the time of election and qualification until the third annual meeting following election. As a result, only one class of directors
will be elected at each annual meeting of our stockholders, with the other classes continuing for the remainder of their respective three-year
terms. Each director’s term continues until the election and qualification of his or her successor, or the earlier of his or her
death, resignation or removal. The classification of our Board may have the effect of delaying or preventing changes in our control or
management.
Board Leadership Structure
Al Altomari is the Chairperson
of our Board, and Seth H.Z. Fischer is our Lead Independent Director. Our Lead Independent Director chairs the executive sessions of
our Board meetings, oversees the Board’s annual self-evaluation process, provides feedback to the Chief Executive Officer, and
works with the Chief Executive Officer to set agendas for Board meetings. We have a separate chairperson for each committee of our Board,
all of whom are independent directors. The chairpersons of each committee report on the activities of their committees in fulfilling
their responsibilities at the meetings of our Board.
Our Board has concluded that
our current leadership structure is appropriate at this time. However, our Board will continue to periodically review our leadership
structure and may make such changes in the future as it deems appropriate.
Board Committees
Our Board has established
an audit committee, a compensation committee, a nominating and corporate governance committee, and a science and technology committee,
each of which operates pursuant to a charter adopted by our Board. The composition and functioning of all of our committees complies
with all applicable requirements of the Sarbanes-Oxley Act of 2002, Nasdaq and SEC rules and regulations.
Audit committee. Ms. Barbari,
Mr. Fischer and Dr. Hubbard currently serve on the audit committee, which is chaired by Ms. Barbari. Our Board has determined
that each member of the audit committee is “independent” for audit committee purposes as that term is defined in the rules of
the SEC and the applicable Nasdaq rules, and has sufficient knowledge in financial and auditing matters to serve on the audit committee.
Our Board has determined that Ms. Barbari qualifies as an “audit committee financial expert,” as defined under the applicable
rules of the SEC. The audit committee operates under a written charter that satisfies the applicable standards of the SEC and Nasdaq
and which is available on our website at www.agiletherapeutics.com. The inclusion of our website address here and elsewhere in this prospectus
does not include or incorporate by reference the information on our website into this prospectus. Our audit committee met eight (8) times
during the year ended December 31, 2023. The audit committee’s responsibilities include:
| |
● |
appointing, approving the compensation of, and assessing the independence
of our independent registered public accounting firm on an annual basis; |
| |
● |
pre-approving auditing and permissible non-audit services, and the
terms of such services, to be provided by our independent registered public accounting firm; |
| |
● |
reviewing the overall audit plan with the independent registered public
accounting firm and members of management responsible for preparing our financial statements; |
| |
● |
reviewing and discussing with management and the independent registered
public accounting firm our annual and quarterly financial statements and related disclosures as well as critical accounting policies
and practices used by us; |
| |
● |
coordinating the oversight and reviewing the adequacy of our internal
control over financial reporting; |
| |
● |
establishing policies and procedures for the receipt and retention
of accounting-related complaints and concerns; |
| |
● |
recommending based upon the audit committee’s review and discussions
with management and the independent registered public accounting firm whether our audited financial statements shall be included
in our Annual Report on Form 10-K; |
| |
● |
monitoring the integrity of our financial statements and our compliance
with legal and regulatory requirements as they relate to our financial statements and accounting matters; |
| |
● |
preparing the audit committee report required by SEC rules to
be included in our annual proxy statement; |
| |
● |
reviewing all related person transactions for potential conflict of
interest situations and approving all such transactions; |
| |
● |
reviewing the Company’s risk assessment and risk management policies
and procedures, and discussing with management the Company’s significant financial risk exposures and the actions management
has taken to limit, monitor or control such exposures, including, but not limited to, reviewing, on an on-going basis, cybersecurity;
and |
| |
● |
reviewing quarterly earnings releases. |
Compensation committee.
Mr. Fischer, Dr. Carson and Sharon Barbari currently serve on the compensation committee, which is chaired by Mr. Fischer.
Dr. Carson replaced Dr. Shetty on the committee when Dr. Shetty did not stand for re-election to the Board at the Company’s annual
meeting of stockholders on June 8, 2023. Our Board has determined that each member of the compensation committee is “independent”
as defined in applicable Nasdaq rules. The compensation committee operates under a written charter that satisfies the applicable standards
of Nasdaq and which is available on our website at www.agiletherapeutics.com. The inclusion of our website address here and elsewhere
in this prospectus does not include or incorporate by reference the information on our website into this prospectus. Our compensation
committee met six (6) times during the year ended December 31, 2023. The compensation committee’s responsibilities
include:
| |
● |
annually reviewing and making recommendations to the Board with respect
to corporate goals and objectives relevant to the compensation of our Chief Executive Officer; |
| |
● |
evaluating the performance of our chief executive officer in light
of such corporate goals and objectives and making recommendations to the Board with respect to the compensation of our Chief Executive
Officer; |
| |
● |
reviewing and approving the compensation of our other executive officers; |
| |
● |
reviewing and establishing our overall management compensation philosophy
and policy; |
| |
● |
overseeing and administering our compensation and similar plans; |
| |
● |
reviewing and approving our policies and procedures for the grant of
equity-based awards; |
| |
● |
reviewing and making recommendations to the Board with respect to director
compensation; |
| |
● |
reviewing and discussing with management the compensation discussion
and analysis, if any, to be included in our annual proxy statement or Annual Report on Form 10-K; and |
| |
● |
reviewing and discussing with the Board corporate succession plans
for the Chief Executive Officer and other key officers. |
Certain of our executive
officers may provide information and assist our compensation committee in carrying out its functions, however, the Committee considers
potential compensation actions and makes decisions independently. Our compensation committee has engaged the services of Pay Governance LLC,
a compensation consulting firm, to advise the compensation committee regarding the amount and types of compensation that we provide to
our executives and directors and how our compensation practices compare to the compensation practices of other companies. Pay Governance LLC
reports directly to the compensation committee. Pay Governance LLC does not provide any services to us other than the services provided
to the compensation committee. The compensation committee believes that Pay Governance LLC does not have any conflicts of interest
in advising the compensation committee under applicable SEC rules or Nasdaq listing standards.
Nominating and corporate
governance committee. Dr. Hubbard, Ms. Barbari, Dr. Carson, and Ms. Torrente currently serve on the nominating
and corporate governance committee, which is chaired by Dr. Hubbard. Our Board has determined that each member of the nominating
and corporate governance committee is “independent” as defined in applicable Nasdaq rules. The nominating and corporate governance
committee operates under a written charter that satisfies the applicable standards of Nasdaq and which is available on our website at
www.agiletherapeutics.com. The inclusion of our website address here and elsewhere in this prospectus does not include or incorporate
by reference the information on our website into this prospectus Our nominating and corporate governance committee met three (3) times
during the year ended December 31, 2023. The nominating and corporate governance committee’s responsibilities include:
| |
● |
developing and recommending to the Board criteria for board and committee
membership; |
| |
● |
establishing procedures for identifying and evaluating board of director
candidates, including nominees recommended by stockholders; |
| |
● |
reviewing the size and composition of the Board to
ensure that it is composed of members containing the appropriate skills and expertise to advise us; |
| |
● |
identifying individuals qualified to become members
of the Board; |
| |
● |
recommending to the Board the persons to be nominated
for election as directors and to each of the Board’s committees; |
| |
● |
developing and recommending to the Board a code of
business conduct and ethics and a set of corporate governance guidelines; |
| |
● |
developing a mechanism by which violations of the
code of business conduct and ethics can be reported in a confidential manner; |
| |
● |
overseeing the evaluation of the Board and management;
and |
| |
● |
reviewing and reporting to the Board concerning our
corporate social responsibility and sustainability efforts, including the impact of environmental and social issues on our company. |
Science and Technology
Committee. Ms. Torrente, Dr. Carson, and Dr. Hubbard currently serve on the science and technology committee, which
is chaired by Ms. Torrente. Dr. Hubbard replaced Dr. Shetty on the committee when Dr. Shetty did not stand for re-election to the
Board at the Company’s annual meeting of stockholders on June 8, 2023. The science and technology committee operates under a written
charter, which is available on our website at www.agiletherapeutics.com. The inclusion of our website address here and elsewhere in this
prospectus does not include or incorporate by reference the information on our website into this prospectus. Our science and technology
committee met two (2) times during the year ended December 31, 2023. The science and technology committee’s responsibilities
include:
| |
● |
identifying and discussing new and emerging trends
in pharmaceutical science, technology, pharmaceutical regulation and manufacturing, and, as necessary, reporting to the Board on
such trends and the committee’s deliberations; |
| |
● |
reviewing, evaluating and advising the Board regarding
the quality, direction and competitiveness of our research and development and manufacturing programs; |
| |
● |
reviewing, evaluating and advising the Board regarding
our progress in achieving our long-term strategic research and development goals and objectives; reviewing and making recommendations
to the Board on our internal and external investments in science, technology and manufacturing. For any external investments in research
and development (e.g., potential acquisitions, alliances, collaborations, equity investments, contracts and grants) that require
approval by the Board, the committee provides the Board with its recommendation prior to any action by the Board unless time does
not permit; |
| |
● |
regularly reviewing our pipeline of product candidates
and clinical development performance; |
| |
● |
providing assistance to the compensation committee
in setting any pipeline or development performance metric(s) under our incentive compensation programs and reviewing the performance
results; |
| |
● |
evaluating its own performance annually and delivering
a report to the Board setting forth the results of the evaluation; |
| |
● |
reviewing and reassessing the adequacy of its charter
annually and recommending any proposed changes to the Board for its approval; and |
| |
● |
performing any other activities consistent with its
charter, our amended and restated bylaws and governing law or regulation, as the committee or the Board deems necessary or appropriate. |
In December 2020, the
board established an ad hoc finance committee to act on its behalf on matters relating to potential transactions to raise capital and
to review potential business development transactions. Ms. Barbari, Mr. Fischer and Dr. Hubbard serve on the committee,
which is chaired by Ms. Barbari. The committee met one (1) time during the year ended December 31, 2023. Our Board may
from time to time establish other committees.
Meetings of the Board of Directors
The full Board met twelve
(12) times during the year ended December 31, 2023. No director attended fewer than 75% of the total number of meetings
of the Board and of any committees of the Board of which he or she was a member during our year ended December 31, 2023.
It is our policy that directors
are invited and encouraged to attend our annual meetings of stockholders. We have scheduled our Annual Meeting on the same day as a regularly
scheduled Board meeting in order to facilitate attendance by the members of our Board. All of our directors at the time of our 2023 Annual
Meeting of Stockholders attended the 2023 Annual Meeting of Stockholders.
Board Oversight of Risk
Risk is inherent with every
business, and how well a business manages risk can ultimately determine its success. We face a number of risks, including risks relating
to our operations, strategic direction and intellectual property as more fully discussed under “Risk Factors” in our Annual
Report on Form 10-K. Management is responsible for the day-to-day management of risks we face, while our Board, as a whole and through
its committees, has responsibility for the oversight of risk management. In its risk oversight role, our Board has the responsibility
to satisfy itself that the risk management processes designed and implemented by management are adequate and functioning as designed.
The role of the Board in
overseeing the management of our risks is conducted primarily through committees of the Board, as disclosed in the descriptions of each
of the committees above and in the charters of each of the committees. For example, our audit committee is responsible for overseeing
the management of risks associated with our financial reporting, accounting and auditing matters, regulatory and legal compliance, and
cyber-security; our compensation committee oversees major risks associated with our compensation policies and programs; and our nominating
and governance committee oversees the management of risks associated with director independence, conflicts of interest, composition and
organization of our Board and director succession planning. The full Board (or the appropriate board committee in the case of risks that
are under the purview of a particular committee) discusses with management our major risk exposures, their potential impact on Agile,
and the steps we take to manage them. When a Board committee is responsible for evaluating and overseeing the management of a particular
risk or risks, the chairperson of the relevant committee reports on the discussion to the full Board during the committee reports portion
of the next Board meeting. This enables the Board and its committees to coordinate the risk oversight role, particularly with respect
to risk interrelationships.
Director Nomination Process
In considering whether to
recommend any candidate for inclusion in our Board’s slate of recommended directors, including candidates recommended by stockholders,
the nominating and corporate governance committee applies a certain set of criteria, including, but not limited to, the candidate’s
integrity, business acumen, experience, commitment, diligence, conflicts of interest and his or her ability to act in the interests of
all stockholders. We believe that the value of diversity on the board should be considered by the nominating and corporate governance
committee in the director identification and nomination process. The committee seeks nominees with a broad diversity of experience, professions,
skills, geographic representation and backgrounds. The committee does not assign specific weights to particular criteria and no particular
criterion is necessarily applicable to all prospective nominees. We believe that the backgrounds and qualifications of the directors,
considered as a group, should provide a significant breadth of experience, knowledge and abilities that will allow the Board to fulfill
its responsibilities. Nominees are not discriminated against on the basis of race, religion, national origin, sex, sexual orientation,
gender identity or expression, disability or any other basis proscribed by law or Company policy.
Stockholders may also nominate
persons to be elected as directors. Our nominating and corporate governance committee will consider director candidates recommended by
our stockholders, in accordance with our bylaws, as amended. If a stockholder wishes to nominate a person for election as director, he
or she must follow the procedures contained in our bylaws, as amended. In evaluating candidates recommended by our stockholders, our
nominating and corporate governance committee applies the same criteria as discussed above. To nominate a person to stand for election
as a director, a stockholder must provide our Secretary with timely notice of the nomination and the notice must include the information
required by Section 2.4 of our bylaws, as amended.
Limitation of Liability and Indemnification
Arrangements
As permitted by the Delaware
General Corporation Law (the “DGCL”), we have adopted provisions in our amended and restated certificate of incorporation
and amended and restated by-laws that limit or eliminate the personal liability of our directors. Consequently, a director will not be
personally liable to us or our stockholders for monetary damages for breach of fiduciary duty as a director, except for liability for:
| |
● |
any breach of the director’s duty of loyalty
to us or our stockholders; |
| |
● |
any act or omission not in good faith or that involves
intentional misconduct or a knowing violation of law; |
| |
● |
any unlawful payments related to dividends or unlawful
stock repurchases, redemptions or other distributions; or |
| |
● |
any transaction from which the director derived an
improper personal benefit. |
These limitations of liability
do not alter director liability under the federal securities laws and do not affect the availability of equitable remedies such as an
injunction or rescission.
In addition, our amended
and restated by-laws provide that we will:
| |
● |
indemnify our directors, officers and, in the discretion
of our Board, certain employees to the fullest extent permitted by the DGCL; and |
| |
● |
advance expenses, including attorneys’ fees,
to our directors and, in the discretion of our Board, to our officers and certain employees, in connection with legal proceedings,
subject to limited exceptions. |
We have entered into separate
indemnification agreements with our directors and executive officers. These agreements, among other things, provide for indemnification
of our directors and executive officers for certain expenses, judgments, fines and settlement amounts, among others, incurred by such
persons in any action or proceeding arising out of such person’s services as a director or executive officer in any capacity with
respect to any employee benefit plan or as a director, partner, trustee or agent of another entity at our request. We believe that these
indemnification agreements, along with the above-referenced provisions of our amended and restated certificate of incorporation and amended
and restated bylaws, are necessary to attract and retain qualified persons as directors and executive officers.
We also maintain general
liability insurance to provide insurance coverage to our directors and officers for losses arising out of claims based on acts or omissions
in their capacities as directors or officers, including liabilities under the Securities Act of 1933, as amended, or the Securities Act.
Insofar as indemnification for liabilities arising under the Securities Act may be permitted to directors, officers, or persons controlling
the registrant pursuant to the foregoing provisions, we have been informed that in the opinion of the Securities and Exchange Commission
such indemnification is against public policy as expressed in the Securities Act and is therefore unenforceable.
These provisions may discourage
stockholders from bringing a lawsuit against our directors and officers in the future for any breach of their fiduciary duties. These
provisions may also have the effect of reducing the likelihood of derivative litigation against directors and officers, even though such
an action, if successful, might otherwise benefit us and our stockholders. Furthermore, a stockholder’s investment may be adversely
affected to the extent we pay the costs of settlement and damage awards against directors, officers and certain employees pursuant to
these indemnification provisions. We believe that these provisions, the indemnification agreements and the insurance are necessary to
attract and retain talented and experienced directors and officers.
Stockholder Communications with the Board
of Directors
Stockholders wishing to communicate
with the Board or with an individual member of the Board may do so by writing to the Board or to the particular member of the Board,
care of the Corporate Secretary by mail to our principal executive offices, Attention: Corporate Secretary. The envelope should indicate
that it contains a stockholder communication. All such stockholder communications will be forwarded to the director or directors to whom
the communications are addressed.
EXECUTIVE OFFICERS AND DIRECTORS
The following table provides information concerning
our executive officers and directors as of January 9, 2024:
| Name |
|
Age |
|
Position |
| Alfred (Al) Altomari |
|
64 |
|
Chairperson, Chief
Executive Officer and Director |
| Scott M. Coiante |
|
57 |
|
Senior Vice President,
Chief Financial Officer |
| Robert G. Conway |
|
67 |
|
Senior Vice President, Chief Corporate Planning &
Supply Chain Officer |
| Geoffrey P. Gilmore |
|
57 |
|
Senior Vice President, Chief Administrative Officer |
| Paul Korner |
|
58 |
|
Senior Vice President, Chief Medical Officer |
| Amy Welsh |
|
52 |
|
Senior Vice President,
Chief Commercial Officer |
| Sharon Barbari |
|
69 |
|
Director |
| Sandra Carson, M.D., FACOG |
|
70 |
|
Director |
| Seth H.Z. Fischer |
|
67 |
|
Director |
| John Hubbard, Ph.D., FCP |
|
67 |
|
Director |
| Josephine Torrente |
|
57 |
|
Director |
Al Altomari. Alfred
Altomari has served as our Chairperson of the Board and Chief Executive Officer since October 2016 and has been a member of our
Board since February 2004. Mr. Altomari served as President and Chief Executive Officer from 2010 to 2016. Previously Mr. Altomari
served as Agile’s Executive Chairperson from 2004 to 2010. From 2003 to 2008, Mr. Altomari held multiple senior management
positions at Barrier Therapeutics, Inc., including Chief Commercial Officer, Chief Operating Officer, and Chief Executive Officer.
In 2008, in his role as Chief Executive Officer and as a member of Barrier’s Board of Directors, Mr. Altomari completed the
successful sale of Barrier to Stiefel Laboratories, which was subsequently acquired by GlaxoSmithKline plc. From 1982 to 2003, Mr. Altomari
held numerous executive roles in general management, commercial operations, business development, product launch preparation, and finance
with Johnson & Johnson. Mr. Altomari also serves on the Board of Directors of Insmed Inc. He previously served on the Board
of Directors of Recro Pharm, Inc. and Baudax Bio. Mr. Altomari received an M.B.A. from Rider University and his B.S. from Drexel
University. He currently serves as a Trustee of Drexel University and is on the LeBow College of Business Advisory Board and the Board
of the Charles D. Close School of Entrepreneurship at Drexel University. We believe Mr. Altomari’s qualifications to sit on
our Board of Directors include his extensive pharmaceutical industry leadership and operations experience.
Scott
M. Coiante. Mr. Coiante has been our Chief Financial Officer since August of 2023. He joined the Company from Aprea
Therapeutics, Inc. where he served as the Chief Financial Officer from August 2019 until January 2023 and remained as an employee through
March 31, 2023. Prior to joining Aprea Therapeutics, Inc., Mr. Coiante served as our Senior Vice President and Chief Financial Officer
from 2010 to August 2019. From 2002 to 2010, Mr. Coiante was Vice President of Finance and Treasurer at Medarex, Inc., formerly a Nasdaq
listed biotech company that was acquired in 2009 by Bristol Myers Squibb. From 1989 to 2002, Mr. Coiante held management positions of
increasing responsibility at Ernst & Young LLP. Mr. Coiante received a B.S. in Accounting from Villanova University.
Robert
G. Conway. Mr. Conway has served as our Senior Vice President and Chief Corporate Planning and Supply Chain Officer since
November 2022. Prior to that, he served as our Senior Vice President and Chief Supply Chain Officer since January 2020, as
our Senior Vice President, Enterprise Planning and Information Management from 2017 to 2020, as our Senior Vice President of Operations
from 2014 to 2017 and as our Chief Development Officer and Vice President of Operations from 2004 to 2014. Mr. Conway has over thirty years
of practice in U.S. and international operations with an extensive background in the medical device, pharmaceutical and consumer products
industries. Mr. Conway has also previously served as Principal of R. G. Conway and Associates, an independent engineering and project
management-consulting firm. Prior to consulting in 1997, Mr. Conway began his career in healthcare with Johnson & Johnson
and later joined a Johnson & Johnson supported venture-backed medical device company operating as President and Chief Operating
Officer. Mr. Conway holds a B.S. degree in Mechanical Engineering from New Jersey Institute of Technology.
Geoffrey
P. Gilmore. Mr. Gilmore has been our Senior Vice President and Chief Administrative Officer since November 2022.
Prior to that, Mr. Gilmore served as our General Counsel since August 2014. From 2012 to April 2016, Mr. Gilmore
was a principal of Life Sciences Advisory Services, providing consulting services to the pharmaceutical and life science industries.
He served as Senior Vice President, General Counsel and Corporate Secretary of Amicus Therapeutics, Inc., a public bio-pharmaceutical
company, from 2008 to 2012. Prior to joining Amicus, Mr. Gilmore spent 10 years at Bristol-Myers Squibb where he held roles
of increasing responsibility in the legal department with the Commercial, Intellectual Property, and R&D legal groups, served
in the Office of the Corporate Secretary and then reported to the General Counsel as Vice-President and Senior Counsel, Corporate Securities.
Mr. Gilmore began his legal career in the business and finance groups at Philadelphia based law firms, Ballard Spahr LLP and
Montgomery McCracken Walker & Rhoads LLP. Mr. Gilmore received his B.A. degree from Franklin and Marshall College
and his J.D. from the University of Michigan Law School.
Paul Korner. Dr. Korner
has been our Chief Medical Officer since August 2020. He has served in various leadership positions of increasing responsibility
at several global companies focused on female reproductive health, including Solvay Pharmaceuticals, Wyeth Research, Bayer, and Ferring
Pharmaceuticals. Dr. Korner has led more than 50 clinical trials that include multiple engagements with the U.S. Food and Drug Administration,
European Medicines Agency, Pharmaceuticals and Medical Devices Agency in Japan, and Health Canada. Most recently Dr. Korner brought
his clinical and development acumen to the gene therapy space as a Senior Vice President of Clinical Development & Medical Affairs
at Axovant Gene Therapies, Ltd. He currently serves on the Laidlaw Venture Partners Scientific Advisory Board and the Board of Directors
of Voltron Therapeutics as an Independent Director. Dr. Korner received his MD from the Stritch School of Medicine at Loyola University
and also holds an MBA from the Michael J. Coles College of Business at Kennesaw State University.
Amy Welsh. Amy Welsh
has been our Senior Vice President and Chief Commercial Officer since November 2022. Prior to that Ms. Welsh served as our
Vice President, Marketing since May 2020. She brings over 25 years of industry marketing experience to Agile with a proven track
record in product launch and lifecycle management. Prior to joining Agile, Ms. Welsh served for 3 years as the Vice President of
Marketing at Antares Pharma Inc., a public specialty pharmaceutical company, where she oversaw the launch of its flagship brand XYOSTED,
as well as the commercialization of products, strategic planning of developmental assets, as well as new M&A opportunities. Before
Antares, she spent over eight years at AstraZeneca in brand management and marketing roles of increasing responsibility. Ms. Welsh
also brings perspective and experience from her time working at a digital advertising agency dedicated to serving the life sciences industry.
