China Approves Merck's Molnupiravir for Emergency Use, Regulator Says -- Update
2022年12月30日 - 9:33PM
Dow Jones News
China's top drug regulator said Friday that it approved Merck
& Co. and Ridgeback Biotherapeutics' Molnupiravir for emergency
use on Thursday, as the country grapples with waves of infections
after Beijing abruptly reversed its stringent Covid-19 restrictions
earlier this month.
The National Medical Products Administration said it is
requiring the approval holder to continue relevant research,
complete conditional requirements and submit follow-up research
results in a timely manner, according to a statement posted on its
website Friday.
Molnupiravir and Pfizer Inc.'s Paxlovid are rival drugs for the
treatment of Covid-19. Though Paxlovid pill was found in clinical
trials to be more effective than Molnupiravir, the latter is also
in high demand as an easy-to-use treatment that can be taken at
home.
Write to Singapore Editors at singaporeeditors@dowjones.com
(END) Dow Jones Newswires
December 30, 2022 07:18 ET (12:18 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
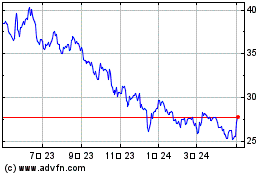
Pfizer (NYSE:PFE)
過去 株価チャート
から 3 2024 まで 4 2024
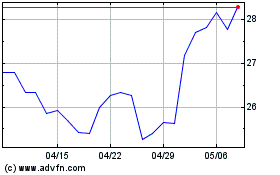
Pfizer (NYSE:PFE)
過去 株価チャート
から 4 2023 まで 4 2024