false
0001535955
0001535955
2023-11-13
2023-11-13
iso4217:USD
xbrli:shares
iso4217:USD
xbrli:shares
UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
WASHINGTON,
D.C. 20549
FORM
8-K
CURRENT
REPORT
Pursuant
to Section 13 or 15(d) of
The
Securities Exchange Act of 1934
Date
of Report (Date of Earliest Event Reported):
November
13, 2023

LIPOCINE
INC.
(Exact
name of registrant as specified in its charter)
Commission
File No. 001-36357
| Delaware |
|
99-0370688 |
(State
or other jurisdiction
of
incorporation) |
|
(IRS
Employer
Identification
Number) |
675
Arapeen Drive, Suite 202
Salt
Lake City, Utah 84108
(Address
of principal executive offices) (Zip Code)
Registrant’s
telephone number, including area code: (801) 994-7383
Former
name or former address, if changed since last report: Not Applicable
Check
the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under
any of the following provisions:
| ☐ |
Written
communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| |
|
| ☐ |
Soliciting
material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| |
|
| ☐ |
Pre-commencement
communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| |
|
| ☐ |
Pre-commencement
communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities
registered pursuant to Section 12(b) of the Act:
| Title
of each class |
|
Trading
Symbol(s) |
|
Name
of each exchange on which registered |
| Common
Stock, par value $0.0001 per share |
|
LPCN |
|
The
NASDAQ Stock Market LLC |
Indicate
by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (17 CFR §
230.405) or Rule 12b-2 of the Securities Exchange Act of 1934 (17 CFR § 240.12b-2).
Emerging
growth company ☐
If
an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying
with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item
8.01 Other Events
On
November 13, 2023, the Company issued a press release announcing a late breaking presentation on LPCN 1148 Phase 2 Results at The American
Association for the Study of Liver Diseases (AASLD) The Liver Meeting® 2023. The press release is filed as Exhibit 99.1 and is incorporated
herein by reference.
The
Company’s materials to be presented are filed as Exhibits 99.2 and 99.3.
Item
9.01 Financial Statements and Exhibits.
(d)
Exhibits
The
following exhibits are filed with this report:
SIGNATURES
Pursuant
to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by
the undersigned hereunto duly authorized.
| |
|
|
LIPOCINE
INC. |
| |
|
|
|
|
| Date: |
November
13, 2023 |
|
By: |
/s/
Mahesh V. Patel |
| |
|
|
|
Mahesh
V. Patel |
| |
|
|
|
President
and Chief Executive Officer |
Exhibit
99.1

Lipocine
Releases Late Breaking Presentation on LPCN 1148 Phase 2 Results at The Liver Meeting® 2023
Results
demonstrate improvements in sarcopenia and other clinically meaningful outcomes
Patients
converting from placebo to LPCN 1148 in the ongoing open label extension also showed improvement in sarcopenia
SALT
LAKE CITY, November 13, 2023 — Lipocine Inc. (NASDAQ: LPCN), a biopharmaceutical company focused on treating Central Nervous System
(CNS) disorders, today announced that results of its Phase 2 study evaluating LPCN 1148 are being presented at The American Association
for the Study of Liver Diseases (AASLD) – The Liver Meeting® 2023, taking place in Boston MA. The results are featured in a
late-breaking oral presentation and e-poster by Dr. Arun Sanyal, MD, Director, Stravitz-Sanyal
Institute for Liver Disease and Metabolic Health, Virginia Commonwealth University, advisor to Lipocine.