Ms. Welsh earned a dual B.A. degree in Marketing Communications and English Literature from LaSalle University.
Sharon Barbari. Ms. Barbari
has served as a member of our Board since June 2020. Ms. Barbari has over 40 years of pharmaceutical and biotechnology experience.
From 2004 to 2017, she was Chief Financial Officer at Cytokinetics. From 2002 to 2004, she was employed as Chief Financial Officer and
Senior Vice President of Finance and Administration at InterMune. Ms. Barbari spent four years from 1998 to 2002 in senior financial
roles at Gilead Sciences, including Chief Financial Officer. In this capacity, she led the finance, accounting, and information technology
functions during a period of significant growth for Gilead. Ms. Barbari was also employed as Vice President of Strategic Planning
at Foote, Cone & Belding Healthcare. She began her career at Syntex Corporation/Roche Pharmaceuticals, where she held various
roles of increasing responsibility from 1972 to 1996. Ms. Barbari currently serves as a board member for Vyne Therapeutics (formerly
Menlo Pharmaceuticals and Foamix Pharmaceuticals). She previously was a board member for the Association of Bioscience Finance Officers
Northern California Chapter and Phytogen Life Sciences. In 2017, Ms. Barbari was a recipient of the YWCA Silicon Valley Tribute
to Women Awards. She received her B.S. in accounting from San Jose State University. We believe Ms. Barbari’s qualifications
to sit on our Board include her extensive pharmaceutical industry leadership and financial operations experience.
Sandra Carson, M.D., FACOG.
Dr. Carson has served as a member of our Board since June 2020. Dr. Carson is a Professor of Obstetrics, Gynecology
and Reproductive Sciences and Director, Reproductive Endocrinology and Infertility at Yale University. Most recently, she served as the
Emeritus Vice President for Education at the American College of Obstetricians & Gynecologists (ACOG) from August 2018
to February 2019 and Vice President for Education from March 2013 to August 2018. Prior to joining ACOG she served as
Professor and Director of the Division of Reproductive Endocrinology and Infertility at Brown University from January 2007 to February 2013.
She had previously directed the Assisted Reproductive Technology programs at the University of Tennessee (1986-1998) and the Baylor College
of Medicine from October 1998 to June 2007. Dr. Carson is one of the foremost leaders in the field of reproductive endocrinology
and infertility and she has published over 150 papers, received numerous competitive grants and had a major impact on the field. She
has held multiple national leadership positions including President of the American Society for Reproductive Medicine (ASRM), Member
of the council of the National Institutes of Child Health and Human Development, Head of the Reproductive Endocrinology and Infertility
Division of the American Board of Obstetrics and Gynecology (ABOG), Director and Vice President of the ABOG board and Vice President
of ACOG. Dr. Carson served on the U. S. Food and Drug Administration’s Fertility and Maternal Health Drugs Advisory Committee
from July 1992 to July 1996 and then from 2007 to 2009. Dr. Carson is a board-certified OB/GYN. She received her Doctor
of Medicine degree and completed her residency fellowship training at Northwestern University Medical School and fellowship training
at Michael Reese Hospital Medical Center, University of Chicago. We believe Dr. Carson’s qualifications to sit on our Board
include her extensive medical and clinical experience in the area of Obstetrics, Gynecology and Reproductive Sciences and her significant
educational background.
Seth
H.Z. Fischer. Mr. Fischer has served as a member of our Board since July 2016, and as our Lead Independent Director
since June 2020. Mr. Fischer currently serves as a member of the Board of Directors of Marinus Pharmaceuticals, Inc.,
Spectrum Pharmaceuticals, Inc., and Esperion Therapeutics, Inc. and is also an advisor to MedHab, LLC. Previously, from 2013
to 2017, Mr. Fischer served as the Chief Executive Officer and as a Director of Vivus, Inc., a publicly traded biopharmaceutical
company commercializing and developing innovative, next-generation therapies to address unmet needs. He has served in positions of increasing
responsibility with Johnson & Johnson from 1983 until his retirement in 2012. Most recently, Mr. Fischer served as Company
Group Chairman, Johnson & Johnson and Worldwide Franchise Chairman, Cordis Corporation from 2008 to 2012, and as Company Group
Chairman, North America Pharmaceuticals from 2004 to 2007, which included responsibilities for Ortho-McNeil Pharmaceuticals, Janssen
and Scios. Prior to that, Mr. Fischer served as President of Ortho-McNeil Pharmaceuticals from 2000 to 2004. His operating responsibilities
encompassed the commercialization of products in multiple therapeutic categories including Topamax® for epilepsy and migraine and
products in the analgesic, anti-infective, cardiovascular, neurologic, psychiatric and women's health areas, including ORTHO EVRA®,
one of the most successful contraceptive launches in the U.S. and the first ever contraceptive patch. He earned a Bachelor of General
Studies from Ohio University and served as a captain in the U.S. Air Force. We believe Mr. Fischer’s qualifications to sit
on our Board include his extensive pharmaceutical industry leadership and operations experience.
John Hubbard, Ph.D., FCP.
Dr. Hubbard has served as a member of our Board since November 2014. Dr. Hubbard currently serves on the Strategic Advisory
Board of Genstar Capital and is responsible for advising the private equity firm on investments in the area of healthcare. He is the
Chairman of Precision Medicine Group, a privately owned clinical research organization, and of Signant Health, a privately owned specialty
clinical trials and technology service provider. He also serves as a non-Executive Director on the Board of Directors of Science 37 Holdings, Inc.,
a public telemedicine and clinical trial technology provider and Advarra, a privately owned institutional review board. Dr. Hubbard
was President and the Chief Executive Officer of BioClinica, a leading privately owned provider of medical imaging, clinical technology
and research services until January 1, 2018. Prior to joining BioClinica, he was Senior Vice President and Worldwide Head of Development
Operations for Pfizer Inc. and was responsible for the global clinical trial operations and management of more than 450 clinical projects
from Phase I to IV. He was a founding member of the Board of Directors of TransCelerate Biopharma, Inc., a leading biopharma industry
consortium, and served on the Executive, Audit and Finance Committees. Dr. Hubbard has been leading pharmaceutical research and
development activity for over thirty years and held positions of increasing responsibility in the biopharmaceutical and clinical research
and development services industries. Prior to joining Pfizer in 2010, he was Group President, Global Clinical Research Services at ICON
plc, a leading global clinical research organization, where he was responsible for the global business and operations. He is an Executive
Committee Member Emeritus on the Clinical Trials Transformation Initiative, former Board Member of Life Sciences Pennsylvania and former
Chairman of the Board of the Association of Clinical Research Organizations (ACRO). During his career, Dr. Hubbard has been directly
responsible for drug discovery and non clinical pharmacology, clinical pharmacology, project management, product development optimization,
commercial assessment of new chemical entities, and clinical development operations. He has led several drug development teams to successful
commercialization of new chemical entities and has participated at FDA meetings to support end of Phase II and end of Phase III data
presentations for psychiatric, neurological, cardiovascular, and anti infective drugs. Dr. Hubbard received a Bachelor of Science
degree in Biopsychology from the University of Santa Clara and a Doctorate from the University of Tennessee, with a research focus on
the genetic basis of hypertension and autonomic dysfunction. He was a National Institute of Health Postdoctoral Fellow in Cardiovascular
and Clinical Pharmacology at the University of Texas Health Sciences Center. We believe Dr. Hubbard’s qualifications to sit
on our Board include his extensive pharmaceutical experience leading research operations and his significant educational background.
Josephine Torrente.
Ms. Torrente has served as a member of our Board since October 2021. Ms. Torrente has more than 30 years of experience
in the pharmaceutical industry, and is currently a Director at Hyman, Phelps & McNamara PC, a law firm focused on advising clients
on FDA matters, where she has practiced since 1998. In addition to her time at Hyman, Phelps & McNamara, Ms. Torrente has
worked at Sprout Pharmaceuticals as Executive Vice President, Corporate and Regulatory Affairs, and Wyeth-Ayerst Research, where she
managed U.S. regulatory affairs and conducted discovery research. Ms. Torrente received her B.S. with honors from Case Western Reserve
University, her M.S. from the University of Alabama at Birmingham, and her J.D. summa cum laude from Temple University School of Law.
We believe Ms. Torrente’s qualifications to sit on our Board include her extensive pharmaceutical experience advising Companies
on FDA regulatory and drug development matters and her significant educational background.
EXECUTIVE COMPENSATION
Compensation Philosophy and Programs
We are a “smaller reporting
company” as defined in Rule 405 of the Securities Act of 1933, as amended (the “Securities Act”), and Item 10
of Regulation S-K, and in accordance with relevant SEC rules and guidance. We have elected, with respect to the disclosures required
by Item 402 (Executive Compensation) of Regulation S-K, to comply with the disclosure requirements applicable to smaller reporting companies.
Our intent with this overview is to enhance our stockholders’ understanding of our compensation philosophy and programs for our
executive officers as well as the compensation committee’s role in the design and administration of these programs and as it relates
to specific compensation decisions for our named executive officers.
Our executive compensation
philosophy is centered around attracting and retaining talented executives who are essential for our continued growth and success and
aligning the interests of these executives with those of our stockholders so that we can build long-term stockholder value. To achieve
this objective, in addition to annual base salaries, our executive compensation program utilizes a combination of annual incentives through
structured cash bonuses based on pre-defined goals as well as long-term incentives through equity-based compensation. On the whole, we
follow a “market median” target compensation philosophy approach, reviewing each compensation component (base salary, target
AIP and LTI awards) against the market median with flexibility to target an individual’s base and/or total compensation opportunity
above or below the market median based on performance, critical skills and recognition for current and/or anticipated future contributions.
In establishing overall executive compensation levels, our compensation committee may consider the following factors, including (i) the
executive’s scope of responsibilities, (ii) the strategic importance of the executive’s role, (iii) relevant peer
group data, (iv) attainment of individual and overall company performance objectives, (v) recruitment and/or retention concerns,
and (vi) the results of the advisory vote of the stockholders on the “say-on-pay” proposal at the prior years’
annual meeting of the stockholders. A substantial portion of executive compensation is directly tied to the achievement of corporate
performance objectives, and the contribution of these individuals to build a profitable business and stockholder value over time.
Our executive compensation
program is administered by the compensation committee of our Board (the “Compensation Committee”), with guidance and input
from our Chief Executive Officer and our independent compensation consultant, Pay Governance LLC (“Pay Governance”). The
Compensation Committee oversees the governance, design and administration of our executive compensation programs and evaluates these
programs against competitive practice, legal and regulatory developments and corporate governance trends. We are committed to continuing
to incorporate leading design and governance practices into our compensation programs. The Compensation Committee, in addition to other
activities mentioned above, establishes the new performance objectives for the upcoming fiscal year and approves merit, bonus and equity
awards for the current year. The Compensation Committee solicits and considers evaluations and recommendations submitted by the Chief
Executive Officer for the Compensation Committee’s review and approval. In the case of the Chief Executive Officer, the evaluation
of his performance is conducted by the Compensation Committee in consultation with the Board, and the Compensation Committee approves
any adjustments to his compensation as well as equity awards to be granted. The Compensation Committee obtains and considers input from
Pay Governance, including benchmarking data discussed below. Mr. Altomari, our Chief Executive Officer, plays no role in determining
his own salary, annual cash performance bonus or equity compensation.
Our Peer Group and the Use of Peer Group Compensation Data
Since 2016, the Compensation
Committee has periodically engaged Pay Governance as its independent consultant to assist the Compensation Committee in evaluating its
executive compensation program. The Compensation Committee has assessed the independence of Pay Governance, considering the factors required
by the Nasdaq Global Market Listing Rules and concluded that Pay Governance is independent from the Company and there are no conflicts
of interest.
Each year the Compensation
Committee has engaged Pay Governance LLC to provide compensation market data, provide updates on executive compensation trends and regulatory
development and provide recommendations to be used to establish compensation levels and plans for our executive officers for the following
year. In doing so, Pay Governance develops a relevant peer group of companies (described below) in order to perform a thorough compensation
review and analysis of our executive compensation levels and plans when compared to this group of peers, including base salary, annual
cash bonus and long-term equity incentive awards.
In August 2023, at the
recommendation of Pay Governance, the Compensation Committee approved modest changes to the peer group to align with our evolution as
a commercial organization, and removed two companies that were either acquired, restructured, or had fallen outside the financial evaluation
criteria, and added one company at similar commercial stage. In 2023 our peer group consisted of the following 19 companies:
| AcelRx Pharmaceuticals |
|
Femays |
|
Spectrum Pharmaceuticals |
| Aziyo Biologics |
|
Kala Pharma |
|
Sutro Biopharma |
| Aytu BioPharma |
|
Lipocine Inc. |
|
Trevena |
| Baudex Bio, Inc. |
|
Marinus Pharmaceuticals |
|
VYNE Therapeutics |
| Cidara Therapeutics, Inc. |
|
OptiNose Inc. |
|
Zynerba Pharma |
| Dare Bioscience |
|
Palatin Technologies |
|
|
| Evofem Biosciences |
|
SCYNEXIS |
|
|
We believe that our selected
peer group provides useful information to help us establish competitive compensation practices and levels of compensation that allow
us to attract, retain and motivate a talented executive team and, at the same time, aligns the interests of our executives with those
of our stockholders. We believe our executive compensation must be competitive within such peer groups, yet fully aligned with our current
stage of development and our responsibilities to stockholders.
While the target goal for
annual cash salary, annual cash bonus and long-term equity remains around or at the 50th percentile of our peer group, given our need
to preserve cash, the Board and Compensation Committee did not award performance-based bonuses to our Named Executive Officers for the
2022 fiscal year, as further described below. Annual bonuses which were approved at 70% of the target were also not awarded as the Company
did not have sufficient funds available to pay them. For the 2023 fiscal year, the Board and the Compensation Committee have deferred
any decision on performance-based bonuses and/or annual bonuses until the Committee meets to review 2023 performance against corporate
goals and assess available funds. The Named Executive Officers will need to be employed on the date any such payment is made. The Board
and Committee did award annual equity grants in the normal course in June 2023. No merit based salary adjustments were awarded to our
Named Executive Officers in 2023 given our need to preserve cash, although Messrs. Altomari and Gilmore elected to take salary increases
in the form of restricted stock unit (“RSU”) awards. A decision has not been taken yet on merit based salary adjustments
for 2024.
Summary Compensation Table
The following table provides
information concerning the compensation paid to our President and Chief Executive Officer, and our other three most highly compensated
individuals, for the fiscal years ended December 31, 2023 and 2022. We refer to these individuals as our named executive officers.
Name and
Principal | |
| | |
| | |
Restricted
Stock Unit
Awards | | |
Option | | |
All Other | | |
| |
| Position | |
Year | | |
Salary | | |
(RSU)(2) | | |
Awards(1) | | |
Compensation | | |
Total | |
| Al Altomari | |
2023 | | |
$ | 600,554 | | |
| 115,228 | | |
$ | 26,958 | | |
$ | 45,978 | (3) | |
$ | 788,718 | |
| Chairperson & Chief Executive Officer | |
2022 | | |
$ | 600,554 | | |
| | | |
$ | 196,396 | | |
$ | 43,842 | | |
$ | 840,762 | |
| | |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Geoffrey P. Gilmore | |
2023 | | |
$ | 450,000 | | |
| 50,136 | | |
$ | 9,705 | | |
$ | 44,851 | (4) | |
$ | 554,692 | |
| Sr. VP, Chief Administrative Officer | |
2022 | | |
| 429,492 | | |
| | | |
$ | 65,458 | | |
$ | 49,428 | | |
$ | 544,378 | |
| | |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Paul Korner | |
2023 | | |
$ | 442,000 | | |
| 33,480 | | |
$ | 9,705 | | |
$ | 52,596 | (5) | |
$ | 537,781 | |
| Chief Medical Officer | |
2022 | | |
| 442,000 | | |
| | | |
| 65,458 | | |
| 38,402 | | |
| 545,860 | |
| Scott M. Coiante | |
2023 | | |
$ | 161,250 | | |
| 42,000 | | |
| | | |
| 13,148 | (6) | |
| 216,398 | |
| Chief Financial Officer | |
2022 | | |
$ | - | | |
| - | | |
| - | | |
| - | | |
| - | |
| (1) |
In accordance with SEC rules, this column reflects the aggregate grant
date fair value of the option awards granted computed in accordance with Financial Accounting Standard Board Accounting Codification
Topic 718 for stock-based compensation transactions (ASC 718). Assumptions used in the calculation of these amounts are included
in Note 11 to our financial statements in our Annual Report on Form 10-K for the year ended December 31, 2022.
These amounts do not reflect the actual economic value that will be realized by the named executive officer upon the vesting of the
stock options, the exercise of the stock options, or the sale of the common stock underlying such stock options. |
| (2) |
Represents total grant date value of RSUs that were granted on June 28, 2023, which is based on the
closing price of Agile common stock on the grant date and the total number of restricted stock units granted. 37,500 RSU awards for
Mr. Altomari and 13,500 RSU awards for Mr. Gilmore are expected to vest 25% on each four year anniversary. This amount does not reflect
the actual economic value that will be realized by the named executive officer. 8,963 RSU awards for Mr. Altomari, and 6716 RSU awards
for Mr. Gilmore are each expected to vest in one year, June 28, 2024 and were awarded in lieu of a cash increase in their base salaries. |
| (3) |
Represents $29,112 for premiums paid by us for health and group life
insurance, $12,200 paid by us for safe harbor match and $4,666 paid by us for life insurance. |
| (4) |
Represents $31,581 for premiums paid by us for health and group life
insurance, $12,200 paid by us for safe harbor match and $1,070 paid by us for life insurance. |
| (5) |
Represents $39,589 for premiums paid by us for health and group life
insurance, $12,200 paid by us for safe harbor match and $807 paid by us for life insurance. |
| (6) |
Represents base salary and $13,148 for premiums paid by us for health
and group life insurance for time worked from September through December 2023. Mr. Coiante joined Agile Therapeutics on
August 16, 2023, so no data is available for 2022. |
Narrative Explanation of Certain Aspects of
the Summary Compensation Table
Pursuant to employment agreements
entered into with us, each of our named executive officers is eligible to receive (i) a base salary and (ii) an annual performance
bonus payable in cash, stock or a combination of both at the discretion of the compensation committee of the Board. The target amount
of each named executive officer’s annual performance bonus is a percentage of his or her base salary, as set forth in the
table below, and the actual amount payable is based on the achievement of individual and corporate objectives.
On August 16, 2023, Mr. Coiante
became our Senior Vice President, Chief Financial Officer (“CFO”). In connection with his appointment as CFO, Mr. Coiante
entered into an employment agreement and was awarded a grant of an RSU for 20,000 shares of the Company’s common stock vesting
over four years. Pursuant to the terms of his employment agreement, Mr. Coiante will receive an initial annual base salary of $430,000
and will be eligible to participate in the Company’s benefit and compensation plans, including the Company’s annual bonus
plan and the 2023 Equity Incentive Compensation Plan. Mr. Coiante’s target annual bonus is 45% of his base salary, which, for 2023,
shall be pro-rated for Mr. Coiante’s actual period of service in 2023.
Base Salary and Annual Performance Bonus
The base salary and target
annual performance bonus for each of our named executive officers for our fiscal year ended December 31, 2023, is listed in the
table below:
| | |
| | |
2023 Target | |
| Name | |
2023
Base Salary | | |
Performance
Bonus | |
| Al Altomari | |
| 600,554 | | |
| 60 | % |
| Scott M. Coiante | |
| 430,000 | | |
| 45 | % |
| Geoffrey P. Gilmore | |
| 450,000 | | |
| 45 | % |
| Paul Korner | |
| 442,000 | | |
| 45 | % |
For 2022, the Compensation
Committee, based on a recommendation from management, did not approve a merit-based increase in base salary for any officer in connection
with our overall performance. The Compensation Committee had initially intended to award bonus payments for our named executive officers
to reflect the commercial performance of Twirla, the management of our financial resources and progress on the Company’s Phase
4 commitments in 2021 at 50% of each executive’s target. However, given our need to preserve cash, the named executive officers
were not granted these bonuses. In addition, the long-term equity incentives were reduced in value compared to the awards granted in
2021 because of the reduced number of shares available in the Company’s incentive plan. Mr. Altomari’s long-term equity
incentive grants were reduced by 88%, Mr. Gilmore’s grants were reduced by 89% and Dr. Korner’s grants were reduced
by 84% in 2022. In November 2022, Mr. Gilmore was promoted to Senior Vice President, Chief Administrative Officer and his salary was
increased to $450,000.
For 2023, the Compensation
Committee, based on a recommendation from management, again did not approve a merit-based increase in base salary for any officer based
on our continued need to preserve cash. Messrs. Altomari and Gilmore elected to take salary increases in the form of restricted stock
unit (“RSU”) awards and were granted RSUs for 8,963 shares and 6,716 shares respectively. Objectives for the named executive
officers’ target bonuses for our fiscal year ending December 31, 2022 included three general categories: (1) the commercial
performance of Twirla, as measured by net revenue, weighted at 50% of the total bonus potential; (2) management of our financial
position for the 2022 fiscal year, as measured by our pre-tax earnings and cash position, collectively weighted at 40% of the total bonus
potential; and (3) activities related to evaluating its pipeline and implementing the post approval commitments to the FDA in connection
with the FDA’s approval of Twirla, and business development activity either through acquisition of additional product or similar
transaction, both collectively weighted at 10% of the total bonus potential.
Objectives for the 2022
named executive officer target bonuses were deemed to be achieved at 70%, which at the discretion of the Compensation Committee reflected
that we had (i) not met our net revenue target, (ii) partially achieved our goals to raise capital but had successfully managed
our expenses, (iii) met the objectives for evaluating our pipeline and fully met the timelines for our obligations to the FDA, and
(iv) reviewed and identified multiple business development candidates throughout the course of the year. The Compensation Committee,
based on a recommendation from management did not approve bonuses for our named executive officers in a continued effort to conserve
cash.
For fiscal year ending December 31, 2023, the
Compensation Committee had established the following goals which must be achieved in order for the named executive officers to fully
realize their potential annual cash bonus targets:
| · | Twirla
Growth: Achieving growth of Twirla net revenue as measured by established targets; |
| · | Financial
Management: Reducing our loss and financing the Company’s operations through 2023
as measured by targets for pre-tax earnings and cash; and |
| · | Building
Agile: Pursuing business development opportunities to grow our business and meet our
Phase 4 commitments to the U.S. Food and Drug Administration for 2023. |
The Compensation Committee will meet in the first
quarter of 2024 to review our performance against the Company’s corporate goals and the achievement of any merit-based salary increase
and/or annual bonus incentive for the named executive officers.
Equity Compensation
We have historically offered
stock options as the primary long-term incentive vehicle to our employees, including our named executive officers, as the long-term incentive
component of our compensation program. Stock options allow employees to purchase shares of our common stock at a price per share equal
to the fair market value of our common stock on the date of grant and may or may not be intended to qualify as “incentive stock
options” for U.S. federal income tax purposes. We typically grant stock options to new hires upon their commencing employment with
us. Awards to newly hired employees generally vest with respect to 25% of the total number of option shares on the first anniversary
of the grant date and in equal monthly installments over the following 36 months.
