Pfizer, Valneva Initiate Phase 3 Study of Lyme Disease Vaccine VLA15
2022年8月9日 - 6:36AM
Dow Jones News
By Denny Jacob
Drug giant Pfizer Inc. and specialty vaccine company Valneva SE
said Monday they initiated a Phase 3 clinical study for VLA15, a
Lyme disease vaccine candidate.
The study will enroll about 6,000 participants ages five and up
who will receive three doses of VLA15 or a placebo as a primary
vaccine series followed by one booster dose of either the vaccine
or the placebo, the companies said. The study will be conducted at
up to 50 sites in areas where Lyme disease, an infection caused by
bacteria passed to humans by infected ticks, is highly endemic
including Finland, Germany and the U.S., among others.
Pfizer said it could potentially submit a biologics license
application to the U.S. Food and Drug Administration and marketing
authorization application to the European Medicines Agency in 2025
pending a successfully completed Phase 3 study.
As part of its collaboration agreement, Pfizer said it will make
a $25 million milestone payment to Valneva upon initiation of the
study.
Write to Denny Jacob at denny.jacob@wsj.com
(END) Dow Jones Newswires
August 08, 2022 17:21 ET (21:21 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
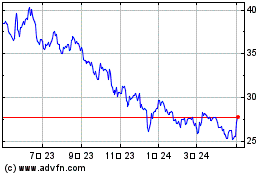
Pfizer (NYSE:PFE)
過去 株価チャート
から 3 2024 まで 4 2024
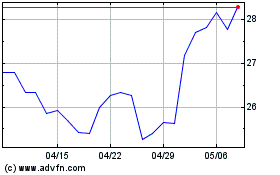
Pfizer (NYSE:PFE)
過去 株価チャート
から 4 2023 まで 4 2024