“We
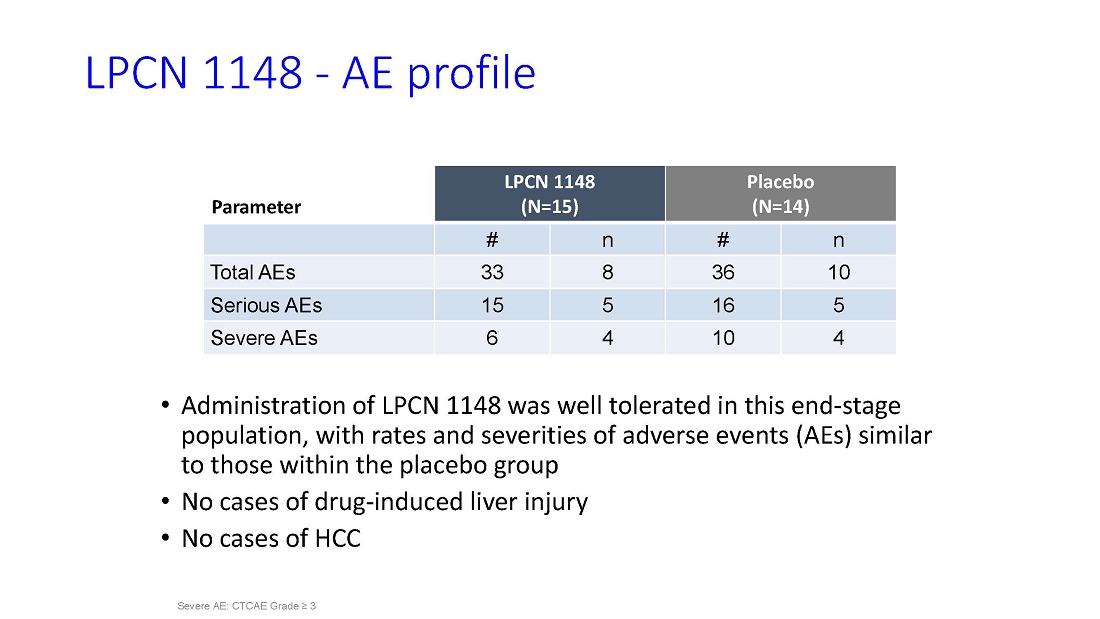
are very pleased to share the data from our successful Phase 2 study of LPCN 1148 with the hepatology community at this year’s
Liver Meeting,” said Dr. Mahesh Patel, President and CEO of Lipocine. “The data show that LPCN 1148 improved multiple additional
clinically meaningful and surrogate outcomes, including hepatic encephalopathy. An Open Label Extension is now ongoing, and we look forward
to reporting further data in Q1 2024.”
The
Phase 2 proof of concept study (NCT # 04874350) is a 52-week ongoing randomized placebo-controlled study in sarcopenic male patients
with cirrhosis on the liver transplant waitlist. After week 24, all patients enter a single arm open-label extension (“OLE”)
stage of the study and receive LPCN 1148. The purposes of the OLE are 1) to evaluate the safety and efficacy of continued exposure with
LPCN 1148 in this population, and 2) to increase the number of participants on LPCN 1148 treatment.
A
copy of the presentation and e-poster will be available on the Lipocine corporate website.
About
LPCN 1148
Lipocine
is currently evaluating LPCN 1148 comprising testosterone laurate (“TL”) for the management of decompensated cirrhosis. The
Company believes LPCN 1148 targets unmet needs for cirrhosis subjects including improvement in the quality of life of patients while
on the liver transplant waiting list, prevention or reduction in the occurrence of new decompensation events such as hepatic encephalopathy
(“HE”), and improvement in post liver transplant survival, including outcomes and costs.
About
Lipocine
Lipocine
is a biopharmaceutical company leveraging its proprietary technology platform to augment therapeutics through effective oral delivery
to develop differentiated products for CNS disorders. Lipocine has drug candidates in development as well as drug candidates for which
we are exploring partnering. Our drug candidates represent enablement of differentiated, patient friendly oral delivery options for favorable
benefit to risk profile which target large addressable markets with significant unmet medical needs.
Lipocine’s
clinical development candidates include: LPCN 1154, oral brexanolone, for the potential treatment of postpartum depression, LPCN 2101
for the potential treatment of epilepsy and LPCN 1148, a novel androgen receptor agonist prodrug for oral administration targeted for
the management of symptoms associated with liver cirrhosis. Lipocine is exploring partnering opportunities for LPCN 1107, our candidate
for prevention of preterm birth, LPCN1154, for rapid relief of postpartum depression, LPCN 1148, for the management of decompensated
cirrhosis, LPCN 1144, our candidate for treatment of non-cirrhotic NASH, and LPCN 1111, a once-a-day therapy candidate for testosterone
replacement therapy (TRT). TLANDO, a novel oral prodrug of testosterone containing testosterone undecanoate developed by Lipocine, is
approved by the FDA for conditions associated with a deficiency of endogenous testosterone, also known as hypogonadism, in adult males.
For more information, please visit www.lipocine.com.