As part of an annual compensation
evaluation of our named executive officers at the beginning of each year, the compensation committee of the Board considers granting
stock options and other long-term incentive vehicles to our named executive officers based on such executive’s individual performance
for the preceding year and as an incentive for future performance. Stock options are granted under our 2023 Equity Incentive Compensation
Plan, or the 2023 Plan, and generally vest with respect to 25% of the total number of option shares on the first anniversary of the grant
date and in equal monthly installments over the following 36 months. RSUs can also be granted under the 2023 Plan, and generally
vest with respect to 25% of the total number of restricted stock units on the first four anniversaries of the grant date.
Effective January 19,
2022, the Compensation Committee granted each of Mr. Altomari, Mr. Gilmore, and Dr. Korner options to purchase shares
of our common stock in the amount of 337, 112, and 112 (respectively), each at an exercise price of $684 per share, which was the closing
price of our common stock on January 19, 2022. The Compensation Committee made its decision to grant stock options to our named
executive officers based on (i) their individual performance for 2021 and contribution to the Company’s performance against
its corporate goals in 2021 and (ii) to incentivize future performance.
On November 9, 2022,
the Compensation Committee approved an off-cycle grant for all active employees, including our named executive officers. The Compensation
Committee granted each of Mr. Altomari, Mr. Gilmore, and Dr. Korner options to purchase shares of our common stock in
the amount of 600, 200 and 200 (respectively), each at an exercise price of $10.22 per share, which was the closing price of our common
stock on November 9, 2022.
On June 28, 2023, following
the Company’s Annual Meeting, the Compensation Committee approved an annual grant for all active employees including our named
executive officers. The Compensation Committee granted each of Mr. Altomari, Mr. Gilmore, and Dr. Korner a mix of options and RSUs. The
options to purchase shares of our common stock were in the amount of 12,500, 4,500 and 4,500 (respectively) at an exercise price of $2.48
per share, which was the closing price of our common stock on June 28, 2023. The RSUs were granted under the 2023 Plan equivalent to
shares of the Company’s common stock in the amount of 37,500, 13,500 and 13,500 (respectively). At the same time, Messrs. Altomari
and Gilmore were awarded RSUs in the amount of 8,963 shares and 6,716 shares, respectively, in lieu of cash salary increases.
On October 3, 2023, Scott
Coiante was granted an equity award (“Inducement Grant”) in the form of an RSU for 20,000 shares of the Company’s common
stock (“Shares”). The Inducement Grant is governed by the terms of an individual award agreement, and the 2023 Equity Incentive
Compensation Plan. The Inducement Grant will vest on a four-year vesting schedule, with 25% of the Shares subject to the option vesting
on the first four anniversaries of the date that the Inducement Grant is granted, subject to Mr. Coiante’s continued employment
with the Company on each vesting date and subject to accelerated vesting in the event of a change in control.
As described under “Outstanding
Equity Awards as of December 31, 2023” below, all outstanding and unvested equity awards held by our named executive officers
are subject to accelerated vesting in the event we experience a change in control and the equity awards are not assumed by the successor
corporation, or at the discretion of the Board. In addition, all outstanding equity awards held by our named executive officers are subject
to accelerated vesting in the event of a termination without cause or resignation for good reason that occurs in connection with a change
in control, as described under “Severance and Change in Control Benefits” below.
Outstanding Equity Awards as of December 31, 2023
The following table sets
forth information regarding each outstanding equity award held by each of our named executive officers as of December 31, 2023.
The number of shares subject to each award and, where applicable, the exercise price per share, reflect all changes as a result of our
capitalization adjustments.
| | |
Option Awards | |
Stock Awards | |
| | |
| |
Number of | | |
Number of | | |
| | |
| | |
Number of | | |
Market Value | |
| | |
| |
Securities | | |
Securities | | |
| | |
| | |
Shares or | | |
of Shares or | |
| | |
| |
Underlying | | |
Underlying | | |
| | |
| | |
Units of | | |
Units of | |
| | |
| |
Unexercised | | |
Unexercised | | |
Option | | |
| | |
Stock That | | |
Stock That | |
| | |
| |
Options | | |
Options | | |
Exercise | | |
Option | | |
Have Not | | |
Have Not | |
| | |
Grant | |
Exercisable | | |
Unexercisable | | |
Price | | |
Expiration | | |
Vested | | |
Vested | |
| Name | |
Date(1) | |
(#) | | |
(#) | | |
($)(2) | | |
Date | | |
(#) | | |
($)(3) | |
| Al Altomari | |
| |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| | |
6/24/2014 | |
| 36 | | |
| — | | |
$ | 21,500 | | |
| 6/23/2024 | | |
| | | |
| | |
| | |
2/19/2015 | |
| 45 | | |
| — | | |
$ | 18,900 | | |
| 2/18/2025 | | |
| | | |
| | |
| | |
2/19/2015 | |
| 45 | | |
| — | | |
$ | 21,500 | | |
| 2/18/2025 | | |
| | | |
| | |
| | |
2/8/2016 | |
| 49 | | |
| — | | |
$ | 11,860 | | |
| 2/7/2026 | | |
| | | |
| | |
| | |
1/25/2017 | |
| 120 | | |
| — | | |
$ | 4,520 | | |
| 1/24/2027 | | |
| | | |
| | |
| | |
1/24/2018 | |
| 127 | | |
| — | | |
$ | 6,920 | | |
| 1/23/2028 | | |
| | | |
| | |
| | |
6/20/2018 | |
| 75 | | |
| — | | |
$ | 1,160 | | |
| 6/19/2028 | | |
| | | |
| | |
| | |
1/29/2019 | |
| 269 | | |
| — | | |
$ | 1,680 | | |
| 1/28/2029 | | |
| | | |
| | |
| | |
1/29/2019 | |
| 76 | | |
| — | | |
$ | 5,000 | | |
| 1/28/2029 | | |
| | | |
| | |
| | |
1/22/2020 | |
| 407 | | |
| 8 | | |
$ | 5,660 | | |
| 1/21/2030 | | |
| | | |
| | |
| | |
1/27/2021 | |
| 255 | | |
| 90 | | |
$ | 5,640 | | |
| 1/26/2031 | | |
| | | |
| | |
| | |
1/19/2022 | |
| 163 | | |
| 174 | | |
$ | 684 | | |
| 1/18/2032 | | |
| | | |
| | |
| | |
11/9/2022 | |
| 164 | | |
| 435 | | |
$ | 10.22 | | |
| 11/8/2032 | | |
| | | |
| | |
| | |
6/28/2023 | |
| — | | |
| 12,500 | | |
| 2.48 | | |
| 6/27/2033 | | |
| | | |
| | |
| | |
6/28/2023 | |
| | | |
| | | |
| | | |
| | | |
| 46,463 | | |
| 90,603 | |
| | |
| |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Scott M. Coiante | |
10/3/2023 | |
| | | |
| | | |
| | | |
| | | |
| 20,000 | | |
| 39,000 | |
| | |
| |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Geoffrey P. Gilmore | |
8/1/2014 | |
| 15 | | |
| — | | |
$ | 12,000 | | |
| 7/31/2024 | | |
| | | |
| | |
| | |
2/19/2015 | |
| 10 | | |
| — | | |
$ | 18,900 | | |
| 2/18/2025 | | |
| | | |
| | |
| | |
2/8/2016 | |
| 14 | | |
| — | | |
$ | 11,860 | | |
| 2/7/2026 | | |
| | | |
| | |
| | |
4/18/2016 | |
| 14 | | |
| — | | |
$ | 12,920 | | |
| 4/17/2026 | | |
| | | |
| | |
| | |
1/25/2017 | |
| 69 | | |
| — | | |
$ | 4,520 | | |
| 1/24/2027 | | |
| | | |
| | |
| | |
1/24/2018 | |
| 53 | | |
| — | | |
$ | 6,920 | | |
| 1/23/2028 | | |
| | | |
| | |
| | |
6/20/2018 | |
| 75 | | |
| — | | |
$ | 1,160 | | |
| 6/19/2028 | | |
| | | |
| | |
| | |
1/29/2019 | |
| 86 | | |
| — | | |
$ | 1,680 | | |
| 1/28/2029 | | |
| | | |
| | |
| | |
1/29/2019 | |
| 19 | | |
| — | | |
$ | 5,000 | | |
| 1/28/2029 | | |
| | | |
| | |
| | |
7/3/2019 | |
| 75 | | |
| — | | |
$ | 2,960 | | |
| 7/2/2029 | | |
| | | |
| | |
| | |
1/22/2020 | |
| 154 | | |
| 3 | | |
$ | 5,660 | | |
| 1/21/2030 | | |
| | | |
| | |
| | |
1/27/2021 | |
| 101 | | |
| 28 | | |
$ | 5,640 | | |
| 1/26/2031 | | |
| | | |
| | |
| | |
1/19/2022 | |
| 59 | | |
| 53 | | |
$ | 684 | | |
| 1/18/2032 | | |
| | | |
| | |
| | |
11/9/2022 | |
| 56 | | |
| 143 | | |
$ | 10.22 | | |
| 11/8/2032 | | |
| | | |
| | |
| | |
6/28/2023 | |
| — | | |
| 4,500 | | |
| 2.48 | | |
| 6/27/2033 | | |
| | | |
| | |
| | |
6/28/2023 | |
| | | |
| | | |
| | | |
| | | |
| 20,216 | | |
| 39,421 | |
| | |
| |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Paul Korner | |
8/17/2020 | |
| 67 | | |
| 7 | | |
$ | 5,740 | | |
| 8/16/2030 | | |
| | | |
| | |
| | |
1/27/2021 | |
| 68 | | |
| 17 | | |
$ | 5,640 | | |
| 1/26/2031 | | |
| | | |
| | |
| | |
1/19/2022 | |
| 59 | | |
| 53 | | |
$ | 684 | | |
| 1/18/2032 | | |
| | | |
| | |
| | |
11/9/2022 | |
| 56 | | |
| 143 | | |
$ | 10.22 | | |
| 11/8/2032 | | |
| | | |
| | |
| | |
6/28/2023 | |
| — | | |
| 4,500 | | |
| 2.48 | | |
| 6/27/2033 | | |
| | | |
| | |
| | |
6/28/2023 | |
| | | |
| | | |
| | | |
| | | |
| 13,500 | | |
| 26,325 | |
| (1) |
Each of the option awards vest with respect to 25% of the shares one year
following the date of grant and with respect to 1/36th of the remaining shares on each monthly anniversary thereafter
over the following three years, subject to the executive’s continuous service with us through the vesting date. |
| (2) |
All of the option awards listed in the table above were granted with
a per share exercise price equal to or above the fair market value of our common stock on the date of grant. |
| (3) |
Market value is based on the closing price of $1.95 on December 29,
2023, the last trading day of 2023. |
Severance and Change in Control Benefits
Each of our named executive
officers is eligible to receive certain benefits if his or her employment is terminated under certain circumstances, as described under
“Severance and Change in Control Benefits” below.
Al Altomari
We entered into an employment
agreement with Mr. Altomari on October 11, 2010, which was amended on December 18, 2012, April 12, 2016, and amended
and restated on August 14, 2020, and November 22, 2022. Pursuant to the terms of the agreement, Mr. Altomari is entitled
to receive certain benefits in the event his employment is terminated.
Payments Upon Termination Absent
a Change in Control.
If Mr. Altomari terminates
his employment for good reason or if we terminate his employment without reasonable cause (other than due to death or disability), in
either case in the absence of a change in control, he is entitled to receive the following severance benefits: (i) base salary continuation
for a period of 12 months, and (ii) reimbursement of Mr. Altomari’s health insurance premiums for a period of 12 months
following the date of his termination or until Mr. Altomari obtains other employment, whichever is sooner. In the event of a change
in control following his termination, any base salary continuation payments still due to Mr. Altomari shall be paid in full upon
the change in control.
In the event Mr. Altomari’s
employment terminates as a result of his disability, he will be entitled to receive (i) base salary continuation for a period of
12 months following the date of his termination, and (ii) reimbursement of Mr. Altomari’s health insurance premiums
for a period of 12 months following the date of his termination due to his disability or until Mr. Altomari obtains other employment,
whichever is sooner.
Payments Upon Termination in Connection
with a Change in Control.
If Mr. Altomari terminates
his employment for good reason or if we terminate his employment without reasonable cause (other than due to death or disability), in
either case upon or within 12 months following a change in control, he is entitled to receive the following severance benefits:
(i) a lump-sum cash payment in the amount of 2.0 times his then annual rate of base salary, (ii) a lump sum cash payment
equal to Mr. Altomari’s target annual bonus for the year in which his termination occurs, (iii) reimbursement of
Mr. Altomari’s health insurance premiums for a period of 24 months following the date of his termination or until Mr. Altomari
obtains other employment, whichever is sooner and (iv) each outstanding equity award shall automatically vest in full.
Notwithstanding the foregoing,
any payments and benefits that would otherwise be paid to Mr. Altomari (whether or not under his employment agreement) in connection
with a change in control of the Company will be reduced to the extent necessary to ensure that he is not subject to any excise tax under
Internal Revenue Code Section 4999 in connection with any change in control of the Company or his subsequent termination of employment.
However, such reduction will not be made if Mr. Altomari would be better off (on an after-tax basis) receiving all payments and
benefits and paying all excise and income taxes under Internal Revenue Code Section 4999.
Under Mr. Altomari’s
employment agreement, the terms below are generally defined as follows:
“Change in
Control” means (i) a merger or consolidation in which more than 50% of the voting securities of the Company are transferred
and the composition of the board after such transaction constitutes less than 50% of the members of the board prior to the transaction;
(ii) any acquisition, directly or indirectly, of beneficial ownership of more than 50% of the total combined voting power of the
Company, other than in a capital-raising transaction; or (iii) the sale, transfer, exclusive worldwide license or other disposition
of all or substantially all of the assets of the Company.
“Good reason”
means Mr. Altomari’s resignation following notice to the Company of, and failure by the Company to cure, the occurrence of
any of the following: (i) an office relocation of more than 50 miles; (ii) failure by the Company to comply with any material
term of the employment agreement; or (iii) the demotion to a lesser position or substantial diminution of authority, duties or responsibilities,
except for a reduction in title, position, responsibilities or duties solely by virtue of the Company being acquired and made part of,
or operated as a subsidiary of, a larger company, so long as the new duties and responsibilities are reasonably commensurate with Mr. Altomari’s
experience.
“Reasonable
cause” means (i) an act or omission that constitutes dishonesty, disloyalty, fraud, deceit, gross negligence, willful misconduct
or recklessness, including, but not limited to Mr. Altomari’s willful violation of the Company’s bylaws or code of conduct,
and that is directly or indirectly materially detrimental to the Company’s best interest; (ii) intentional failure to perform
any lawful duties assigned by the board after receiving notice and an opportunity to cure; (iii) the commission of any act that
constitutes a felony; or (iv) any material breach of certain sections of the employment agreement.
The payment of any severance
compensation described above is subject to Mr. Altomari’s execution and non-revocation of a general release of claims against
the Company, and his compliance with non-competition and non-solicitation restrictive covenants for a 24-month period after his termination
without cause or for good reason upon or within 12 months following a change of control and a 12-month period following his termination
date in all other cases.
Geoffrey P. Gilmore
We entered into an employment
agreement with Mr. Gilmore on April 12, 2016, which was amended on July 3, 2019, August 14, 2020, and November 1,
2022. Pursuant to the terms of the agreement, as amended, Mr. Gilmore is entitled to receive certain benefits in the event his employment
is terminated.
Payments Upon Termination
Absent a Change in Control.
If Mr. Gilmore terminates
his employment for good reason or if we terminate his employment without reasonable cause (other than due to death or disability), in
either case in the absence of a change in control, he is entitled to receive the following severance benefits: (i) base salary continuation
for a period of 12 months, and (ii) reimbursement of Mr. Gilmore’s health insurance premiums for a period of 12 months
following the date of his termination or until he obtains other employment, whichever is sooner. In the event of a change in control
following his termination, any base salary continuation payments still due to Mr. Gilmore shall be paid in full upon the change
in control.
In the event Mr. Gilmore’s
employment terminates as a result of his disability, he will be entitled to receive (i) base salary continuation for a period of
12 months following the date of his termination, and (ii) reimbursement of his health insurance premiums for a period of 12 months
following the date of his termination due to his disability or until he obtains employment, whichever is sooner.
Payments Upon Termination in Connection
with a Change in Control.
If Mr. Gilmore terminates
his employment for good reason or if we terminate his employment without reasonable cause (other than due to death or disability), in
either case upon or within 12 months following a change in control, he is entitled to receive the following severance benefits:
(i) a lump-sum cash payment in the amount of 1.5 times his then annual rate of base salary, (ii) a lump sum cash payment equal
to his target annual bonus for the year in which his termination occurs, (iii) reimbursement of his health insurance premiums
for a period of 18 months following the date of his termination or until he obtains other employment, whichever is sooner and (iv) each
outstanding equity award shall automatically vest in full.
Notwithstanding the foregoing,
any payments and benefits that would otherwise be paid to Mr. Gilmore (whether or not under his employment agreement) in connection
with a change in control of the Company will be reduced to the extent necessary to ensure that he is not subject to any excise tax under
Internal Revenue Code Section 4999 in connection with any change in control of the Company or his subsequent termination of employment.
However, such reduction will not be made if Mr. Gilmore would be better off (on an after-tax basis) receiving all payments and benefits
and paying all excise and income taxes under Internal Revenue Code Section 4999.
Under Mr. Gilmore’s
employment agreement, the terms below are generally defined as follows:
“Change in
Control” means (i) a merger or consolidation in which more than 50% of the voting securities of the Company are transferred
and the composition of the board after such transaction constitutes less than 50% of the members of the board prior to the transaction;
(ii) any acquisition, directly or indirectly, of beneficial ownership of more than 50% of the total combined voting power of the
Company, other than in a capital-raising transaction; or (iii) the sale, transfer, exclusive worldwide license or other disposition
of all or substantially all of the assets of the Company.
“Good reason”
means Mr. Gilmore’s resignation following notice to the Company of, and failure by the Company to cure, the occurrence of
any of the following: (i) an office relocation of more than 50 miles; (ii) failure by the Company to comply with any material
term of the employment agreement; or (iii) the demotion to a lesser position or substantial diminution of authority, duties or responsibilities,
except for a reduction in title, position, responsibilities or duties solely by virtue of the Company being acquired and made part of,
or operated as a subsidiary of, a larger company, so long as the new duties and responsibilities are reasonably commensurate with Mr. Gilmore’s
experience.
“Reasonable
cause” means (i) an act or omission that constitutes dishonesty, disloyalty, fraud, deceit, gross negligence, willful misconduct
or recklessness, including, but not limited to Mr. Gilmore’s willful violation of the Company’s bylaws or code of conduct,
and that is directly or indirectly materially detrimental to the Company’s best interest; (ii) intentional failure to perform
any lawful duties assigned by the Board after receiving notice and an opportunity to cure; (iii) the commission of any act that
constitutes a felony; or (iv) any material breach of certain sections of the employment agreement.
The payment of any severance
compensation described above is subject to Mr. Gilmore’s execution and non-revocation of a general release of claims against
the Company, and his compliance with non-competition and non-solicitation restrictive covenants for an 12-month period after his termination
without cause or for good reason upon or within 18 months following a change of control.
Paul Korner, M.D.
We entered into an employment
agreement with Dr. Korner on August 17, 2020, which was amended on November 1, 2022. Pursuant to the terms of the agreement,
Dr. Korner is entitled to receive certain benefits in the event his employment is terminated.
Payments Upon Termination
Absent a Change in Control.
If Dr. Korner terminates
his employment for good reason or if we terminate his employment without reasonable cause (other than due to death or disability), in
either case in the absence of a change in control, he is entitled to receive the following severance benefits: (i) base salary continuation
for a period of 12 months, and (ii) reimbursement of his health insurance premiums for a period of 12 months following
the date of his termination or until he obtains other employment, whichever is sooner. In the event of a change in control following
his termination, any base salary continuation payments still due to Dr. Korner shall be paid in full upon the change in control.
In the event Dr. Korner’s
employment terminates as a result of his disability, he will be entitled to receive (i) base salary continuation for a period of
12 months following the date of his termination, and (ii) reimbursement of his health insurance premiums for a period of 12 months
following the date of his termination due to his disability or until he obtains employment, whichever is sooner.
Payments Upon Termination in Connection with
a Change in Control.
If Dr. Korner terminates
his employment for good reason or if we terminate his employment without reasonable cause (other than due to death or disability), in
either case upon or within 12 months following a change in control, he is entitled to receive the following severance benefits:
(i) a lump-sum cash payment in the amount of 1.5 times his then annual rate of base salary, (ii) a lump sum cash payment equal
to his target annual bonus for the year in which his termination occurs, (iii) reimbursement of his health insurance premiums
for a period of 18 months following the date of his termination or until he obtains other employment, whichever is sooner and (iv) each
outstanding equity award shall automatically vest in full.
Notwithstanding the foregoing,
any payments and benefits that would otherwise be paid to Dr. Korner (whether or not under his employment agreement) in connection
with a change in control of the Company will be reduced to the extent necessary to ensure that he is not subject to any excise tax under
Internal Revenue Code Section 4999 in connection with any change in control of the Company or his subsequent termination of employment.
However, such reduction will not be made if Dr. Korner would be better off (on an after-tax basis) receiving all payments and benefits
and paying all excise and income taxes under Internal Revenue Code Section 4999.
Under Dr. Korner’s
employment agreement, the terms below are generally defined as follows:
“Change in
Control” means (i) a merger or consolidation in which more than 50% of the voting securities of the Company are transferred
and the composition of the board after such transaction constitutes less than 50% of the members of the board prior to the transaction;
(ii) any acquisition, directly or indirectly, of beneficial ownership of more than 50% of the total combined voting power of the
Company, other than in a capital-raising transaction; or (iii) the sale, transfer, exclusive worldwide license or other disposition
of all or substantially all of the assets of the Company.
“Good reason”
means Dr. Korner’s resignation following notice to the Company of, and failure by the Company to cure, the occurrence of any
of the following: (i) an office relocation of more than 50 miles; (ii) failure by the Company to comply with any material term
of the employment agreement; or (iii) the demotion to a lesser position or substantial diminution of authority, duties or responsibilities,
except for a reduction in title, position, responsibilities or duties solely by virtue of the Company being acquired and made part of,
or operated as a subsidiary of, a larger company, so long as the new duties and responsibilities are reasonably commensurate with Dr. Korner’s
experience.
“Reasonable
cause” means (i) an act or omission that constitutes dishonesty, disloyalty, fraud, deceit, gross negligence, willful misconduct
or recklessness, including, but not limited to Dr. Korner’s willful violation of the Company’s bylaws or code of conduct,
and that is directly or indirectly materially detrimental to the Company’s best interest; (ii) intentional failure to perform
any lawful duties assigned by the board after receiving notice and an opportunity to cure; (iii) the commission of any act that
constitutes a felony; or (iv) any material breach of certain sections of the employment agreement.
The payment of any severance
compensation described above is subject to Dr. Korner’s execution and non-revocation of a general release of claims against
the Company, and his compliance with non-competition and non-solicitation restrictive covenants for a 12-month period after his termination
without cause or for good reason upon or within 18 months following a change of control.
Scott M. Coiante.
We entered into an employment
agreement with Mr. Coiante on August 16, 2023. Pursuant to the terms of the agreement, Mr. Coiante is entitled to receive certain
benefits in the event his employment is terminated.
Payments Upon Termination
Absent a Change in Control.
If Mr. Coiante terminates
his employment for good reason or if we terminate his employment without reasonable cause (other than due to death or disability), in
either case in the absence of a change in control, he is entitled to receive the following severance benefits: (i) base salary continuation
for a period of 12 months, and (ii) reimbursement of his health insurance premiums for a period of 12 months following
the date of his termination or until he obtains other employment, whichever is sooner. In the event of a change in control following
his termination, any base salary continuation payments still due to Mr. Coiante shall be paid in full upon the change in control.
In the event Mr. Coiante’s
employment terminates as a result of his disability, he will be entitled to receive (i) base salary continuation for a period of
12 months following the date of his termination, and (ii) reimbursement of his health insurance premiums for a period of 12 months
following the date of his termination due to his disability or until he obtains employment, whichever is sooner.
Payments Upon Termination in Connection with
a Change in Control.
If Mr. Coiante terminates
his employment for good reason or if we terminate his employment without reasonable cause (other than due to death or disability), in
either case upon or within 12 months following a change in control, he is entitled to receive the following severance benefits:
(i) a lump-sum cash payment in the amount of 1.5 times his then annual rate of base salary, (ii) a lump sum cash payment equal
to his target annual bonus for the year in which his termination occurs, (iii) reimbursement of his health insurance premiums
for a period of 18 months following the date of his termination or until he obtains other employment, whichever is sooner and (iv) each
outstanding equity award shall automatically vest in full.
Notwithstanding the foregoing,
any payments and benefits that would otherwise be paid to Mr. Coiante (whether or not under his employment agreement) in connection with
a change in control of the Company will be reduced to the extent necessary to ensure that he is not subject to any excise tax under Internal
Revenue Code Section 4999 in connection with any change in control of the Company or his subsequent termination of employment. However,
such reduction will not be made if Mr. Coiante would be better off (on an after-tax basis) receiving all payments and benefits and paying
all excise and income taxes under Internal Revenue Code Section 4999.
Under Mr. Coiante’s
employment agreement, the terms below are generally defined as follows:
“Change in
Control” means (i) a merger or consolidation in which more than 50% of the voting securities of the Company are transferred
and the composition of the board after such transaction constitutes less than 50% of the members of the board prior to the transaction;
(ii) any acquisition, directly or indirectly, of beneficial ownership of more than 50% of the total combined voting power of the
Company, other than in a capital-raising transaction; or (iii) the sale, transfer, exclusive worldwide license or other disposition
of all or substantially all of the assets of the Company.
“Good reason”
means Mr. Coiante’s resignation following notice to the Company of, and failure by the Company to cure, the occurrence of any of
the following: (i) an office relocation of more than 50 miles; (ii) failure by the Company to comply with any material term
of the employment agreement; or (iii) the demotion to a lesser position or substantial diminution of authority, duties or responsibilities,
except for a reduction in title, position, responsibilities or duties solely by virtue of the Company being acquired and made part of,
or operated as a subsidiary of, a larger company, so long as the new duties and responsibilities are reasonably commensurate with Mr.
Coiante’s experience.
“Reasonable
cause” means (i) an act or omission that constitutes dishonesty, disloyalty, fraud, deceit, gross negligence, willful misconduct
or recklessness, including, but not limited to Mr. Coiante’s willful violation of the Company’s bylaws or code of conduct,
and that is directly or indirectly materially detrimental to the Company’s best interest; (ii) intentional failure to perform
any lawful duties assigned by the board after receiving notice and an opportunity to cure; (iii) the commission of any act that
constitutes a felony; or (iv) any material breach of certain sections of the employment agreement.
The payment of
any severance compensation described above is subject to Mr. Coiante’s execution and non-revocation of a general release of claims
against the Company, and his compliance with non-competition and non-solicitation restrictive covenants for a 12-month period after his
termination without cause or for good reason upon or within 18 months following a change of control.
Employee Benefits and Perquisites
We also provide group life
insurance, health, vision, and dental care insurance to all employees, including our named executive officers. These benefits do not
discriminate in scope, terms, or operation in favor of the named executive officers. All such benefits terminate at the time each individual
is no longer employed with us or as otherwise provided in the applicable employment agreement. All our named executive officers are eligible
to participate in all of our employee benefit plans, in each case on the same basis as other employees. We maintain a 401(k) defined
contribution plan, which is our primary retirement benefit for employees, including executives. We make a safe-harbor contribution of
100% of the first 3% of an employee’s elected salary deferral, plus 50% of the next 2% of salary deferral subject to Internal Revenue
Service limitations. Although permitted under the plan, we have not matched employee contributions to the 401(k) plan. We provide
a life insurance benefit for our named executive officers in an amount equal to up to twice the named executive officer’s base
salary and target bonus capped at $1,000,000.
We do not generally provide
our named executive officers with perquisites or other personal benefits (other than severance benefits, and the life insurance benefit,
as described above).
DIRECTOR COMPENSATION
During our fiscal year
ended December 31, 2023, our non-employee directors who served on our Board were entitled to receive cash fees and were granted
restricted stock units (RSUs) and options to purchase shares of our common stock for their service . A non-employee director is a director
who is not employed by us and who does not receive compensation from us (other than for services as a director) or has a business relationship
with us that would require disclosure under certain SEC rules. Due to our cash constraints, we paid the directors their cash fees for
the first quarter of 2023 but did not pay the directors their fees for the rest of 2023. Mr. Altomari, our Chief Executive Officer
and Chairperson of our Board, did not receive any compensation from us during our fiscal year ended December 31, 2023 for his
service as a director and is not included in the table below.
| |
|
Fees
Earned or |
|
|
|
|
|
Restricted |
|
|
|
|
| Name |
|
Paid
in
Cash (4) |
|
|
Option
Awards(1)(2) |
|
|
Stock
Awards(1)(2) |
|
|
Total |
|
| Sharon Barbari |
|
$ |
77,500 |
|
|
$ |
3,344 |
|
|
$ |
3,345 |
|
|
$ |
84,190 |
|
| Sandra Carson, M.D., FACOG |
|
$ |
55,000 |
|
|
$ |
3,344 |
|
|
$ |
3,345 |
|
|
$ |
61,690 |
|
| Seth H.Z. Fischer |
|
$ |
95,000 |
|
|
$ |
3,344 |
|
|
$ |
3,345 |
|
|
$ |
101,690 |
|
| John Hubbard, Ph.D. |
|
$ |
65,000 |
|
|
$ |
3,344 |
|
|
$ |
3,345 |
|
|
$ |
71,690 |
|
| Ajit S. Shetty, Ph.D. (3) |
|
$ |
14,375 |
|
|
$ |
- |
|
|
$ |
- |
|
|
$ |
14,375 |
|
| Josephine Torrente |
|
$ |
55,000 |
|
|
$ |
3,344 |
|
|
$ |
3,345 |
|
|
$ |
61,690 |
|
| (1) |
In accordance with SEC rules, this column reflects the aggregate grant
date fair value of the awards granted computed in accordance with Financial Accounting Standard Board Accounting Codification Topic 718
for stock-based compensation transactions (ASC 718). Assumptions used in the calculation of these amounts are included in Note 11
to our financial statements in our Annual Report on Form 10-K for the year ended December 31, 2022. These amounts
do not reflect the actual economic value that will be realized by the director upon the vesting of the stock options, the exercise
of the stock options, or the sale of the common stock underlying such stock options. |
| (2) |
As of December 31, 2023, our non-employee directors held the following
aggregate number of shares under outstanding stock options (representing unexercised option awards—both exercisable and unexercisable)
and unvested restricted stock unit awards: |
| | |
Aggregate Number of | | |
Aggregate Number of | |
| | |
Shares Underlying | | |
Shares Underlying | |
| Name | |
Option Awards | | |
RSU Awards | |
| Sharon Barbari | |
| 1117 | | |
| 935 | |
| Sandra Carson, M.D., FACOG | |
| 1117 | | |
| 935 | |
| Seth H.Z. Fischer | |
| 1185 | | |
| 935 | |
| John Hubbard, Ph.D. | |
| 1199 | | |
| 935 | |
| Ajit S. Shetty, Ph.D. (3) | |
| 127 | | |
| 0 | |
| Josephine Torrente | |
| 1129 | | |
| 948 | |
| (3) |
The compensation provided for Mr. Shetty represents payment until
June 8, 2023, when he did not stand for re-election to the Board at the Company’s annual meeting of stockholders. |
| (4) |
The amounts represent the fees earned by Board members in 2023;
however, in an effort to preserve cash, the Company has only paid a portion of the fees for 2023 at this time. |
Non-Employee Director Compensation
During 2023, each non-employee member of our Board
is entitled to receive the following cash compensation for board services, as applicable:
| |
● |
$45,000 per year for service as a Board member; |
| |
● |
$25,000 per year for service as lead independent director; |
| |
● |
$20,000 per year for service as chairperson of the audit committee; |
| |
● |
$15,000 per year for service as chairperson of the compensation committee; |
| |
● |
$12,500 per year for service as chairperson of the finance committee; |
| |
● |
$10,000 per year for service as chairperson of the nominating
and corporate governance committee; |
| |
● |
$12,500 per year for service as chairperson of the science and
technology committee; and |
| |
● |
$5,000 per year for service as a member on each committee, except
each member of the compensation committee is entitled to receive $7,500 per year for service in that role and each member of
the audit committee is entitled to receive $10,000 per year for service in that role. |
Non-employee members of our
Board receive automatic grants of equity awards under our 2023 Equity Incentive Compensation Plan (the “2023 Plan”). Each
non-employee director joining our Board will automatically be granted equity awards totaling $150,000 in value split equally between
a non-statutory stock option to purchase shares of common stock with an exercise price equal to the fair market value of our common stock
on the grant date and a restricted stock unit, or RSU. The shares subject to each initial option grant and RSU will vest in three successive
equal annual installments over the 3-year period measured from the date of the non-employee director’s election to our Board, subject
to the non-employee director’s continued board service through each vesting date and provided that the director attends at least
75% of the board meetings held during each respective year of board service.
In addition, on the date
of each annual meeting of our stockholders, each non-employee director will automatically be granted equity awards totaling $75,000 in
value split equally between a non-statutory stock option to purchase shares of our common stock on that date with an exercise price equal
to the fair market value of our common stock on the grant date and an RSU. In 2023, because of the limited number of shares of common
stock available for issuance pursuant to the 2023 Plan, each non-employee director received equity awards totaling approximately $6,690
each, which was evenly split into (i) a stock option to purchase 1,065 shares of common stock, which shares will vest on the first
anniversary of the grant date, subject to the non-employee director’s continued board service through such date and provided that
the non-employee director attends at least 75% of the board meetings held during such year of board service, and (ii) 935 RSUs,
which will vest on the first anniversary of the grant date, subject to the non-employee director’s continued board service through
such date and provided that the non-employee director attends at least 75% of the board meetings held during such year of board
service. The decision to establish a value of equity awards and to equally split the awards between stock option shares and RSUs was
based on a review of director equity compensation by our Board that included participation by the Compensation Committee’s independent
compensation advisor, Pay Governance LLC. In making this determination, our Board considered our business plan, our stock price
and potential dilution to stockholders, and the need to adequately compensate our directors for their service.
The shares subject to each
option grant or RSU under the director compensation policy will immediately vest upon (i) an acquisition of the Company by merger
or asset sale, (ii) the successful completion of a tender offer for more than 50% of our outstanding voting stock or (iii) a
change in the majority of our Board effected through one or more proxy contests for board membership, or a Change in Control. All automatic
director options have a maximum term of ten years.
We will also continue to
reimburse our non-employee directors for their reasonable out-of-pocket expenses incurred in attending board and committee meetings.
Director Independence
Our common stock is listed
on Nasdaq. The listing rules of this stock exchange generally require that a majority of the members of a listed company’s
Board be independent. In addition, the rules of Nasdaq require that, subject to specified exceptions, each member of a listed company’s
audit, compensation, and nominating and corporate governance committees be independent. The Nasdaq director independence definition includes
a series of objective tests, such as that the director is not also one of our employees and has not engaged in various types of business
dealings with us. In addition, as further required by Nasdaq rules, our Board has made a subjective determination as to each independent
director that no relationships exist, which, in the opinion of our Board, would interfere with the exercise of independent judgment in
carrying out the responsibilities of a director. In making these determinations, our directors reviewed and discussed information provided
by the directors and us regarding each director’s business and personal activities as they may relate to us and our management.
Our Board has determined
that none of our non-employee directors or director nominees has a relationship that would interfere with the exercise of independent
judgment in carrying out the responsibilities of a director and that each of our current non-employee directors is “independent”
as that term is defined under Nasdaq rules. The independent members of our Board hold separate regularly scheduled executive session
meetings at which only independent directors are present.
Audit committee members must
also satisfy the independence criteria set forth in Rule 10A-3 under the Exchange Act. In order to be considered independent for
purposes of Rule 10A-3, a member of an audit committee of a listed company may not, other than in his or her capacity as a member
of the audit committee, the Board, or any other board committee, accept, directly or indirectly, any consulting, advisory, or other compensatory
fee from the listed company or any of its subsidiaries; or be an affiliated person of the listed company or any of its subsidiaries.
Each of the current members of our audit committee, Sharon Barbari, Seth H.Z. Fischer, and John Hubbard, Ph.D., FCP, qualifies as an
independent director pursuant to Rule 10A-3.
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS
AND MANAGEMENT
The following table sets
forth certain information with respect to the beneficial ownership of our common stock as of December 31, 2023 (except as otherwise noted)
for:
| |
● |
based on reports filed with the SEC, each person, or group of persons,
who beneficially owns more than five percent (5%) of our common stock; |
| |
● |
each of our named executive officers; |
| |
● |
each of our directors; and |
| |
● |
all of our executive officers and directors as a group. |
We have determined beneficial
ownership in accordance with the rules of the SEC. Except as indicated by the footnotes below, we believe, based on the information
furnished to us, that the persons and entities named in the table below have sole voting and investment power with respect to all shares
of common stock that they beneficially own, subject to applicable community property laws.
Applicable percentage
ownership is based on 2,963,657 shares of common stock outstanding at December 31, 2023. In computing the number of shares of common
stock beneficially owned by a person and the percentage ownership of that person, we deemed to be outstanding all shares of common
stock subject to options or warrants held by that person or entity that are currently exercisable or that will become exercisable or
releasable within 60 days of December 31, 2023. We did not deem these shares outstanding, however, for the purpose of computing
the percentage ownership of any other person. Unless otherwise indicated, the address of each beneficial owner listed in the table
below is c/o Agile Therapeutics, Inc., 500 College Road East, Suite 310, Princeton, New Jersey 08540.
| |
|
Number of Shares
Beneficially |
|
|
Percentage of Shares |
|
| Name
of Beneficial Owner(1) |
|
Owned |
|
|
Beneficially Owned |
|
| Named Executive Officers and Directors |
|
|
|
|
|
|
|
|
| Al Altomari (2) |
|
|
3,178 |
|
|
|
0.1 |
% |
| Scott M. Coiante (3) |
|
|
80 |
|
|
|
* |
|
| Geoffrey P. Gilmore (4) |
|
|
851 |
|
|
|
* |
|
| Paul Korner, M.D. (5) |
|
|
270 |
|
|
|
* |
|
| Sharon Barbari (6) |
|
|
90 |
|
|
|
* |
|
| Sandra Carson, M.D., FACOG (7) |
|
|
90 |
|
|
|
* |
|
| Seth H.Z. Fischer (8) |
|
|
160 |
|
|
|
* |
|
| John Hubbard, Ph.D., FCP (9) |
|
|
174 |
|
|
|
* |
|
| Josephine Torrente (10) |
|
|
88 |
|
|
|
* |
|
| |
|
|
|
|
|
|
|
|
| All current executive officers and directors as
a group (11 persons) (11) |
|
|
5,662 |
|
|
|
0.2 |
% |
| * |
Represents beneficial ownership of less than 1% of the outstanding
shares of our common stock. |
| (1) |
This table is based upon information supplied by officers, directors
and stockholders known by us to be beneficial owners of more than five percent of our common stock as well as Schedules 13G or 13D
and Section 16 reports filed with the SEC. We have not independently verified such information. |
| (2) |
Includes (a) 1,286 shares of common stock owned by Mr. Altomari
and (b) 1,892 shares of common stock that Mr. Altomari has the right to acquire from us within 60 days of December 31,
2023. |
| (3) |
Represents 80 shares of common stock owned by Mr. Coiante. |
| (4) |
Includes (a) 30 shares of common stock owned by Mr. Gilmore
and (b) 821 shares of common stock that Mr. Gilmore has the right to acquire from us within 60 days of December 31, 2023. |
| (5) |
Includes (a) zero shares of common stock owned by Dr. Korner
and (b) 270 shares of common stock that Dr. Korner has the right to acquire from us within 60 days of December 31, 2023. |
| (6) |
Includes (a) 38 shares of common stock owned by Ms. Barbari
and (b) 52 shares of common stock that Ms. Barbari has the right to acquire from us within 60 days of December 31, 2023. |
| (7) |
Includes (a) 38 shares of common stock owned by Dr. Carson
and (b) 52 shares of common stock that Dr. Carson has the right to acquire from us within 60 days of December 31, 2023. |
| (8) |
Includes (a) 40 shares of common stock owned by Mr. Fischer
and (b) 120 shares of common stock that Mr. Fischer has the right to acquire from us within 60 days of December 31, 2023. |
| (9) |
Includes (a) 40 shares of common stock owned by Dr. Hubbard
and (b) 134 shares of common stock that Dr. Hubbard has the right to acquire from us within 60 days of December 31,
2023. |
| (10) |
Includes (a) 39 shares of common stock owned by Ms. Torrente
and (b) 49 shares of common stock that Ms. Torrente has the right to acquire from us within 60 days of December 31, 2023. |
| (11) |
Includes (a) 1,632 shares of common stock owned and (b) 4,030 shares
of common stock that these individuals have the right to acquire from us within 60 days of December 31, 2023. |
CERTAIN RELATIONSHIPS AND RELATED PARTY TRANSACTIONS
In addition to the cash and
equity compensation arrangements of our directors and named executive officers discussed under “Director Compensation” and
“Executive Compensation,” the following is a description of transactions since December 31, 2022, to which we have been
a party in which the amount involved exceeded or will exceed $120,000 and in which any of our directors, executive officers, beneficial
holders of more than 5% of our capital stock, or entities affiliated with or immediate family members of any of the foregoing, had or
will have a direct or indirect material interest.
Employment Agreements
We have entered into employment
agreements with certain of our executive officers. For more information regarding these agreements, see the section of this prospectus
entitled “Executive Compensation.”
Equity Awards and Payments to Executive Officers
and Directors
We have granted stock options
to our executive officers and certain of our directors and paid fees to certain of our directors as more fully described in the section
entitled “Director Compensation” and “Executive Compensation.”
Credit Agreement and Guaranty
In February 2020, we
entered into a Credit Agreement and Guaranty with Perceptive Credit Holdings III, LP (“Perceptive”), an affiliate of a beneficial
holder of more than 5% of our capital stock, for a senior secured term loan credit facility of up to $35.0 million (the “Perceptive
Credit Agreement”). A first tranche of $5.0 million was funded on execution of the Perceptive Credit Agreement. A second tranche
of $15.0 million tranche was to be available to us based on the achievement of a revenue milestone by December 31, 2021, which was
not achieved. The other tranches of debt under the Amended Perceptive Credit Agreement are no longer available to us. On January 7,
2022, we prepaid $5.0 million of the outstanding debt, and Perceptive waived the prepayment premium. On July 8, 2022, we prepaid
$5.0 million of the outstanding debt, and Perceptive waived the prepayment premium. On July 25, 2022, we entered into a fifth amendment
to the Amended Perceptive Credit Agreement, as amended (the “Fifth Amendment”). Pursuant to the Fifth Amendment, Perceptive
agreed to release its security interest in certain assets being transferred from us to Corium in connection with an amendment to our
Manufacturing and Commercialization Agreement with Corium and waive our obligations to comply with certain financial covenants through
the end of 2022. In exchange, we agreed to prepay $7.0 million of outstanding principal under the Amended Perceptive Credit Agreement
using the proceeds of recent sales under our ATM program with H.C. Wainwright & Co., LLC. Such payment was made on July 25,
2022. On March 21, 2023, we entered into a sixth amendment to the Amended Perceptive Credit Agreement (the “Sixth Amendment”).
The Sixth Amendment waived the our obligations to (1) comply with certain financial covenants relating to minimum revenue requirements
and minimum liquidity through June 30, 2023, and (2) file financial statements along with our Annual Report on Form 10-K for the fiscal
year ended December 31, 2022 that are not subject to any “going concern” qualification. On October 30, 2023, we entered into
a seventh amendment to the Perceptive Credit Agreement (the “Seventh Amendment”). The Seventh Amendment: (1) amends our obligations
to comply with certain financial covenants relating to minimum revenue requirements, (2) amends and waives our obligations to comply
with certain financial covenants relating to minimum liquidity through December 31, 2023, and (3) requires us to make principal payments
on its outstanding loan balance of $150,000 per month beginning on December 1, 2023.
The facility will mature
on February 10, 2024 (“Maturity Date”). Pursuant to the Amended Perceptive Credit Agreement, beginning August 31,
2022, we began making monthly principal payments in an amount equal to $75,000. Beginning on December 1, 2023 and pursuant to the Seventh
Amendment, we began making monthly payments of $150,000, and will continue to do so until the Maturity Date, at which time all remaining
principal amount outstanding is due.
Borrowings under the Amended
Perceptive Credit Agreement will accrue interest at an annual rate equal to he Secured Overnight Financing Rate, or SOFR, plus 10.25%,
provided that SOFR shall not be less than 1.5%. The rate of interest in effect as of December 31, 2023 was 15.6%. Upon the occurrence
and during the continuance of any event of default under the Fifth Amendment, the interest rate automatically increases by 3.0% per annum.
We may prepay any outstanding loans in whole or in part. Any such prepayment of the loans is subject to a prepayment premium of 2.0%.
All of our obligations under
the Perceptive Credit Agreement are secured by a first-priority lien and security interest in substantially all of our tangible and intangible
assets, including intellectual property. The Perceptive Credit Agreement contains certain representations and warranties, affirmative
covenants, negative covenants and conditions that are customary for similar financings. The negative covenants restrict or limit our
ability to, among other things and subject to certain exceptions contained in the Perceptive Credit Agreement, incur new indebtedness;
create liens on assets; engage in certain fundamental corporate changes, such as mergers or acquisitions, or changes to our business
activities; make certain investments or restricted payments (each as defined in the Perceptive Credit Agreement); change its fiscal year;
pay dividends; repay other certain indebtedness; engage in certain affiliate transactions; or enter into, amend or terminate any other
agreements that have the impact of restricting our ability to make loan repayments under the Perceptive Credit Agreement. In addition,
as amended by the Seventh Amendment, we must (i) at all times for the period from June 30, 2023 to October 31, 2023 maintain a minimum
cash balance of $0.5 million for the period from November 1, 2023 to December 31, 2023 maintain a minimum cash balance of $1.0 million,
and after December 31, 2023 to the Maturity Date maintain a minimum cash balance of $3.0 million; and (ii) as of the last day of each
fiscal quarter commencing with the fiscal quarter ending September 30, 2023, report net revenues for the trailing 12-month period that
are not less than $5.0 million. Pursuant to the Seventh Amendment, we have received a waiver of certain financial covenants through December
31, 2023.
In connection with the Perceptive
Credit Agreement, we issued to Perceptive two warrants to purchase an aggregate of 700 shares of our common stock (together, the “2020
Perceptive Warrants”). The first warrant is exercisable for 350 shares of common stock at an exercise price of $7,480.00 per share.