Forward-Looking
Statements
This
release contains “forward-looking statements” that are made pursuant to the safe harbor provisions of the Private Securities
Litigation Reform Act of 1995 and include statements that are not historical facts regarding our product development efforts, the application
of our proprietary platform in developing new treatments for CNS disorders, our product candidates and related clinical trials, our development
of and filing of a NDA with the FDA for LPCN 1148,and the potential uses and benefits of our product candidates. Investors are cautioned
that all such forward-looking statements involve risks and uncertainties, including, without limitation, the risks that we may not be
successful in developing product candidates to treat CNS disorders, we may not have sufficient capital to complete the development processes
for our product candidates, we may not be able to enter into partnerships or other strategic relationships to monetize our non-core assets,
the FDA will not approve any of our products, risks related to our products, expected product benefits not being realized, clinical and
regulatory expectations and plans not being realized, new regulatory developments and requirements, risks related to the FDA approval
process including the receipt of regulatory approvals and our ability to utilize a streamlined approval pathway for LPCN 1154, the results
and timing of clinical trials, patient acceptance of Lipocine’s products, the manufacturing and commercialization of Lipocine’s
products, and other risks detailed in Lipocine’s filings with the SEC, including, without limitation, its Form 10-K and other reports
on Forms 8-K and 10-Q, all of which can be obtained on the SEC website at www.sec.gov. Lipocine assumes no obligation to update or revise
publicly any forward-looking statements contained in this release, except as required by law.
For
further information:
Krista
Fogarty
Phone:
(801) 994-7383
kf@lipocine.com
Investors:
PJ
Kelleher
Phone:
(617) 430-7579
pkelleher@lifesciadvisors.com
Exhibit 99.2
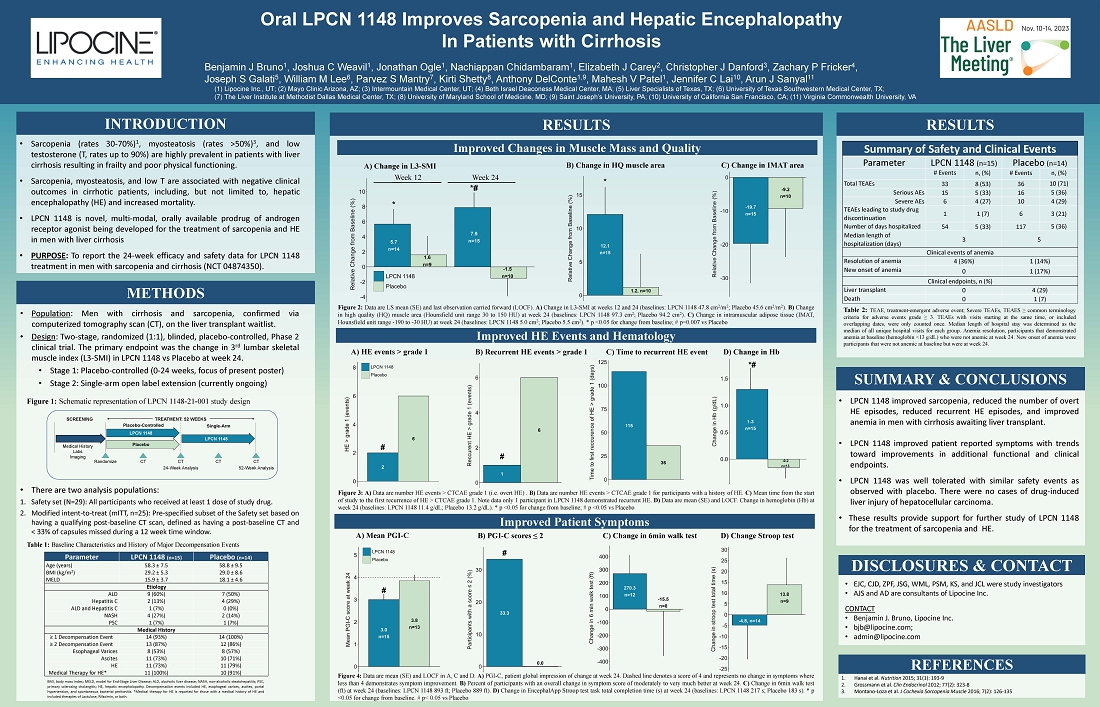
Exhibit 99.3





















v3.23.3
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 2 such as Street or Suite number
| Name: |
dei_EntityAddressAddressLine2 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the state or province.
| Name: |
dei_EntityAddressStateOrProvince |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:stateOrProvinceItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 13e
-Subsection 4c
| Name: |
dei_PreCommencementIssuerTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14d
-Subsection 2b
| Name: |
dei_PreCommencementTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Section 14a
-Number 240
-Subsection 12
| Name: |
dei_SolicitingMaterial |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Securities Act
-Number 230
-Section 425
| Name: |
dei_WrittenCommunications |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
Lipocine (NASDAQ:LPCN)
過去 株価チャート
から 8 2024 まで 9 2024
Lipocine (NASDAQ:LPCN)
過去 株価チャート
から 9 2023 まで 9 2024