The second warrant is exercisable for 350 shares of common stock at an exercise price of $9,340.00 per share. The 2020 Perceptive Warrants
expire on February 10, 2027. In connection with the Perceptive Credit Agreement, we issued to Perceptive a warrant to purchase 225
shares of our common stock (the “2021 Perceptive Warrant” and, together with the 2020 Perceptive Warrants, the “Perceptive
Warrants”) at an exercise price of $5,740.00 per share. The 2021 Perceptive Warrant expires on February 26, 2028. The Perceptive
Warrants contain anti-dilution provisions and other warrant holder protections and are not exercisable to the extent that Perceptive
would beneficially own more than 19.99% of the Company’s common stock because of the exercise. In connection with the Sixth Amendment
and Seventh Amendments, we amended and restated the Perceptive Warrants to reset the strike price of the Perceptive Warrants.
Review and Approval of Related Party Transactions
Our Audit Committee Charter
requires that our Audit Committee review and approve or ratify transactions involving us and any executive officer, director, director
nominee, 5% stockholder and certain of their immediate family members, also referred to herein as a related person. The policy and procedures
cover any transaction involving a related person, also referred to herein as a related person transaction, in which the related person
has a material interest, and which does not fall under an explicitly stated exception set forth in the applicable disclosure rules of
the SEC.
A related person transaction
will be considered approved or ratified if it is authorized by the Audit Committee after full disclosure of the related person’s
interest in the transaction. In considering related person transactions, the Audit Committee will consider any information considered
material to investors and the following factors:
| |
● |
the related person’s interest in the transaction; |
| |
● |
the approximate dollar value of the transaction; |
| |
● |
whether the transaction was undertaken in the ordinary course of our
business; |
| |
● |
whether the terms of the transaction are no less favorable to us than
terms that we could have reached with an unrelated third party; and |
| |
● |
the purpose and potential benefit to us of the transaction. |
DESCRIPTION OF CAPITAL STOCK
The following description
is a general summary of the terms of the shares of common stock or shares of preferred stock, does not include all of the terms of the
shares of common stock or shares of preferred stock and should be read together with our Amended and Restated Certificate of Incorporation
and Amended and Restated Bylaws, copies of which have been filed previously with the SEC. For more information on how you can obtain
copies of our Amended and Restated Certificate of Incorporation and Amended and Restated Bylaws, see “Where You Can Find More Information”
on page 69 of this prospectus.
Common Stock
General
Our Amended and Restated
Certificate of Incorporation, as amended, provides the authority to issue 300,000,000 shares of common stock, par value $0.0001 per share.
At December 31, 2023, there were 2,963,657 shares of common stock outstanding. Each share of our common stock has the same relative rights
and is identical in all respects to each other share of our common stock. The rights, preferences and privileges of holders of our common
stock are subject to the rights, preferences and privileges of the holders of shares of any series of preferred stock that we have issued
or may issue in the future.
Voting Rights
The holders of our common
stock are entitled to one vote per share on any matter to be voted upon by our stockholders. Our Amended and Restated Certificate of Incorporation
does not permit cumulative voting in connection with the election of directors.
Dividends
The holders of our common
stock are entitled to dividends, if any, as our Board may declare from time to time from funds legally available for that purpose, subject
to the holders of other classes of stock, if any, at the time outstanding having prior rights as to dividends, if any.
Liquidation Rights
Upon any voluntary or involuntary
liquidation, dissolution, or winding up of our affairs, the holders of our common stock are entitled to share ratably in all assets remaining
after the payment of creditors, subject to any prior liquidation distribution rights of holders of other classes of stock, if any, at
the time outstanding.
Miscellaneous
Holders of our common stock
have no preemptive, conversion, redemption or sinking fund rights. The outstanding shares of our common stock are, and the shares of common
stock to be offered hereby when issued will be, validly issued, fully paid and non-assessable.
Nasdaq Listing
Our common stock is listed
on the Nasdaq Capital Market under the symbol “AGRX.”
Transfer Agent and Registrar
The transfer agent and registrar
for our common stock is Broadridge Corporate Issuer Solutions, Inc. whose address is 51 Mercedes Way, Edgewood, New York 11717.
Preferred Stock
Our Amended and Restated Certificate
of Incorporation, as amended, authorizes the issuance of up to 10,000,000 shares of preferred stock, par value $0.0001 per share, none
of which are issued and outstanding as of the date of this prospectus. We may issue, from time to time in one or more series, the terms
of which may be determined at the time of issuance by our Board, without further action by our stockholders, shares of preferred stock
and such shares may include voting rights, preferences as to dividends and liquidation, conversion rights, redemption rights and sinking
fund provisions. The shares of each series of preferred stock shall have preferences, limitations and relative rights, including voting
rights, identical with those of other shares of the same series and, except to the extent provided in the description of such series,
of those of other series of preferred stock.
Delaware Law and Certain Amended and Restated Certificate of Incorporation
and Amended and Restated Bylaws Provisions
Our Amended and Restated Certificate
of Incorporation, as amended, and Amended and Restated Bylaws, as amended, to be in effect upon completion of this offering contain provisions
that could delay or prevent a change of control of our company or changes in our Board that our stockholders might consider favorable.
Some of these provisions:
| |
· |
Authorize the issuance of preferred stock which can be created and issued by the Board without prior stockholder approval, with rights senior to those of our common stock; |
| |
· |
Provide for a classified board of directors, with each director serving a staggered three-year term; |
| |
· |
Prohibit our stockholders from filling board vacancies, calling special stockholder meetings or taking action by written consent; |
| |
· |
Provide for the removal of a director only with cause and by the affirmative vote of the holders of 75% or more of the shares then entitled to vote at an election of our directors; |
| |
· |
Require advance written notice of stockholder proposals and director nominations; and |
| |
· |
Require any action instituted against our officers or directors in connection with their service to the Company to be brought in the state of Delaware. |
In addition, we are subject
to the provisions of Section 203 of the Delaware General Corporation Law, which may prohibit certain business combinations with stockholders
owning 15% or more of our outstanding voting stock. These and other provisions in our Amended and Restated Certificate of Incorporation,
as amended, Amended and Restated Bylaws, as amended, and Delaware law could make it more difficult for stockholders or potential acquirors
to obtain control of our Board or initiate actions that are opposed by our then-current board of directors, including a merger, tender
offer or proxy contest involving our company. This provision could have the effect of delaying or preventing a change of control, whether
or not it is desired by or beneficial to our stockholders. Any delay or prevention of a change of control transaction or changes in our
Board could cause the market price of our common stock to decline.
Indemnification
Our Amended and Restated Certificate
of Incorporation, as amended, contains provisions permitted under the General Corporation Law of Delaware relating to the liability of
directors. The provisions eliminate, to the extent legally permissible, a director’s liability for monetary damages for a breach
of fiduciary duty, except in circumstances involving wrongful acts, such as the breach of a director’s duty of loyalty or acts or
omissions that involve intentional misconduct or a knowing violation of law. The limitation of liability described above does not alter
the liability of our directors and officers under federal securities laws. Furthermore, our Amended and Restated Certificate of Incorporation,
as amended, contains provisions to indemnify our directors and officers to the fullest extent permitted by the General Corporation Law
of Delaware. These provisions do not limit or eliminate our right or the right of any stockholder of ours to seek non-monetary relief,
such as an injunction or rescission in the event of a breach by a director or an officer of his or her duty of care to us. We believe
that these provisions assist us in attracting and retaining qualified individuals to serve as directors.
DESCRIPTION OF SECURITIES WE
ARE OFFERING
We are offering up to
shares of common stock, together with Series E-1 warrants to purchase up to
shares of common stock (the “Series E-1 warrants”) and Series E-2 warrants to purchase up to
shares of common stock (the “Series E-2 warrants”, and together with the Series E-1 warrants, the “Series E
warrants”). We are also offering up to pre-funded warrants (the
“Series F pre-funded warrants” and collectively with the Series E warrants, the “warrants”) to those
purchasers, whose purchase of shares of common stock in this offering would result in the purchaser, together with its affiliates and
certain related parties, beneficially owning more than 4.99% (or, at the election of the purchaser, 9.99%) of our outstanding common
stock following the consummation of this offering in lieu of the shares of our common stock that would result in ownership in excess
of 4.99% (or, at the election of the purchaser, 9.99%). Each Series F pre-funded warrant will be exercisable for one share of common
stock. Each Series F pre-funded warrant is being issued together with the same Series E warrants described above being issued
with each share common stock. The shares of common stock or Series F pre-funded warrants, as the case may be, and the Series E
warrants, can only be purchased together in this offering, but the Series F pre-funded warrants and Series E warrants are immediately
separable and will be issued separately in this offering. We are also registering the shares of common stock and the shares of common
stock issuable from time to time upon exercise of the Series F pre-funded warrants and Series E warrants offered hereby.
Common Stock
The description of our common
stock under the section “Description of Our Capital Stock” in this prospectus is incorporated herein by reference.
Series F Pre-Funded Warrants
The following summary of certain
terms and provisions of the Series F pre-funded warrants that are being issued hereby is not complete and is subject to, and qualified
in its entirety by, the provisions of the Series F pre-funded warrant, the form of which will be filed as an exhibit to the registration
statement of which this prospectus forms a part. Prospective investors should carefully review the terms and provisions of the form of
Series F pre-funded warrant for a complete description of the terms and conditions of the Series F pre-funded warrants.
Duration and Exercise Price
Each Series F pre-funded
warrant offered hereby will have an initial exercise price per share equal to $0.0001. The Series F pre-funded warrants will be immediately
exercisable and may be exercised at any time until all of the pre-funded warrants are exercised in full. The exercise price and number
of shares of common stock issuable upon exercise is subject to appropriate adjustment in the event of stock dividends, stock splits, reorganizations
or similar events affecting our common stock and the exercise price. The Series F pre-funded warrants will be issued separately from
the accompanying Series E warrants with Series F pre-funded warrants.
Exercisability
The Series F pre-funded
warrants will be exercisable, at the option of each holder, in whole or in part, by delivering to us a duly executed exercise notice accompanied
by payment in full for the number of shares of our common stock purchased upon such exercise (except in the case of a cashless exercise
as discussed below). A holder (together with its affiliates) may not exercise any portion of the Series F pre-funded warrant to the
extent that the holder would own more than 4.99% of the outstanding common stock immediately after exercise, except that upon at least
61 days’ prior notice from the holder to us, the holder may increase the amount of beneficial ownership of outstanding stock after
exercising the holder’s Series F pre-funded warrants up to 9.99% of the number of shares of our common stock outstanding immediately
after giving effect to the exercise, as such percentage ownership is determined in accordance with the terms of the Series F pre-funded
warrants and in accordance with the rules and regulations of the SEC. Purchasers of Series F pre-funded warrants in this offering
may also elect prior to the issuance of the Series F pre-funded warrants to have the initial exercise limitation set at 9.99% of
our outstanding common stock.
Cashless Exercise
In lieu of making the cash
payment otherwise contemplated to be made to us upon such exercise in payment of the aggregate exercise price, the holder may elect instead
to receive upon such exercise (either in whole or in part) the net number of shares of common stock determined according to a formula
set forth in the Series F pre-funded warrants.
Transferability
Subject to applicable laws,
a Series F pre-funded warrant may be transferred at the option of the holder upon surrender of the Series F pre-funded warrant
to us together with the appropriate instruments of transfer.
Fractional Shares
No fractional shares of common
stock will be issued upon the exercise of the Series F pre-funded warrants. Rather, the number of shares of common stock to be issued
will be rounded to the nearest whole number.
Trading Market
There is no trading market
available for the Series F pre-funded warrants on any securities exchange or nationally recognized trading system, and we do not
expect a trading market to develop. We do not intend to list the Series F pre-funded warrants on any securities exchange or other
trading market. Without a trading market, the liquidity of the Series F pre-funded warrants will be extremely limited. The common
stock issuable upon exercise of the Series F pre-funded warrants is currently listed on the Nasdaq Capital Market.
Right as a Stockholder
Except as otherwise provided
in the Series F pre-funded warrants or by virtue of such holder’s ownership of shares of our common stock, the holders of the
Series F pre-funded warrants do not have the rights or privileges of holders of our common stock, including any voting rights, until
they exercise their Series F pre-funded warrants. The Series F pre-funded warrants will provide that holders have the right
to participate in distributions or dividends paid on our common stock.
Fundamental Transaction
In the event of a fundamental
transaction, as described in the Series F pre-funded warrants and generally including any reorganization, recapitalization or reclassification
of our common stock, the sale, transfer or other disposition of all or substantially all of our properties or assets, our consolidation
or merger with or into another person, the acquisition of more than 50% of our outstanding common stock, or any person or group becoming
the beneficial owner of more than 50% of the voting power represented by our outstanding common stock, the holders of the Series F
pre-funded warrants will be entitled to receive upon exercise of the Series F pre-funded warrants the kind and amount of securities,
cash or other property that the holders would have received had they exercised the Series F pre-funded warrants immediately prior
to such fundamental transaction.
Series E Warrants
The following summary of certain
terms and provisions of the Series E warrants included with the shares of common stock and the Series F pre-funded warrants
that are being issued hereby is not complete and is subject to, and qualified in its entirety by, the provisions of the Series E
warrants, the forms of which will be filed as an exhibit to the registration statement of which this prospectus forms a part. Prospective
investors should carefully review the terms and provisions of the forms of Series E warrants for a complete description of the terms
and conditions of the Series E warrants. The Series E-1 warrants and Series E-2 warrants have the same terms and provisions,
other than the exercise period of each, as described below.
Duration and Exercise Price
Each Series E warrant
offered hereby will have an initial exercise price equal to $ per share of
common stock. The Series E-1 warrants will be exercisable beginning on the Initial Exercise Date and will expire five years from
the Initial Exercise Date. The Series E-2 warrants will be exercisable beginning on the Initial Exercise Date and will expire eighteen
months from the Initial Exercise Date. The exercise price and number of shares of common stock issuable upon exercise is subject to appropriate
adjustment in the event of stock dividends, stock splits, reorganizations or similar events affecting our common stock and the exercise
price. The Series E warrants will be issued separately from the common stock, or the Series F pre-funded warrants, as the case
may be.
We intend to promptly, and
in no event later than 90 days after the consummation of this offering, seek stockholder approval for the issuance of shares of common
stock issuable upon exercise of the Series E warrants but we cannot assure you that such stockholder approval will be obtained, provided,
however, that, if and only if the Pricing Conditions are met, then we will not seek Warrant Stockholder Approval. We have agreed with
the investors in this offering that, if we do not obtain stockholder approval for the issuance of the shares of common stock upon exercise
of the warrants at the first stockholder meeting for such purpose after this offering, we will call a stockholder meeting every 90 days
thereafter until the earlier of the date we obtain such approval or the warrants are no longer outstanding, provided, however, that, if
and only if the Pricing Conditions are met, then we will not seek Warrant Stockholder Approval.
Exercisability
The Series E warrants
will be exercisable, at the option of each holder, in whole or in part, by delivering to us a duly executed exercise notice accompanied
by payment in full for the number of shares of our common stock purchased upon such exercise (except in the case of a cashless exercise
as discussed below). A holder (together with its affiliates) may not exercise any portion of the Series E warrant to the extent that
the holder would own more than 4.99% of the outstanding common stock immediately after exercise, except that upon at least 61 days’
prior notice from the holder to us, the holder may increase the amount of beneficial ownership of outstanding stock after exercising the
holder’s Series E warrants up to 9.99% of the number of shares of our common stock outstanding immediately after giving effect
to the exercise, as such percentage ownership is determined in accordance with the terms of the Series E warrants and in accordance
with the rules and regulations of the SEC.
Cashless Exercise
If, at the time a holder exercises
its Series E warrants, a registration statement registering the issuance of the shares of common stock underlying the Series E
warrants under the Securities Act is not then effective or available for the issuance of such shares, then in lieu of making the cash
payment otherwise contemplated to be made to us upon such exercise in payment of the aggregate exercise price, the holder may elect instead
to receive upon such exercise (either in whole or in part) the net number of shares of common stock determined according to a formula
set forth in the Series E warrants.
Fractional Shares
No fractional shares of common
stock will be issued upon the exercise of the Series E warrants. Rather, the number of shares of common stock to be issued will be
rounded to the nearest whole number.
Transferability
Subject to applicable laws,
a Series E warrant may be transferred at the option of the holder upon surrender of the Series E warrant to us together with
the appropriate instruments of transfer.
Trading Market
There is no trading market
available for the Series E warrants on any securities exchange or nationally recognized trading system, and we do not expect a trading
market to develop. We do not intend to list the Series E warrants on any securities exchange or other trading market. Without a trading
market, the liquidity of the Series E warrants will be extremely limited. The common stock issuable upon exercise of the Series E
warrants is currently listed on the Nasdaq Capital Market.
Right as a Stockholder
Except as otherwise provided
in the Series E warrants or by virtue of such holder’s ownership of shares of our common stock, the holders of the Series E
warrants do not have the rights or privileges of holders of our common stock, including any voting rights, until they exercise their Series E
warrants.
Fundamental Transaction
In the event of a fundamental
transaction, as described in the Series E warrants and generally including any reorganization, recapitalization or reclassification
of our common stock, the sale, transfer or other disposition of all or substantially all of our properties or assets, our consolidation
or merger with or into another person, the acquisition of more than 50% of our outstanding common stock, or any person or group becoming
the beneficial owner of more than 50% of the voting power represented by our outstanding common stock, the holders of the Series E
warrants will be entitled to receive upon exercise of the Series E warrants the kind and amount of securities, cash or other property
that the holders would have received had they exercised the Series E warrants immediately prior to such fundamental transaction.
In addition, in the event of a fundamental transaction which is approved by our Board, the holders of the Series E warrants have
the right to require us or a successor entity to redeem the Series E warrant for cash in the amount of the Black-Scholes value of
the unexercised portion of the Series E warrant on the date of the consummation of the fundamental transaction. In the event of a
fundamental transaction which is not approved by our Board, the holders of the Series E warrants have the right to require us or
a successor entity to redeem the Series E warrant for the consideration paid in the fundamental transaction in the amount of the
Black Scholes value of the unexercised portion of the Series E warrant on the date of the consummation of the fundamental transaction.
Placement Agent Warrants
The following summary of certain
terms and provisions of the Placement Agent Warrants that are being issued hereby is not complete and is subject to, and qualified in
its entirety by, the provisions of the Placement Agent Warrants, the form of which will be filed as an exhibit to the registration statement
of which this prospectus forms a part. Prospective investors should carefully review the terms and provisions of the form of Placement
Agent Warrant for a complete description of the terms and conditions of the Placement Agent Warrant.
Duration and Exercise Price
Each Placement Agent Warrant
offered hereby will have an initial exercise price equal to $ per share of
common stock (125% of the combined public offering price per share of common stock and Series E warrants). The Placement Agent Warrants
will be exercisable beginning on the Initial Exercise Date and will expire five years from the commencement of sales in this offering.
The exercise price and number of shares of common stock issuable upon exercise is subject to appropriate adjustment in the event of stock
dividends, stock splits, reorganizations or similar events affecting our common stock and the exercise price.
Exercisability
The Placement Agent Warrants
will be exercisable, at the option of each holder, in whole or in part, by delivering to us a duly executed exercise notice accompanied
by payment in full for the number of shares of our common stock purchased upon such exercise (except in the case of a cashless exercise
as discussed below). A holder (together with its affiliates) may not exercise any portion of the Placement Agent Warrant to the extent
that the holder would own more than 4.99% of the outstanding common stock immediately after exercise, except that upon at least 61 days’
prior notice from the holder to us, the holder may increase the amount of beneficial ownership of outstanding stock after exercising the
holder’s Placement Agent Warrant up to 9.99% of the number of shares of our common stock outstanding immediately after giving effect
to the exercise, as such percentage ownership is determined in accordance with the terms of the Placement Agent Warrants and in accordance
with the rules and regulations of the SEC.
Cashless Exercise
If, at the time a holder exercises
its Placement Agent Warrants, a registration statement registering the issuance of the shares of common stock underlying the Placement
Agent Warrants under the Securities Act is not then effective or available for the issuance of such shares, then in lieu of making the
cash payment otherwise contemplated to be made to us upon such exercise in payment of the aggregate exercise price, the holder may elect
instead to receive upon such exercise (either in whole or in part) the net number of shares of common stock determined according to a
formula set forth in the Placement Agent Warrants.
Fractional Shares
No fractional shares of common
stock will be issued upon the exercise of the Placement Agent Warrants. Rather, the number of shares of common stock to be issued will
be rounded to the nearest whole number.
Transferability
Subject to applicable laws,
a Placement Agent Warrant may be transferred at the option of the holder upon surrender of the Placement Agent Warrant to us together
with the appropriate instruments of transfer.
Trading Market
There is no trading market
available for the Placement Agent Warrants on any securities exchange or nationally recognized trading system, and we do not expect a
trading market to develop. We do not intend to list the Placement Agent Warrants on any securities exchange or other trading market. Without
a trading market, the liquidity of the Placement Agent Warrants will be extremely limited. The common stock issuable upon exercise of
the Placement Agent Warrants is currently listed on the Nasdaq Capital Market.
Right as a Stockholder
Except as otherwise provided
in the Placement Agent Warrants or by virtue of such holder’s ownership of shares of our common stock, the holders of the Placement
Agent Warrants do not have the rights or privileges of holders of our common stock, including any voting rights, until they exercise their
Placement Agent Warrants.
Fundamental Transaction
In the event of a fundamental
transaction, as described in the Placement Agent Warrants and generally including any reorganization, recapitalization or reclassification
of our common stock, the sale, transfer or other disposition of all or substantially all of our properties or assets, our consolidation
or merger with or into another person, the acquisition of more than 50% of our outstanding common stock, or any person or group becoming
the beneficial owner of more than 50% of the voting power represented by our outstanding common stock, the holders of the Placement Agent
Warrants will be entitled to receive upon exercise of the Placement Agent Warrants the kind and amount of securities, cash or other property
that the holders would have received had they exercised the Placement Agent Warrants immediately prior to such fundamental transaction.
In addition, in the event of a fundamental transaction which is approved by our Board, the holders of the Placement Agent Warrants have
the right to require us or a successor entity to redeem the Placement Agent Warrant for cash in the amount of the Black-Scholes value
of the unexercised portion of the Placement Agent Warrant on the date of the consummation of the fundamental transaction. In the event
of a fundamental transaction which is not approved by our Board, the holders of the Placement Agent Warrants have the right to require
us or a successor entity to redeem the Placement Agent Warrants for the consideration paid in the fundamental transaction in the amount
of the Black Scholes value of the unexercised portion of the Placement Agent Warrant on the date of the consummation of the fundamental
transaction.
MATERIAL U.S. FEDERAL TAX CONSIDERATIONS
FOR HOLDERS OF OUR COMMON STOCK, SERIES F PRE-FUNDED WARRANTS AND SERIES E WARRANTS
The following discussion describes
certain material U.S. federal income tax consequences of the acquisition, ownership and disposition of our common stock, Series F
pre-funded warrants and Series E warrants acquired in this offering. This discussion is based on the current provisions of the Internal
Revenue Code of 1986, as amended (referred to as the “Code”), existing and proposed U.S. Treasury regulations promulgated
thereunder, and administrative rulings and court decisions in effect as of the date hereof, all of which are subject to change at any
time, possibly with retroactive effect. No ruling has been or will be sought from the Internal Revenue Service, or IRS, with respect to
the matters discussed below, and there can be no assurance the IRS will not take a contrary position regarding the tax consequences of
the acquisition, ownership or disposition of our common stock, Series F pre-funded warrants or Series E warrants, or that any
such contrary position would not be sustained by a court.
We assume in this discussion
that the shares of our common stock, Series F pre-funded warrants and Series E warrants will be held as capital assets (generally,
property held for investment). This discussion does not address all aspects of U.S. federal income taxes, does not discuss the potential
application of the Medicare contribution tax and does not deal with state or local taxes or U.S. federal gift and estate tax laws, or
any non-U.S. tax consequences that may be relevant to holders in light of their particular circumstances. This discussion also does not
address the special tax rules applicable to particular holders, such as:
| |
· |
a bank, insurance company, or other financial institution; |
| |
· |
a tax-exempt entity, organization, or arrangement; |
| |
· |
a government or any agency, instrumentality, or controlled entity thereof; |
| |
· |
a real estate investment trust; |
| |
· |
an S corporation or other pass-through entity (or an investor in an S corporation or other pass-through entity); |
| |
· |
a regulated investment company; |
| |
· |
a “controlled foreign corporation” or a “passive foreign investment company”; |
| |
· |
a dealer or broker in stocks and securities, or currencies; |
| |
· |
a trader in securities that elects mark-to-market treatment or any other holder subject to mark-to-market treatment; |
| |
· |
a holder of our common stock, Series F pre-funded warrants, or Series E warrants that is liable for the alternative minimum tax; |
| |
· |
a holder of our common stock, Series F pre-funded warrants, or Series E warrants that received such security through the exercise of options, warrants, or similar derivative securities or otherwise as compensation; |
| |
· |
a holder of our common stock, Series F pre-funded warrants, or Series E warrants that holds such security in a tax-deferred account (such as an individual retirement account or a plan qualifying under Section 401(k) of the Code); |
| |
· |
a holder of our common stock, Series F pre-funded warrants, or Series E warrants that has a functional currency other than the U.S. dollar; |
| |
· |
a holder of our common stock, Series F pre-funded warrants, or Series E warrants that holds such security as part of a hedge, straddle, constructive sale, conversion or other integrated transaction; |
| |
· |
a holder of our common stock, Series F pre-funded warrants, or Series E warrants required to accelerate the recognition of any item of gross income with respect to such security, as a result of such income being recognized on an applicable financial statement; |
| |
· |
a holder of our common stock, Series F pre-funded warrants, or Series E warrants that is a U.S. expatriate or former citizen or long-term resident of the United States; |
| |
· |
a holder of our common stock, Series F pre-funded warrants, or Series E warrants that does not hold such security as a capital asset within the meaning of Section 1221 of the Code (generally, for investment purposes); |
| |
· |
a holder of our common stock, Series F pre-funded warrants, or Series E warrants whose security may constitute “qualified small business stock” under Section 1202 of the Code or “Section 1244 stock” for purposes of Section 1244 of the Code; or |
| |
· |
a holder of our common stock, Series F pre-funded warrants, or Series E warrants that acquired such security in a transaction subject to the gain rollover provisions of Section 1045 of the Code; |
In addition, this discussion
does not address the tax treatment of partnerships or other pass-through entities or of persons who hold our common stock, Series F
pre-funded warrants or Series E warrants through partnerships or other entities which are pass-through entities for U.S. federal
income tax purposes. A partner in a partnership or other pass-through entity that will hold our common stock, Series F pre-funded
warrants or Series E warrants should consult his, her or its own tax advisor regarding the tax consequences of the ownership and
disposition of our common stock, Series F pre-funded warrants or Series E warrants through a partnership or other pass-through
entity, as applicable.
The discussion of U.S.
federal income tax considerations is for information purposes only and is not tax advice. Investors should consult their own tax advisors
regarding the U.S. federal, state, local and non-U.S. income and other tax considerations of acquiring, holding and disposing of our common
stock, Series F pre-funded warrants and Series E warrants.
For the purposes of this discussion,
a “U.S. Holder” means a beneficial owner of our common stock, Series F pre-funded warrants or Series E warrants
that is for U.S. federal income tax purposes (a) an individual citizen or resident of the United States, (b) a corporation (or
other entity treated as a corporation for U.S. federal income tax purposes), organized in or under the laws of the United States, any
state thereof or the District of Columbia, (c) an estate the income of which is includable in gross income for U.S. federal income
tax purposes regardless of its source, or (d) a trust if it (1) is subject to the primary supervision of a court within the
United States and one or more U.S. persons (within the meaning of Section 7701(a)(30) of the Code) has the authority to control all
substantial decisions of the trust or (2) has a valid election in effect under applicable U.S. Treasury regulations to be treated
as a U.S. person. A “Non-U.S. Holder” is, for U.S. federal income tax purposes, a beneficial owner of common stock, Series F
pre-funded warrants or Series E warrants (other than a partnership or other entity or arrangement classified as a partnership for
U.S. federal income tax purposes) that is not a U.S. Holder.
Treatment of Series F Pre-Funded Warrants
Although it is not entirely
free from doubt, we believe a Series F pre-funded warrant should be treated as a share of our common stock for U.S. federal income
tax purposes and a holder of Series F pre-funded warrants should generally be taxed in the same manner as a holder of common stock
as described below. Accordingly, for U.S. federal income tax purposes, no gain or loss should be recognized upon the exercise of a Series F
pre-funded warrant, and upon exercise, the holding period of the share of common stock received should include the holding period of the
Series F pre-funded warrant. Similarly, the tax basis of a share of common stock received upon exercise of a Series F pre-funded
warrant should include the tax basis of the Series F pre-funded warrant (discussed below) increased by the exercise price of $0.0001.
The balance of this discussion generally assumes that the characterization described above is respected for U.S. federal income tax purposes.
Allocation of Purchase Price to Common Stock,
Series F Pre-Funded Warrants and Series E Warrants
For U.S. federal income tax
purposes, a holder’s acquisition of the Series E warrants and common stock or Series F pre-funded warrants, as applicable,
will be treated as the acquisition of an “investment unit” consisting of one share of common stock (or one Series F pre-funded
warrant, as applicable) and a Series E-1 warrant and a Series E-2 warrant, each to acquire one share of our common stock, subject
to adjustment. The purchase price for each investment unit will be allocated between these two components in proportion to their relative
fair market values at the time the investment unit is purchased by the holder. This allocation of the purchase price for each investment
unit will establish the holder’s initial tax basis for U.S. federal income tax purposes in the common stock (or Series F pre-funded
warrant, as applicable) and the Series E warrants included in each investment unit. The separability of the share of common stock
(or Series F pre-funded warrant, as applicable) and the Series E warrants included in each investment unit should not in itself
result in the recognition of income or gain for U.S. federal income tax purposes. Each holder should consult his, her or its own tax advisor
regarding risks associated with the acquisition of an investment unit pursuant to this offering (including potential alternative characterizations
and the allocation of purchase price for an investment unit).
Tax Considerations Applicable to U.S. Holders
Exercise and Expiration of Series E Warrants
In general, a U.S. Holder
will not recognize gain or loss for U.S. federal income tax purposes upon exercise of a Series E warrant. The U.S. Holder will take
a tax basis in the shares acquired on the exercise of a Series E warrant equal to the exercise price of the Series E warrant,
increased by the U.S. Holder’s adjusted tax basis in the Series E warrant exercised (as determined pursuant to the rules discussed
above). The U.S. Holder’s holding period in the shares of our common stock acquired on exercise of the Series E warrant will
begin on the date of exercise of the Series E warrant, and will not include any period for which the U.S. Holder held the Series E
warrant.
In certain limited circumstances,
a U.S. Holder may be permitted to undertake a cashless exercise of Series E warrants into our common stock. The U.S. federal income
tax treatment of a cashless exercise of Series E warrants into our common stock is unclear, and the tax consequences of a cashless
exercise could differ from the consequences upon the exercise of a Series E warrant described in the preceding paragraph. U.S. Holders
should consult their own tax advisors regarding the U.S. federal income tax consequences of a cashless exercise of Series E warrants.
The lapse or expiration of
a Series E warrant will be treated as if the U.S. Holder sold or exchanged the Series E warrant and recognized a capital loss
equal to the U.S. Holder’s tax basis in the Series E warrant. The deductibility of capital losses is subject to limitations.
Certain Adjustments to and Distributions on
the Series E Warrants
Under Section 305 of
the Code, an adjustment to the number of shares of common stock issued on the exercise of the Series E warrants, or an adjustment
to the exercise price of the Series E warrants, may be treated as a constructive distribution to a U.S. Holder of the Series E
warrants if, and to the extent that, such adjustment has the effect of increasing such U.S. Holder’s proportionate interest in our
“earnings and profits” or assets, depending on the circumstances of such adjustment (for example, if such adjustment is to
compensate for a distribution of cash or other property to our shareholders). Any such constructive distribution would be taxable whether
or not there is an actual distribution of cash or other property. In addition, if we were to make a distribution in cash or other property
with respect to our common stock after the issuance of the Series E warrants, then we may, in certain circumstances, make a corresponding
distribution to a Series E warrant holder. The taxation of a distribution received with respect to a Series E warrant is unclear.
It is possible such a distribution would be treated as a distribution (or constructive distribution), although other treatments are possible.
For more information regarding the tax considerations related to distributions, see the discussion below regarding “Distributions.”
U.S. Holders should consult their tax advisors regarding the proper treatment of any adjustments to and distributions on the Series E
warrants.
Distributions
As discussed above in the
section captioned “Dividend Policy”, we currently anticipate that we will retain all available funds and any future earnings
for use in the operation of our business and do not anticipate declaring or paying any cash dividends on our common stock for the foreseeable
future. In the event that we do make distributions on our common stock or Series F pre-funded warrants to a U.S. Holder, those distributions
generally will constitute dividends for U.S. tax purposes to the extent paid out of our current or accumulated earnings and profits (as
determined under U.S. federal income tax principles). Distributions to a U.S. Holder that are not derived from our current or accumulated
earnings and profits will constitute a return of capital that is applied against and reduces, but not below zero, the U.S. Holder’s
adjusted tax basis in our common stock or Series F pre-funded warrant, as applicable, and to the extent in excess of such basis,
will be treated as gain realized on the sale or exchange of our common stock or Series F pre-funded warrants, as applicable, as described
below under the section titled “Disposition of Our Common Stock, Series F Pre-Funded Warrants or Series E Warrants.”
Disposition of Our Common Stock, Series F Pre-Funded Warrants or
Series E Warrants
Upon a sale or other taxable disposition of our common stock, Series F pre-funded warrants or Series E warrants, a U.S. Holder generally
will recognize capital gain or loss in an amount equal to the difference between the amount realized and the U.S. Holder’s adjusted
tax basis in the applicable common stock, Series F pre-funded warrants or Series E warrants. Capital gain or loss will constitute long-term
capital gain or loss if the U.S. Holder’s holding period for the applicable common stock, Series F pre-funded warrant or Series
E warrant exceeds one year. The deductibility of capital losses is subject to certain limitations. U.S. Holders who recognize losses with
respect to a disposition of our common stock, Series F pre-funded warrants or Series E warrants should consult their own tax advisors
regarding the tax treatment of such losses.
Information Reporting and Backup Withholding
Information reporting requirements
generally will apply to payments of dividends (including constructive dividends) on our common stock, Series F pre-funded warrants
and Series E warrants and to the proceeds of a sale or other disposition of common stock, Series E warrants and Series F
pre-funded warrants by a U.S. Holder unless such U.S. Holder is an exempt recipient, such as a corporation. Backup withholding will apply
to those payments if the U.S. Holder fails to provide the holder’s taxpayer identification number, or certification of exempt status,
or if the holder otherwise fails to comply with applicable requirements to establish an exemption. Backup withholding is not an additional
tax. Rather, amounts withheld as backup withholding may be credited against a person’s U.S. federal income tax liability, and a
holder generally may obtain a refund of any excess amounts withheld under the backup withholding rules by timely filing the appropriate
claim for refund with the IRS and furnishing any required information.
Tax Considerations Applicable to Non-U.S. Holders
Exercise and Expiration of Series E Warrants
In general, a Non-U.S. Holder
will not be subject to U.S. federal income tax on the exercise of the Series E warrants into shares of common stock. As described
under “U.S. Holders — Exercise and Expiration of Series E Warrants,” the U.S. federal income tax
treatment of a cashless exercise of Series E warrants into our common stock is unclear. A Non-U.S. Holder should consult his, her,
or its own tax advisor regarding the U.S. federal income tax consequences of a cashless exercise of Series E warrants.
The expiration of a Series E
warrant will be treated as if the Non-U.S. Holder sold or exchanged the Series E warrant and recognized a capital loss equal to the
Non-U.S. Holder’s tax basis in the Series E warrant. However, a Non-U.S. Holder will not be able to utilize a loss recognized
upon expiration of a Series E warrant against the Non-U.S. Holder’s U.S. federal income tax liability unless the loss is effectively
connected with the Non-U.S. Holder’s conduct of a trade or business within the United States (and, if an income tax treaty applies,
is attributable to a permanent establishment or fixed base in the United States) or is treated as a U.S.-source loss and the Non-U.S.
Holder is an individual nonresident and present 183 days or more in the taxable year of disposition in the United States and certain other
conditions are met.
Certain Adjustments to and Distributions on
the Series E Warrants
As described under “U.S.
Holders — Certain Adjustments to and Distributions on the Series E Warrants,” an adjustment to the Series E
warrants could result in a constructive distribution to a Non-U.S. Holder, which would be treated as described under “Distributions”
below, and the tax treatment of a distribution on a Series E warrant is unclear. Any resulting withholding tax attributable to deemed
dividends would be collected from other amounts payable or distributable to the Non-U.S. Holder. Non-U.S. Holders should consult their
tax advisors regarding the proper treatment of any adjustments to or distributions on the Series E warrants.
Distributions
As discussed above in the
section captioned “Dividend Policy”, we currently anticipate that we will retain all available funds and any future earnings
for use in the operation of our business and do not anticipate declaring or paying any cash dividends on our common stock for the foreseeable
future. In the event that we do make distributions on our common stock or Series F pre-funded warrants to a Non-U.S. Holder, those
distributions generally will be treated as dividends, as return of capital or as gain on the sale or exchange of common stock or Series F
pre-funded warrants for U.S. federal income tax purposes as described in “U.S. Holders — Distributions.”
Subject to the discussions
below under the sections titled “Information Reporting and Backup Withholding” and “Foreign Accounts,” any distribution
(including constructive distributions) on our common stock or Series F pre-funded warrants that is treated as a dividend paid to
a Non-U.S. Holder that is not effectively connected with the holder’s conduct of a trade or business in the United States will generally
be subject to withholding tax at a 30% rate or such lower rate as may be specified by an applicable income tax treaty between the United
States and the Non-U.S. Holder’s country of residence. To obtain a reduced rate of withholding under a treaty, a Non-U.S. Holder
generally will be required to provide the applicable withholding agent with a properly executed IRS Form W-8BEN, IRS Form W-8BEN-E
or other appropriate form, certifying the Non-U.S. Holder’s entitlement to benefits under that treaty. Such form must be provided
prior to the payment of dividends and must be updated periodically and when otherwise required by law.
We generally are not required
to withhold tax on dividends paid (or constructive dividends deemed paid) to a Non-U.S. Holder that are effectively connected with such
holder’s conduct of a trade or business within the United States (and, if required by an applicable income tax treaty, are attributable
to a permanent establishment or fixed base that the holder maintains in the United States) if a properly executed IRS Form W-8ECI,
stating that the dividends are so connected, is furnished to us. In general, such effectively connected dividends will be subject to U.S.
federal income tax on a net income basis at the regular rates applicable to U.S. persons. A corporate Non-U.S. Holder receiving effectively
connected dividends may also be subject to an additional “branch profits tax,” which is imposed, under certain circumstances,
at a rate of 30% (or such lower rate as may be specified by an applicable treaty) on the corporate Non-U.S. Holder’s effectively
connected earnings and profits, subject to certain adjustments.
Distributions to a Non-U.S.
Holder that are not derived from our current or accumulated earnings and profits generally will be treated as a return of capital that
will be applied against and reduce (but not below zero) the Non-U.S. Holder’s basis in its common stock or Series F pre-funded
warrants, as applicable, and to the extent in excess of such basis, will be treated as gain from the sale or exchange of such common stock
or Series F pre-funded warrants, as applicable, as described under “Disposition of Our Common Stock, Series F Pre-Funded
Warrants or Series E Warrants” below.
If a Non-U.S. Holder holds
stock through a financial institution or other agent acting on the holder’s behalf, the holder will be required to provide appropriate
documentation to such agent. The holder’s agent may then be required to provide certification to the applicable withholding agent,
either directly or through other intermediaries.
Disposition of Our Common
Stock, Series F Pre-Funded Warrants or Series E Warrants
Subject to the discussions
below under the sections titled “Information Reporting and Backup Withholding” and “Foreign Accounts,” a Non-U.S.
Holder generally will not be subject to U.S. federal income or withholding tax with respect to gain realized on a sale or other disposition
of our common stock, Series F pre-funded warrants or Series E warrants unless:
| |
· |
the gain is effectively connected with the Non-U.S. Holder’s conduct of a trade or business in the United States (and, if required by an applicable income tax treaty between the United States and such Non-U.S. Holder’s country of residence, the gain is attributable to a permanent establishment or fixed base maintained by the Non-U.S. Holder in the U.S.), in which case the Non-U.S. Holder will be taxed on a net income basis at the regular rates and in the manner applicable to U.S. persons, and if the Non-U.S. Holder is a corporation, an additional branch profits tax at a rate of 30%, or a lower rate as may be specified by an applicable income tax treaty, may also apply; |
| |
· |
the Non-U.S. Holder is a nonresident alien present in the United States for 183 days or more in the taxable year of the disposition and certain other requirements are met, in which case the Non-U.S. Holder will be subject to a 30% tax (or such lower rate as may be specified by an applicable income tax treaty between the United States and such holder’s country of residence) on the net gain derived from the disposition, which may be offset by certain U.S.-source capital losses of the Non-U.S. Holder, if any, provided that the Non-U.S. Holder has timely filed U.S. federal income tax returns reporting those losses; or |
| |
· |
we are, or have been, a “United States real property holding corporation,” or USRPHC, for U.S. federal income tax purposes during the five-year period preceding such disposition (or the Non-U.S. Holder’s holding period, if shorter). We do not believe that we are or have been a USRPHC and, even if we are or were a USRPHC, as long as our common stock is regularly traded on an established securities market, dispositions will not be subject to tax for a Non-U.S. Holder that has not held more than 5% of our common stock, actually or constructively, during the five-year period preceding such Non-U.S. Holder’s disposition (or the Non-U.S. Holder’s holding period, if shorter). Special rules may apply to the determination of the 5% threshold in the case of a holder of a Series F pre-funded warrant or Series E warrant. |
See the sections titled “Information
Reporting and Backup Withholding” and “Foreign Accounts” below for additional information regarding withholding rules that
may apply to proceeds of a disposition of our common stock, Series F pre-funded warrants or Series E warrants paid to foreign
financial institutions or non-financial foreign entities.
Information Reporting and Backup Withholding
We must report annually to
the IRS and to each Non-U.S. Holder the gross amount of the distributions (including constructive distributions) on our common stock,
Series F pre-funded warrants or Series E warrants paid to such holder and the tax withheld, if any, with respect to such distributions.
Non-U.S. Holders may have to comply with specific certification procedures to establish that the holder is not a U.S. person (as defined
in the Code) in order to avoid backup withholding at the applicable rate, currently 24%. Generally, a Non-U.S. Holder will comply with
such procedures if it provides a properly executed applicable IRS Form W-8 or by otherwise establishing an exemption. Dividends paid
to Non-U.S. Holders subject to withholding of U.S. federal income tax, as described above under the heading “Distributions,”
will generally be exempt from U.S. backup withholding.
Information reporting and
backup withholding generally will apply to the proceeds of a disposition of our common stock, Series F pre-funded warrants or Series E
warrants by a Non-U.S. Holder effected by or through the U.S. office of any broker, U.S. or foreign, unless the Non-U.S. Holder certifies
its status as a Non-U.S. Holder and satisfies certain other requirements, or otherwise establishes an exemption. Generally, information
reporting and backup withholding will not apply to a payment of disposition proceeds to a Non-U.S. Holder where the transaction is effected
outside the United States through a non-U.S. office of a non-U.S. broker. However, for information reporting purposes, dispositions effected
through a non-U.S. office of a broker with substantial U.S. ownership or operations generally will be treated in a manner similar to dispositions
effected through a U.S. office of a broker. Non-U.S. Holders should consult their own tax advisors regarding the application of the information
reporting and backup withholding rules to them.
Copies of information returns
may be made available to the tax authorities of the country in which the Non-U.S. Holder resides or is incorporated under the provisions
of a specific treaty or agreement.
Backup withholding is not
an additional tax. Any amounts withheld under the backup withholding rules from a payment to a Non-U.S. Holder can be refunded or
credited against the Non-U.S. Holder’s U.S. federal income tax liability, if any, provided that an appropriate claim is timely filed
with the IRS.
Foreign Accounts
Legislation commonly referred
to as the “Foreign Account Tax Compliance Act,” or “FATCA,” generally imposes a 30% withholding tax on dividends
on common stock, Series F pre-funded warrants and Series E warrants if paid to a non-U.S. entity unless (i) if the non-U.S.
entity is a “foreign financial institution,” the non-U.S. entity undertakes certain due diligence, reporting, withholding,
and certification obligations, (ii) if the non-U.S. entity is not a “foreign financial institution,” the non-U.S. entity
identifies certain of its U.S. investors, if any, or (iii) the non-U.S. entity is otherwise exempt under FATCA.
Intergovernmental agreements
between the United States and foreign countries with respect to FATCA may significantly modify the requirements described in this section
for Non-U.S. Holders. Holders should consult their own tax advisors regarding the possible implications of FATCA on their investment in
our common stock, Series F pre-funded warrants or Series E warrants.
The preceding discussion
of material U.S. federal tax considerations is for information only. It is not tax advice. Prospective investors should consult their
own tax advisors regarding the particular U.S. federal, state, local and non-U.S. tax consequences of purchasing, holding and disposing
of our common stock, Series F pre-funded warrants or Series E warrants, including the consequences of any proposed changes in
applicable laws.
PLAN OF DISTRIBUTION
We engaged
(“ ” or the “placement agent”) to act as our exclusive placement agent to solicit offers to purchase
the securities offered by this prospectus on a reasonable best efforts basis. The placement agent is not purchasing or selling any securities,
nor are they required to arrange for the purchase and sale of any specific number or dollar amount of securities, other than to use their
“reasonable best efforts” to arrange for the sale of the securities by us. Therefore, we may not sell the entire amount of
securities being offered.
There is no minimum amount
of proceeds that is a condition to closing of this offering. The placement agent does not guarantee that it will be able to raise new
capital in this offering. Investors purchasing securities offered hereby will have the option to execute a securities purchase agreement
with us. In addition to rights and remedies available to all purchasers in this offering under federal securities and state law, the
purchasers which enter into a securities purchase agreement will also be able to bring claims of breach of contract against us. The ability
to pursue a claim for breach of contract is material to larger purchasers in this offering as a means to enforce the following covenants
uniquely available to them under the securities purchase agreement: (i) a covenant to not enter into variable rate financings for
a period of one year following the closing of the offering, subject to an exception; and (ii) a covenant to not enter into any equity
financings for ( ) days from closing of the offering, subject to certain exceptions. The placement agent may engage one or more sub-placement
agents or selected dealers to assist with the offering.
The nature of the representations,
warranties and covenants in the securities purchase agreements shall include:
| |
· |
standard issuer representations and warranties on matters such as organization, qualification, authorization, no conflict, no governmental filings required, current in SEC filings, no litigation, labor or other compliance issues, environmental, intellectual property and title matters and compliance with various laws such as the Foreign Corrupt Practices Act; |
| |
|
|
| |
· |
standard issuer representations and warranties on matters such as organization, qualification, authorization, no conflict, no governmental filings required, current in SEC filings, no litigation, labor or other compliance issues, environmental, intellectual property and title matters and compliance with various laws such as the Foreign Corrupt Practices Act; and |
| |
|
|
| |
· |
covenants regarding matters such as registration of warrant shares, no integration with other
offerings, filing of an 8-K to disclose entering into these securities purchase agreements, no shareholder rights plans, no material
nonpublic information, use of proceeds, indemnification of purchasers, reservation and listing of common stock, and no subsequent
equity sales for ( ) days, subject to certain exceptions. |
Fees and Expenses
The following table shows
the per share and Series E warrants and Series F pre-funded warrant and Series E warrants placement agent fees and total
placement agent fees we will pay in connection with the sale of the securities in this offering, assuming the purchase of all of the securities
we are offering.
| Per share and Series E warrants placement agent cash fees |
|
$ |
|
|
| Per Series F pre-funded warrant and Series E warrants placement agent cash fees |
|
$ |
|
|
| Total |
|
$ |
|
|
We have agreed to pay
the placement agent a total cash fee equal to 7.0% of the gross proceeds of this offering and a management fee equal to 1.0% of the gross
proceeds raised in this offering. We will also pay the placement agent a non-accountable expense allowance of $10,000, $15,950 for the
expenses of its clearing firm, and will reimburse the placement agent’s legal fees and expenses in an amount up to $100,000. We
estimate the total offering expenses of this offering that will be payable by us, excluding the placement agent fees and expenses, will
be approximately $ million. After deducting the placement agent fees and
our estimated offering expenses, we expect the net proceeds from this offering to be approximately $ million.
Placement Agent Warrants
We have agreed to grant
placement agent warrants to the placement agent, or its designees, (the “Placement Agent Warrant”) to purchase a number of
shares of our common stock equal to 5.0% of the aggregate number of shares of common stock and Series F pre-funded warrants sold
to the investors in this offering. The placement agent warrants will have an exercise price of $
(125% of the combined public offering price per share of common stock and Series E warrants) and will terminate on the five year
anniversary of commencement of sales in this offering. The Placement Agent Warrants and the shares of common stock exercisable under
the Placement Agent Warrants are registered on the registration statement of which this prospectus is a part. The form of the Placement
Agent Warrant will be included as an exhibit to this registration statement of which this prospectus forms a part.
Tail
We have also agreed to pay
the placement agent a tail fee equal to the cash and warrant compensation in this offering, if any investor, who was contacted or introduced
to us by the placement agent during the term of its engagement, provides us with capital in any public or private offering or other financing
or capital raising transaction during the 12-month period following expiration or termination of our engagement of the placement agent.
Determination of Offering Price
The combined public offering
price per share and Series E warrants and the combined public offering price per Series F pre-funded warrant and Series E
warrants we are offering and the exercise prices and other terms of the warrants were negotiated between us and the investors, in consultation
with the placement agent based on the trading of our common stock prior to this offering, among other things. Other factors considered
in determining the public offering prices of the securities we are offering and the exercise prices and other terms of the warrants include
the history and prospects of our company, the stage of development of our business, our business plans for the future and the extent to
which they have been implemented, an assessment of our management, general conditions of the securities markets at the time of the offering
and such other factors as were deemed relevant.
Lock-up Agreements
We and each of our officers
and directors have agreed with the placement agent to be subject to a lock-up period of ( ) days following the date of this prospectus.
This means that, during the applicable lock-up period, we and such persons may not offer for sale, contract to sell, or sell any shares
of our common stock or any securities convertible into, or exercisable or exchangeable for, shares of our common stock subject to certain
customary exceptions. The placement agent may, in its sole discretion and without notice, waive the terms of any of these lock-up agreements.
In addition, we have agreed to not issue any securities that are subject to a price reset based on trading prices of our common stock
or upon a specified or contingent event in the future, or enter into an agreement to issue securities at a future determined price, until
one (1) year following the closing date of this offering, subject to an exception. The placement agent may waive this prohibition
in its sole discretion and without notice.
Transfer Agent and Registrar
The transfer agent and registrar
for our common stock is Broadridge Corporate Issuer Solutions, Inc. whose address is 51 Mercedes Way, Edgewood, New York 11717.
Nasdaq Listing
Our common stock is currently
listed on the Nasdaq Capital Market under the symbol “AGRX.” On , 2024, the reported closing price per share of our
common stock was $ . We do not plan to list the warrants on the Nasdaq Capital
Market or any other securities exchange or trading market.
Indemnification
We have agreed to indemnify
the placement agent against certain liabilities, including liabilities under the Securities Act of 1933, as amended (the “Securities
Act”), or to contribute to payments the placement agent may be required to make with respect to any of these liabilities.
Regulation M
The placement agent may be
deemed to be an underwriter within the meaning of Section 2(a)(11) of the Securities Act and any fees received by it and any profit
realized on the sale of the securities by it while acting as principal might be deemed to be underwriting discounts or commissions under
the Securities Act. The placement agent will be required to comply with the requirements of the Securities Act and the Exchange Act of
1934, as amended (the “Exchange Act”), including, without limitation, Rule 10b-5 and Regulation M under the Exchange
Act. These rules and regulations may limit the timing of purchases and sales of our securities by the placement agent. Under these
rules and regulations, the placement agent may not (i) engage in any stabilization activity in connection with our securities;
and (ii) bid for or purchase any of our securities or attempt to induce any person to purchase any of our securities, other than
as permitted under the Exchange Act, until they have completed their participation in the distribution.
Electronic Offer, Sale and Distribution of
Securities
A prospectus in electronic
format may be made available on the websites maintained by the placement agent, if any, participating in this offering and the placement
agent may distribute prospectuses electronically. Other than the prospectus in electronic format, the information on these websites is
not part of this prospectus or the registration statement of which this prospectus forms a part, has not been approved or endorsed by
us or the placement agent, and should not be relied upon by investors.
LEGAL MATTERS
The validity of the securities
being offered in this offering will be passed upon for us by Morgan, Lewis & Bockius LLP, Princeton, New Jersey.
EXPERTS
The financial statements of
Agile Therapeutics, Inc. appearing in Agile Therapeutics, Inc.’s Annual Report (Form 10-K) for the year ended December 31, 2022, have been audited by Ernst & Young LLP, our independent registered public accounting firm, as set forth in their report
thereon included therein, and incorporated herein by reference which contains explanatory paragraphs describing conditions that raise
substantial doubt about the Company’s ability to continue as a going concern as described in Note 1 to the financial statements
and describing the restatement of the 2021 financial statements as described in Note 2 to the financial statements. Such financial statements
are, and audited financial statements to be included in subsequently filed documents will be, incorporated herein in reliance upon the
report of Ernst & Young LLP pertaining to such financial statements (to the extent covered by consents filed with the Securities
and Exchange Commission) given on the authority of such firm as experts in accounting and auditing.
WHERE YOU CAN FIND MORE INFORMATION
This prospectus forms part
of a registration statement on Form S-1 that we filed with the SEC. This prospectus does not contain all of the information set forth
in the registration statement and the exhibits to the registration statement or the documents incorporated by reference herein and therein.
For further information with respect to us and the securities that we are offering under this prospectus, we refer you to the registration
statement and the exhibits and schedules filed as a part of the registration statement and the documents incorporated by reference herein
and therein. You should rely only on the information contained in this prospectus or incorporated by reference herein or therein. We have
not authorized anyone else to provide you with different information. We are not making an offer of these securities in any state where
the offer is not permitted. You should not assume that the information in this prospectus is accurate as of any date other than the date
on the cover page of this prospectus, regardless of the time of delivery of this prospectus or any sale of the securities offered
hereby. We file annual, quarterly and current reports, proxy statements and other information with the SEC. The SEC maintains a website
that contains reports, proxy statements and other information regarding issuers that file electronically with the SEC, including Agile
Therapeutics. The address of the SEC website is www.sec.gov.
We maintain a website at www.agiletherapeutics.com.
Information contained in or accessible through our website does not constitute a part of this prospectus.
INFORMATION INCORPORATED BY REFERENCE
The SEC allows us to “incorporate
by reference” information into this prospectus, which means that we can disclose important information to you by referring you to
another document filed separately with the SEC. The SEC file number for the documents incorporated by reference in this prospectus is
001-36464. The documents incorporated by reference into this prospectus contain important information that you should read about us.
The following documents are
incorporated by reference into this document:
| 6. | our Current Reports on Form 8-K or 8-K/A filed with
the SEC on January 26, 2023; February 27, 2023; March 14, 2023; March 30, 2023; April 10, 2023; April 26, 2023; May 17, 2023; May 25, 2023; June 5, 2023; June 8, 2023; June 14, 2023; June 27, 2023; June 30, 2023; August 22, 2023; September 28, 2023; October 2, 2023; December 4, 2023; and December 6, 2023; and |
We also incorporate by reference
into this prospectus all documents (other than current reports furnished under Item 2.02 or Item 7.01 of Form 8-K and exhibits filed
on such form that are related to such items) that are filed by us with the SEC pursuant to Sections 13(a), 13(c), 14 or 15(d) of
the Exchange Act after the date of this prospectus but prior to the termination of the offering. These documents include periodic reports,
such as Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q and Current Reports on Form 8-K, as well as proxy statements.
We will provide to each person,
including any beneficial owner, to whom a prospectus is delivered, without charge upon written or oral request, a copy of any or all of
the documents that are incorporated by reference into this prospectus but not delivered with the prospectus, including exhibits which
are specifically incorporated by reference into such documents. Requests should be directed to: Agile Therapeutics, Inc., Attn: Investor
Relations, 500 College Road East, Suite 310, Princeton, New Jersey 08540. Our telephone number is (609) 683-1880.
Any statement contained herein
or in a document incorporated or deemed to be incorporated by reference into this document will be deemed to be modified or superseded
for purposes of the document to the extent that a statement contained in this document or any other subsequently filed document that is
deemed to be incorporated by reference into this document modifies or supersedes the statement.

$15,000,000
Up to
Shares of Common Stock and
Accompanying Series E-1 Warrants to
Purchase up to Shares of Common Stock and
Series E-2 Warrants
to Purchase up to Shares of Common Stock
or
Series F Pre-funded
Warrants to Purchase up to Shares of Common Stock and
Accompanying Series E-1 Warrants to
Purchase up to Shares of Common Stock and
Series E-2 Warrants
to Purchase up to Shares of Common Stock
Placement Agent Warrants
to Purchase up to Shares of Common Stock
Preliminary Prospectus
, 2024
PART II
INFORMATION NOT REQUIRED IN PROSPECTUS
Item 13. Other Expenses of Issuance and
Distribution.
The following table sets forth
the fees and expenses in connection with the issuance and distribution of the securities being registered (excluding the underwriting
discount). Except for the Securities and Exchange Commission registration fee and the FINRA filing fee, all amounts are estimates.
| | |
Amount Paid
or to be Paid | |
| SEC registration fee | |
$ | 2,214.00 | |
| FINRA filing fee | |
$ | 2,750.00 | |
| Legal fees and expenses | |
| * | |
| Accounting fees and expenses | |
| * | |
| Printing expenses | |
| * | |
| Transfer agent fees and expenses | |
| * | |
| Miscellaneous | |
| * | |
| Total | |
| * | |
* To be completed by amendment
Each of the amounts set forth
above, other than the SEC registration fee and FINRA filing fee, is an estimate.
Item 14. Indemnification of Directors
and Officers.
Section 145 of the Delaware
General Corporation Law authorizes a corporation’s board of directors to grant, and authorizes a court to award, indemnity to officers,
directors, and other corporate agents.
As permitted by Delaware law,
our Amended and Restated Certificate of Incorporation, as amended, provides that, to the fullest extent permitted by Delaware law, no
director will be personally liable to us or our stockholders for monetary damages for breach of fiduciary duty as a director. Pursuant
to Delaware law such protection would be not available for liability:
| |
· |
for any breach of a duty of loyalty to us or our stockholders; |
| |
· |
for acts or omissions not in good faith or that involve intentional misconduct or a knowing violation of law; |
| |
· |
for any transaction from which the director derived an improper benefit; or |
| |
· |
for an act or omission for which the liability of a director is expressly provided by an applicable statute, including unlawful payments of dividends or unlawful stock repurchases or redemptions as provided in Section 174 of the Delaware General Corporation Law. |
Our Amended and Restated Certificate
of Incorporation, as amended, also provides that if Delaware law is amended after the approval by our stockholders of the amended and
restated certificate of incorporation to authorize corporate action further eliminating or limiting the personal liability of directors,
then the liability of our directors will be eliminated or limited to the fullest extent permitted by Delaware law.
Our amended and restated bylaws,
as amended, further provide that we must indemnify our directors and officers to the fullest extent permitted by Delaware law. The amended
and restated bylaws also authorize us to indemnify any of our employees or agents and permit us to secure insurance on behalf of any officer,
director, employee or agent for any liability arising out of his or her action in that capacity, whether or not Delaware law would otherwise
permit indemnification.
In addition, our amended and
restated bylaws provide that we are required to advance expenses to our directors and officers as incurred in connection with legal proceedings
against them for which they may be indemnified and that the rights conferred in the amended and restated bylaws are not exclusive.
At present, there is no pending
litigation or proceeding involving any of our directors, officers or employees for which indemnification is sought, and we are not aware
of any threatened litigation that may result in claims for indemnification.
We have entered into indemnification
agreements with each of our directors and executive officers. These agreements, among other things, require us to indemnify each director
and officer to the fullest extent permitted by Delaware law, the amended and restated certificate of incorporation and amended and restated
bylaws, for expenses such as, among other things, attorneys’ fees, judgments, fines, and settlement amounts incurred by the director
or executive officer in any action or proceeding, including any action by or in our right, arising out of the person’s services
as our director or executive officer or as the director or executive officer of any subsidiary of ours or any other company or enterprise
to which the person provides services at our request. We also maintain directors’ and officers’ liability insurance.
The SEC has taken the position
that personal liability of directors for violation of the federal securities laws cannot be limited and that indemnification by us for
any such violation is unenforceable. The limitation of liability and indemnification provisions in our amended and restated certificate
of incorporation and amended and restated bylaws may discourage stockholders from bringing a lawsuit against our directors and officers
for breach of their fiduciary duty. They may also reduce the likelihood of derivative litigation against our directors and officers, even
though an action, if successful, might benefit us and other stockholders. Further, a stockholder’s investment may be adversely affected
to the extent that we pay the costs of settlement and damage awards against directors and officers as required by these indemnification
provisions.
Item 15. Recent Sales of Unregistered
Securities
Set forth below is information
regarding securities we have issued within the past three years that were not registered under the Securities Act:
On February 10, 2020,
in connection with the Perceptive Credit Agreement, we issued to Perceptive two warrants to purchase an aggregate of 700 shares of our
common stock (together, the “2020 Perceptive Warrants”). The first warrant is exercisable for 350 shares of common stock at
an exercise price of $7,480.00 per share. The second warrant is exercisable for 350 shares of common stock at an exercise price of $9,340.00
per share. The 2020 Perceptive Warrants expire on February 10, 2027. In connection with the Amended Perceptive Credit Agreement,
we issued to Perceptive a warrant to purchase 225 shares of our common stock (the “2021 Perceptive Warrant” and, together
with the 2020 Perceptive Warrants, the “Perceptive Warrants”) at an exercise price of $5,740.00 per share. The 2021 Perceptive
Warrant expires on February 26, 2028. The Perceptive Warrants contain anti-dilution provisions that expired on December 31,
2022 and other warrant holder protections and are not exercisable to the extent that Perceptive would beneficially own more than 19.99%
of our common stock as a result of the exercise. In connection with the Sixth Amendment, we amended and restated the Perceptive Warrants
to reset the strike price of the Perceptive Warrants.
On December 3, 2023, we entered into a Warrant
Amendment and Additional Issuance Agreement (“Warrant Amendment and Additional Issuance Agreement”) relating to the amendment
of warrants to purchase shares of our common stock that were issued in transactions on March 14, 2022, April 25, 2022, and May 25,
2023 (collectively, the “Warrants”). Collectively, the Warrants represent the right to purchase approximately 3.8 million
shares of our common stock. Under the terms of the Warrant Amendment and Additional Issuance Agreement, the holder agreed to revise provisions
related to the use of a Black-Scholes model to value the Warrants in the event of a change of control transaction. The holder also agreed
to revise provisions related to the cashless exercise of the Warrants. In exchange for the holder’s agreement to amend the Warrants,
we agreed to issue an additional new warrant (the “New Warrant”) to purchase 1,005,560 shares of our common stock. The New
Warrant has an exercise price of $2.09 per share. The New Warrant is exercisable six months after issuance and will expire five years
from the date that the New Warrant is initially exercisable. The exercise price of the New Warrant is subject to adjustment for stock
splits, reverse splits, and similar capital transactions as described in the New Warrant.
Our issuances of the warrants
were deemed to be exempt from registration under the Securities Act pursuant to Section 4(a)(2) of the Securities Act in that
such issuances did not involve a public offering. The recipient of securities in each of these transactions acquired the securities
for investment only and not with a view to or for sale in connection with any distribution thereof and appropriate legends were affixed
to the securities issued in these transactions. The recipient of securities in these transactions was an accredited investor within the
meaning of Rule 501 of Regulation D under the Securities Act and had adequate access, through employment, business or other relationships,
to information about us. No underwriters were involved in these transactions.
Item 16. Exhibits and Financial Statement
Schedules.
| 3.4 |
|
Certificate of Amendment to Amended and Restated Certificate of Incorporation filed with the Secretary of State of Delaware on April 10, 2023 (Incorporated by reference, Exhibit 3.1 to Company’s Current Report on Form 8-K, file number 001-36464, filed on April 10, 2023). |
| |
|
|
| 3.5 |
|
Amended and Restated Bylaws of the Registrant (Incorporated by reference, Exhibit 3.1 to Company’s Current Report on Form 8-K, file number 001-36464, filed on January 26, 2023.) |
| |
|
|
| 3.6 |
|
Certificate of Designation of Preferences, Rights and Limitations of Series A Convertible Preferred Stock filed with the Secretary of State of the State of Delaware on March 14, 2022 (Incorporated by reference, Exhibit 3.1 to Company’s Current Report on Form 8-K, file number 001-36464, filed on March 15, 2022.) |
| |
|
|
| 3.7 |
|
Certificate of Designation of Preferences, Rights and Limitations of Series B Convertible Preferred Stock filed with the Secretary of State of the State of Delaware on March 14, 2022 (Incorporated by reference, Exhibit 3.2 to Company’s Current Report on Form 8-K, file number 001-36464, filed on March 15, 2022.) |
| |
|
|
| 3.8 |
|
Certificate of Designation of Preferences, Rights, and Limitations of Series C Preferred Stock filed with the Secretary of State of the State of Delaware on January 26, 2023 (Incorporated by reference, Exhibit 3.2 to Company’s Current Report on Form 8-K, file number 001-36464, filed on January 26, 2023.) |
| |
|
|
| 4.1 |
|
Specimen Certificate evidencing shares of Registrant’s common stock (Incorporated by reference, Exhibit 4.1 to Company’s Third Amendment of Registration Statement on Form S-1, file number 333-194621, filed on May 9, 2014.) |
| |
|
|
| 4.2 |
|
Amended and Restated Common Stock Purchase Warrant between Agile Therapeutics, Inc. and Perceptive Credit Holdings III, LP, dated as of March 21, 2023 (Incorporated by reference, Exhibit 4.2 to Company’s Annual Report on Form 10-K, file number 001-36464, filed on March 23, 2023.) |
| |
|
|
| 4.3 |
|
Amended and Restated Common Stock Purchase Warrant between Agile Therapeutics, Inc. and Perceptive Credit Holdings III, LP, dated as of March 21, 2023 (Incorporated by reference, Exhibit 4.3 to Company’s Annual Report on Form 10-K, file number 001-36464, filed on March 23, 2023.) |
| |
|
|
| 4.4 |
|
Amended and Restated Common Stock Purchase Warrant between Agile Therapeutics, Inc. and Perceptive Credit Holdings III, LP, dated as of March 21, 2023 (Incorporated by reference, Exhibit 4.4 to Company’s Annual Report on Form 10-K, file number 001-36464, filed on March 23, 2023.) |
| |
|
|
| 4.5 |
|
Form of Warrant (Incorporated by reference, Exhibit 4.1 to Company’s Current Report on Form 8-K, file number 001-36464, filed on October 8, 2021.) |
| |
|
|
| 4.6 |
|
Form of Series A Warrant (Incorporated by reference, Exhibit 4.1 to Company’s Current Report on Form 8-K, file number 001-36464, filed on March 15, 2022.) |
| |
|
|
| 4.7 |
|
Form of Series B Warrant (Incorporated by reference, Exhibit 4.2 to Company’s Current Report on Form 8-K, file number 001-36464, filed on March 15, 2022.) |
| |
|
|
| 4.8 |
|
Form of Placement Agent Warrant (Incorporated by reference, Exhibit 4.3 to Company’s Current Report on Form 8-K, file number 001-36464, filed on March 15, 2022.) |
| |
|
|
| 4.9 |
|
Form of Series A-1 Warrant (Incorporated by reference, Exhibit 4.1 to Company’s Current Report on Form 8-K, file number 001-36464, filed on July 8, 2022.) |
| |
|
|
| 4.10 |
|
Form of Series A-2 Warrant (Incorporated by reference, Exhibit 4.2 to Company’s Current Report on Form 8-K, file number 001-36464, filed on July 8, 2022.) |
| |
|
|
| 4.11 |
|
Form of Series B Pre-Funded Warrant (Incorporated by reference, Exhibit 4.3 to Company’s Current Report on Form 8-K, file number 001-36464, filed on July 8, 2022.) |
| |
|
|
| 4.12 |
|
Form of Placement Agent Warrant (Incorporated by reference, Exhibit 4.4 to Company’s Current Report on Form 8-K, file number 001-36464, filed on July 8, 2022.) |
| 4.13 |
|
Description of Capital Stock (Incorporated by reference, Exhibit 4.4 to Company’s
Annual Report on Form 10-K, file number 001-36464, filed on February 20, 2020.) |
| |
|
|
| 4.14 |
|
Form of
Series C-1 Warrant (Incorporated by reference, Exhibit 4.1 to Company’s Current Report on Form 8-K, file number
001-36464, filed on May 25, 2023.) |
| |
|
|
| 4.15 |
|
Form of
Series C-2 Warrant (Incorporated by reference, Exhibit 4.2 to Company’s Current Report on Form 8-K, file number
001-36464, filed on May 25, 2023.) |
| |
|
|
| 4.16 |
|
Form of
Series D Pre-funded Warrant (Incorporated by reference, Exhibit 4.3 to Company’s Current Report on Form 8-K,
file number 001-36464, filed on May 25, 2023.) |
| |
|
|
| 4.17 |
|
Form of
Placement Agent Warrant (Incorporated by reference, Exhibit 4.4 to Company’s Current Report on Form 8-K, file number
001-36464, filed on May 25, 2023.) |
| |
|
|
| 4.18 |
|
Form of Amended and
Restated Common Stock Purchase Warrant between Agile Therapeutics, Inc. and an institutional investor, dated October 8,
2021(Incorporated by reference, Exhibit 4.5 to Company’s Current Report on Form 8-K, file number 001-36464, filed
on May 25, 2023.) |
| |
|
|
| 4.19 |
|
Form of Amended and
Restated Series A Common Stock Purchase Warrant between Agile Therapeutics, Inc. and an institutional investor, dated March 14,
2022 (Incorporated by reference, Exhibit 4.6 to Company’s Current Report on Form 8-K, file number 001-36464, filed
on May 25, 2023.) |
| |
|
|
| 4.20 |
|
Form of Amended and
Restated Series A Common Stock Purchase Warrant between Agile Therapeutics, Inc. and an institutional investor, dated April 25,
2022 (Incorporated by reference, Exhibit 4.7 to Company’s Current Report on Form 8-K, file number 001-36464, filed
on May 25, 2023.) |
| |
|
|
| 4.21 |
|
Form of Amended and
Restated Series A-1 Common Stock Purchase Warrant between Agile Therapeutics, Inc. and an institutional investor, dated
July 6, 2022 (Incorporated by reference, Exhibit 4.8 to Company’s Current Report on Form 8-K, file number 001-36464,
filed on May 25, 2023.) |
| |
|
|
| 4.22 |
|
Form of Amended and
Restated Series A-2 Common Stock Purchase Warrant between Agile Therapeutics, Inc. and an institutional investor, dated
July 6, 2022 (Incorporated by reference, Exhibit 4.9 to Company’s Current Report on Form 8-K, file number 001-36464,
filed on May 25, 2023.) |
| |
|
|
| 4.23 |
|
Form of Amended and
Restated Series B Common Stock Purchase Warrant between Agile Therapeutics, Inc. and an institutional investor, dated March 14,
2022 (Incorporated by reference, Exhibit 4.10 to Company’s Current Report on Form 8-K, file number 001-36464, filed
on May 25, 2023.) |
| |
|
|
| 4.24 |
|
Form of New Warrant.
(Incorporated by reference, Exhibit 4.1 to Company’s Current Report on Form 8-K, file number 001-36464, filed
on December 4, 2023.) |
| |
|
|
| 4.25** |
|
Amendment to Warrants and Certificate of Adjustment between Agile Therapeutics, Inc. and Perceptive
Credit Holdings III, LP, dated as of October 30, 2023. |
| 4.26** |
|
Form of
Series E-1 Warrant |
| |
|
|
| 4.27** |
|
Form of Series E-2 Warrant |
| |
|
|
| 4.28** |
|
Form of Series F Pre-Funded Warrant |
| |
|
|
| 4.29** |
|
Form of Placement Agent Warrant |
| |
|
|
| 5.1++ |
|
Opinion of Morgan, Lewis &
Bockius LLP |
| |
|
|
| 10.1+ |
|
Form of Indemnification
Agreement. (Incorporated by reference, Exhibit 10.1 to Company’s Second Amendment of Registration Statement on Form S-1,
file number 333-194621, filed on May 5, 2014.) |
| |
|
|
| 10.2+ |
|
Agile Therapeutics, Inc.
Amended and Restated 1997 Equity Incentive Plan, as amended, and form of Stock Option Agreement thereunder. (Incorporated by reference,
Exhibit 10.2 to Company’s Registration Statement on Form S-1, file number 333-194621, filed on March 17, 2014.) |
| |
|
|
| 10.3+ |
|
Agile Therapeutics, Inc.
Amended and Restated 2008 Equity Incentive Plan and form of Nonqualified Stock Option Agreement and form of Incentive Stock Option
Agreement thereunder. (Incorporated by reference, Exhibit 10.3 to Company’s Registration Statement on Form S-1, file
number 333-194621, filed on March 17, 2014.) |
| |
|
|
| 10.4+ |
|
Form of Performance
Unit Issuance Agreement (Incorporated by reference, Exhibit 10.1 to Company’s Current Report on Form 8-K, file number
001-36464, filed on January 26, 2018.) |
| |
|
|
| 10.5 |
|
Lease Agreement, dated
August 6, 2021 by and between Agile Therapeutics, Inc. and 500 College Road Venture, LLC (Incorporated by reference, Exhibit 10.1
to Company’s Quarterly Report on Form 10-Q, file number 001-36464, filed on November 2, 2021.) |
| |
|
|
| 10.6 |
|
Common Stock Sales Agreement
dated November 8, 2019 by and between the Registrant and H.C. Wainwright & Co., LLC (Incorporated by reference, Exhibit 1.1
to Company’s Current Report on Form 8-K, file number 001-36464, filed on November 8, 2019.) |
| |
|
|
| 10.7 |
|
Common Stock Sales Agreement
dated March 18, 2021, by and between Agile Therapeutics, Inc. and H.C. Wainwright & Co., LLC (Incorporated by
reference, Exhibit 1.1 to the Company’s Current Report on Form 8-K, file number 001-036464, filed on March 18,
2021.) |
| |
|
|
| 10.8 |
|
Common Stock Sales Agreement
dated April 27, 2022 by and between Agile Therapeutics, Inc. and H.C. Wainwright & Co., LLC (Incorporated by reference,
Exhibit 1.1 to Company’s Current Report on Form 8-K, file number 001-036464, filed on April 27, 2022.) |
| |
|
|
| 10. 9 |
|
Controlled Equity OfferingSM
Sales Agreement dated January 10, 2022 by and among Agile Therapeutics, Inc. and Cantor Fitzgerald & Co.
and H.C. Wainwright & Co., LLC (Incorporated by reference, Exhibit 1.1 to Company’s Current Report on Form 8-K,
file number 001-36464, filed on January 10, 2022.) |
| |
|
|
| 10.10 |
|
Engagement Agreement, by
and between Agile Therapeutics, Inc. and H.C. Wainwright & Co., LLC (Incorporated by reference, Exhibit 10.32
to Company’s Amendment No. 1 to Registration Statement on Form S-1/A, file number 333-264960, filed on June 29,
2022.) |
| |
|
|
| 10.11 |
|
Amendment to Engagement
Agreement, by and between Agile Therapeutics, Inc. and H.C. Wainwright & Co., LLC (Incorporated by reference, Exhibit 10.33
to Company’s Amendment No. 1 to Registration Statement on Form S-1/A, file number 333-264960, filed on June 29,
2022.) |
| |
|
|
| 10.12 |
|
Credit Agreement and Guaranty
among Agile Therapeutics, Inc., the guarantors from time to time party thereto, the lenders from time to time party thereto
and Perceptive Credit Holdings III, LP, dated as of February 10, 2020 (Incorporated by reference, Exhibit 10.1 to Company’s
Current Report on Form 8-K, file number 001-36464, filed on February 12, 2020.) |
| |
|
|
| 10.13 |
|
Waiver and First Amendment
to Credit Agreement and Guaranty among Agile Therapeutics, Inc., the guarantors from time to time party thereto, the lenders
from time to time party thereto and Perceptive Credit Holdings III, LP, dated as of February 26, 2021(Incorporated by reference,
Exhibit 10.11 to Company’s Annual Report on Form 10-K, file number 001-36464, filed on March 1, 2021.) |
| 10.14 |
|
Waiver and
Second Amendment to Credit Agreement and Guaranty among Agile Therapeutics, Inc., the guarantors from time to time party thereto,
the lenders from time to time party thereto and Perceptive Credit Holdings III, LP, dated as of January 7, 2022 (Incorporated
by reference, Exhibit 10.1 to Company’s Current Report on Form 8-K, file number 001-36464, filed on January 10,
2022.) |
| 10.15 |
|
Waiver and
Third Amendment to Credit Agreement and Guaranty among Agile Therapeutics, Inc., the guarantors from time to time party thereto,
the lenders from time to time party thereto and Perceptive Credit Holdings III, LP, dated as of March 10, 2022 (Incorporated
by reference, Exhibit 10.1 to Company’s Current Report on Form 8-K, file number 001-36464, filed on March 11,
2022.) |
| |
|
|
| 10.16 |
|
Waiver and Fourth Amendment
to Credit Agreement and Guaranty among Agile Therapeutics, Inc., the guarantors from time to time party thereto, the lenders
from time to time party thereto and Perceptive Credit Holdings III, LP, dated as of May 11, 2022 (Incorporated by reference,
Exhibit 10.1 to Company’s Current Report on Form 8-K, file number 001-36464, filed on May 12, 2022.) |
| |
|
|
| 10.17 |
|
Fifth Amendment to Credit
Agreement and Guaranty among Agile Therapeutics, Inc., the guarantors from time to time party thereto, the lenders from time
to time party thereto and Perceptive Credit Holdings III, L.P. dated as of July 25, 2022 (Incorporated by reference, Exhibit 10.1
to Company’s Quarterly Report on form 10-Q, file number 001-36464, filed on November 7, 2022.) |
| |
|
|
| 10.18 |
|
Waiver and Sixth Amendment to Credit Agreement and Guaranty among Agile Therapeutics, Inc., the guarantors from time to time party thereto,
the lenders from time to time party thereto and Perceptive Credit Holdings III, LP, dated as of March 21, 2023 (Incorporated by reference,
Exhibit 10.18 to Company’s Registration Statement on Form S-1, file number 333-271249, filed on April 14, 2023.) |
| |
|
|
| 10.19 |
|
Seventh Amendment to Credit Agreement and Guaranty among Agile Therapeutics, Inc., the guarantors from time to time party thereto, the lenders from time to time party thereto and Perceptive Credit Holdings III, L.P., dated as of October 30, 2023. |
| |
|
|
| 10.20 |
|
Form of Securities
Purchase Agreement, dated March 13, 2022, by and between Agile Therapeutics, Inc. and the purchaser signatory thereto (Incorporated
by reference, Exhibit 10.1 to Company’s Current Report on Form 8-K, file number 001-36464, filed on March 15,
2022.) |
| |
|
|
| 10.21 |
|
Form of Securities
Purchase Agreement by and between Agile Therapeutics, Inc. and the purchasers signatory thereto (Incorporated by reference,
Exhibit 10.31 to Company’s Amendment No. 1 to Registration Statement on Form S-1/A, file number 333-264960,
filed on June 29, 2022.) |
| |
|
|
| 10.22 |
|
Form of Securities
Purchase Agreement, by and between Agile Therapeutics, Inc., and certain purchasers (Incorporated by reference, Exhibit 10.1
to Company’s Current Report on Form 8-K, file number 001-36464, filed on July 8, 2022.) |
| |
|
|
| 10.23* |
|
Project Agreement, dated
April 30, 2020, by and between Agile Therapeutics, Inc. and inVentiv Commercial Services, LLC (Incorporated by reference,
Exhibit 10.1 to Company’s Quarterly Report on Form 10-Q, file number 001-36464, filed on August 11, 2020.) |
| |
|
|
| 10.24* |
|
First Amendment to Project
Agreement, dated June 1, 2020, by and between Agile Therapeutics, Inc. and inVentiv Commercial Services, LLC (Incorporated
by reference, Exhibit 10.13 to Company’s Annual Report on Form 10-K, file number 001-36464, filed on March 1,
2021.) |
| |
|
|
| 10.25* |
|
Second Amendment to Project
Agreement, dated January 1, 2021, by and between Agile Therapeutics, Inc. and inVentiv Commercial Services, LLC (Incorporated
by reference, Exhibit 10.2 to Company’s Quarterly Report on Form 10-Q, file number 001-36464, filed on November 2,
2021.) |
| |
|
|
| 10.26* |
|
Third Amendment to Project
Agreement, dated July 1, 2021, by and between Agile Therapeutics, Inc. and inVentiv Commercial Services, LLC. (Incorporated
by reference, Exhibit 10.3 to Company’s Quarterly Report on Form 10-Q, file number 001-36464, filed on November 2,
2021.) |
| |
|
|
| 10.27* |
|
Fourth Amendment to Project
Agreement, dated September 1, 2021, by and between Agile Therapeutics, Inc. and inVentiv Commercial Services, LLC. (Incorporated
by reference, Exhibit 10.24 to Company’s Annual Report on Form 10-K, file number 001-36464, filed on March 30,
2022.) |
| 10.29* |
|
Sixth Amendment to Project Agreement, dated January 3, 2023, by and between Agile Therapeutics, Inc. and inVentiv Commercial Services, LLC (Incorporated by reference, Exhibit 10.27 to the Company’s Annual Report on Form 10-K, file number 001-36464, filed on March 23, 2023.) |
| |
|
|
| 10.30* |
|
Seventh Amendment to Project Agreement, dated September 28, 2023, by and between Agile Therapeutics, Inc. and Syneos Health Commercial Services, LLC (Incorporated by reference, Exhibit 10.1 to Company’s Current Report on Form 8-K, file number 001-36464, filed on September 28, 2023.) |
| 10.31* |
|
Master Service
Agreement, dated October 11. 2017, by and between Agile Therapeutics, Inc. and inVentiv Commercial Services, LLC (Incorporated
by reference, Exhibit 10.2 to Company’s Quarterly Report on Form 10-Q, file number 001-36464, filed on August 11,
2020.) |
| |
|
|
| 10.32* |
|
First Amendment to Master
Service Agreement, dated April 30, 2020, by and between Agile Therapeutics, Inc. and inVentiv Commercial Services, LLC
(Incorporated by reference, Exhibit 10.3 to Company’s Quarterly Report on Form 10-Q, file number 001-36464, filed
on August 11, 2020.) |
| |
|
|
| 10.33* |
|
Manufacturing and Commercialization
Agreement, dated April 30, 2020, by and between Agile Therapeutics, Inc. and Corium, Inc. (Incorporated by reference,
Exhibit 10.4 to Company’s Quarterly Report on Form 10-Q, file number 001-36464, filed on August 11, 2020.) |
| |
|
|
| 10.34 |
|
Amendment No. 1 to
Manufacturing and Commercialization Agreement, by and between Corium, Inc. and Agile Therapeutics, Inc., dated as of July 25,
2022, and Bill of Sale by Agile Therapeutics, Inc. to Corium Inc., dated as of July 25, 2022 (Incorporated by reference,
Exhibit 10.2 to Company’s Quarterly Report on Form 10-Q, file number 001-36464, filed on November 7, 2022.) |
| |
|
|
| 10.35+ |
|
Agile Therapeutics, Inc.
Amended and Restated 2014 Incentive Compensation Plan (Incorporated by reference, Appendix A to Registrant’s Proxy Statement
pursuant to Section 14(a) of the Securities Exchange Act of 1934, file number 001-36464, filed on April 25, 2018.) |
| |
|
|
| 10.36+ |
|
Amended and Restated Employment
Agreement, dated November 22, 2022 by and between Agile Therapeutics, Inc. and Alfred Altomari (Incorporated by reference,
Exhibit 10.33 to the Company’s Annual Report on Form 10-K, file number 001-36464, filed on March 23, 2023.) |
| |
|
|
| 10.37+ |
|
Amended and Restated Employment
Agreement, dated November 1, 2022 by and between Agile Therapeutics, Inc. and Geoffrey P. Gilmore (Incorporated by reference,
Exhibit 10.34 to the Company’s Annual Report on Form 10-K, file number 001-36464, filed on March 23, 2023.) |
| |
|
|
| 10.38+ |
|
Amended and Restated Employment
Agreement, dated November 1, 2022 by and between Agile Therapeutics, Inc. and Paul Korner, M.D. (Incorporated by reference,
Exhibit 10.35 to the Company’s Annual Report on Form 10-K, file number 001-36464, filed on March 23, 2023.) |
| |
|
|
| 10.39 |
|
Form of Securities
Purchase Agreement, dated May 22, 2023, by and between Agile Therapeutics, Inc. and certain purchasers (Incorporated by
reference, Exhibit 10.1 to Company’s Current Report on Form 8-K, file number 001-36464, filed on May 25, 2023.) |
| |
|
|
| 10.40 |
|
Form of Warrant Amendment Agreement, dated May 22,
2023, by and between Agile Therapeutics, Inc. and certain holders (Incorporated by reference, Exhibit 10.2 to Company’s
Current Report on Form 8-K, file number 001-36464, filed on May 25, 2023.) |
| |
|
|
| 10.41 |
|
Agile Therapeutics, Inc.
2023 Equity Incentive Plan effective June 8, 2023 (Incorporated by reference, Exhibit 99.1 to the Company’s Registration
Statement on Form S-8, file number 333-272576, filed on June 9, 2023.) |
| |
|
|
| 10.42+ |
|
Employment Agreement, dated
August 16, 2023, by and between Agile Therapeutics, Inc. and Scott Coiante (Incorporated by reference, Exhibit 10.1
to Company’s Current Report on Form 8-K/A, file number 001-36464, filed August 22, 2023.) |
| |
|
|
| 10.43 |
|
Form of
Warrant Amendment and Additional Issuance Agreement. (Incorporated by reference, Exhibit 10.1 to the
Company’s Current Report on Form 8-K, file number 001-36464, filed on December 4, 2023.) |
| |
|
|
| 10.44** |
|
Form of Securities
Purchase Agreement, by and among Agile Therapeutics, Inc. and the purchasers signatory thereto. |
| |
|
|
| 23.1** |
|
Consent of Independent Registered Public Accounting Firm. |
| |
|
|
| 23.2++ |
|
Consent of Morgan, Lewis &
Bockius LLP (included in Exhibit 5.1) |
| |
|
|
| 24.1** |
|
Power of Attorney (contained
on the signature page to the Registration Statement). |
| |
|
|
| 107** |
|
Filing
Fee Table |
| + |
|
Indicates management contract or compensatory plan or arrangement. |
| |
|
|
| ++ |
|
To be filed by amendment. |
| |
|
|
| * |
|
Portions of this exhibit have been redacted in accordance with Regulation S-K Item 601(b)(10). |
| |
|
|
| ** |
|
Previously filed. |
Item 17. Undertakings.
The undersigned registrant
hereby undertakes:
| |
(a) |
(1) To file, during any period in which offers or sales are being made, a post-effective amendment to this registration statement: |
(i) To include any prospectus required
by Section 10(a)(3) of the Securities Act of 1933, as amended (the “Securities Act”);
(ii) To reflect in the prospectus
any facts or events arising after the effective date of the registration statement (or the most recent post-effective amendment thereof)
which, individually or in the aggregate, represent a fundamental change in the information set forth in the registration statement. Notwithstanding
the foregoing, any increase or decrease in the volume of securities offered (if the total dollar value of securities offered would not
exceed that which was registered) and any deviation from the low or high end of the estimated maximum offering range may be reflected
in the form of prospectus filed with the Commission pursuant to Rule 424(b) if, in the aggregate, the changes in volume and
price represent no more than 20% change in the maximum aggregate offering price set forth in the “Calculation of Registration Fee”
table in the effective registration statement; and
(iii) To include any material information
with respect to the plan of distribution not previously disclosed in the registration statement or any material change to such information
in the registration statement.
provided,
however, that paragraphs (1)(i), (1)(ii) and (1)(iii) do not apply if the information required to be included in
a post-effective amendment by those paragraphs is contained in reports filed with or furnished to the Commission by the registrant pursuant
to Section 13 or Section 15(d) of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), that
are incorporated by reference in the registration statement, or is contained in a form of prospectus filed pursuant to Rule 424(b) that
is part of the registration statement.
| |
(2) |
That, for the purposes of determining any liability under the Securities Act, each such post-effective amendment shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at the time shall be deemed to be the initial bona fide offering thereof. |
| |
(3) |
To remove from registration by means of a post-effective amendment any of the securities being registered which remain unsold at the termination of the offering. |
| |
(4) |
That, for the purpose of determining liability of the registrant under the Securities Act to any purchaser in the initial distribution of the securities, the undersigned registrant undertakes that in a primary offering of securities of the undersigned registrant pursuant to this registration statement, regardless of the underwriting method used to sell the securities to the purchaser, if the securities are offered or sold to such purchaser by means of any of the following communications, the undersigned registrant will be a seller to the purchaser and will be considered to offer or sell such securities to such purchaser: |
(i) Any preliminary
prospectus or prospectus of the undersigned registrant relating to the offering required to be filed pursuant to Rule 424;
(ii) Any free writing prospectus
relating to the offering prepared by or on behalf of the undersigned registrant or used or referred to by the undersigned registrant;
(iii) The portion of any other free
writing prospectus relating to the offering containing material information about the undersigned registrant or its securities provided
by or on behalf of the undersigned registrant; and
(iv) Any other communication that
is an offer in the offering made by the undersigned registrant to the purchaser.
| |
(5) |
That, for purposes of determining any liability under the Securities Act: |
(i) the information omitted from
the form of prospectus filed as part of the registration statement in reliance upon Rule 430A and contained in the form of prospectus
filed by the Registrant pursuant to Rule 424(b)(1) or (4) or 497(h) under the Securities Act shall be deemed to be
part of the registration statement as of the time it was declared effective; and
(ii) each post-effective amendment
that contains a form of prospectus shall be deemed to be a new registration statement relating to the securities offered therein, and
the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof.
| |
(b) |
The undersigned registrant hereby undertakes that, for purposes of determining any liability under the Securities Act, each filing of the registrant’s annual report pursuant to Section 13(a) or 15(d) of the Exchange Act (and, where applicable, each filing of an employee benefit plan’s annual report pursuant to Section 15(d) of the Exchange Act) that is incorporated by reference in the registration statement shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. |
| |
|
|
| |
(c) |
Insofar as indemnification for liabilities arising under the Securities Act may be permitted to directors, officers and controlling persons of the registrant pursuant to the indemnification provisions described herein, or otherwise, the registrant has been advised that in the opinion of the SEC such indemnification is against public policy as expressed in the Securities Act and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by the registrant of expenses incurred or paid by a director, officer or controlling person of the registrant in the successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities being registered, the registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public policy as expressed in the Securities Act and will be governed by the final adjudication of such issue. |
SIGNATURES
Pursuant to the requirements
of the Securities Act of 1933, as amended, the Registrant has duly caused this Registration Statement to be signed on its behalf by the
undersigned, thereunto duly authorized, in the City of Princeton, State of New Jersey on February 2, 2024.
| |
Agile Therapeutics, Inc. |
| |
|
| |
By: |
| |
|
/s/ Alfred Altomari |
| |
|
Alfred Altomari |
| |
|
Chief Executive Officer |
Pursuant to the requirements
of the Securities Act of 1933, as amended, this registration statement has been signed by the following persons in the capacities and
on the dates indicated.
| Name |
|
Title |
|
Date |
| |
|
|
|
|
| /s/
Alfred Altomari |
|
|
|
February 2,
2024 |
| Alfred
Altomari |
|
Chief Executive Officer
and Director (Principal Executive Officer) |
|
|
| |
|
|
|
|
| /s/
Scott Coiante |
|
|
|
February
2, 2024 |
| Scott
Coiante |
|
Chief Financial Officer
(Principal Financial and Accounting Officer) |
|
|
| |
|
|
|
|
| * |
|
|
|
February
2, 2024 |
| Sharon
Barbari |
|
Director |
|
|
| |
|
|
|
|
| * |
|
|
|
February
2, 2024 |
| Sandra
Carson, M.D., FACOG |
|
Director |
|
|
| |
|
|
|
|
| * |
|
|
|
February 2,
2024 |
| Seth
H.Z. Fischer |
|
Director |
|
|
| |
|
|
|
|
| * |
|
|
|
February 2,
2024 |
| John
Hubbard, Ph.D. |
|
Director |
|
|
| |
|
|
|
|
| * |
|
|
|
February 2,
2024 |
| Josephine
Torrente |
|
Director |
|
|
| * By: |
/s/ Alfred Altomari |
|
| |
Alfred Altomari |
|
| |
Attorney-In-Fact |
|
Agile Therapeutics (NASDAQ:AGRX)
過去 株価チャート
から 5 2024 まで 6 2024
Agile Therapeutics (NASDAQ:AGRX)
過去 株価チャート
から 6 2023 まで 6 2024